Taking the Next Step: a Neural Coaptation Orthotopic Hind Limb Transplant Model to Maximize Functional Recovery in Rat
Summary
This protocol presents a robust, reproducible model of vascularized composite allotransplant (VCA) geared toward simultaneous study of immunology and functional recovery. The time invested in meticulous technique in a right mid-thigh hind limb orthotopic transplant with hand sewn vascular anastomoses and neural coaptation yields the ability to study functional recovery.
Abstract
Limb transplant in particular and vascularized composite allotransplant (VCA) in general have wide therapeutic promise that have been stymied by current limitations in immunosuppression and functional neuromotor recovery. Many animal models have been developed for studying unique features of VCA, but here we present a robust reproducible model of orthotopic hind limb transplant in rats designed to simultaneously investigate both aspects of current VCA limitation: immunosuppression strategies and functional neuromotor recovery. At the core of the model rests a commitment to meticulous, time-tested microsurgical techniques such as hand sewn vascular anastomoses and hand sewn neural coaptation of the femoral nerve and the sciatic nerve. This approach yields durable limb reconstructions that allow for longer lived animals capable of rehabilitation, resumption of daily activities, and functional testing. With short-term treatment of conventional immunosuppressive agents, allotransplanted animals survived up to 70 days post-transplant, and isotransplanted animals provide long lived controls beyond 200 days post-operatively. Evidence of neurologic functional recovery is present by 30 days post operatively. This model not only provides a useful platform for interrogating immunological questions unique to VCA and nerve regeneration, but also allows for in vivo testing of new therapeutic strategies specifically tailored for VCA.
Introduction
Limb transplant under the broader category of vascularized-composite allotransplant (VCA) or composite tissue allotransplant (CTA) has yet to fulfill its therapeutic promise. Since the first successful human hand transplants in Lyon, France and Louisville, Kentucky in 1998 and 1999, over 100 upper extremity transplants have been performed worldwide in carefully selected patients1. Wider applicability has been stymied by substantial immunosuppression and limited functional neuromotor recovery. Current immunosuppression strategies result in 85% incidence of acute rejection in the face of 77% incidence of opportunistic infection2. On the other hand, functional recovery after hand transplant occurs; mean Disability of Arm Shoulder and Hand (DASH) scores improve from 71 to 43, but that level of function may still qualify as a disability2. Given the nonlife saving nature of limb transplant, current techniques must be refined in animal models to take the next step in VCA.
Since the first rat model of limb transplant in 19783, many innovative animal models have been developed to advance the field of VCA4, incorporating vascular cuffed anastomoses to minimize operative time5,6, heterotopic osteomyocutaneous transplants to minimize physiologic insult to the recipient animal7,8,9,10,11, and novel immunologic approaches7,12,13,14. The rat model of orthotopic right hind limb mid-thigh transplant presented here emphasizes meticulous, time-tested microsurgical techniques such as hand sewn vascular anastomoses and neural coaptation as an upfront investment in a robust, reproducible model platform to simultaneously investigate both aspects of current VCA limitation: immunosuppression strategies and functional neuromotor recovery.
Protocol
All experiments were conducted in accordance with the Guide for the Care and Use of Laboratory Animals of the National Institutes of Health (NIH) and were approved by the Northwestern University Animal Care and Use Committee. The specific procedures were performed under protocol IS00001663.
NOTE: Two strains of rats were used, Lewis rats and August Copenhagen x Irish (ACI) rats. Animals were divided into three treatment groups: allotransplant without immune suppression (ACI to Lewis), allotransplant with conventional immune suppression (ACI to Lewis), and isotransplant (Lewis to Lewis or ACI to ACI). Lewis is an inbred strain, while ACI rats represent an out-bred wild-type, therefore this combination was chosen to model the worse-case rejection response. Conventional immunosuppression was administered subcutaneously either as rapamycin 1 mg/kg from post-operative day (POD) minus 1 to POD 28 or as FK506 3 mg/kg from POD 0 to POD 14, and then once weekly thereafter. Both male and female rats were eligible recipients from 8 to 16 weeks old, weighing between 250 and 400 grams at the time of surgery.
1. Donor right hind limb harvest
- Induce general anesthesia with 5% isoflurane in pure oxygen through a vaporizer with an appropriate scavenging system.
- Confirm adequate depth of anesthesia with toe pinch, and then use hair clippers to trim the fur off of the right hind limb and right groin surgical site
- Down-titrate the isoflurane through a rodent nose cone to 2-2.5%.
- Position the rat supine with spread limbs taped out to the sides on an operating board with a heating pad underneath. Disinfect the hairless skin with 70% rubbing alcohol and protect the surgical field with sterile gauze.
- Using an appropriate microsurgical microscope, microsurgical instruments, and with easy access to bipolar and monopolar electrocautery, begin the dissection.
- Use scissors to make a circumferential skin/subcutaneous tissue incision around the right hindlimb. Start in the inguinal crease medially at roughly the same level as the inguinal ligament and extend dorsal-laterally to complete the circumferential incision.
- Having exposed the muscular layer directly beneath the incision, dissect and cauterize the superficial epigastric vessels that lead from the muscular layer to the proximal skin/subcutaneous flap just created.
- Reflect the proximal flap superomedially to the inguinal ligament and the distal skin/subcutaneous flap inferolaterally to the knee.
- Use a wire retractor or rolled gauze to help expose the field.
- Observe that the inguinal anatomy of the rat is similar to humans; from lateral to medial lie the nerve, artery, and vein.
- Dissect out the femoral nerve, divide it sharply at the inguinal ligament, proximal to the bifurcation if possible. Retract the divided nerve inferiorly, keeping it safely out of the way, covered beneath moist gauze.
- Turning attention to the femoral artery and vein, use 4 cm 7-0 silk ties to atraumatically retract the vessels instead of handling them directly.
- Ligate all branches of the femoral vessels as they arise with 7-0 silk ties; divide the branches between the ties. For very small branches, bipolar cautery may be used instead of ties.
NOTE: Arterial and venous branches which require division include the superficial circumflex iliac and the muscular vessels. The superficial circumflex iliac is usually largest and appears to dive deep as would the profunda femoral in humans, but the profunda is absent in the rat15. More distal branches of the femoral vessels such as the highest genicular and the saphenous branch do not usually require division. - Systemically inject 500 international units of heparin through the penile vein in a male rat donor. Use the superficial epigastric vein if the donor rat is female.
- Allow the heparin to circulate systemically for 2 min before proceeding with the next steps.
- Ligate the femoral artery with 7-0 silk ties as proximal to the inguinal ligament as possible and divide between the ties.
- Similar to the artery, ligate and divide the femoral vein.
- Reflect both artery and vein inferiorly, safely out of the way, covered beneath moist gauze together with the femoral nerve covered previously. Dissect the ventral muscle groups, taking care to cauterize any visible vessel that arises. Attention to hemostasis here will minimize recipient blood loss after reperfusion.
- Deep to the ventral muscle groups, identify and sharply divide the sciatic nerve proximal to its branches. Three sciatic branches are usually visible: tibial, peroneal and sural. All three should all be preserved in the donor limb. A fourth cutaneous branch is not typically seen in this dissection15,16.
- Finish dividing the remaining ventral and dorsal muscle groups at mid-thigh level with meticulous hemostasis. It may be necessary to retract the limb medially to complete dividing the muscles.
- Transect the femur bone at midshaft using a hand-held cordless rotary saw.
- Having removed the limb graft from the donor, cut the silk tied ends from the graft side femoral artery and vein stumps, thereby re-opening the vessels.
- Insert a 24-gauge angiocatheter into the graft artery stump and flush the graft with 250 international units of heparin diluted in 5 mL of ice-cold normal saline, watching it flow out clear through the opened vein.
- Slowly, gently flush the graft for around 3 min. Excess forceful flushing may damage the endothelium.
- Place the graft in a chilled saline dish nested in an ice bucket until transplantation.
- Euthanize the donor rat with bilateral thoracotomy.
- Clean all surgical instruments appropriately.
2. Recipient native right hind limb amputation
- Induce anesthesia with isoflurane at 5%, confirm depth, trim the fur, position the animal, and disinfect the skin with alcohol as described for the donor rat.
- Down-titrate isoflurane to 2-2.5% and inject subcutaneous preoperative analgesia with buprenorphine 1.2 mg/kg, and preoperative prophylaxis with enrofloxacin 7.5 mg/kg.
- Same as for the donor, make a circumferential incision in the inguinal crease, reflect skin flaps assuring hemostasis, and dissect out the femoral nerve, artery and vein, ligating the same branch vessels as above.
- Divide the femoral nerve more distally than for the donor, but proximally to the bifurcation if possible.
- Dissect out the femoral artery and vein with enough space to clamp each separately at the level of the inguinal ligament. Clamp the vein and artery with microsurgical bulldog clamps. Once clamped, divide each vessel sharply with scissors.
- Divide the ventral and dorsal muscles of the thigh at mid-thigh level with meticulous hemostasis, retracting the limb medially as necessary.
- Identify and divide the sciatic nerves proximal to their branch points as above.
- Transect the femur at midshaft using the saw.
- Remove the recipient native right hind limb and dispose appropriately.
- Down-titrate the isoflurane to 1-1.5% through the nose cone.
3. Donor to recipient limb implantation
- Using the hand-held power saw, shave off any irregularities from both donor and recipient femur cut ends.
- Using the saw, cut off the hub end of an 18-gauge needle, which will become the femur intramedullary rod.
- Before manipulating the bone, apply a small amount of bone wax to the recipient cut end of femur bone to reduce marrow bleeding during the reaming process.
- Coapt the donor and recipient femoral bones using the 18-gauge needle as an intramedullary rod. Some force is necessary, but do not ream either bone so far as to fracture the cortex.
- As needed, remove the needle and trim it to an appropriate length so that both bones fit smoothly over the needle with no needle showing in between the bone.
- Place a small support such as a pad of gauze or a small rock or modelling clay underneath the donor limb to keep it off tension.
- Reapproximate the ventral muscle groups with eight to ten simple interrupted 5-0 polyglactin sutures so that the graft does not rotate around the femur needle. This gives the limb stability for the anastomoses.
- Periodically irrigate the graft and surgical field with ice-cold saline for better visualization and to reduce warm ischemic reperfusion injury.
- Align the donor and recipient femoral arteries and anastomose them in end to end fashion using simple interrupted 10-0 nylon suture, avoiding both tension and looping. The artery requires an average of six sutures.
- Similar to the artery, anastomose the donor and recipient femoral veins in end to end fashion. The vein requires six to eight sutures.
NOTE: Generous cold saline irrigation, atraumatic vessel handling technique, and leaving long tails to serve as stay sutures for vessel retraction are important tools for effective microsurgical anastomoses. - Place a small amount of hemostatic cellulose powder around both anastomoses, and then remove the proximal microsurgical bulldog clamps on the vein and the artery.
- Inspect both anastomoses for good patency and flow. Use cotton swab sticks to gently prod the vein and assure good hemostasis of both anastomoses. Hold pressure over bleeding sites and place more hemostatic cellulose powder if needed. Another suture may be placed through a bleeding hole at the risk of “back-walling" the needle only as a last resort.
- When both anastomoses are confirmed satisfactory, trim any remaining long stay suture tails short to match the others.
- Reposition the rat to the left lateral decubitus position, use liberal electrocautery to attain meticulous hemostasis of any reperfusion muscle bleeding.
- Turn attention to the nerve anastomoses once muscle hemostasis is assured. Trim back any nerve cut ends that appear ragged.
- Reapproximate the dorsal muscle groups under sciatic nerve with simple interrupted 5-0 polyglactin sutures.
- Reapproximate the sciatic nerve. Eight to ten 10-0 nylon neural simple interrupted sutures will usually suffice.
- Reapproximate the reminding dorsal muscle groups and then close the dorsal skin with 4-0 polyglactin continuous suture.
- Reposition the rat back to supine position and reapproximate the femoral nerve. Two to three 10-0 nylon neural simple interrupted sutures will usually suffice.
- Close the ventral skin with 4-0 polyglactin continuous suture. Avoid excess suture tail, which can be irritating to the rat once awake.
4. Post-operative care
- Recover animals in their cages with a heating pad under the cage and ready access to food and water, monitoring for early complications daily for the first week.
- Provide post-operative analgesia with subcutaneous meloxicam 1 mg/kg daily injection through POD 2. Provide post-operative antibiotic prophylaxis dilute enrofloxacin spray. Provide disincentive for autotomy (self-mutilation) with Bitter Safe Mist sprayed twice daily to the graft through POD 7.
- Maintain transplanted rats in cages with other rats, to stimulate return to daily activities and rehabilitate the transplanted limb.
5. Post-operative sensation testing
- Apply the Hargreaves testing of thermal sensation protocol, also described elsewhere17,18.
- Place the rat in the testing container and allowed it to acclimatize for 20 minutes. The apparatus glass is confirmed clean, and the heat source confirmed to be working with the investigator’s finger.
- Before testing, confirm that the rat is awake and the tested paw is positioned over the infrared motion detector.
- Transmit thermal energy at intensity level 90. Time delay in the animal moving its paw away from the heat source is recorded. If no movement occurs within 20 seconds, the test is aborted to prevent injury.
- Obtain five trials per tested limb, excluding the highest and lowest value before calculating the mean withdrawal latency time for each animal.
6. Post-operative motor testing
- Using a gait analysis treadmill and integrated software analysis platform, select candidates for treadmill testing at four to six weeks post-surgery.
- Trim all rat toenails one or two days before testing.
- Acclimatize animals to the testing room for one hour before testing, and allow for one minute of pre-test petting to calm anxiety.
- Placing the rat inside the treadmill, run the treadmill at trials of increasing speed, from 10 cm/s, to 14 cm/s, to the goal 18 cm/s. If the rat is reticent and cannot be coaxed to walk, abort the testing that day to avoid negative conditioning. Allow high performers to walk up to 24 cm/s.
- Rinse the treadmill apparatus with 70% ethanol in between tested animals.
- Gait parameters are output from the analysis platform’s proprietary software.
Representative Results
Survival and recovery depend on meticulous surgical technique. Attention to the vascular anastomoses and the neural anastomoses, as well as the bone coaptation as described above is crucial maximizing the success of this model. Operative design and representative anastomotic results are shown in Figure 1.
Overall mortality was dependent on immunosuppression strategy, with the majority of isotransplanted animals attaining the study endpoint of 100-200 post-operative days as seen in Figure 2. Once out of the acute post-operative window, treated allotransplanted animals could experience survival up to 58 post-operative days. Isografted rats lived indefinitely over the course of study while allograft transplanted rats had variable mortalities from rapamycin and FK506. Out of the treatments FK506 promoted the longest viability (day 57), while rapamycin was second best (day 20) over the untreated control (day 10).
Sensory and motor recovery can be shown in Figure 3. Animals were shown to have recovered sensory nerve function of the transplanted paw using the Hargreaves apparatus by day 30. Animals displayed significant recovery by four weeks after surgery (Aii). Animals shown marked improvements in motor function of the transplanted limb using a gait analysis treadmill and integrated software analysis platform. Example gait parameter based on specific limbs are shown (Bii) and a Sciatic Function Index (SFI) are also presented (Biii).
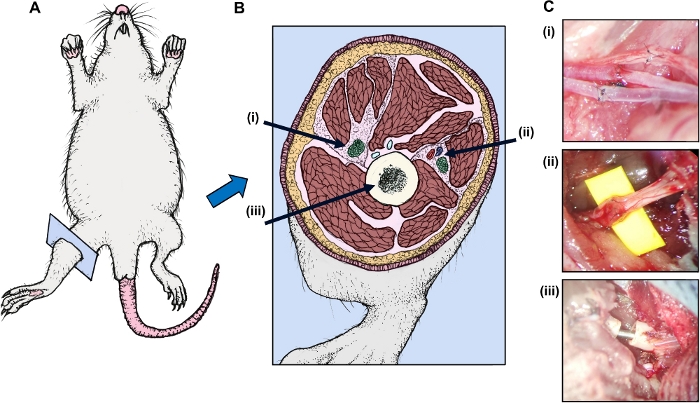
Figure 1: Operative design is depicted in cartoon format. (A) The rat is shown with (B) right hind leg cross-section depicting (i) the femoral bundle (nerve, artery, and vein) (ii) the sciatic nerve, and (iii) the bone. (C) Representative micrographs from the operating microscope (donor left and recipient right) were taken of the (i) sciatic nerve anastomosis (ii) the femoral nerve artery, and vein anastomoses (shown from top to bottom), and (iii) the 18-gauge needle intramedullary rod-femur bone coaptation. Note the donor structures appear to the left in each photo. Also note the femur is shown before full coaptation when both bones are opposed and the needle is concealed within. Please click here to view a larger version of this figure.
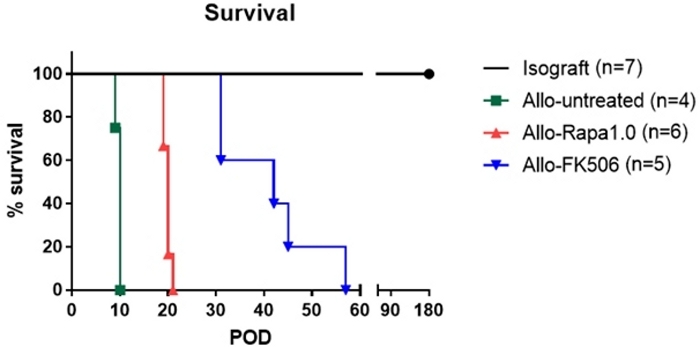
Figure 2: Percent survival of animals as presented days post-surgery (POD). Groups shown include isograft, allografts with no treatment, rapamycin, and FK506 immunosuppressant drugs. Please click here to view a larger version of this figure.
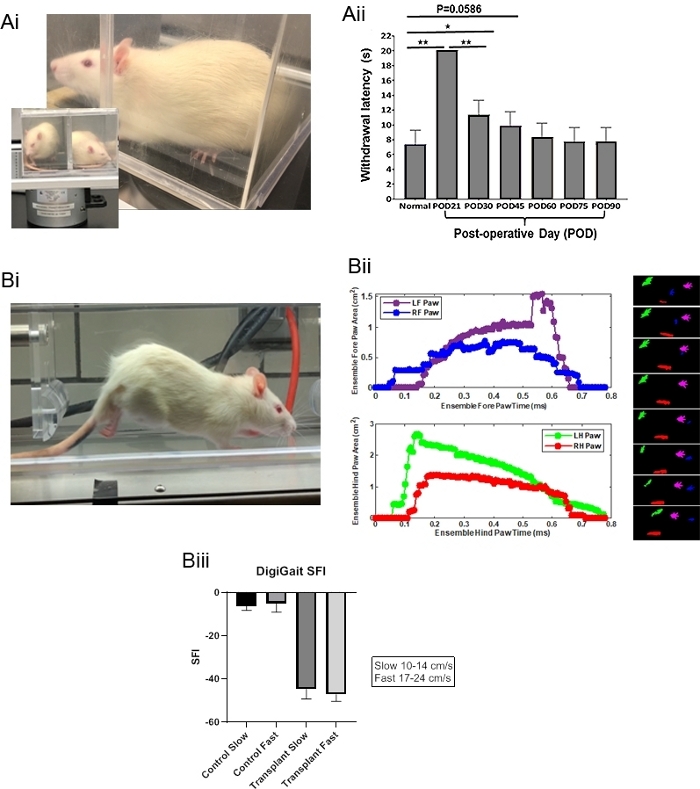
Figure 3: Sensory nerve recovery is demonstrated in (A) Hargreaves testing transplanted animals each at six post-operative time points and in (B) still shot of treadmill testing using DigiGait. (i) Representative pictures are shown with (ii) respective paw data. Respective color-coded images of paws are also in 0.025 ms frames. Digigate models (iii) are also shown. Significance was determined using a one-way ANOVA with a Bonferroni's multiple comparison test and SEM, where n=7 and p< 0.05. These particular DigiGait data were taken from an isogeneic animal tested at post-operative day 28. Please click here to view a larger version of this figure.
Discussion
Limb transplant, under the broader category of vascularized component allotransplantation (VCA), has widely applicable therapeutic promise as yet unfulfilled. The main roadblocks lie in unsolved immunological issues unique to VCA and neuromotor recovery techniques used currently. Development of new techniques will depend on animal modeling that is flexible, robust, and reproducible.
Many animal models have been established in VCA, each with specific advantages4. Non-human primate models offer attractive translatability to human patients, but have been hampered by cost concerns and toxic levels of immunosuppression required4. Canine models have been seen as advantageous for specific similarities of muscular structure as humans as well as a more experienced immune system19,20. Porcine models offer the benefits of a large animal model where the immune system is increasingly well studied21,22. Mouse model systems present the most advanced techniques to study immunology, but despite important advances in cuffed vascular microsurgical anastomosis23, mouse limb transplant remains technically challenging and has some limitations in functional recovery assessment5,24,25,26.
Rat models in VCA have been utilized since 19783, providing a mature platform to investigate both immunological and neuromotor hypotheses6,9,13,14,17,27,28,38. The model here combines the advantages of hindlimb orthotopic approach, suture anastomosis, nerve re-approximation, and potential for gait analysis. Hindlimb orthotopic as opposed to forelimb transplant is less of an encumbrance to the rat during the recovery process and allows for continued normal grooming and feeding behaviors post operatively. Suture anastomosis although painstaking may potentially offer less technical confounding for long term studies. Nerve re-approximation allows for future investigation17,18 and gait analysis. This protocol relies on meticulous, time-tested microsurgical techniques well-described elsewhere29, requiring constant attention to avoid the immediate pitfalls of anesthetic overdose, anastomotic failure, anastomotic thrombosis, and excessive surgical blood loss. Although multiple microsurgeons can improve the workflow, we have described a method by which a single operating microsurgeon can achieve sufficient experimental output.
Autotomy or self-mutilation has been a phenomenon noted in several microsurgical models, and it has been hypothesized to inversely correlate with nerve healing30,31. Autotomy was overall controlled in this model, possibly related meticulous neural anastomotic technique. Autotomy also decreased farther into the learning curve. Bitter Safe Mist was a valuable adjunct in controlling this phenomenon.
Gait analysis in rats has been studied for multiple models of injury32,33,34, most relevantly for sciatic nerve injury35,36. Rats even when not limb transplant recipients are known to be heterogenous subjects for gait analysis, and investigators still debate which analysis parameters describe recovery37. In this model we have described several methods to obtaining the best data from transplanted recipients who are willing and able to walk. Preselection of adequate walkers was not predictive of post-operative cooperation. Although animals are able to move about their housing as soon as several hours after surgery, they are not ready for treadmill ambulation until at least four to six weeks after surgery.
A protocol’s ability to measure nerve recovery in VCA is dependent on its strategy for rehabilitation. This protocol explicitly promotes transplant recipients interacting with other rats as inducement to function. This strategy is cognizant of the importance of modeling rehabilitation, yet is simple, economical, and is largely standard. Future strategies may include more active rehabilitation such as treadmill training.
The immunologic techniques applicable to this model are beyond the scope of this discussion, but in particular, comparing isotransplant versus allotransplanted animals provides a useful control to differentiate allograft immunologic phenomena and rejection from the ischemic reperfusion injury, inflammation, revascularization, and post-surgical infection processes inherent in the transplant surgery itself. Isotransplants provide a similar control for nerve function studies for the same reason.
Using this platform, investigators may be able to advance both VCA immunology and neuromotor recovery.
Declarações
The authors have nothing to disclose.
Acknowledgements
This work was funded by the Frankel Foundation and the Northwestern Memorial Hospital McCormick Grant (Operation RESTORE). Research reported in this publication was supported by the National Institute of General Medicial Sciences of the National Institutes of Health under Award Number T32GM008152. This work was supported by the Northwestern University Microsurgery Core and Behavioral Phenotyping Core.
Materials
| Anesthesia machine | Vet Equip | 911103 | |
| 0.5cc syringe | Exel | 26018 | |
| 18-gauge needle | BD | 305196 | |
| 1cc syringe | BD | 309659 | |
| 22-gauge needle | BD | 305156 | |
| 24-gauge angiocatheter | Sur-Vet | SROX2419V | |
| 25-gauge needle | Exel | 26403 | |
| 3 cc syringe | BD | 309657 | |
| 5cc syringe | Exel | 26230 | |
| Alcohol | Fisher Scientific | HC-600-1GAL | |
| Anesthesia induction chamber | Vet Equip | 941443 | |
| Anesthetic gas scavenger system | Vet Equip | 931401 | |
| Bipolar electrocautery | Aura | 26-500 | |
| Bitter Spray Mist | Henry Schein | 5553 | |
| Bone wax | CP Medical | CPB31A | |
| Breathing circuit | Vet Equip | 921413 | |
| Buprenophine | Reckitt Benckiser | 12496075705 | |
| Castro-Viejos needle drivers | Roboz | RS-6416 | |
| Cordless rotary saw | Dremel | 8050-N/18 | |
| Cotton swab stick | Fisher Scientific | 23-400-101 | For hemostasis |
| DigiGait Appparatus and Software | Mouse Specifics | MSI-DIG, DIG-SOFT | |
| Dumont forceps (#4) | Roboz | RS-4972 | |
| Dumont forceps (#5) | Roboz | RS-5035 | |
| Enrofloxacin | Norbrook | ANADA 200-495 | |
| FK-506 | Astellas | 301601 | |
| Gauze | Kendall | 1903 | |
| Gauze | Covidien | 8044 | |
| Gloves | Microflex | DGP-350-M | |
| Hair clippers | Oster | 078005-010-003 | |
| Handheld monopolar electrocautery | Bovie | AA00 | |
| Hargreaves Apparatus | Ugo Basile S.R.L. Gemonio, Italy | 37370 | |
| Heating pad | Walgreens | 126987 | |
| Heparin | Fresenius Kabi | 42592K | |
| Hot plate | Corning | PC-351 | For warming resusscitation fluid |
| Isoflurane | Henry Schein | 29405 | |
| Lactated ringers | Baxter | 2B2074 | |
| Large petri dish | Fisher Scientific | FB0875713 | For donor graft while in chilled saline |
| Meloxicam | Henry Schein | 49755 | |
| micro Collin Hartmann retractor | |||
| Micro dissecting scissors | Roboz | RS-5841 | |
| Microfibrillar collagen powder | BD | 1010590 | For hemostasis |
| Microvascular clips | Roboz | RS-5420 | |
| Normal saline | Baxter | 2F7124 | |
| Opthalmic lube | Dechra | IS4398 | |
| Rapmycin | MedChem Express | HY-10219 | |
| Small petri dish | Fisher Scientific | FB0875713A | For warmed resusscitation fluid |
| Sterile drapes | ProAdvantage | N207100 | |
| Surgical gown | Cardinal Health | 9511 | |
| Surgical mask | 3M | 1805 | |
| Surgical microscope, optic model OPMIMD | Zeiss | 169756 | |
| Surgical microscope, Universal S3 | Zeiss | 243188 | |
| Suture 10-0 nylon | Covidien | N2512 | |
| Suture 5-0 vicryl | Ethicon | J213H | |
| Suture 7-0 silk tie | Teleflex | 103-S | |
| Tape | 3M | 1530-1 | |
| Ultrasonic instrument cleaner | Roboz | RS-9911 | |
| Vessel dilation forceps | Roboz | RS-5047 |
Referências
- Elliott, R. M., Tintle, S. M., Levin, L. S. Upper extremity transplantation: current concepts and challenges in an emerging field. Current Reviews in Musculoskeletal Medicine. 7 (1), 83-88 (2014).
- Petruzzo, P., et al. The International Registry on Hand and Composite Tissue Transplantation. Transplantation. 90 (12), 1590-1594 (2010).
- Shapiro, R. I., Cerra, F. B. A model for reimplantation and transplantation of a complex organ: the rat hind limb. Journal of Surgical Research. 24 (6), 501-506 (1978).
- Brandacher, G., Grahammer, J., Sucher, R., Lee, W. P. Animal models for basic and translational research in reconstructive transplantation. Birth Defects Research Part C: Embryo Today. 96 (1), 39-50 (2012).
- Furtmuller, G. J., et al. Orthotopic Hind Limb Transplantation in the Mouse. Journal of Visualized Experiments. (108), e53483 (2016).
- Sucher, R., et al. Orthotopic hind-limb transplantation in rats. Journal of Visualized Experiments. (41), e2022 (2010).
- Horner, B. M., et al. In vivo observations of cell trafficking in allotransplanted vascularized skin flaps and conventional skin grafts. Journal of Plastic, Reconstructive & Aesthetic Surgery. 63 (4), 711-719 (2010).
- Fleissig, Y., et al. Modified Heterotopic Hindlimb Osteomyocutaneous Flap Model in the Rat for Translational Vascularized Composite Allotransplantation Research. Journal of Visualized Experiments. (146), e59458 (2019).
- Zhou, X., et al. A series of rat segmental forelimb ectopic implantation models. Scientific Reports. 7 (1), (2017).
- Adamson, L. A., et al. A modified model of hindlimb osteomyocutaneous flap for the study of tolerance to composite tissue allografts. Microsurgery. 27 (7), 630-636 (2007).
- Ulusal, A. E., Ulusal, B. G., Hung, L. M., Wei, F. C. Heterotopic hindlimb allotransplantation in rats: an alternative model for immunological research in composite-tissue allotransplantation. Microsurgery. 25 (5), 410-414 (2005).
- Fries, C. A., et al. enzyme responsive, tacrolimus-eluting hydrogel enables long-term survival of orthotopic porcine limb vascularized composite allografts: A proof of concept study. PLoS One. 14 (1), 0210914 (2019).
- Cottrell, B. L., et al. Neuroregeneration in composite tissue allografts: effect of low-dose FK506 and mycophenolate mofetil immunotherapy. Plastic and Reconstructive Surgery. 118 (3), 5 (2006).
- Benhaim, P., Anthony, J. P., Lin, L. Y., McCalmont, T. H., Mathes, S. J. A long-term study of allogeneic rat hindlimb transplants immunosuppressed with RS-61443. Transplantation. 56 (4), 911-917 (1993).
- Greene, E. C. . Anatomy of the rat. Volume N.S. American Philos. Soc. , 27 (1935).
- Schmalbruch, H. Fiber composition of the rat sciatic nerve. Anatomical Record. 215 (1), 71-81 (1986).
- Cheah, M., Fawcett, J. W., Andrews, M. R. Assessment of Thermal Pain Sensation in Rats and Mice Using the Hargreaves Test. Bio-protocol. 7 (16), (2017).
- Hargreaves, K., Dubner, R., Brown, F., Flores, C., Joris, J. A new and sensitive method for measuring thermal nociception in cutaneous hyperalgesia. Pain. 32 (1), 77-88 (1988).
- Hong, S. H., Eun, S. C. Experimental Forelimb Allotransplantation in Canine Model. BioMed Research International. 2016, 1495710 (2016).
- Mathes, D. W., et al. A preclinical canine model for composite tissue transplantation. Journal of Reconstructive Microsurgery. 26 (3), 201-207 (2010).
- Ibrahim, Z., et al. A modified heterotopic swine hind limb transplant model for translational vascularized composite allotransplantation (VCA) research. Journal of Visualized Experiments. (80), e50475 (2013).
- Fries, C. A., et al. A Porcine Orthotopic Forelimb Vascularized Composite Allotransplantation Model: Technical Considerations and Translational Implications. Plastic and Reconstructive Surgery. 138 (3), 71 (2016).
- Kim, M., Fisher, D. T., Powers, C. A., Repasky, E. A., Skitzki, J. J. Improved Cuff Technique and Intraoperative Detection of Vascular Complications for Hind Limb Transplantation in Mice. Transplantation Direct. 4 (2), 345 (2018).
- Chong, A. S., Alegre, M. L., Miller, M. L., Fairchild, R. L. Lessons and limits of mouse models. Cold Spring Harbor Perspectives in Medicine. 3 (12), 015495 (2013).
- Sucher, R., et al. Mouse hind limb transplantation: a new composite tissue allotransplantation model using nonsuture supermicrosurgery. Transplantation. 90 (12), 1374-1380 (2010).
- Tung, T. H., Mohanakumar, T., Mackinnon, S. E. Development of a mouse model for heterotopic limb and composite-tissue transplantation. Journal of Reconstructive Microsurgery. 17 (4), 267-273 (2001).
- Tang, J., et al. A vascularized elbow allotransplantation model in the rat. Journal of Shoulder and Elbow Surgery. 24 (5), 779-786 (2015).
- Yan, Y., et al. Nerve regeneration in rat limb allografts: evaluation of acute rejection rescue. Plastic and Reconstructive Surgery. 131 (4), 511 (2013).
- Georgiade, N. G., Serafin, D. . A Laboratory Manual of Microsurgery. Fourth Printing. , (1986).
- Tseng, S. H. Suppression of autotomy by N-methyl-D-aspartate receptor antagonist (MK-801) in the rat. Neuroscience Letters. 240 (1), 17-20 (1998).
- Haselbach, D., et al. Regeneration patterns influence hindlimb automutilation after sciatic nerve repair using stem cells in rats. Neuroscience Letters. 634, 153-159 (2016).
- Kloos, A. D., Fisher, L. C., Detloff, M. R., Hassenzahl, D. L., Basso, D. M. Stepwise motor and all-or-none sensory recovery is associated with nonlinear sparing after incremental spinal cord injury in rats. Experimental Neurology. 191 (2), 251-265 (2005).
- Berryman, E. R., Harris, R. L., Moalli, M., Bagi, C. M. Digigait quantitation of gait dynamics in rat rheumatoid arthritis model. Journal of Musculoskeletal and Neuronal Interactions. 9 (2), 89-98 (2009).
- Hamers, F. P., Lankhorst, A. J., van Laar, T. J., Veldhuis, W. B., Gispen, W. H. Automated quantitative gait analysis during overground locomotion in the rat: its application to spinal cord contusion and transection injuries. Journal of Neurotrauma. 18 (2), 187-201 (2001).
- Deumens, R., Marinangeli, C., Bozkurt, A., Brook, G. A. Assessing motor outcome and functional recovery following nerve injury. Methods in Molecular Biology. 11622, 179-188 (2014).
- Bozkurt, A., et al. Aspects of static and dynamic motor function in peripheral nerve regeneration: SSI and CatWalk gait analysis. Behavioural Brain Research. 219 (1), 55-62 (2011).
- Neckel, N. D. Methods to quantify the velocity dependence of common gait measurements from automated rodent gait analysis devices. Journal of Neuroscience Methods. 253, 244-253 (2015).
- Yeh, L. S., Gregory, C. R., Theriault, B. R., Hou, S. M., Lecouter, R. A. A functional model for whole limb transplantation in the rat. Plastic and Reconstructive Surgery. 105 (5), 1704-1711 (2000).

