Protocols of 3D Bioprinting of Gelatin Methacryloyl Hydrogel Based Bioinks
Summary
Presented here is a method for the 3D bioprinting of gelatin methacryloyl.
Abstract
Gelatin methacryloyl (GelMA) has become a popular biomaterial in the field of bioprinting. The derivation of this material is gelatin, which is hydrolyzed from mammal collagen. Thus, the arginine-glycine-aspartic acid (RGD) sequences and target motifs of matrix metalloproteinase (MMP) remain on the molecular chains, which help achieve cell attachment and degradation. Furthermore, formation properties of GelMA are versatile. The methacrylamide groups allow a material to become rapidly crosslinked under light irradiation in the presence of a photoinitiator. Therefore, it makes great sense to establish suitable methods for synthesizing three-dimensional (3D) structures with this promising material. However, its low viscosity restricts GelMA’s printability. Presented here are methods to carry out 3D bioprinting of GelMA hydrogels, namely the fabrication of GelMA microspheres, GelMA fibers, GelMA complex structures, and GelMA-based microfluidic chips. The resulting structures and biocompatibility of the materials as well as the printing methods are discussed. It is believed that this protocol may serve as a bridge between previously applied biomaterials and GelMA as well as contribute to the establishment of GelMA-based 3D architectures for biomedical applications.
Introduction
Hydrogels are thought to be a suitable material in the field of biofabrication1,2,3,4. Among them, gelatin methacryloyl (GelMA) has become one of the most versatile biomaterials, initially proposed in 2000 by Van Den Bulcke et al.5. GelMA is synthesized by the direct reaction of gelatin with methacrylic anhydride (MA). The gelatin, which is hydrolyzed by the mammal collagen, is composed of target motifs of matrix metalloproteinase (MMP). Thus, in vitro three-dimensional (3D) tissue models established by GelMA can ideally mimic the interactions between cells and extracellular matrix (ECM) in vivo. Furthermore, arginine-glycine-aspartic acid (RGD) sequences, which are absent in some other hydrogels such as alginates, remain on the molecular chains of GelMA. This makes it possible to realize the attachment of encapsulated cells inside the hydrogel networks6. Additionally, the formation capability of GelMA is promising. The methacrylamide groups on the GelMA molecular chains react with the photoinitiator under mild reaction conditions and form covalent bonds upon exposure to light irradiation. Therefore, the printed structures can be rapidly crosslinked to maintain the designed shapes in a simple way.
Based on these properties, a series of fields utilize GelMA to carry out various applications, such as tissue engineering, basic cytology analysis, drug screening, and biosensing. Accordingly, various fabrication strategies have been also demonstrated7,8,9,10,11,12,13,14. However, it is still challenging to carry out 3D bioprinting based on GelMA, which is due to its fundamental properties. GelMA is a temperature-sensitive material. During the printing process, the temperature of the printing atmosphere has to be strictly controlled in order to maintain the physical state of the bioink. Besides, the viscosity of GelMA is generally lower than other common hydrogels (i.e., alginate, chitosan, hyaluronic acid, etc.). However, other obstacles are faced when building 3D architectures with this material15.
This article summarizes several approaches for the 3D bioprinting of GelMA proposed by our lab and describes the printed samples (i.e., the synthesis of GelMA microspheres, GelMA fibers, GelMA complex structures, and GelMA-based microfluidic chips). Each method has specialized functions and can be adopted in different situations with different requirements. GelMA microspheres are generated by an electroassisted module, which forms extra external electric force to shrink the droplet size. In terms of GelMA fibers, they are extruded by a coaxial bioprinting nozzle with the help of viscous sodium alginate. In addition, the establishment of complex 3D structures is achieved with a digital light processing (DLP) bioprinter. Finally, a twice crosslinking strategy is proposed to build GelMA-based microfluidic chips, combining GelMA hydrogel and traditional microfluidic chips. It is believed that this protocol is a significant summary of the GelMA bioprinting strategies used in our lab and may inspire other researchers in relative fields.
Protocol
1. Cell culturing
- Prepare Dulbecco's modified Eagle medium (DMEM), supplemented with 10% fetal bovine serum (FBS) and 1% penicillin/streptomycin, used to culture human breast cancer cell (MDA-MB-231) lines and human umbilical vein endothelial cell (HUVEC) lines.
- Prepare DMEM with L-glutamine (DMEM/F-12), supplemented with 10% FBS and 1% penicillin/streptomycin, used to culture bone marrow mesenchymal stem cell (BMSC) lines.
- Set the culturing environment as 37 °C and 5% CO2. Culture MDA-MB-231, HUVEC, and BMSC, and passage the cells in a 1:2 ratio when 90% confluence is reached.
2. Fabrication of GelMA microspheres
- Print the fixture as Figure 1A with polylactic acid (PLA) on a fused deposition modeling (FDM) printer. Place two metal ring electrodes in the fixture.
- Connect the two metal ring electrodes with ground and positive poles, respectively. Place the metal plate connected with the high voltage below the ring electrode and place a Petri dish with silicon oil on the metal plate as a droplet receiver.
- Dissolve freeze-dried GelMA (5% w/v) and lithium phenyl-2,4,6-trimethylbenzoylphosphinate (LAP, 0.5% w/v) in Dulbecco’s phosphate-buffered saline (DPBS) as the bioink (10 mL). Filter the bioink through a 0.22 μm filter for sterility and heat it in a 37 °C water bath for 15 min.
- Detach MDA-MB-231 cells with 3 mL of 0.25% trypsin-0.02% EDTA solution for 3 min at 37 °C. Centrifuge cells in a 15 mL centrifugal tube at 100 x g for 5 min to obtain a cell pellet.
- Remove the supernatant. Mix the cell pellet with 1 mL of prepared bioink by slowly pipetting it to prevent the production of bubbles.
- Place 1 mL of bioink (MDA-MB-231) into a 3 mL sterile syringe. Feed the bioink by the force of compressed air (~0.5 kPa). Put the syringe on the fixture.
NOTE: The printing environment should be strictly controlled at a temperature of 30 °C and humidity of 50%. - Switch on the high voltage power and set the voltage as 0−4 kV. Simultaneously, turn on the 405 nm wavelength light to crosslink the GelMA droplets in 5 mL of silicon oil.
- Pour the most part of the silicon oil away by decanting the Petri dish. Transfer the remained silicon oil and the GelMA microspheres into a 15 mL centrifugal tube using a spoon.
- Add 5 mL of DPBS and shake the mixture uniformly. Centrifuge the tube at 100 x g for 5 min and remove the supernatant fluid.
- Repeat step 2.9 3x.
- Take out the GelMA microspheres with a spoon and culture them in DMEM in a Petri dish at 37 °C and 5% CO2 for 3 days.
- Discard the medium and wash the microspheres with DPBS. Fix with 2 mL of 4% paraformaldehyde (PFA) for 30 min at room temperature (RT).
- Discard the PFA and wash the microspheres with DPBS. Permeabilize with 2 mL of 0.5% nonionic surfactant (i.e., Triton X-100) for 5 min at RT.
- Discard the nonionic surfactant and wash the microspheres with DPBS. Stain them with 2 mL of tetramethylrhodamine (TRITC) phalloidin for 30 min in darkness at RT.
- Discard the TRITC and wash the microspheres with DPBS. Stain them with 2 mL of 4–,6-diamidino-2-phenylindole (DAPI) for 10 min in darkness at RT.
- Discard the DAPI and wash the microspheres with DPBS. Capture the morphology with a confocal fluorescence microscope.
3. Fabrication of GelMA fibers
- Prepare a coaxial nozzle as shown in Figure 2A. Fix an inner nozzle (25 G, OD = 510 μm, ID = 250 μm) and outer nozzle (18 G, OD = 1200 μm, ID = 900 μm) with soldering. Connect a glass tube (length = 50 mm, inside diameter = 1.2 mm) to the end of the coaxial nozzle.
- Dissolve sodium alginate (Na-Alg) powder that is sterilized under ultraviolet (UV) light for 30 min in deionized water at 2% (w/v).
- Prepare a sterile bioink solution following step 2.3. Heat the GelMA bioink and Na-Alg solution in a 37 °C water bath for 15 min.
- Detach BMSCs cells with 3 mL of 0.25% trypsin-0.02% EDTA solution for 3 min at 37 °C. Centrifuge cells in a 15 mL centrifugal tube at 100 x g for 5 min to obtain a cell pellet.
- Remove the supernatant fluid. Mix the cell pellet with 2 mL of prepared GelMA bioink by slowly pipetting it to prevent the production of bubbles.
- Place 2 mL of bioink (BMSCs) into a 10 mL syringe. Place 2 mL of Na-Alg solution into another syringe (10 mL). Feed them with two syringe pumps, respectively (here, bioink at 50 μm/min and Na-Alg solution at 350 μm/min).
NOTE: The printing environment should be strictly controlled at a temperature of 30 °C and humidity of 50%. - Turn on the 405 nm wavelength light to irradiate the transparent tube to crosslink the GelMA fibers. Use a Petri dish with DPBS to receive the fibers.
- Take out the GelMA fibers with a spoon from DPBS and culture them for 3 days in the prepared DMEM/F-12 in 37 °C and 5% CO2.
- Follow steps 2.12−2.16 to prepare the GelMA fibers for morphological observation with a confocal fluorescence microscope.
4. Fabrication of complex 3D GelMA structures
NOTE: Figure 3A shows the fabrication sketch of the complex 3D GelMA structures.
- Wipe the DLP bioprinter (Figure 3E) with 75% alcohol and expose it to the UV irradiation for 30 min for sterility.
- Dissolve the freeze-dried GelMA (10% w/v) and LAP (0.5% w/v) in DPBS. Add magenta edible pigment into the solution (3% v/v) to improve the printing accuracy.
- Filter the solution through a 0.22 μm filter for sterility and heat it in a 37 °C water bath for 15 min.
- Build the 3D models with computer-aided design (CAD) software. Import the model documents to the upper software (EFL) of the applied DLP bioprinter.
- Add 10 mL of prepared bioink into the trough of the DLP bioprinter.
- Set the printing parameters in the upper software as follows: light intensity = 12 mW/cm2, irradiation duration = 30 s, and slice height = 100 μm. Start printing.
- Remove the printed structure from the bioprinter and immerse it in DPBS in a Petri dish.
- Detach the MDA-MB-231s cells with 3 mL of 0.25% trypsin-0.02% EDTA solution for 3 min at 37 °C. Centrifuge cells at 100 x g for 5 min in a 15 mL tube to obtain a cell pellet.
- Remove the supernatant fluid and mix the cell pellet with 2 mL of DMEM.
- Add 100 μL of cell suspension on the printed structures. Culture them for 3 days in the prepared DMEM at 37 °C and 5% CO2.
- Follow steps 2.12−2.16 to prepare the complex 3D structures for morphological observation with a confocal fluorescence microscope.
5. Fabrication of GelMA-based microfluidic chips
NOTE: Figure 4A shows the fabrication sketch of the GelMA-based microfluidic chip.
- Dissolve the freeze-dried GelMA 10% (w/v) and LAP (0.5% w/v) in DPBS. Filter the GelMA solution through a 0.22 μm filter for sterility.
- Sterilize the gelatin powder under UV light for 30 min and add it to the GelMA-LAP solution prepared in step 5.1 to a final concentration of gelatin of 5% (w/v). Heat the mixture in a 37 °C water bath for 15 min.
- Design a group of molds (Figure 4B,C) with CAD software and manufacture them with photopolymer resin on a DLP printer.
- Fill the molds fully with the prepared bioink.
- Put the molds into a 4 °C refrigerator to crosslink the gelatin for 30 min.
- Remove the molds and demold with a blade the partially (physically) crosslinked hydrogel sheets from the molds.
- Combine the two demolded hydrogel sheets and bond them with the help of GelMA by irradiating at 405 nm for 1 min.
- Detach the HUVECs cells with 3 mL of 0.25% trypsin-0.02% EDTA solution for 3 min at 37 °C. Centrifuge cells in a 15 mL centrifugal tube to obtain a cell pellet at 100 x g for 5 min.
- Remove the supernatant fluid and mix the cell pellet with 2 mL of DMEM.
- Fill the microchannel fully by injecting the cell suspension with a nozzle and syringe.
- Flip the chip upside down every 15 min during the next 3 h to achieve uniform and complete cell seeding. Culture the chips in the Petri dish for 3 days in the prepared DMEM at 37 °C and 5% CO2.
- Follow steps 2.12−2.16 to prepare the microfluidic chips for morphological observation with a confocal fluorescence microscope.
Representative Results
During the fabrication of GelMA microspheres, the GelMA droplets were separated by the external electric field force. When the droplets fell into the receiving silicon oil, they remained standard spheroid shape without tails. This is because the GelMA droplets were in an aqueous phase, while the silicon oil was in an oil phase. The surface tension that formed between the two phases caused the GelMA droplets to maintain a standard spheroid shape. In terms of the cell-laden microspheres, cells experienced the high voltage electric field force in this process. From the morphology of the stained MDA-MB-231s (Figure 1B–E), it was found that the encapsulated MDA-MB-231s maintained its spreading capability, verifying the biocompatibility of this electroassisted fabrication method.
In terms of the GelMA fibers, GelMA and sodium alginate solution flowed in the inner and outer nozzles of the coaxial nozzle, respectively. As the sodium alginate had higher viscosity than GelMA, GelMA was restricted in the sodium alginate solution and maintained a line shape. The irradiation by light (405 nm wavelength) caused the inner GelMA to become crosslinked, forming the GelMA fibers (Figure 2B). Besides, BMSCs were encapsulated in the GelMA fibers (Figure 2C,D). As shown, the encapsulated BMSCs maintained spreading capability in the GelMA hydrogel networks after the fabrication process (Figure 2E).
A DLP bioprinter was chosen to fabricate GelMA structures with more complex shapes. As shown in Figure 3B–D, the structures of “nose”, “ear”, and “multichamber” were established. On the surface of the crosslinked GelMA structures, the seeded HUVECs attached to the GelMA materials and spread (Figure 3F). This demonstrated the possibility that the establishment of GelMA complex 3D structures with the help of a DLP bioprinter holds great potential in applications in the field of tissue engineering.
Unlike the traditional microfluidic chip that is based on materials without biodegradation properties16,17,18,20 (i.e, resin, glass, polydimethylsiloxane [PDMS], and polymethyl methacrylate [PMMA]), a GelMA-based microfluidic chip was fabricated here using a twice cross-linking strategy. Two components in the bioink were crosslinked successively. Chips with various microchannels were built by designing different molds on demand (Figure 4B,C). Besides, it was verified that HUVECs were seeded in the channels and attached to the channel wall, forming the macroscopic vessel shape (Figure 4D,E).
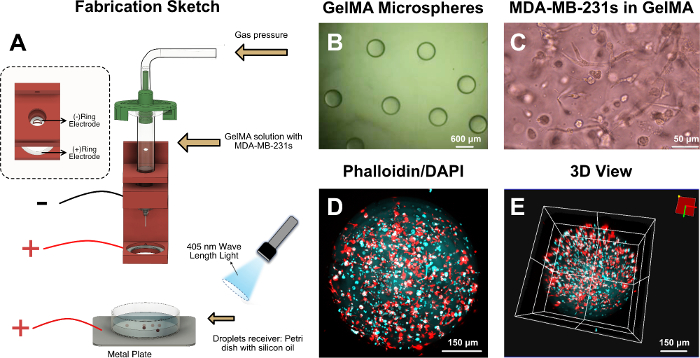
Figure 1: GelMA microspheres. (A) Fabrication sketch of the GelMA microspheres. (B) Optical microscope image of the GelMA microspheres. (C) Optical microscope image of the MDA-MB-231s in GelMA. (D) 2D view of the F-actin and nucleus of the encapsulated MDA-MB-231s. (E) 3D view of the F-actin and nucleus of the encapsulated MDA-MB-231s. Please click here to view a larger version of this figure.
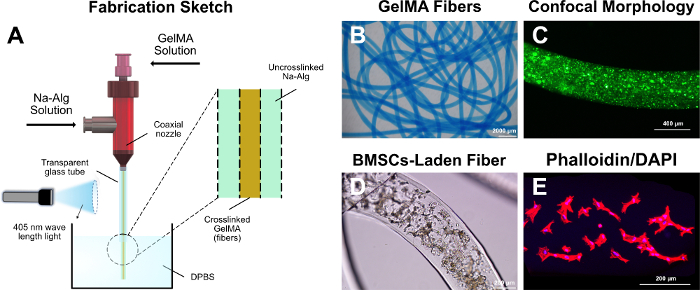
Figure 2: GelMA fibers. (A) Fabrication sketch of the GelMA fibers. (B) Optical microscope image of the GelMA fibers (with blue ink). (C) Confocal fluorescence microscope image of the GelMA fibers (with green fluorescence particles). (D) Optical microscope image of the BMSCs in GelMA fibers. (E) The F-actin and nucleus of the encapsulated BMSCs. Please click here to view a larger version of this figure.

Figure 3: GelMA complex 3D structures. (A) Fabrication sketch of the complex GelMA 3D structures. (B) Optical microscope image of the GelMA “nose”. (C) Optical microscope image of the GelMA “ear”. (D) Optical microscope image of the GelMA “multichamber”. (E) The applied DLP bioprinter. (F) The F-actin and nucleus of the seeded MDA-MB-231s. Please click here to view a larger version of this figure.
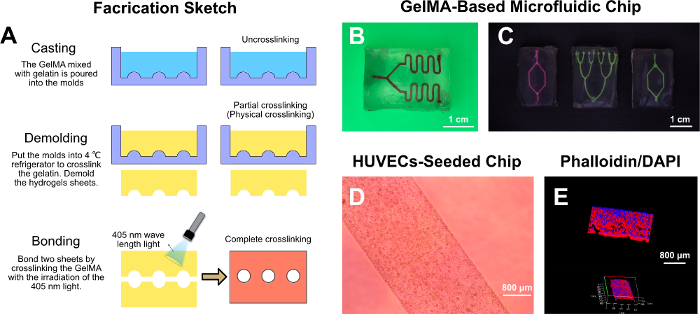
Figure 4: GelMA-based microfluidic chip. (A) Fabrication sketch of the GelMA-based microfluidic chip. (B,C) Optical microscope images of the GelMA-based microfluidic chip. (D) Optical microscope image of the seeded HUVECs on the channel wall. (E) The F-actin and nucleus of the seeded HUVECs on the channel wall. Please click here to view a larger version of this figure.
Discussion
This article describes several strategies to fabricate GelMA 3D structures, namely GelMA microspheres, GelMA fibers, GelMA complex structures, and GelMA-based microfluidic chips. GelMA has promising biocompatibility and formation capability and is widely used in the field of biofabrication. Microsphere structures are suitable for controlled drug release, tissue culturing, and injection into organisms for further therapy21,22,23,24,25. Because the viscosity of GelMA solution is low, its formation is challenging. Thus, during the fabrication of the GelMA microspheres, the electrohydrodynamic (EHD) principle was chosen to solve this problem. The voltage applied was relatively low, and the microdroplets were generated one-by-one. To fabricate microspheres of a smaller size, the applied voltage can be increased, and the fluid would be in another state with the Taylor cone26.
Because of the Coulomb explosion phenomenon, the dropping droplets were further separated by their excessive electric density, resulting in smaller GelMA microspheres. Furthermore, monocomponent GelMA fibers were fabricated with the help of a coaxial nozzle and sodium alginate solution. A coaxial nozzle was applied here. As mentioned above, because of the low viscosity of GelMA, sodium alginate provided resistance to help maintain the shape of fiber. Hydrogel fiber structures are suitable for mimicking the fiber-shaped tissues in vivo (i.e., muscles, vessels, etc.27,28,29,30,31,32). For GelMA fibers with more complicated components, the applied bioprinting nozzle can be further modified. For example, a three-layer coaxial nozzle can be assembled to generate multilayer GelMA fibers.
In the establishment of complex GelMA 3D structures, it was found that the DLP bioprinter breaks through the printing obstacle caused by the low viscosity of GelMA. With the help of CAD software, GelMA 3D structures were fabricated on demand. Finally, a new GelMA fabrication method, the twice cross-linking strategy, was demonstrated and applied to the combination of GelMA and a traditional microfluidic chip. The hydrogels have higher biocompatibility, and researchers can encapsulate cells inside the chip body. The proposed GelMA-based microfluidic chip can be further improved by encapsulating cells in the chips to serve as suitable models in vitro for drug screening, cellular interaction studies, etc. We believe that the methods for fabrication of GelMA described here will increase the rate of development in this field and can be applied in further biomedical research.
Declarações
The authors have nothing to disclose.
Acknowledgements
This work was sponsored by the National Key Research and Development Program of China (2018YFA0703000), the National Nature Science Foundation of China (No.U1609207, 81827804), the Science Fund for Creative Research Groups of the National Natural Science Foundation of China (No. 51821093).
Materials
| 0.22 μm filter membrane | Millipore | ||
| 2-(4-amidinophenyl)-6-indolecarbamidine dihydrochloride (DAPI) | Yeasen Biological Technology Co., Ltd., Shanghai, China | ||
| 3D bioprinter | SuZhou Intelligent Manufacturing Research Institute, SuZhou, China | ||
| 405nm wavelength light | SuZhou Intelligent Manufacturing Research Institute, SuZhou, China | ||
| co-axial nozzle | SuZhou Intelligent Manufacturing Research Institute, SuZhou, China | ||
| confocal fluorescence microscope | OLYMPUS FV3000 | ||
| digital light processing (DLP) bioprinter | SuZhou Intelligent Manufacturing Research Institute, SuZhou, China | ||
| DLP printer | SuZhou Intelligent Manufacturing Research Institute, SuZhou, China | ||
| Dulbecco's Phosphate Buffered Saline (DPBS) | Tangpu Biological Technology Co., Ltd., Hangzhou, China | ||
| Dulbecco's Modified Eagle Medium (DMEM) | Tangpu Biological Technology Co., Ltd., Hangzhou, China | ||
| Dulbecco's Modified Eagle Medium with L-glutamine (DMEM/F-12) | Tangpu Biological Technology Co., Ltd., Hangzhou, China | ||
| EFL Software | SuZhou Intelligent Manufacturing Research Institute, SuZhou, China | ||
| fetal bovine serum (FBS) | Tangpu Biological Technology Co., Ltd., Hangzhou, China | ||
| gelatin | Sigma-Aldrich, Shanghai, China | ||
| gelatin methacryloyl (GelMA) | SuZhou Intelligent Manufacturing Research Institute, SuZhou, China | ||
| high voltage power | SuZhou Intelligent Manufacturing Research Institute, SuZhou, China | ||
| lithium phenyl-2, 4, 6-trimethylbenzoylphosphinate (LAP) | SuZhou Intelligent Manufacturing Research Institute, SuZhou, China | ||
| paraformaldehyde | Tangpu Biological Technology Co., Ltd., Hangzhou, China | ||
| penicillin/streptomycin | Tangpu Biological Technology Co., Ltd., Hangzhou, China | ||
| sodium alginate (Na-Alg) | Sigma-Aldrich, Shanghai, China | ||
| TRITC phalloidin | Yeasen Biological Technology Co., Ltd., Shanghai, China | ||
| Triton X-100 | Solarbio Co., Ltd., Shanghai, China |
Referências
- Ahmed, E. M. Hydrogel: Preparation, characterization, and applications: A review. Journal of Advanced Research. 6 (2), 105-121 (2015).
- Ashton, R. S., Banerjee, A., Punyani, S., Schaffer, D. V., Kane, R. S. Scaffolds based on degradable alginate hydrogels and poly(lactide-co-glycolide) microspheres for stem cell culture. Biomaterials. 28 (36), 5518-5525 (2007).
- Billiet, T., Vandenhaute, M., Schelfhout, J., Van Vlierberghe, S., Dubruel, P. A review of trends and limitations in hydrogel-rapid prototyping for tissue engineering. Biomaterials. 33 (26), 6020-6041 (2012).
- Saroia, J., et al. A review on biocompatibility nature of hydrogels with 3D printing techniques, tissue engineering application and its future prospective. Bio-Design and Manufacturing. 1 (4), 265-279 (2018).
- Van Den Bulcke, A. I., et al. Structural and Rheological Properties of Methacrylamide Modified Gelatin Hydrogels. Biomacromolecules. 1 (1), 31-38 (2000).
- Sun, M., et al. Synthesis and Properties of Gelatin Methacryloyl (GelMA) Hydrogels and Their Recent Applications in Load-Bearing Tissue. Polymers. 10 (11), 1290 (2018).
- Gao, Q., et al. 3D printing of complex GelMA-based scaffolds with nanoclay. Biofabrication. 11 (3), 035006 (2019).
- Hassanzadeh, P., et al. Ultrastrong and flexible hybrid hydrogels based on solution self-assembly of chitin nanofibers in gelatin methacryloyl (GelMA). Journal of Materials Chemistry B. 4 (15), 2539-2543 (2016).
- McBeth, C., et al. 3D bioprinting of GelMA scaffolds triggers mineral deposition by primary human osteoblasts. Biofabrication. 9 (1), 015009 (2017).
- Nie, J., et al. Vessel-on-a-chip with Hydrogel-based Microfluidics. Small. 14 (45), 1802368 (2018).
- Shao, L., et al. Bioprinting of Cell-Laden Microfiber: Can It Become a Standard Product. Advanced Healthcare Materials. 8 (9), 1900014 (2019).
- Shao, L., et al. Fiber-Based Mini Tissue with Morphology-Controllable GelMA Microfibers. Small. 14 (44), 1802187 (2018).
- Xie, M., et al. Electro-Assisted Bioprinting of Low-Concentration GelMA Microdroplets. Small. 15 (4), 1804216 (2019).
- Yue, K., et al. Synthesis, properties, and biomedical applications of gelatin methacryloyl (GelMA) hydrogels. Biomaterials. 73, 254-271 (2015).
- Schuurman, W., et al. Gelatin-Methacrylamide Hydrogels as Potential Biomaterials for Fabrication of Tissue-Engineered Cartilage Constructs. Macromolecular Bioscience. 13 (5), 551-561 (2013).
- Barbot, A., Decanini, D., Hwang, G. On-chip Microfluidic Multimodal Swimmer toward 3D Navigation. Scientific Reports. 6, 19041 (2016).
- Esmaeilsabzali, H., et al. An integrated microfluidic chip for immunomagnetic detection and isolation of rare prostate cancer cells from blood. Biomedical Microdevices. 18 (1), 22 (2016).
- Lee, J. M., Zhang, M., Yeong, W. Y. Characterization and evaluation of 3D printed microfluidic chip for cell processing. Microfluidics and Nanofluidics. 20 (1), 5 (2016).
- Picot, J., et al. A biomimetic microfluidic chip to study the circulation and mechanical retention of red blood cells in the spleen. American Journal of Hematology. 90 (4), 339-345 (2015).
- Ren, K., Zhou, J., Wu, H. Materials for Microfluidic Chip Fabrication. Accounts of Chemical Research. 46 (11), 2396-2406 (2013).
- Chen, H., et al. Covalently antibacterial alginate-chitosan hydrogel dressing integrated gelatin microspheres containing tetracycline hydrochloride for wound healing. Materials Science and Engineering: C. 70, 287-295 (2017).
- Fan, M., et al. Covalent and injectable chitosan-chondroitin sulfate hydrogels embedded with chitosan microspheres for drug delivery and tissue engineering. Materials Science and Engineering: C. 71, 67-74 (2017).
- Feng, J., et al. Preparation of black-pearl reduced graphene oxide-sodium alginate hydrogel microspheres for adsorbing organic pollutants. Journal of Colloid and Interface Science. 508, 387-395 (2017).
- Park, K. S., Kim, C., Nam, J. O., Kang, S. M., Lee, C. S. Synthesis and characterization of thermosensitive gelatin hydrogel microspheres in a microfluidic system. Macromolecular Research. 24 (6), 529-536 (2016).
- Zheng, Y., et al. Injectable Hydrogel-Microsphere Construct with Sequential Degradation for Locally Synergistic Chemotherapy. ACS Applied Materials, Interfaces. 9 (4), 3487-3496 (2017).
- Fernández de la Mora, J. The Fluid Dynamics of Taylor Cones. Annual Review of Fluid Mechanics. 39 (1), 217-243 (2006).
- Hsiao, A. Y., et al. Smooth muscle-like tissue constructs with circumferentially oriented cells formed by the cell fiber technology. PLoS ONE. 10, 0119010 (2015).
- Meng, Z. J., et al. Microfluidic generation of hollow Ca-alginate microfibers. Lab on a Chip. 16 (14), 2673-2681 (2016).
- Peng, L., Liu, Y., Gong, J., Zhang, K., Ma, J. Continuous fabrication of multi-stimuli responsive graphene oxide composite hydrogel fibres by microfluidics. RSC Advances. 7 (31), 19243-19249 (2017).
- Sugimoto, M., et al. Micropassage-embedding composite hydrogel fibers enable quantitative evaluation of cancer cell invasion under 3D coculture conditions. Lab on a Chip. 18 (9), 1378-1387 (2018).
- Yamada, M., Sugaya, S., Naganuma, Y., Seki, M. Microfluidic synthesis of chemically and physically anisotropic hydrogel microfibers for guided cell growth and networking. Soft Matter. 8 (11), 3122-3130 (2012).
- Gao, G., et al. Tissue engineered bio-blood-vessels constructed using a tissue-specific bioink and 3D coaxial cell printing technique: a novel therapy for ischemic disease. Advanced Functional Materials. 27 (33), 1700798 (2017).

