Developing Photosensitizer-Cobaloxime Hybrids for Solar-Driven H2 Production in Aqueous Aerobic Conditions
Summary
We have directly incorporated a stilbene-based organic dye into a cobaloxime core to generate a photosensitizer-catalyst dyad for photocatalytic H2 production. We have also developed a simple experimental setup for evaluating the light-driven H2 production by photocatalytic assemblies.
Abstract
Developing photocatalytic H2 production devices is the one of the key steps for constructing a global H2-based renewable energy infrastructure. A number of photoactive assemblies have emerged where a photosensitizer and cobaloxime-based H2 production catalysts work in tandem to convert light energy into the H-H chemical bonds. However, the long-term instability of these assemblies and the need for hazardous proton sources have limited their usage. Here, in this work, we have integrated a stilbene-based organic dye into the periphery of a cobaloxime core via a distinct axial pyridine linkage. This strategy allowed us to develop a photosensitizer-catalyst hybrid structure with the same molecular framework. In this article, we have explained the detailed procedure of the synthesis of this hybrid molecule in addition to its comprehensive chemical characterization. The structural and optical studies have exhibited an intense electronic interaction between the cobaloxime core and the organic photosensitizer. The cobaloxime was active for H2 production even in the presence of water as the proton source. Here, we have developed a simple airtight system connected with an online H2 detector for the investigation of the photocatalytic activity by this hybrid complex. This photosensitizer-catalyst dyad present in the experimental setup continuously produced H2 once it was exposed in the natural sunlight. This photocatalytic H2 production by the hybrid complex was observed in aqueous/organic mixture media in the presence of a sacrificial electron donor under complete aerobic conditions. Thus, this photocatalysis measurement system along with the photosensitizer-catalyst dyad provide valuable insight for the development of next generation photocatalytic H2 production devices.
Introduction
In the modern world, fossil fuels such as coal, oil, and natural gas supply a majority share of the energy. However, they produce copious amount of CO2 during the energy harvesting to negatively impact the global climate1. In coming years, a steep rise in energy demand is predicted worldwide following the continuous growth of population and constant improvement in human lifestyle. Thus, there is an active search for a suitable alternative energy resource to match the global energy requirement. Renewable energy resources like solar, wind, and tidal power have emerged as one of the best solutions due to their environment-friendly zero carbon energy transduction process2. However, the intermittent nature of these energy resources has so far limited their extensive application. A possible solution of this problem can be found in biology; solar energy is efficiently transformed into chemical energy during photosynthesis3. Following this clue, researchers have developed artificial photosynthetic strategies for storing solar energy into chemical bonds following a number of small molecule activation reactions4,5. The H2 molecule has been considered one of the most appealing chemical vectors due to their high energy density and simplicity of their chemical transformation6,7.
The presence of a photosensitizer and a H2 production catalyst are essential for an active solar-driven H2 production setup. Here in this work, we will focus on the cobalt-based molecular complex cobaloxime for the catalytic segment. Typically, a hexa-coordinated cobalt center is bound in a square planar N4 geometry, derived from the dimethylglyoxime (dmg) ligands, in cobaloximes. The complementary Cl– ions, solvent molecules (such as water or acetonitrile) or pyridine derivatives ligate in the residual axial positions8. Cobaloximes are long known for active H2 production electrocatalysis and their reactivity can be tuned by appending variable functionalities on the axial pyridine9,10,11,12. The relatively uncomplicated syntheses, oxygen tolerance under catalytic conditions, and moderate catalytic response of cobaloximes have prompted researchers to explore their photocatalytic H2 production reactivity. The Hawecker group was the pioneer in demonstrating the light-driven H2 production activity of cobaloximes by utilizing Ru(polypyridyl)-based photosensitizers13. Eisenberg and his coworkers utilized platinum (Pt)-based inorganic photosensitizers to induce photocatalytic H2 production in tandem with cobaloxime catalysts14,15. Later, the Che group utilized organo-gold photosensitizer to replicate similar activity16. Fontecave and Artero expanded the range of photosensitizers by applying iridium (Ir)-based molecules17. The practical applications of these photocatalytic systems were heading towards a roadblock due to the use of expensive metal-based photosensitizers. The Eisenberg and Sun research groups have countered that by independently devising organic dye-based photo-driven H2 production systems18,19. Despite the successful photo-driven H2 production by all these systems, it was observed that the overall catalytic turnovers were relatively slow20. In all these cases, the photosensitizer and cobaloxime molecules were added as separate moieties in the solution, and the lack of direct communication between them might have hindered the overall efficiency of the system. A number of photosensitizer-cobaloxime dyads were developed to rectify this issue, where a variety of photosensitizers were directly linked with the cobaloxime core via the axial pyridine ligand21,22,23,24,25,26. Sun and co-workers were even successful in developing a noble-metal free device by introducing a Zn-porphyrin motif as a photosensitizer24. Recently, Ott and coworkers have successfully incorporated the cobaloxime catalyst within an metal organic framework (MOF) that displayed photocatalytic H2 production in the presence of organic dye27. However, the inclusion of the high molecular weight photosensitizers into the cobaloxime framework reduced the water solubility while affecting the long-term stability of the dyads under catalytic conditions. The stability of the active dyads under aqueous conditions during the catalysis is crucial as the omnipresent water is an attractive source of protons during the catalysis. Thus, there is a serious need for developing an aqueous soluble, air-stable photosensitizer-cobaloxime dyad system to establish an efficient and economical photo-driven H2 production setup.
Here in this work, we have anchored a stilbene-based organic dye28 as photosensitizer to the cobaloxime core via the axial pyridine linker (Figure 1). The light molecular weight of the dye ensured improved water solubility of the dyad. This stilbene-cobaloxime hybrid molecule was characterized in detail via optical and 1H NMR spectroscopy along with its single crystal structure elucidation. The electrochemical data revealed the active electrocatalytic H2 production by the cobaloxime motif even with the appended organic dye. This hybrid complex exhibited significant photo-driven H2 production when exposed to direct sunlight in the presence of an appropriate sacrificial electron donor in a 30:70 water/DMF (N,N′-dimethylformamide) solution without any degradation of the hybrid structure as complemented by optical spectroscopy studies. A simple photocatalytic device, consisting of a H2 detector, was employed during the photocatalysis of the hybrid complex that demonstrated continuous production of H2 gas under aqueous aerobic condition without any preliminary lag period. Thus, this hybrid complex has the potential to become the base for developing the next generation of solar-driven H2 production catalysts for efficient renewable energy utilization.
Protocol
1. Synthesis of the photosensitizer-catalyst hybrid
- Synthesis of catalyst precursor Co(dmg)2Cl2 complex
NOTE: This complex was synthesized following the modified version of the reported procedure29.- Dissolve 232 mg (1 mmol) of dimethylglyoxime (dmg) ligand (two equivalents in this reaction) in 27 mL of acetone.
- Dissolve 118 mg (0.5 mmol) of CoCl2∙6H2O (one equivalent in this reaction) in 3 mL of deionized water separately that produces a pink color solution.
- Add the aqueous CoCl2 solution drop wise to the acetone solution containing dmg with continuous stirring at room temperature.
- Closely monitor the change in the solution color, which will sequentially turn to bluish green color following the metal addition.
- Continue the reaction for 2 h.
- Filter the reaction mixture through a Grade 40 filter paper and keep the filtrate at 4 °C overnight.
- The next day, obtain the green colored precipitate of Co(dmg)2Cl2 complex (cobaloxime) from the solution and filter it through grade 40 filter paper.
- Dry the sample under air.
- Synthesis of the photosensitizer (PS)-cobaloxime hybrid
NOTE: The stilbene based photosensitizer (PS) was synthesized as per the reported method28. The following steps were followed for the PS-catalyst hybrid complex synthesis.- Add 100 mg (0.277 mmol) of cobaloxime (one equivalent) (synthesized in Step 1) in 5 mL of methanol. It will form a green suspension.
- Add 38 µL (0.277 mmol) of triethylamine (TEA) base (one equivalent) to the green suspension with continuous stirring. The solution will turn transparent brown after within 1 min.
- Add 65 mg (0.277 mmol) of solid stilbene dye (one equivalent) to the previously mentioned TEA added cobalt solution in methanol.
- Continue the stirring for 3 h. Closely monitor the change in the solution, which will sequentially produce the reddish-brown precipitate of the PS-cobaloxime hybrid.
- Filter the reddish-brown precipitate with Grade 40 filter paper and wash it with copious amount of cold methanol (20 mL).
- Dissolve the precipitate in chloroform (10 mL) and collect the reddish-brown filtrate.
- Evaporate the filtrate under reduced pressure using a rotavapor at room temperature.
- Collect the solid reddish-brown product [Observed yield: 76 mg (65%)].
- Recrystallize the product from chloroform solution at room temperature, where the chloroform evaporates slowly to produce reddish-brown crystals of the complex.
2. Characterization of the photosensitizer-cobaloxime hybrid
- NMR characterization
- Dissolve 5.0 mg of the purified PS-Cobaloxime hybrid complex in 650 μL of d6-DMSO.
- Record the 1H NMR in NMR spectrometer at room temperature.
NOTE: 1H NMR signals, in δ (ppm) units with the corresponding number of protons, their identity, and the splitting pattern in parentheses (s = singlet, d = doublet, m = multiplet), are as following: 1H NMR: 2.34 (12H, -dmg-CH3, s), 2.97 (6H, -dye-N-(CH3)2, s), 6.74 (2H, dye-aromatic, d), 6.84 (1H, allylic-H, d), 7.48 (5H, four dye-aromatic, one allylic-H, m), 7.82(2H, dye-aromatic, d), 18.47(2H,dmg-NOH, s).
- UV-Vis spectroscopy
- Prepare a 1.0 mM solution of the PS-cobaloxime complex in N,N′-dimethylformamide (DMF) by adding the appropriately weighed amount of the complex in the solvent.
- Dilute the solution 10 times with blank DMF to generate 0.1 mM solution of the hybrid complex in DMF.
- Further dilute it 5 times with blank DMF to generate 20 μM solution of the hybrid complex in DMF.
- Record the optical spectra of the 20 μM PS-cobaloxime complex solution using a spectrophotometer.
NOTE: UV-Vis peaks (λ/nm), with the corresponding molar extinction coefficient (ε /M-1cm-1) in parentheses, are as follows: 266 (13400) and 425 (14600).
- Single crystal structure determination
- Prepare a concentrated 0.2 M sample of the PS-catalyst hybrid complex in 5 mL of chloroform. Grow reddish-brown (cubic) crystals of the complex from that chloroform solution over 3 days.
- Select a suitable crystal of the complex and mount on a cryo-loop using cryoprotectant (e.g., Paratone oil).
- Collect the single crystal diffraction data for the hybrid complex at 298 K on the diffractometer.
- Apply the empirical absorption correction to the data by employing the multi-scan method in SADABS programming30.
- Resolve the structure by direct methods with SHELXS-97 and refine by the full matrix least square methods on F2 using the SHELXL-201431.
- Electrochemical studies
- Sample preparation
- Prepare a 1 mM solution of the PS-catalyst hybrid complex in HPLC grade DMF containing 0.1 M tetra-N-butyl ammonium fluoride (n-Bu4N+F−/TBAF).
- Place 2 mL of the sample solution prepared in step 1 in the electrochemical cell (volume 5 mL).
- Purge N2 gas through the solution for 30 min to remove oxygen.
- Electrode preparation
- Polish the 1 mm diameter glassy carbon-disc working electrode with 0.25 μm alumina paste prepared in water on a polishing pad.
- Rinse the polished electrode thoroughly with a copious amount of deionized water.
- Place the clean working electrode in the electrochemical cell.
- Place the Ag/AgCl (in 1.0 M AgNO3) reference electrode and the platinum (Pt)-wire counter electrode in the electrochemical cell.
- Connect all the electrodes accordingly to the potentiostat.
- Collecting data
- Stop the N2 gas purging before the electrochemical experiment.
- Keep a continuous flow of N2 above the sample solution in the electrochemical cell.
- Record cyclic voltammograms (CV) of the sample starting from the anodic direction to cathodic direction with appropriate scan rate (0.1 V/s scan rate was used in this experiment).
- Repeat the above experiment by adding appropriate amounts of water (30% water in DMF) and trifluoroacetic acid (TFA) (8 μL of 10x diluted neat TFA), respectively.
- Add ferrocene to the sample solution and record the corresponding CV. Adjust the potential scale with the ferrocene couple (FeCp2+/0 = 0V vs. Ferrocene) for all the collected data. Thus, all the potential values mentioned in this work was internally referenced against Ferrocene couple.
- Sample preparation
3. Catalytic H2 production by the photosensitizer-catalyst hybrid in sunlight
- Photocatalytic H2 production by the PS-catalyst hybrid complex
- Prepare 0.2 mM PS-catalyst hybrid complex in 10 mL of 70:30 DMF water (pH 7, 0.1 MES buffer) in a two-neck test tube.
- Add 1 mL of triethanolamine (TEOA) as the sacrificial electron donor to the sample solution.
- Close the two openings of the test tube with the air-tight septum.
- Connect this setup with the H2 detector with appropriate tubing connections.
NOTE: The H2 detector has two tube connections. One of them acts as the input that goes through an in-built detector to measure the amount of H2 (in ppm units) present in the sample. The measured gas sample then connects back to the reaction vessel by the output tubing. - Place the set up under sunlight for 30 min and monitor the H2 production rate via the detector.
- Monitoring the solar-driven H2 production via gas chromatography (GC)
- Collect 1 mL of headspace gas via gas-tight syringe.
- Inject the collected gas in the gas chromatography (GC) instrument.
- Monitor the resulted gas chromatograph.
- Inject 1 mL of headspace gas collected from a control sample placed under dark.
- Inject 1 mL of gas from a calibrated standard gas mixture containing 1% H2.
Representative Results
In this work, a stilbene photosensitizer-cobaloxime hybrid complex (C1) was synthesized successfully by anchoring the organic dye (L1) derived pyridine motif as the axial ligand to the cobalt core. The 1H NMR data of the hybrid complex clearly demonstrated the presence of both the cobaloxime and organic dye protons in the same complex. As shown in Figure 2, the up-fielded aliphatic region highlighted the presence of both oxime-bound methyl and stilbene N-dimethyl proton signals in appropriate proportions at δ (ppm) 2.34 and 2.97, respectively. The aromatic and unique allylic proton signals from the stilbene skeleton were sighted in the 6.74-7.82 δ (ppm) region, which was highlighted in detail in the inset of Figure 2. The stability of the cobaloxime core was exemplified by the presence of the intra-molecular hydrogen bonding in the oxime moiety in the far down field region (~12.47 δ (ppm))11. The optical spectra of the hybrid complex C1 exhibited two major signals (Figure 3). In the UV region, a distinct signal was observed at 266 nm. This signal resembled the characteristic π‒π* transition originated from the oxime scaffold. Another optical transition was noticed for C1 in the visible region at 425 nm. This signal is significantly red-shifted compared to the typical π‒π* transition observed for the stilbene compound (λmax 385 nm) (Figure 3)32. This transition observed in C1 possibly has significant contribution from the Npyrdine‒Co(III) ligand to metal charge transfer (LMCT) transition, analogous to similar axial pyrine bound cobaloximes29,33. The ligation between stilbene-derived pyridine motif and cobaloxime was definitively verified with the single crystal structure data of C1. As shown in Figure 4, the critical Npyrdine‒Co bond distance was measured at 1.965 Å, similar to typical axial Npyrdine‒Co bonds9. The aromatic rings along with the allylic group remained in the same plane in the hybrid complex C1 that ensure an elongated conjugation in the stilbene moiety. The details of the crystal data collections and data refinement parameters are given in Table 1. The complete crystallographic information file (CIF) of the PS-catalyst hybrid complex was deposited in the Cambridge crystallographic data centre (CCDC No: 1883987)34.
A cyclic voltammetry (CV) experiment was performed with the PS-catalyst-hybrid complex C1 staring with a cathodic scan in the range of 0.5 V to -1.8 V in DMF (Figure 5). An irreversible reduction signal was observed at -1.0 V (vs. Fc+/0) followed by two successive reversible signals at -1.3 and -1.5 V. The first reductive signal can be assigned as the metal based Co(III/II) reduction while the reversible signals were attributed to the stoichiometric redox processes at aromatic organic dye framework32. C1 demonstrated a distinct catalytic signal at -1.25 V when water was added to the solution. Electrocatalytic H2 production was possibly responsible for this cathodic catalytic behavior. This hypothesis was corroborated by a gradual increase in that catalytic response following the addition of TFA in the same solution (Figure 5). The turnover frequency (TOF) for these catalytic responses was tabulated using the following equation:
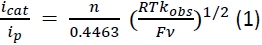
where icat = catalytic current, ip = stoichiometric current, n = number of electrons involved in this process, R = universal gas constant, T = temperature in K, F = 1 Faraday, and ν = scan rate. The TOF for H2 production in the presence of water and aqueous TFA were 30 s-1 and 172 s-1, respectively. The complementary chronocoulometric (bulk electrolysis) experiment was used along with the complementary gas chromatography (GC) to provide further evidence of H2 production during the catalytic step with 70% Faradaic efficiency (details in supplementary section, Figure S1).
The H2 production activity of the cobaloxime core in C1 was further investigated during the photo-catalytic studies. In this experiment, C1 was loaded in an airtight container containing 30:70 water/DMF solvent along with TEOA sacrificial electron donor. This system was connected to the H2 sensor and exposed to natural sunlight (power density ~ 100 mW/cm2) (Figure 6). As shown in Figure 7, the PS-catalyst hybrid complex C1 displayed catalytic H2 production immediately following the sunlight exposure. In this case, an almost linear increase in photocatalytic H2 production was observed over time. The identity and purity of the photo-generated gas accumulated in the headspace of the set-up was validated by gas chromatography (GC). As illustrated in Figure 8, solar-driven, H2 production was confirmed by the GC results. The minimal change in the comparative optical spectra demonstrated the stability of C1 during this experiment (Figure S2).
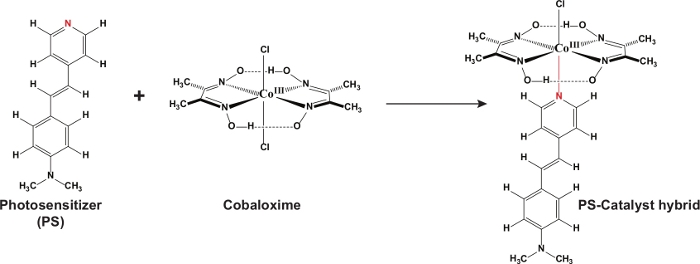
Figure 1: Reaction scheme. The scheme represents the synthetic route for the PS-catalyst hybrid complex. Please click here to view a larger version of this figure.
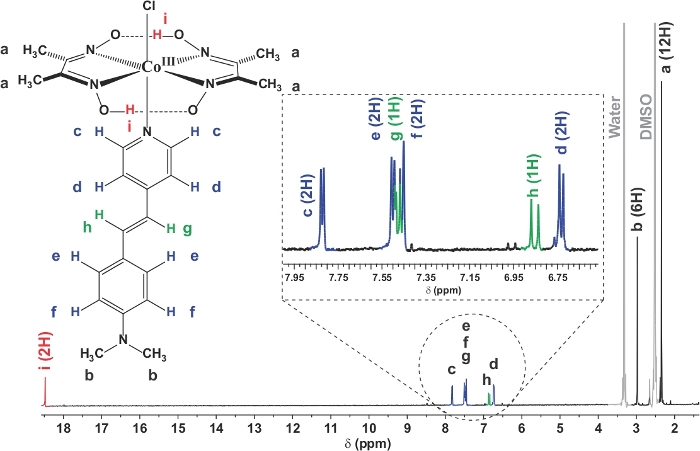
Figure 2: 1H NMR spectra of PS-catalyst hybrid complex C1. This figure displays the 1H NMR of PS-catalyst hybrid complex recorded in d6-DMSO at room temperature. The aliphatic region consists of oxime-methyl groups (12 H, a), and PS-bound N-methyl groups (6 H, b) (black trace). The aromatic region consists of 10 H, containing both aromatic (c, d, e, f) and allylic (g and h) protoms. The oxime (-NOH) protons are the most down-shielded protons (i) (red trace). The inset highlights the detailed splitting pattern of the aromatic (blue trace) and allylic protons (green trace). Please click here to view a larger version of this figure.
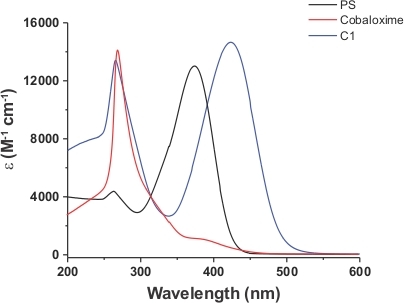
Figure 3: Comparative optical spectra. The comparative Uv-vis spectra of PS (black trace), cobaloxime precursor (red trace), and PS-catalyst dyad C1 (blue trace) recorded in DMF at room temperature. The formation of the hybrid complex distinctly red-shifted the LMCT band, while the π‒π* transition remained same. Please click here to view a larger version of this figure.

Figure 4: Single crystal structure of photosensitizer-Cobaloxime hybrid C1. ORTEP representation for C1 with 50% thermal ellipsoids probability. The carbon (grey), hydrogen (white), oxygen (red), nitrogen (sky-blue), chlorine (green), and cobalt (deep blue) atoms are shown in the figures accordingly. One chloroform molecule was found inside the crystal lattice, but it is omitted here for clarity. Please click here to view a larger version of this figure.
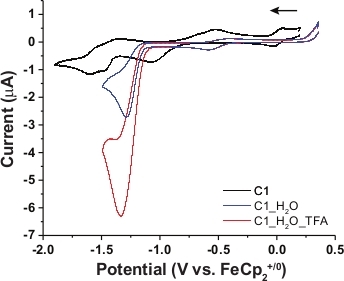
Figure 5: Comparative cyclic voltammograms. The comparative cyclic voltammograms (CVs) of 1 mM C1 in only DMF (black trace), in the presence of 30:70 water/DMF (blue trace), and in the presence of 16 equivalent TFA in 30:70 water/DMF(red trace) were shown in the figure. The scans were performed in the presence of 0.1 M tetra-N-butyl ammonium fluoride (n-Bu4N+F−/TBAF) as supporting electrolyte utilizing 1mm glassy carbon disc working electrode, Ag/AgCl (in 1.0 M AgNO3) reference electrode and platinum (Pt)-wire counter electrode at room temperature with 0.1 V/s scan rate. The initial scan direction is shown with the horizontal black arrow. Please click here to view a larger version of this figure.
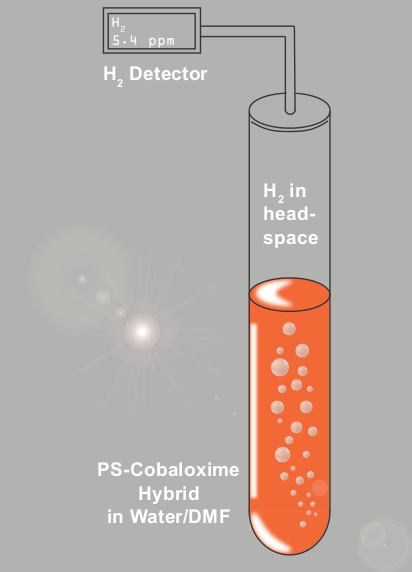
Figure 6: The photocatalytic H2 production monitoring system. The schematic representation of the experimental set-up, consisting of an online H2 detector, used for continuous monitoring H2 production by photosensitizer-cobaloxime dyad C1 under natural sunlight and complete aerobic condition. Please click here to view a larger version of this figure.
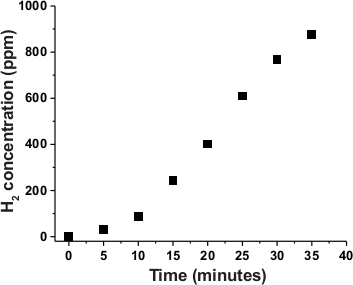
Figure 7: Photocatalytic H2 production by C1 over time. The accumulation of H2 over time during the natural sunlight-driven photocatalysis by photosensitizer-cobaloxime hybrid complex C1 as detected by the online H2 detector. Please click here to view a larger version of this figure.
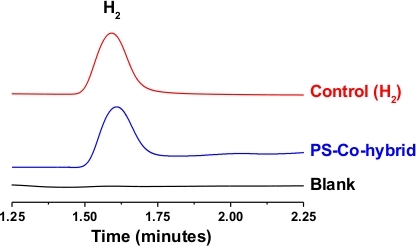
Figure 8: Comparative gas chromatography data. Comparative gas chromatography (GC) data recorded for the head space gas collected from the photosensitizer-cobaloxime dyad C1 placed under dark (black trace) and natural sunlight (blue trace). The red trace signified the signal from the 1% H2 calibration gas mixture sample. Please click here to view a larger version of this figure.
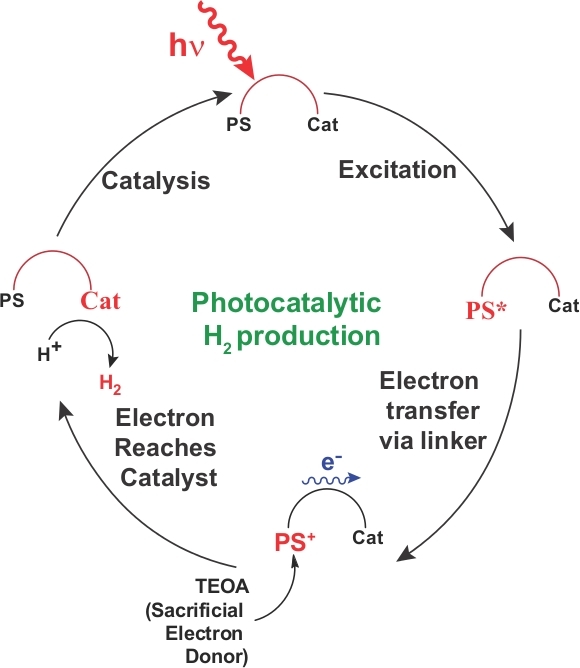
Figure 9: Photocatalytic scheme for H2 production by C1. Possible photo-catalytic H2 production cycle for the PS-catalyst hybrid complex C1. This mechanism presumably follows the sequence of excitation of the photosensitizer, transfer the excited electron to the catalyst via linker, and H2 production catalysis at the reduced catalytic centre. The cationic photosensitizer returns to the ground state by accepting electron from the sacrificial electron donor. Please click here to view a larger version of this figure.
Supplementary Materials. Please click here to download this file.
Discussion
The organic photosensitizer stilbene moiety was successfully incorporated into the cobaloxime core via the axial pyridine linkage (Figure 1). This strategy allowed us to devise a photosensitizer-cobaloxime hybrid complex C1. The presence of both the oxime and organic dye in the same molecular framework was evident from the single crystal structure of the C1 (Figure 4). The phenyl and pyridine functionalities of the stilbene motif existed in the same plane via an elongated conjugation through the allylic group. The interaction between these variable groups of the organic dye continued even in the solution phase as corroborated by the 1H NMR data (Figure 2). The stilbene molecule contained a dimethyl amine group that can exhibit a strong electron push via the conjugated aromatic-allylic network to the pyridine N-terminal32. This electronic interaction was expected to improve the σ-donation property of the N-pyridine towards the cobalt center in the axially coordinated Cobaloxime complex C1. The distinct alteration in the LMCT band of cobaloxime core along with red-shift of the π‒π* transition of the stilbene motif in C1 indicated that the electronic interaction between the metal and photosensitizer modules (Figure 3).
The electrochemical data highlighted active H2 production by this photosensitizer-cobaloxime hybrid C1 in the presence of water (Figure 5). This data suggested that (a) the cobaloxime core in C1 retained its intrinsic H2 production activity even in the presence of organic dye in its periphery and (b) water can act as a proton source during the catalysis. These results led to the investigation of photocatalytic H2 production by C1. During this experiment, an aqueous/DMF solution of C1, containing a TEOA sacrificial electron donor, was exposed to natural sunlight under aerobic condition and the complete air-tight setup was connected with an online H2 detector (Figure 6). A continuous accumulation of H2 was noticed during this experiment without any lag period, highlighting the photo-driven H2 production by C1 (Figure 7). The production of H2 during the photocatalytic conditions was further corroborated by the complementary GC experiments (Figure 8). This solar-driven H2 production by C1 possibly follows the typical catalytic cycle observed for cobaloxime-based photocatalytic devices that is illustrated in Figure 921. Earlier studies by Eisenberg et al. also supported the proposed photocatalytic cycle35,36,37.
The experimental setup developed during this project can be utilized to screen a number of photocatalytic systems by varying the combinations of photosensitizers, catalysts, sacrificial electron donor, and the solution ingredients. There is a potential application of this system under broad reaction conditions as it is functional in the presence of natural sunlight. This simple setup can also be employed in couple with variable laser configurations for the in-depth analysis of the photocatalytic activity. Here, we have incorporated stilbene dye with the cobaloxime complex to generate moderate photocatalytic H2 production hybrid. Their reactivity can be modified further by installing enzyme-inspired basic functionalities on the complex skeleton to further enhance the proton exchange rate, a critical step for the catalytic cycle38,39,40. This first generation photosensitizer-catalyst adduct provides an efficient, inexpensive, and green solar H2 production pathway compared to the other existing H2 generation techniques41. Hence, both the photocatalysts design strategy and solar-driven H2 production detection technique will pave the way for the development of next generation photo-active assemblies to renovate the renewable energy circuit.
Declarações
The authors have nothing to disclose.
Acknowledgements
Financial support was provided by IIT Gandhinagar and Government of India. We would also like to thank the extramural funding provided by Science and Engineering Research Board (SERB) (File no. EMR/2015/002462).
Materials
| 1 mm diameter glassy carbon disc electrode | ALS Co., Limited, Japan | 2412 | 1 |
| Acetone | SD fine chemicals | 25214L10 | 27 mL |
| Ag/AgCl reference electrode | ALS Co., Limited, Japan | 12171 | 1 |
| Co(dmg)2Cl2 | Lab synthesised | NA | 100 mg |
| CoCl2.6H2O | Sigma Aldrich | C2644 | 118 mg |
| d6 dmso | Leonid Chemicals | D034EAS | 650 µL |
| Deionized water from water purification system | NA | NA | 500 mL |
| Dimethyl formamide | SRL Chemicals | 93186 | 5 mL |
| Dimethyl glyoxime | Sigma Aldrich | 40390 | 232 mg |
| Gas-tight syringe | SGE syringe Leur lock | 21964 | 1 |
| MES Buffer | Sigma | M8250 | 195 mg |
| Methanol | Finar | 67-56-1 | 15 mL |
| Platinum counter electrode | ALS Co., Limited, Japan | 2222 | 1 |
| Stilbene Dye | Lab synthesised | NA | 65 mg |
| TBAF(Tetra-n-butylammonium fluoride) | TCI Chemicals | T1338 | 20 mg |
| Triethanolamine | Finar | 102-71-6 | 1 mL |
| Triethylamine | Sigma Aldrich | T0886 | 38 µL |
| Trifluoroacetic acid | Finar | 76-05-1 | 10 µL |
| Whatman filter paper | GE Healthcare | 1001125 | 2 |
Referências
- Chu, S., Majumdar, A. Opportunities and challenges for a sustainable energy future. Nature. 488 (7411), 294-303 (2012).
- Lewis, N. S., Nocera, D. G. Powering the planet: Chemical challenges in solar energy utilization. Proceedings of the National Academy of Sciences of the United States of America. 103 (43), 15729-15735 (2006).
- Faunce, T. A., et al. Energy and environment policy case for a global project on artificial photosynthesis. Energy and Environmental Science. 6 (3), 695-698 (2013).
- Artero, V., Fontecave, M. Solar fuels generation and molecular systems: is it homogeneous or heterogeneous catalysis. Chemical Society Reviews. 42 (6), 2338-2356 (2013).
- Artero, V. Bioinspired catalytic materials for energy-relevant conversions. Nature Energy. 2, 17131 (2017).
- Ball, M., Wietschel, M. The future of hydrogen – opportunities and challenges. International Journal of Hydrogen Energy. 34 (2), 615-627 (2009).
- da Silva Veras, T., Mozer, T. S., da Costa Rubim Messeder dos Santos, D., da Silva César, A. Hydrogen: Trends, production and characterization of the main process worldwide. International Journal of Hydrogen Energy. 42 (4), 2018-2033 (2017).
- Artero, V., Fontecave, M. Some general principles for designing electrocatalysts with hydrogenase activity. Coordination Chemistry Reviews. 249 (15), 1518-1535 (2005).
- Razavet, M., Artero, V., Fontecave, M. Proton Electroreduction Catalyzed by Cobaloximes: Functional Models for Hydrogenases. Inorganic Chemistry. 44 (13), 4786-4795 (2005).
- Landrou, G., Panagiotopoulos, A. A., Ladomenou, K., Coutsolelos, A. G. Photochemical hydrogen evolution using Sn-porphyrin as photosensitizer and a series of Cobaloximes as catalysts. Journal of Porphyrins and Phthalocyanines. 20, 534-541 (2016).
- Panagiotopoulos, A., Ladomenou, K., Sun, D., Artero, V., Coutsolelos, A. G. Photochemical hydrogen production and cobaloximes: the influence of the cobalt axial N-ligand on the system stability. Dalton Transactions. 45 (15), 6732-6738 (2016).
- Wakerley, D., Reisner, E. Development and understanding of cobaloxime activity through electrochemical molecular catalyst screening. Physical Chemistry Chemical Physics. 16 (12), 5739-5746 (2014).
- Hawecker, J., Lehn, J. M., Ziessel, R. Efficient homogeneous photochemical hydrogen generation and water reduction mediated by cobaloxime or macrocyclic cobalt complexes. Nouveau Journal de Chimie. 7 (5), 271-277 (1983).
- Du, P., Knowles, K., Eisenberg, R. A Homogeneous System for the Photogeneration of Hydrogen from Water Based on a Platinum(II) Terpyridyl Acetylide Chromophore and a Molecular Cobalt Catalyst. Journal of the American Chemical Society. 130 (38), 12576-12577 (2008).
- Du, P., Schneider, J., Luo, G., Brennessel, W. W., Eisenberg, R. Visible Light-Driven Hydrogen Production from Aqueous Protons Catalyzed by Molecular Cobaloxime Catalysts. Inorganic Chemistry. 48 (11), 4952-4962 (2009).
- To, W. P., et al. Luminescent Organogold(III) Complexes with Long-Lived Triplet Excited States for Light-Induced Oxidative C-H Bond Functionalization and Hydrogen Production. Angewandte Chemie International Edition. 51 (11), 2654-2657 (2012).
- Zhang, P., et al. Phosphine Coordination to a Cobalt Diimine–Dioxime Catalyst Increases Stability during Light-Driven H2 Production. Inorganic Chemistry. 51 (4), 2115-2120 (2012).
- McCormick, T. M., et al. Reductive Side of Water Splitting in Artificial Photosynthesis: New Homogeneous Photosystems of Great Activity and Mechanistic Insight. Journal of the American Chemical Society. 132 (44), 15480-15483 (2010).
- Zhang, P., et al. Photocatalytic Hydrogen Production from Water by Noble-Metal-Free Molecular Catalyst Systems Containing Rose Bengal and the Cobaloximes of BFx-Bridged Oxime Ligands. The Journal of Physical Chemistry C. 114 (37), 15868-15874 (2010).
- Dalle, K. E., Warnan, J., Leung, J. J., Reuillard, B., Karmel, I. S., Reisner, E. Electro- and Solar-Driven Fuel Synthesis with First Row Transition Metal Complexes. Chemical Reviews. 119 (4), 2752 (2019).
- Fihri, A., Artero, V., Razavet, M., Baffert, C., Leibl, W., Fontecave, M. Cobaloxime-Based Photocatalytic Devices for Hydrogen Production. Angewandte Chemie International Edition. 47 (3), 564-567 (2008).
- Li, C., Wang, M., Pan, J., Zhang, P., Zhang, R., Sun, L. Photochemical hydrogen production catalyzed by polypyridyl ruthenium-cobaloxime heterobinuclear complexes with different bridges. Journal of Organometallic Chemistry. 694 (17), 2814-2819 (2009).
- Mulfort, K. L., Tiede, D. M. Supramolecular Cobaloxime Assemblies for H2 Photocatalysis: An Initial Solution State Structure-Function Analysis. The Journal of Physical Chemistry B. 114 (45), 14572-14581 (2010).
- Zhang, P., Wang, M., Li, C., Li, X., Dong, J., Sun, L. Photochemical H2 production with noble-metal-free molecular devices comprising a porphyrin photosensitizer and a cobaloxime catalyst. Chemical Communications. 46 (46), 8806-8808 (2009).
- McCormick, T. M., Han, Z., Weinberg, D. J., Brennessel, W. W., Holland, P. L., Eisenberg, R. Impact of Ligand Exchange in Hydrogen Production from Cobaloxime-Containing Photocatalytic Systems. Inorganic Chemistry. 50 (21), 10660-10666 (2011).
- Veldkamp, B., Han, W. S., Dyar, S., Eaton, S., Ratner, M., Wasielewski, M. Photoinitiated multi-step charge separation and ultrafast charge transfer induced dissociation in a pyridyl -linked photosensitizer-cobaloxime assembly. Energy & Environmental Science. 6 (6), 1917-1928 (2013).
- Roy, S., Bhunia, A., Schuth, N., Haumann, M., Ott, S. Light-driven hydrogen evolution catalyzed by a cobaloxime catalyst incorporated in a MIL-101(Cr) metal-organic framework. Sustainable Energy & Fuels. 2 (6), 1148-1152 (2018).
- Song, T., Yu, J., Cui, Y., Yang, Y., Qian, G. Encapsulation of dyes in metal-organic frameworks and their tunable nonlinear optical properties. Dalton Transactions. 45 (10), 4218-4223 (2016).
- Schrauzer, G. N., Parshall, G. W., Wonchoba, E. R. Bis(Dimethylglyoximato)Cobalt Complexes: (“Cobaloximes”). Inorganic Syntheses. , 61-70 (2007).
- Sheldrick, G. M. Program for Empirical Absorption Correction of Area Detector Data. Sadabs. , (1996).
- Gruene, T., Hahn, H. W., Luebben, A. V., Meilleur, F., Sheldrick, G. M. Refinement of macromolecular structures against neutron data with SHELXL2013. Journal of Applied Crystallography. 47, 462-466 (2014).
- Kumari, B., Paramasivam, M., Dutta, A., Kanvah, S. Emission and Color Tuning of Cyanostilbenes and White Light Emission. ACS Omega. 3 (12), 17376-17385 (2018).
- Schrauzer, G. N., Lee, L. P., Sibert, J. W. Alkylcobalamins and alkylcobaloximes. Electronic structure, spectra, and mechanism of photodealkylation. Journal of the American Chemical Society. 92 (10), 2997-3005 (1970).
- Groom, C. R., Bruno, I. J., Lightfoot, M. P., Ward, S. C. The Cambridge Structural Database. Acta Crystallographica Section B, Structural Science, Crystal Engineering and Materials. 72, 171-179 (2016).
- Das, A., Han, Z., Haghighi, M. G., Eisenberg, R. Photogeneration of hydrogen from water using CdSe nanocrystals demonstrating the importance of surface exchange. Proceedings of the National Academy of Sciences of the United States of America. 110 (42), 16716-16723 (2013).
- Das, A., Han, Z., Brennessel, W. W., Holland, P. L., Eisenberg, R. Nickel Complexes for Robust Light-Driven and Electrocatalytic Hydrogen Production from Water. ACS Catalysis. 5 (3), 1397-1406 (2015).
- Eckenhoff, W. T., Eisenberg, R. Molecular systems for light driven hydrogen production. Dalton Transactions. 41 (42), 13004-13021 (2012).
- Dutta, A., Appel, A. M., Shaw, W. J. Designing electrochemically reversible H 2 oxidation and production catalysts. Nature Reviews Chemistry. 2 (9), 244 (2018).
- Savéant, J. M. Proton Relays in Molecular Catalysis of Electrochemical Reactions: Origin and Limitations of the Boosting Effect. Angewandte Chemie International Edition. 58 (7), 2125-2128 (2019).
- Khandelwal, S., Zamader, A., Nagayach, V., Dolui, D., Mir, A. Q., Dutta, A. Inclusion of Peripheral Basic Groups Activates Dormant Cobalt-Based Molecular Complexes for Catalytic H2 Evolution in Water. ACS Catalysis. , 2334-2344 (2019).
- Staffell, I., et al. The role of hydrogen and fuel cells in the global energy system. Energy & Environmental Science. 12 (2), 463-491 (2019).

