Human Peripheral Blood Neutrophil Isolation for Interrogating the Parkinson’s Associated LRRK2 Kinase Pathway by Assessing Rab10 Phosphorylation
Summary
Mutations in the leucine rich repeat kinase 2 gene (LRRK2) cause hereditary Parkinson’s disease. We have developed an easy and robust method for assessing LRRK2-controlled phosphorylation of Rab10 in human peripheral blood neutrophils. This may help identify individuals with increased LRRK2 kinase pathway activity.
Abstract
The leucine rich repeat kinase 2 (LRRK2) is the most frequently mutated gene in hereditary Parkinson’ disease (PD) and all pathogenic LRRK2 mutations result in hyperactivation of its kinase function. Here, we describe an easy and robust assay to quantify LRRK2 kinase pathway activity in human peripheral blood neutrophils by measuring LRRK2-controlled phosphorylation of one of its physiological substrates, Rab10 at threonine 73. The immunoblotting analysis described requires a fully selective and phosphospecific antibody that recognizes the Rab10 Thr73 epitope phosphorylated by LRRK2, such as the MJFF-pRab10 rabbit monoclonal antibody. It uses human peripheral blood neutrophils, because peripheral blood is easily accessible and neutrophils are an abundant and homogenous constituent. Importantly, neutrophils express relatively high levels of both LRRK2 and Rab10. A potential drawback of neutrophils is their high intrinsic serine protease activity, which necessitates the use of very potent protease inhibitors such as the organophosphorus neurotoxin diisopropylfluorophosphate (DIFP) as part of the lysis buffer. Nevertheless, neutrophils are a valuable resource for research into LRRK2 kinase pathway activity in vivo and should be considered for inclusion into PD biorepository collections.
Introduction
Attempts to slow or stop Parkinson’s disease (PD) have thus far failed. The discovery of hyperactivating mutations in the leucine rich repeat kinase 2 (LRRK2) that cause and/or increase the risk for PD has led to the development of LRRK2 kinase inhibitors1,2,3. These have now entered clinical trials4. The exact function of LRRK2 is unclear, but a major advancement has been the identification of a subset of Rab GTPase proteins, including Rab10, as the first bona fide physiological substrates of the LRRK2 kinase5,6,7. Key challenges in the era of disease-modifying therapeutics are biochemical markers of LRRK2 kinase activation status and target engagement of LRRK2 kinase inhibitors.
So far, the principal pharmacokinetic marker for LRRK2 inhibitors in vivo has been a cluster of constitutively phosphorylated serine residues of LRRK2, in particular serine 935, that become dephosphorylated in response to diverse LRRK2 inhibitors8,9. However, serine 935 phosphorylation does not correlate with intrinsic cellular LRRK2 kinase activity because it is not directly phosphorylated by LRRK2 and is still phosphorylated in kinase-inactive LRRK210. LRRK2 kinase activity correlates well with autophosphorylation of serine 1292, but it is in practical terms not a suitable readout for endogenous LRRK2 kinase activity by immunoblot analysis of whole cell extracts due to the current lack of reliable and phosphospecific antibodies for this site10,11.
We have developed a robust and easy assay to quantify LRRK2 kinase pathway activity in human peripheral blood cells that measures LRRK2-controlled phosphorylation of its physiological target protein Rab10 at threonine 7311. Peripheral blood is easily accessible by venesection, which is a low risk and quick procedure that causes minimal discomfort. We focus on human peripheral blood neutrophils because they constitute an abundant (37–80% of all white blood cells) and homogeneous cell population that expresses relatively high levels of both LRRK2 and Rab1011. Furthermore, peripheral blood neutrophils can be isolated quickly and efficiently by employing an immunomagnetic negative approach. To ensure that the subsequent observed Rab10 phosphorylation is mediated by LRRK2, each batch of neutrophils is incubated with or without a potent and selective LRRK2 kinase inhibitor (we use and recommend MLi-2)2,12. This is then followed by cell lysis in a buffer containing the protease inhibitor diisopropyl fluorophosphate (DIFP), which is necessary for suppressing the intrinsic serine protease activity that is known to be high in neutrophils13. For the final analysis by quantitative immunoblotting, we recommend using the MJFF-pRab10 rabbit monoclonal antibody that specifically detects the Rab10 Thr73-phosphoepitope and does not cross-react with other phosphorylated Rab proteins14. Selectivity and specificity of this antibody has been validated in overexpression models of different Rab proteins and a A549 Rab10 knock-out cell line14. Thus, we measure the difference in Rab10 phosphorylation in neutrophil lysates that have been treated with and without a potent and selective LRRK2 kinase inhibitor2. Alternatively, samples could also be analyzed by other methods, such as quantitative mass spectrometry.
In conclusion, LRRK2-controlled Rab10 phosphorylation is a superior marker of LRRK2 kinase activity to LRRK2 phosphorylation at serine 935 and human peripheral blood neutrophils are a valuable resource for PD research into LRRK2. Our protocol provides a robust and easy assay to interrogate LRRK2 pathway activity in peripheral blood neutrophils and allows biochemical stratification of individuals with increased LRRK2 kinase activity15. Importantly, such individuals may benefit from future LRRK2 kinase inhibitor treatment.
Protocol
According to local UK regulation all manipulations and pipetting of human blood are undertaken in a category 2 biological safety cabinet. All procedures were performed in compliance with local ethics review board and all participants have provided informed consent.
1. Preparation
- Prepare 0.1 mL of EDTA Stock Solution 1 containing 100 mM EDTA in phosphate-buffered saline (PBS).
- Prepare 60 mL of EDTA Stock Solution 2 containing 1 mM EDTA in PBS.
- Prepare lysis buffer containing 50 mM Tris-HCl (pH = 7.5), 1% (v/v) Triton X-100, 1 mM EGTA, 1 mM Na3VO4, 50 mM NaF, 10 mM β-glycerophosphate, 5 mM sodium pyrophosphate, 0.27 M sucrose, 0.1% (v/v) β-mercaptoethanol, 1x protease inhibitor cocktail, 1 μg/mL microcystin-LR, and 0.5 mM diisopropyl fluorophosphate (DIFP).
NOTE: The authors routinely use an EDTA-free product, but an EDTA-containing protease inhibitor cocktail should also work. The lysis buffer can be made in advance without the β-mercaptoethanol, protease inhibitors, microcystin-LR, and DIFP, and stored at 4 °C until use. Ensure that the β-mercaptoethanol, protease inhibitors, microcystin-LR, and DIFP is only added immediately before use.
CAUTION: DIFP is toxic and should be handled with care in a fume hood following local health and safety risk assessment. DIFP can be added to the lysis buffer and used immediately. Alternatively, the complete lysis buffer containing all other components, including DIFP, can be aliquoted and stored at -80 °C for subsequent use for at least 4 weeks.
2. Neutrophil isolation from whole blood
- Collect 10 mL of blood into a blood collection tube. Mix gently by inverting tubes 7−8x.
- Transfer 10 mL of blood into a 50 mL conical tube.
- Add 100 μL of EDTA Stock Solution 1 to the blood. Mix gently.
- Add 500 μL of the isolation cocktail (50 μL/mL) from the neutrophil isolation kit (Table of Materials) to the whole blood sample.
- Vortex the magnetic beads from the neutrophil isolation kit for 30 s before use in order to resuspend the very fine magnetic beads.
- Add 500 μL of the magnetic beads to the blood sample and mix gently by inverting the tube several times.
- Incubate at room temperature (RT) for 5 min.
- Fill the tube to 50 mL with EDTA Stock Solution 2. Mix by very gently pipetting up and down 2–3x.
- Place the tube into the magnet and remove the lid to avoid subsequent agitation of the tube.
- Incubate for 10 min at RT.
- Carefully pipette the enriched cell suspension that contains the neutrophils into a new 50 mL conical tube.
NOTE: Do not touch the side of the tube that is in contact with the magnet and avoid collection and perturbation of the red blood cells at the bottom of the tube. Leave approximately 10 mL of the red blood cell suspension behind at the bottom of the tube. - Vortex the magnetic beads for 30 s before use and add 0.5 mL of the magnetic beads to the tube containing the enriched neutrophils. Mix gently by inverting the tube.
- Incubate at RT for 5 min.
- Place the tube into the magnet and remove the lid to avoid subsequent agitation.
- Incubate at RT for 5 min.
- Carefully pipette the enriched cell suspension that contains the neutrophils into a new 50 mL conical tube.
NOTE: Do not touch the side of the tube that is in contact with the magnet. Leave approximately 5 mL of the suspension at the bottom of the tube. - To ensure the complete removal of magnetic beads from the cell mixture, place the tube containing the enriched cells into the magnet.
- Incubate for 10 min at RT.
- Carefully pipette the enriched cell suspension that now contains pure neutrophils into a new 50 mL conical tube.
NOTE: Do not touch the side of the tube that is in contact with the magnet. Leave approximately 5 mL of the suspension at the bottom of the tube. - Mix the isolated cells with 1 mM EDTA Stock Solution 2 to a final volume of approximately 41 mL. Pipette up and down to mix.
- Divide the solution equally into two tubes with approximately 20 mL in each tube.
- Centrifuge both tubes at 335 x g for 5 min.
- During this centrifugation take MLi-2 inhibitor stock (200 µM/1,000x concentration) out of the -80 °C freezer and leave at RT for subsequent use.
- Immediately after the centrifugation step and without agitation of the tubes, pour off the supernatant without disturbing the neutrophil pellets. Resuspend each cell pellet in 10 mL of cell culture media (Table of Materials) at RT by gently pipetting cells up and down 4x.
3. LRRK2 kinase inhibitor treatment of pure neutrophils
- Label one tube "DMSO" and the other tube "MLi-2".
- To the "DMSO" labeled tube, add 10 µL of DMSO and mix gently by pipetting up and down 2x with a 10 mL pipette. To the "MLi-2" labeled tube, add 10 μL of 200 μM MLi-2 stock solution (final concentration 200 nM) and mix gently by pipetting up and down 2x with a 10 mL pipette.
- Incubate the samples for 30 min at RT. Mix gently by inversion every 10 min during the incubation.
- During the incubation period, remove 0.5 M DIFP stock from the -80 °C freezer and place in a fume hood on ice. Remove 1 mg/mL microcystin-LR stock solution from the -80 °C freezer and leave at RT to thaw. Take an aliquot (0.25 mL) of the lysis buffer out of the freezer, allow it to defrost at RT, and then place it on ice for subsequent use.
- Prepare 1 mL of cell culture medium containing 1 μL of DMSO and call this DMSO resuspension buffer. Prepare 1 mL of RPMI media containing 1 μL of 200 μM MLi-2 and call this MLi-2 resuspension buffer.
- After the 30 min incubation period, centrifuge both tubes at 335 x g for 5 min.
- Carefully discard the supernatant in each tube without disturbing the neutrophil pellet.
- For the DMSO labeled sample gently resuspend the pellet in 1 mL of the DMSO resuspension buffer and for the MLi-2 labeled tube, resuspend the pellet in 1 mL of the MLi-2 resuspension buffer.
- Transfer the resuspended cell pellets to corresponding centrifugation tubes labeled "DMSO" and "MLi-2" and centrifuge both tubes at 335 x g for 3 min.
- During the centrifugation step, prepare the lysis buffer. In the fume hood carefully add 0.25 μL of 0.5 M DIFP solution as well as 0.25 μL of 1 mg/mL microcystin-LR to the 0.25 mL lysis buffer. Mix and leave on ice until use.
NOTE: Add DIFP to the lysis buffer within 15 min of cell lysis, because DIFP is relatively unstable in an aqueous solution. - Immediately after the centrifugation, carefully and completely remove all the supernatant with a pipette without disturbing the neutrophil pellet and place the tubes on ice.
- Immediately add 100 µL of lysis buffer containing DIFP and microcystine-LR to each tube. Using a 100–200 μL pipette, resuspend the cell pellets by pipetting up and down about 5–10x.
- Lyse the cells on ice for 10 min.
- Centrifuge tubes at 20,000 x g for 15 min at 4 °C to remove cell debris.
- Transfer the "DMSO" and "MLi-2" supernatants containing the neutrophil lysates into new centrifugation tubes. Discard the debris pellet.
NOTE: The neutrophil lysates are now ready for use or can be snap frozen in liquid nitrogen and stored at -80 °C for future analysis.
Representative Results
Our assay allows interrogating the activation of the PD-associated LRRK2 kinase in human peripheral blood neutrophils with LRRK2-dependent Rab10 phosphorylation as a readout. Neutrophils are a homogenous and abundant peripheral white blood cell population that expresses high levels of both the LRRK2 and Rab10 proteins (Figure 1). The only other cell population among the remaining peripheral blood mononuclear cells (PBMCs) with high copy numbers of both proteins are monocytes, but these make up only 2–12% of white blood cells. This indicates that peripheral blood neutrophils are a more suitable biomatrix for studying LRRK2-controlled Rab10 phosphorylation.
When isolating peripheral blood neutrophils from 10 mL of blood with our procedure, between 0.5–0.75 mg of total protein lysate per donor was obtained (Figure 2A), which is sufficient for a significant number of immunoblot analysis for which only 10 μg per gel lane are required. While checking purity and viability of cells is not routinely performed, we demonstrated for three healthy donors that the purity of isolated neutrophils is between 94–98% and the viability of cells ~99% as determined by flow cytometry analysis using the CD66b−Fluorescein isothiocyanate neutrophil marker and 4',6-Diamidine-2'-phenylindole dihydrochloride (DAPI) staining for viability (Figure 2A).
While the focus of this publication is the isolation and processing of neutrophils from peripheral blood and not the analysis by quantitative Western blotting, Figure 2B demonstrates that the MJFF-pRab10 monoclonal antibody that specifically detects Rab10 phosphorylated at threonine 73 revealed robust signals in the neutrophil samples, which were markedly suppressed by treatment with a potent and specific LRRK2 kinase inhibitor, in this case MLi-2.
Neutrophils contain high levels of serine proteases that can affect subsequent Western blot analysis. While DIFP effectively suppresses the high protease activity in neutrophils, it is a potent organophosphorus neurotoxin and it would be desirable to replace it with an equally effective but less toxic protease inhibitor, such as phenylmethylsulfonyl fluoride (PMSF). We found that Rab10 phosphorylation was equally well preserved when DIFP was replaced by PMSF at a concentration of 2.5 mM (Figure 2C). However, the integrity of the LRRK2 protein was compromised when using PMSF compared to DIFP, suggesting that the larger LRRK2 protein is more susceptible to degradation (Figure 2C).
We have previously shown that another PD-causing gene mutation VPS35 D620N results in hyperactivation of the LRRK2 kinase by a yet unknown mechanism15. Neutrophils from three people with PD harboring a disease-causing heterozygous VPS35 D620N mutation were isolated using the method described in this article (Figure 3). Neutrophil samples from nine healthy donors were isolated as controls15. Immunoblot analysis using the MJFF-pRab10 monoclonal antibody demonstrates a significant, ~3x increase in Rab10 phosphorylation at Thr 73 in neutrophils from Parkinson’s patients with a VPS35 D620N mutation compared to the controls. The total Rab10 protein expression is similar in all 12 neutrophil samples.
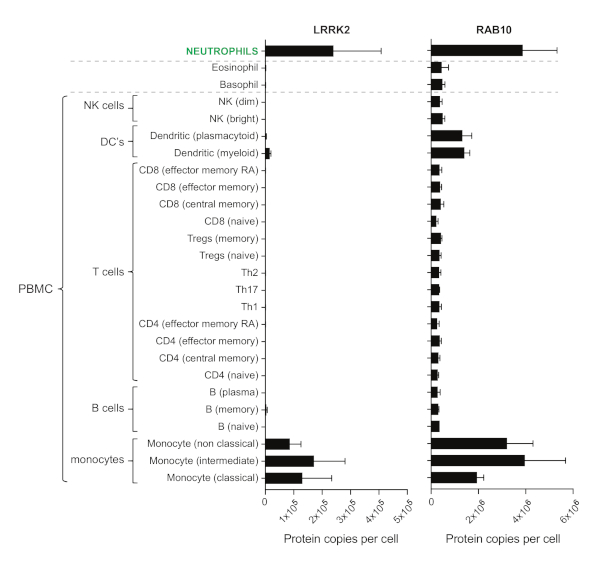
Figure 1: Abundance of LRRK2 and Rab10 proteins in immune cells isolated from human blood using data that is publicly available on the immprot database (http://www.immprot.org)16. The graph shows the number of protein copies per cell for LRRK2 and Rab10 in a range of peripheral blood immune cells, including subsets of T cells, B cells, monocytes, NK cells, dendritic cells, and the granulocytes neutrophils, basophils, and eosinophils. This figure has been modified from Fan et al.11. Please click here to view a larger version of this figure.
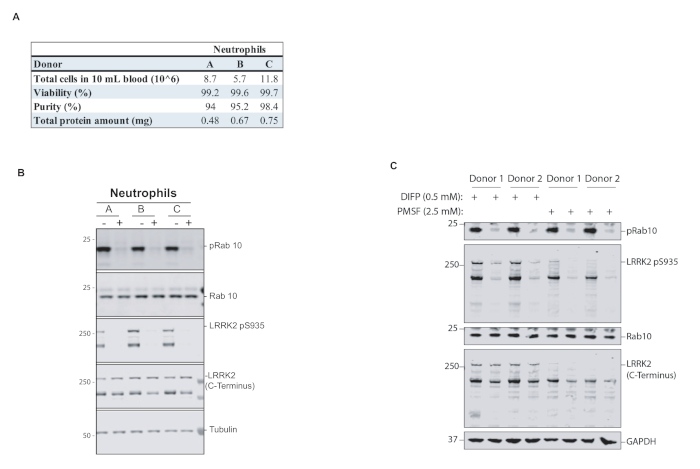
Figure 2: Characterization and analysis of human peripheral blood neutrophils and importance of DIFP for the prevention of proteolytic degradation in neutrophils. (A) Neutrophils were isolated from the whole blood of three healthy donors (A, B, and C) showing purity, viability, and total protein yield after cell lysis. (B) Neutrophils were treated with or without LRRK2 kinase inhibitor (MLi-2), then lysed and subjected to quantitative immunoblot analysis with the indicated antibodies via near-infrared (NIR) fluorescence imaging. (C) Neutrophils were isolated from two healthy donors and treated with 100 nM MLi-2 for 30 min. Cells were lysed in the presence of either 0.5 mM DIFP or 2.5 mM PMSF to block serine protease activity in neutrophils. A and B have been modified from Mir et al.15, while C has been modified from Fan et al.11. Please click here to view a larger version of this figure.
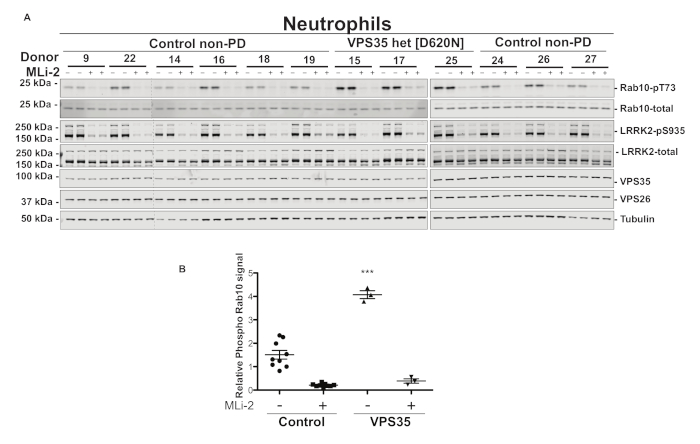
Figure 3: Increased LRRK2 kinase pathway activity in Parkinson’s disease patients harboring a heterozygeous VPS35 D620N mutation. Neutrophils were isolated from nine non-age-matched healthy controls and three PD patients with a disease-causing heterozygous VPS35 D620N mutation. The cells were treated with or without 200 nM MLi-2 for 30 min before cell lysis. (A) A total of 10 µg of whole cell extract subjected to quantitative immunoblot analysis with the indicated antibodies via near-infrared (NIR) fluorescence imaging. Immunoblots were quantified for phospho-Thr73 Rab10:total Rab10 ratio (B). Data were analyzed by one-way ANOVA with Tukey's multiple comparison test. Data presented as means ± SD; ***p < 0.0001. This figure has been modified from Mir et al.15. Please click here to view a larger version of this figure.
Discussion
Compelling clinical, genetic, and biochemical evidence points towards an important role for LRRK2 and in particular its kinase function in Parkinson’s disease7. LRRK2 kinase inhibitors have been developed and are entering clinical trials2,4,12. As such there is a need for exploiting LRRK2 as a biomarker for target engagement as well as patient stratification. Our protocol describes a robust and easy assay for analyzing LRRK2 kinase pathway activation as reflected by the phosphorylation of its physiological substrate Rab10 in the homogenous pool of human peripheral blood neutrophils11,14. For analysis by quantitative immunoblotting, we strongly recommend the use of a highly selective phosphospecific Rab10 antibody (MJFF-pRab10 monoclonal antibody)14,17.
We use human neutrophils because they constitute a homogenous subset of peripheral blood cells that make up the dominant leukocyte population17,18. More importantly, neutrophils also have high protein expression levels of LRRK2 and its substate Rab10 (Figure 1)16. In contrast, the remaining subsets of leukocytes that make up the pool of PBMCs are heterogeneous with variable and predominantly low expression of LRRK2 and Rab10. Monocytes and dendritic cells have high expression levels, but are low in overall abundance16.
While we recommend the use of EDTA vacutainer blood collection tubes, an anticoagulant other than EDTA can be used. However, the presence of EDTA is important for the performance of the neutrophil isolation kit. We therefore recommend adding EDTA to the whole blood sample so that a final concentration of 1 mM EDTA is reached even if an alternative anticoagulant is used. The use of the neutrophil isolation kit allows purifying neutrophils directly from human whole blood by immunomagnetic negative selection in a relatively fast and easy way. The number of available magnets determines how many blood samples can be processed in parallel. It is easily feasible to isolate neutrophils from up to six blood samples in parallel using six magnets. A potential drawback is the associated cost for commercial neutrophil isolation kits and the required magnet. Alternative methods for neutrophil isolation from whole blood have been described and rely on density gradient separation or fluorescence activated cell sorting (FACS), which have significant drawbacks. The former is significantly more time and labor intensive and at least in our hands not as reliable and efficient. The latter requires a FACS machine and additional handling steps would need to be introduced, including the depletion of red blood cells and cell staining, adding time to the cell isolation process. Overall, immunomagnetic isolation is fast, generates a highly pure sample, and avoids excessive handling of the cells. With regards to sample processing, we have previously shown that a delay between blood collection and neutrophil isolation of at least up to 24 h does not result in a significant change or variation in the outcome of our assay, which provides flexibility for future clinical exploitation11.
The neutrophil isolation procedure itself is very easy. We recommend vortexing the magnetic beads before each use. It is also important not to disturb the magnet once the tube is inside the magnet to avoid turbulence and dislodgement of the magnetic beads. Neutrophils are being enriched in suspension by several rounds of immunomagnetic removal of all unwanted cells. Care should be taken not to touch the side of the tube that is in touch with the magnet and during the first round of isolation (step 2.11) so as not to perturb the red blood cells at the bottom of the tube. After the final round of pipetting the enriched cell suspension into a new conical tube (step 2.24), neutrophils are immediately available for onward processing. If neutrophils are used to assess LRRK2 activity in cells, neutrophils are then split into two batches and after pelleting by centrifugation, resuspended in either a LRRK2 kinase inhibitor (here, MLi-2) or DMSO containing cell culture medium. As dephosphorylation in the presence and rephosphorylation in the absence of LRRK2 kinase inhibition are relatively rapid events, care needs to be taken to ensure that the MLi-2 treated neutrophil fraction remains exposed to a LRRK2 kinase inhibitor (e.g., MLi-2) up until cell lysis (steps 3.8–3.10).
There are several centrifugation steps in this protocol using 15 and 50 mL conical tubes. While we routinely use the indicated centrifugation speeds in the protocol, it is possible to increase the centrifugation speed up to 400 x g without adversely affecting the viability of the cells. This results in a slightly firmer neutrophil cell pellet which might help to reduce any potential loss of neutrophil material during the decanting and pipetting off the supernatant steps during this protocol. This will likely increase the yield in terms of total protein lysate obtained. A potential concern is that a higher centrifugation force could activate neutrophils and potentially affect subsequent analysis. Our general recommendation is to handle neutrophils during every step of the protocol as gently as possible.
Our protocol uses the highly potent serine protease inhibitor DIFP (0.5 mM) as part of the neutrophil lysis buffer. DIFP is a potent organophosphorus neurotoxin, and while we have investigated alternative compounds, its addition to the lysis buffer was essential for the analysis of LRRK2-controlled Rab10 phosphorylation in human neutrophils by immunoblotting. For example, replacing DIFP with 1% (w/v) SDS has been used to lyse neutrophils in other studies19 and led to significant protein degradation to the extent that immunoblotting for LRRK2, Rab10, and even the GAPDH loading control did not yield a signal, thus highlighting the importance of including a highly potent protease inhibitor in the lysis buffer (Figure 2C). When replacing DIFP with the less potent, but also less toxic serine protease inhibitor PMSF at 2.5 mM, Rab10 phosporylation was well preserved, but the larger LRRK2 protein underwent significant degradation (Figure 2C). In order to minimize handling of the DIFP stock solution, lysis buffer containing DIFP can be prepared in batches, aliquoted and stored at -20 °C or -80 °C11.With regards to the analysis by quantitative immunoblotting, it is paramount to use a phosphospecific antibody that has been demonstrated to be selective for only a single LRRK2 phosphorylated Rab protein, such as the MJFF-pRab10 antibody used for our studies14.
The protocol can also be scaled up using a maximal volume of whole blood of up to 25 mL, which is the limit that can be processed in one isolation procedure using one magnet, which is able to hold 50 mL conical tubes. The only steps that would need adjustments are step 2.4 (adding 50 µL of isolation cocktail per mL of blood), steps 2.6 and 2.12 (adding 50 µL of magnetic beads per mL of blood), and a proportionate increase in the volume of lysis buffer for neutrophil lysis in step 3.12. All other steps can be kept identical.
In summary, our protocol describes an easy and robust method to isolate human neutrophils from peripheral blood. Neutrophils can then be treated with and without a potent and specific LRRK2 kinase inhibitor to enable the quantification of the LRRK2-controlled phosphorylation of Rab10 in vivo. This can be useful for stratifying individuals according to LRRK2 kinase pathway activity and for identifying those with pathway hyperactivation who might benefit from future LRRK2 kinase inhibitor treatment. While this will be unlikely the case for the majority of people with PD, specifically idiopathic PD, our assay has already been successfully deployed in individuals carrying a rare, heterozygous mutation in another PD-associated gene, VPS35 D620N, where LRRK2 kinase pathway activity is significantly increased by a yet unknown mechanism15. We had previously examined LRRK2-controlled Rab 10 phosphorylation levels in a small number of individuals carrying the more common LRRK2 G2019S mutation that is known to activate LRRK2 kinase activity only modestly by a factor of around two without detecting a significant difference when compared to controls and patients with idiopathic PD with our assay11,14. This and further unpublished data suggest that LRRK2 kinase activity probably requires an increase of >3x in order to yield a significant result using quantitative immunoblotting. However, the sensitivity for detecting LRRK2 kinase pathway activation may likely be increased if deploying state-of-the-art mass spectrometry technology.
We suggest that neutrophils are a valuable resource for Parkinson’s disease research into LRRK2 kinase pathway activity and might help identifying individuals who could benefit from future LRRK2 kinase inhibitor treatment.
Declarações
The authors have nothing to disclose.
Acknowledgements
We thank the healthy volunteers who kindly donated blood for the present study. We thank The Michael J. Fox Foundation for Parkinson’s Research (MJFF) and the Fox BioNet study leadership (FBN) for their support and input towards the written protocol and the video. We thank Professor Alexander Zimprich from the University of Vienna in Austria for testing our protocol and collaboration. We value the contributions of Paul Davies to the project (general manager of the MRC PPU). We also recognize the excellent technical support of the MRC Protein Phosphorylation and Ubiquitylation Unit (PPU) namely Chemical Synthesis (Natalia Shpiro for synthesising MLi-2), MRC PPU Reagents and Services antibody purification teams (coordinated by Hilary McLauchlan and James Hastie). We thank Mhairi Towler and Fraser Murdoch from Vivomotion for their help with making the videos and animations. We thank Steve Soave from 81 films for assistance with the final edits. Esther Sammler is supported by a Scottish Senior Clinical Academic Fellowship and has received funding from Parkinson's UK (K-1706).
Materials
| 1 mL Pipette tips | Sarstedt | 70.762 | or equivalent |
| 1.5 mL Micro tubes | Sarstedt | 72.690.001 | or equivalent |
| 10 mL Pipette tips | Sarstedt | 86.1254.025 | or equivalent |
| 10 μL Pipette tips | Sarstedt | 70.113 | or equivalent |
| 15 mL falcon tube | Cellstar | 188 271 | or equivalent |
| 200 μL Pipette tips | Sarstedt | 70.760.002 | or equivalent |
| 25 mL Pipette tips | Sarstedt | 86.1685.001 | or equivalent |
| 50 mL falcon tube | Cellstar | 227 261 | or equivalent |
| BD Vacutainer Hemogard Closure Plastic K2-EDTA Tube | BD | BD 367525 | or equivalent |
| Beckman Coulter Allegra X-15R centrifuge | Beckman | or equivalent centrifuge with swimging bucket rotator for 15 mL and 50 mL falcon tubes at speed 1000-1200 x g | |
| Category 2 biological safety cabinet. | |||
| cOmplete(EDTA-free) protease inhibitor cocktail | Roche | 11836170001 | |
| DIFP (Diisopropylfluorophosphate) | Sigma | D0879 | Prepare 0.5M stock solution in isopropanol using special precautions |
| Dimethyl sulfoxide | Sigma | 6250 | |
| Dry ice or liquid nitrogene | |||
| Dulbecco's phosphate-buffered saline | ThermoFisher | 14190094 | or equivalent |
| Easy 50 EasySep Magnet | Stemcell | 18002 | for holding 1 x 50ml conical tube |
| EasySep Direct Human Neutrophil Isolation Kit | Stemcell | 19666 | This contains Solutions called “Isolation Cocktail” and “RapidSpheres magnetic beads |
| EGTA | Sigma | E3889 | |
| Eppendorf centrifuge 5417R centrifuge | Eppendorf | ||
| Ethanol, in spray bottle | |||
| Ethylenediaminetetraacetic acid | Sigma | E6758 | |
| Ice | |||
| Isopropanol (anhydrous grade) | Sigma | 278475 | |
| Lysis buffer (50 mM Tris-HCl pH 7.5, 1%(v/v) Triton X-100, 1 mM EGTA, 1 mM Na3VO4, 50 mM NaF, 10 mM β-glycerophosphate, 5 mM sodium pyrophosphate, 0.27 M sucrose, 0.1% (v/v) β-mercaptoethanol, 1x cOmplete(EDTA-free) protease inhibitor cocktail (Roche), 1 μg/ml Microcystin-LR, 0.5 mM diisopropylfluorophosphate (DIFP). | alternatively frozen lysis buffer in aliquots without Microcystin-LR, DIFP available from MRC-PPU Reagents (http://mrcppureagents.dundee.ac.uk/) | ||
| Merck LRRK2 inhibitor II (MLi-2) | Merck | 438194-10MG | or equivalent (potent and selective LRRK2 inhinitor) |
| Microcystin-LR | Enzo Life Sciences | ALX-350-012-M001 | 1 mg/ml stock in DMSO and store at -80 oC. |
| Na3VO4 | Aldrich | 450243 | |
| NaF | Sigma | S7920 | |
| Odyssey CLx scan Western Blot imaging system | Odyssey | ||
| Permanent marker pen | |||
| Personal protection equipment | |||
| RPMI 1640 Medium | ThermoFisher | 21875034 | or equivalent |
| sodium pyrophosphate | Sigma | S22 | |
| sucrose | Sigma | S0389 | |
| β-glycerophosphate | Sigma | 50020 | |
| β-mercaptoethanol | Sigma | M3148 | |
| Suggested antibodies for Western blotting | |||
| Anti-RAB10 (phospho T73) antibody [MJF-R21] | abcam | ab230261 | |
| Anti-α-tubulin | Cell Signaling Technologies | 5174 | used at 1:2000 dilution |
| Goat anti-mouse IRDye 680LT | LI-COR | 926-68020 | used at 1:10,000 dilution |
| Goat anti-mouse IRDye 800CW | LI-COR | 926-32210 | used at 1:10,000 dilution |
| Goat anti-rabbit IRDye 800CW | LI-COR | 926-32211 | used at 1:10,000 dilution |
| MJFF-total Rab10 mouse antibody | generated by nanoTools (nanotools.de) | not applicable* | used at 2 μg/ml final concentration; * The MJFF-total Rab10 antibody generated by nanoTools (www.nanotools.de) [11] will be commercialised by the Michael J Fox Foundation in 2018 |
| Mouse anti-LRRK2 C-terminus antibody | Antibodies Incorporated | 75-253 | used at 1 μg/ml final concentration |
| pS935-LRRK2 | MRC PPU Reagents and Services | UDD2 | MJFF-total Rab10 mouse antibody |
Referências
- Paisan-Ruiz, C., et al. Cloning of the gene containing mutations that cause PARK8-linked Parkinson’s disease. Neuron. 44 (4), 595-600 (2004).
- Fell, M. J., et al. MLi-2, a Potent, Selective, and Centrally Active Compound for Exploring the Therapeutic Potential and Safety of LRRK2 Kinase Inhibition. Journal of Pharmacology and Experimental Therapeutics. 355 (3), 397-409 (2015).
- Zimprich, A., et al. Mutations in LRRK2 cause autosomal-dominant parkinsonism with pleomorphic pathology. Neuron. 44 (4), 601-607 (2004).
- Sardi, S. P., Cedarbaum, J. M., Brundin, P. Targeted Therapies for Parkinson’s Disease: From Genetics to the Clinic. Journal of Movement Disorders. 33 (5), 684-696 (2018).
- Steger, M., et al. Phosphoproteomics reveals that Parkinson’s disease kinase LRRK2 regulates a subset of Rab GTPases. Elife. 5, (2016).
- Ito, G., et al. Phos-tag analysis of Rab10 phosphorylation by LRRK2: a powerful assay for assessing kinase function and inhibitors. Biochemical Journal. 473 (17), 2671-2685 (2016).
- Alessi, D. R., SammLer, E. LRRK2 kinase in Parkinson’s disease. Science. 360 (6384), 36-37 (2018).
- Yue, M., et al. Progressive dopaminergic alterations and mitochondrial abnormalities in LRRK2 G2019S knock-in mice. Neurobiology of Disease. 78, 172-195 (2015).
- Doggett, E. A., Zhao, J., Mork, C. N., Hu, D., Nichols, R. J. Phosphorylation of LRRK2 serines 955 and 973 is disrupted by Parkinson’s disease mutations and LRRK2 pharmacological inhibition. Journal of Neurochemistry. 120 (1), 37-45 (2012).
- Sheng, Z., et al. Ser1292 autophosphorylation is an indicator of LRRK2 kinase activity and contributes to the cellular effects of PD mutations. Science Translational Medicine. 4 (164), (2012).
- Fan, Y., et al. Interrogating Parkinson’s disease LRRK2 kinase pathway activity by assessing Rab10 phosphorylation in human neutrophils. Biochemical Journal. 475 (1), 23-44 (2018).
- Scott, J. D., et al. Discovery of a 3-(4-Pyrimidinyl) Indazole (MLi-2), an Orally Available and Selective Leucine-Rich Repeat Kinase 2 (LRRK2) Inhibitor that Reduces Brain Kinase Activity. Journal of Medicinal Chemistry. 60 (7), 2983-2992 (2017).
- Pham, C. T. Neutrophil serine proteases: specific regulators of inflammation. Nature Reviews Immunology. 6 (7), 541-550 (2006).
- Lis, P., et al. Development of phospho-specific Rab protein antibodies to monitor in vivo activity of the LRRK2 Parkinson’s disease kinase. Biochemical Journal. 475 (1), 1-22 (2018).
- Mir, R., et al. The Parkinson’s disease VPS35[D620N] mutation enhances LRRK2-mediated Rab protein phosphorylation in mouse and human. Biochemical Journal. 475 (11), 1861-1883 (2018).
- Rieckmann, J. C., et al. Social network architecture of human immune cells unveiled by quantitative proteomics. Nature Immunology. 18 (5), 583-593 (2017).
- Borregaard, N. Neutrophils, from marrow to microbes. Immunity. 33 (5), 657-670 (2010).
- Bain, B., Dean, A., Broom, G. The estimation of the lymphocyte percentage by the Coulter Counter Model S Plus III. Clinical & Laboratory Haematology. 6 (3), 273-285 (1984).
- Tomazella, G. G., et al. Proteomic analysis of total cellular proteins of human neutrophils. Proteome Science. 7, 32 (2009).

