Retinal Organoid Induction System for Derivation of 3D Retinal Tissues from Human Pluripotent Stem Cells
Summary
Here we describe an optimized retinal organoid induction system, which is suitable for various human pluripotent stem cell lines to generate retinal tissues with high reproducibility and efficiency.
Abstract
Retinal degenerative diseases are the main causes of irreversible blindness without effective treatment. Pluripotent stem cells that have the potential to differentiate into all types of retinal cells, even mini-retinal tissues, hold huge promises for patients with these diseases and many opportunities in disease modeling and drug screening. However, the induction process from hPSCs to retinal cells is complicated and time-consuming. Here, we describe an optimized retinal induction protocol to generate retinal tissues with high reproducibility and efficiency, suitable for various human pluripotent stem cells. This protocol is performed without the addition of retinoic acid, which benefits the enrichment of cone photoreceptors. The advantage of this protocol is the quantification of EB size and plating density to significantly enhance the efficiency and repeatability of retinal induction. With this method, all major retinal cells sequentially appear and recapitulate the main steps of retinal development. It will facilitate downstream applications, such as disease modeling and cell therapy.
Introduction
Retinal degenerative diseases (RDs), such as age-related macular degeneration (AMD) and retinitis pigmentosa (RP), are characterized by the dysfunction and death of photoreceptor cells and typically lead to irreversible vision loss without effective ways to cure1. The mechanism underlying these diseases is largely unknown partially due to lack of human disease models2. Over the past decades, significant advances have been accomplished in regenerative medicine through stem cell technology. Many researchers, including ourselves, have shown that human pluripotent stem cells (hPSCs), including human embryonic stem cells (hESCs) and human induced pluripotent stem cells (hiPSCs), can differentiate into all types of retinal cells, even mini-retinal tissues through various differentiation approaches3,4,5,6,7,8,9,10,11, providing huge potential in disease modeling and cell therapy12,13,14.
However, the induction process from hPSCs to retinal cells is highly complicated and time-consuming with low repeatability, which requires researchers with rich experience and high skills. During the complex and dynamic induction process, a number of factors will impact the yield of retinal tissues15,16,17. Also, different induction methods often vary considerably in timing and robust expression of retinal markers, which might confound the sample collection and data interpretation3. Therefore, a straightforward protocol of retinal differentiation from hPSCs with step-by-step guidance would be in demand.
Here, based on our published studies18,19,20,21, an optimized retinal induction protocol to generate retinal organoids (ROs) with rich cone photoreceptors from hPSCs is described, which does not require the supplement of retinoic acid (RA). This protocol focuses on the description of the multi-step method to generate neural retina and RPE. EB formation is the essential part of the early induction stage. Both size and plating density of EBs are quantitatively optimized, which scientifically enhances the yield of retinal tissues and promotes repeatability. In the second part of the induction, optic vesicles (OVs) self-organize in the adherence culture and ROs form in the suspension culture; the time courses and efficiencies of this part vary considerably in different hPSC lines. The maturation and specification of retinal cells in ROs mainly occur in the middle and late stage of induction. Without the addition of RA, mature photoreceptors with both rich cones and rods can be produced.
The purpose of this protocol is to quantitatively describe and detail each step for inexperienced researchers to repeat. Various hPSC lines have been successfully induced into ROs by this protocol with a robust yield of cone-rich retinal tissues and high repeatability. HPSCs-derived ROs with this protocol can recapitulate the main steps of retinal development in vivo, and survive long-term, which facilitates downstream applications, such as disease modeling, drug screening, and cell therapy.
Protocol
1. Culture and expansion of hPSCs
- HPSC culture
- Coat two wells of a 6-well plate with extracellular matrix (ECM, hESC-qualified matrix). Prepare 50 mL of an ECM solution containing 8-12 µg/mL of ECM in Dulbecco's Modified Eagle's Medium (DMEM). In 49 mL of DMEM, add 1 mL of the thawed ECM stock solution (50x). Add 1 mL of the ECM solution to each well of a 6-well plate. Incubate it for 1 h in an incubator at 37 °C and 5% CO2.
- Prepare hPSC maintenance medium (MM) according to the manufacturer's instruction.
- Pre-warm MM at room temperature (RT) for 30 min.
- Thaw a cryogenic vial of hPSCs (hiPSCs or hESCs) (about 1 x 106) from a liquid nitrogen tank by incubation in a water bath at 37 °C for 30 s.
- Take out the vial, and carefully disinfect it using a 75% disinfection alcohol spray. Put it in a biosafety cabinet.
- Transfer the cell suspension from the vial to a 15 mL tube, add 5 mL of pre-warmed MM drop by drop to the tube using a 5 mL pipette. Meanwhile, gently shake the tube to blend the hPSCs .
- Centrifuge the tube at 170 x g for 5 min. Remove most of the supernatant using a 1 mL pipette carefully and leave behind about 50 µL of supernatant to avoid losing the cells.
- Add 1 mL of MM to the tube, and resuspend the pellet by gently pipetting up and down once or twice with a 1 mL pipette.
NOTE: The survival of single cells of hPSCs is low. Small cell clumps with 3-5 cells are preferred to keep the hPSCs growing in colonies. - Remove ECM from the pre-coated wells (step 1.1.1), add 1.5 mL of MM to each well, and then distribute 0.5 mL of cell suspension per well.
- Gently shake the plate to distribute the hPSCs uniformly, and put the plate in an incubator at 37 °C and 5% CO2. Do not move the plate for at least 24 h to promote cell adherence.
- Change MM every other day and passage the hPSCs when the confluence has reached about 80%.
- Passaging of hPSCs
NOTE: The maintenance of the undifferentiated state in hPSCs is quite critical for further applications. Under the adherent conditions, hPSCs grow in colonies with a well-defined border. The cells should be passaged when the confluence of hPSCs reaches about 80%.- Observe the cells under a microscope. Mark and mechanically remove the clearly visible differentiated cells (<5%) before passaging.
- Prepare the ECM-coated plate as described in step 1.1.1.
- Pre-warm MM and 1x phosphate buffer saline (PBS) without Ca2+ and Mg2+ at RT.
- Pre-warm the 0.5 mM EDTA (in 1x PBS) solution in a water bath at 37 °C.
- Remove the medium from the culture plate using a vacuum-aspiration system, add 1 mL of 1x PBS in each well to wash the cells using a 1 mL pipette and repeat twice.
- Add 1 mL of EDTA solution per well to dissociate the hPSCs in a cell culture incubator at 37 °C and 5% CO2 for 5 min. Do not exceed the recommended incubation time in order to avoid dissociation to single cells.
- Take out the plate and check for the detachment of cells under a microscope. The confluent hPSCs loosen up and each cell border can be seen, but the cells cannot easily come off by gently shaking the cell plate.
- Remove the EDTA solution with a 1 mL pipette, and add 1 mL of MM to stop the dissociation. Gently pipette the hPSCs once or twice with a 1 mL pipette to resuspend the cells. There is no need to centrifuge to collect the cells.
NOTE: If most of the cells come off from the plate after incubation with EDTA, cells can be collected by centrifuge. - Remove ECM from the pre-coated wells (step 1.2.2), and add 1.5 mL of MM per well.
- Transfer 150-200 µL of cell clumps to each well. Generally, hPSCs can be passaged at a ratio of 1:6. For example, cells from one well of a 6-well plate can be distributed to six new wells.
- Gently shake the plate to distribute the hPSCs uniformly and culture the hPSCs in the incubator at 37 °C and 5% CO2 for at least 24 h without touching the plate.
- Change MM every other day as described in step 1.1.
2. Retinal differentiation from hPSCs
NOTE: When the colonies reach ~80% confluence (Figure 1B), they can be guided to differentiate into retinal organoids following the protocol schematized in Figure 1A. To ensure the hPSCs have high quality and good yield, regularly evaluate the pluripotency with molecular markers such as OCT4 or NANOG using IFC or QPCR. HPSCs should be discarded if differentiated cells account for more than 5% of the total cells. Check for mycoplasma contamination with a mycoplasma detection kit according to the manufacturer's instructions. Use only mycoplasma-free hPSCs as mycoplasma can alter the differentiation capability of hPSCs.
- Prepare media and reagents
- Prepare neural induction medium (NIM) by mixing the following: 500 mL of Dulbecco's Modified Eagle Medium/Nutrient Mixture F-12 (DMEM/F-12, 1:1), 5 mL of 1% N2 supplement, 0.5 mL of 0.1% heparin (2 mg/mL in 1x PBS), and 5 mL of 1% MEM Non-Essential Amino Acids (NEAA).
- Prepare retinal differentiation medium (RDM) containing 300 mL of DMEM/F-12, 200 mL of DMEM basic, 10 mL of 2% B27 supplement, 5 mL of 1% Antibiotic Antimycotic, and 5 mL of 1% MEM NEAA.
NOTE: Both NIM and RDM are not filtered but a sterility test is performed. Take out 1 mL of medium and add it into a 35 mm dish, and culture for 3-7 days in an incubator at 37 °C and 5% CO2. The media can be stored at 4 °C and should be used within 2 weeks to ensure the activity of the components. - Prepare 10 mM Blebbistatin (1,000x) in DMSO. Add 1,710 µL of DMSO to dissolve 5 mg of Blebbistatin to obtain 10 mM stock solution (1,000x), aliquot at 10 µL/tube, and store at -20 °C.
NOTE: All media and reagents should be warmed at RT for 30 min before use, unless otherwise mentioned.
- Embryoid body (EB) formation
- On day 0 (D0), initiate the differentiation. Take out one well of hPSCs from a 6-well plate, which has grown to ~80% confluence. Collect the cells with EDTA dissociation solution as described in steps 1.2.1 to 1.2.6.
- Remove the EDTA solution, add 1 mL of MM containing 10 µM Blebbistatin to stop cell dissociation, and collect the cells with a 1 mL pipette. The size of cell clumps is one of the key factors impacting the yield of EBs. Approximately, five cells per clump are preferred to produce the right size of EBs on D5 to D7.
NOTE: This is a key step. Do not pipette the cells too many times since EB-like aggregates are hard to form from single cells of hPSCs. - Transfer the cell suspension (about 2 x 106 cells) to a 100 mm ultra-low attachment Petri dish and add 9 mL of MM containing 10 µM Blebbistatin to the dish.
- Gently shake the dish twice to distribute the cells uniformly, and put the dish in the incubator at 37 °C and 5% CO2.
- On D1, after the cells are cultured for at least 24 h, take out the dish and observe it under the microscope. A large number of the small cell aggregates will be spontaneously formed by this time (Figure 1C).
- Prepare 12 mL of mixture with MM and NIM at a 3:1 ratio (9 mL of MM and 3 mL of NIM) in a 15 mL tube.
- Transfer the cell cultures to a 15 mL centrifuge tube with a 10 mL pipette perpendicularly, and add 10 mL of the pre-warmed mixture to the dish.
- Centrifuge the tube at 60 x g for 3 min to collect the aggregates, remove the supernatant using a 5 mL pipette and leave behind about 500 µL to avoid losing cells.
- Add 2 mL of the mixture to the tube, and transfer the suspension to the same dish (step 2.2.7).
- Gently shake the dish to uniformly distribute the cell aggregates, and put the dish back in the incubator.
- On D2, prepare 12 mL of a new mixture with MM and NIM at a 1:1 ratio (6 mL of MM and 6 mL of NIM) in a 15 mL tube. Change cell medium with the fresh prepared mixture by repeating the steps from 2.2.5 to 2.2.10.
- On D3, change cell medium with 15 mL of NIM as described above. Culture the cells for at least 5 days under the suspension conditions.
NOTE: During D1 to D3, the medium should be changed each day, providing enough nutrition. Since D3, NIM can be changed every other day. Also, EBs can be divided into several dishes to provide abundant nutrition.
- Seed the EBs
NOTE: On D5 to D7, choose an appropriate time point to plate the EBs on the ECM-coated dishes according to the size of EBs. EBs with an approximate diameter of 200 µm is appropriate for the retinal differentiation. In general, one well of hPSCs in a 6-well plate can produce about 300 to 1,000 EBs. The variation of EB yield is varied by the hPSC lines.- On D4, prepare ECM-coated dishes for EBs adherent culture. Add 5 mL of ECM to each 100 mm tissue culture dish (surface treated), and put them in the incubator overnight.
- On D5, remove ECM from the pre-coated dishes, and add 10 mL of pre-warmed NIM to each dish.
- Take out the dish containing EBs. Check the quality of EBs under the microscope and ensure that they are quite bright and round in shape. The size of the EBs is approximate 200 µm in diameter. Collect all EBs in a 15 mL tube. Transfer the EBs from the dishes to a 15 mL tube with a 5 mL pipette. Let the EBs settle down for 5 min. Remove most of the supernatant, leaving behind about 2 mL of medium.
- Distribute the EBs into the coated dishes containing 10 mL of NIM drop by drop with a 1 mL pipette. Seed the EBs at a density of approximately 2-3 EBs per cm2. For example, add about 120-180 EBs into a 100 mm dish. To roughly judge the EB number, place one drop of EB suspension onto a coverslip, and count the number of EBs under the microscope.
NOTE: The plating density of EBs is one of key factors impacting the efficiency of retinal induction. The density can be also adjusted by each hPSC line. - Gently shake the dishes to distribute the EBs uniformly. Put them in the incubator at 37 °C and 5% CO2.
NOTE: Do not move the dishes for at least 24 h to enhance the adherence of EBs.
- Induction of optic vesicles (OVs) and retinal pigment epithelium (RPE) in adherent conditions
NOTE: After EBs are seeded on the ECM coated surface, hPSCs can develop OV-like structures, which can be observed as early as D20 after differentiation. In this protocol, specific growth factors or signaling molecules are not required to guide the hPSCs into the retinal fate except the addition of N2 and B27 supplements in the media.- On D8-D9, remove the dishes and observe the EBs under the microscope. All EBs will be attached and spread out on the dishes (Figure 1D). Add 10 mL of fresh NIM to each 100 mm dish containing 10 mL of old medium. Put them back in the incubator.
NOTE: Do not remove the old medium. - On D12, change half of the medium with NIM using a 10 mL pipette. Keep the culture in the incubator.
- On D16, remove all NIM from the dishes using a vacuum-aspiration system. Add 20 mL of RDM to each dish. Keep culturing in RDM and change half of the medium every other day.
- During D10-D30, observe the morphological changes of the cells twice a week under a microscope and evaluate the efficiency of retinal differentiation.
NOTE: Since D10, eye field (EF) domains are self-organized in the peripheral zones of adherent EBs. The OV-like structures appear between D20 to D25, gradually protrude from the dish, and self-form an optic cup, which is surrounded by the pigmented RPE (Figure 1E). The OVs can be easily recognized with the bright, refractive, and thick NR ring.
- On D8-D9, remove the dishes and observe the EBs under the microscope. All EBs will be attached and spread out on the dishes (Figure 1D). Add 10 mL of fresh NIM to each 100 mm dish containing 10 mL of old medium. Put them back in the incubator.
- Detach and culture OVs and RPE in suspension to obtain retinal organoids (ROs)
- On D28-D35, most of OVs appear in the dishes. Use a Tungsten needle or a needle with 1 mL syringe to mechanically detach the morphologically identifiable OVs along with the adjacent RPE. Culture them in suspension.
NOTE: The appearance and yield of OVs and RPE vary widely in different hPSC lines. Thus, the time point of detaching OV and RPE is flexible. Obvious OVs with the adjacent RPE can be detached, and then moved to a low attachment culture dish containing RDM. Keep culturing the rest of the cells until all OVs and RPEs are lifted up. - Put 50-60 OVs into each 100 mm low attachment culture dish containing 15 mL of RDM for the ROs formation (Figure 1F).
- Change RDM every 2-3 days until D42, when the ROs are well round-shaped.
- On D28-D35, most of OVs appear in the dishes. Use a Tungsten needle or a needle with 1 mL syringe to mechanically detach the morphologically identifiable OVs along with the adjacent RPE. Culture them in suspension.
3. Retinal development and maturation
NOTE: In this protocol, serum is required to keep the ROs grow and mature for long-term culture.
- Retinal lamination and specification in ROs
- Prepare 10 mL of 100 mM taurine (1,000x) in 1x PBS. Weigh 125 mg of taurine, and dissolve in 10 mL of 1x PBS. Filter the solution with a 0.22 µm syringe filter. Aliquot at 500 µL/tube, and store at -20 °C.
- Prepare retinal culture medium 1 (RC1). Mix the following components: 250 mL of DMEM/F-12, 175 mL of DMEM basic, 50 mL of fetal bovine serum,10 mL of 2% B27 supplement, 5 mL of 1% Antibiotic Antimycotic, 5 mL of 1% MEM NEAA, 0.5 mL of 100 µM taurine, and 5 mL of 2 mM L-alanyl-L-glutamine.
- Prepare retinal culture medium 2 (RC2) containing 450 mL of DMEM/F-12, 50 mL of fetal bovine serum, 5 mL of 1% N2 supplement, 5 mL of 1% Antibiotic Antimycotic, 0.5 mL of 100 µM taurine, 5 mL of 1% MEM NEAA, and 5 mL of 2 mM L-alnyl-L-glutamine.
NOTE: The RC1 and RC2 are not filtered, but undergo a sterility test. Take out 1 mL of medium, add it to a 35 mm dish, and culture for 3-7 days in the incubator at 37 °C and 5% CO2, to ensure sterility before use. The medium can be stored at 4 °C and should be used within 2 weeks to ensure the activity of the components. All media and reagents should be pre-warmed at RT for 30 min before use. - On D42, switch the culture medium from RDM to RC1.
- Tilt the dishes at about 30˚ and let the ROs settle down for 30 s. Remove the old RDM with a 10 mL pipette leaving behind about 1 mL of medium to avoid losing ROs. Add 15 mL of fresh RC1 to each dish.
- Gently shake the dishes to distribute the ROs uniformly. Put the dishes back in the incubator. Change the whole medium twice a week thereafter.
- During D50-D90, select out high quality of ROs for long-term culture, which are round-shaped with a thick and bright NR. Place 30-40 ROs in a 100 mm low attachment dish with 20 mL of RC1,and change the whole medium twice a week.
- For the long-term suspension culture of ROs, pipette the ROs to avoid RO-RO reattaching using a pipette. Transfer ROs to new culture dishes once a month to avoid ROs sticking to the surface of the dishes.
NOTE: Under the suspension culture conditions, ROs are round-shaped, with a bright and thick NR ring attached with more or less RPE at one side. Laminated neural retina develop and retinal cell subtypes sequentially appear with retinal ganglion cells first generated, followed by photoreceptor cells, amacrine cells, and bipolar cells.
- Human photoreceptor maturation with enrichment of cones in ROs
- After D90, switch the medium from RC1 to RC2, which is suitable for photoreceptor maturation.
- Change the medium as described in steps 3.1.7-3.1.8.
NOTE: Under this culture condition, ROs can grow long-term (Figure 1G), up to D300 tested. Retinal cells in ROs become mature, and all cell subtypes of neural retina, including muller glial cells, rods and cones are also acquired. Without any addition of RA, cone photoreceptors are also rich in ROs.
Representative Results
The retinal induction process in this protocol mimics the development of human fetal retina. To initiate the retinal differentiation, hPSCs were dissociated into small clumps and cultured in suspension to induce the formation of EBs. On D1, the uniformed cell aggregates or EBs formed (Figure 1C). The culture medium was gradually transitioned into NIM. On D5, EBs were plated onto the ECM-coated culture dishes. Cells gradually migrated out of the EBs (Figure 1D). From D10, eye fields self-organized in the peripheral zone of adherent EBs. On D16, the induction medium was replaced by RDM. Afterwards, the NR domains gradually formed, protruded from the dish, and self-formed OV-like structures surrounded by the RPE cells (Figure 1E). During D28-D35, OVs along with the adjacent RPE were lifted up with a sharp needle and cultured in suspension. Under the suspension culture conditions, ROs self-formed comprising neural retina (NR) attached with more or less RPE sphere at one side (Figure 1F) and could survive and mature overtime as long as FBS were added to the medium.
As retinal differentiation and specification progressed, hPSCs produced all major retinal cell subtypes sequentially. The subtypes of neural retina gradually lined up in layers, mimicking the architecture features of native human retina (Figure 2A–G). Retinal ganglion cells (RGCs) were first generated from retinal progenitors and accumulated in the basal side of NRs. Photoreceptor cells located in the apical side, while amacrine cells, horizontal cells, bipolar cells, and muller glial cells all located in the intermediate layer of NRs.
With this protocol, ROs developed into the highly mature photoreceptors with both rods and cones (Figure 2G–I). Photoreceptors increased rapidly in the developing out nuclear layer (Figure 2G) after week 8, and gradually matured from week 17 onward. From week 21, all subtypes of photoreceptors including rods, red/green cones, and blue cones can be detected in ROs. Both rich rods and cones can be obtained in this induction protocol without any addition of RA throughout the whole differentiation process.
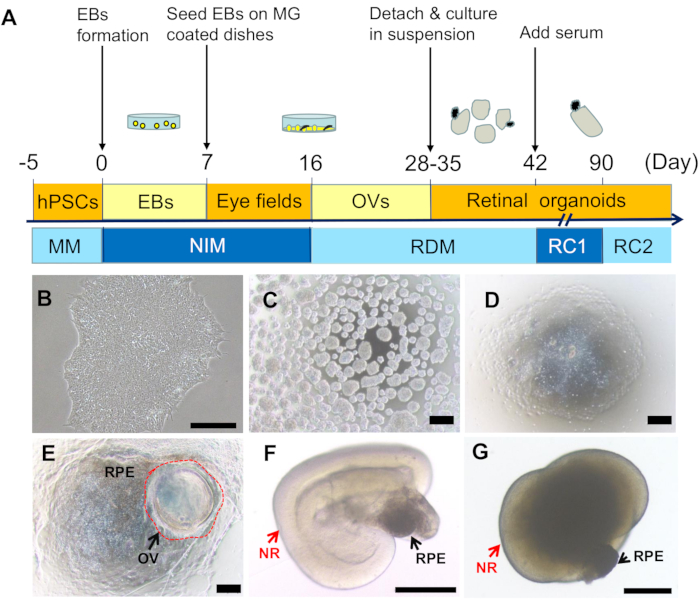
Figure 1: Induction and morphological features of retinal organoids from hPSCs. (A) Schematics of retinal induction from hPSCs. (B) A typical colony of hPSCs (10x). (C) EBs on D1 (4x). (D) On D7, plated EBs were attached and spread out on the dishes (4x). (E) On D25, the optic vesicle like structures (OVs) formed and protruded from the dish (indicated by the red circle), surrounded by pigmented RPE (4x). (F) Retinal organoids self-formed after OVs were lifted up and cultured in suspension conditions (the arrows pointed NR and RPE (4x). (G) A retinal organoid comprising NR (red arrow) and RPE (black arrow) on D180 (4x). Scale bars = 200 µm. Please click here to view a larger version of this figure.
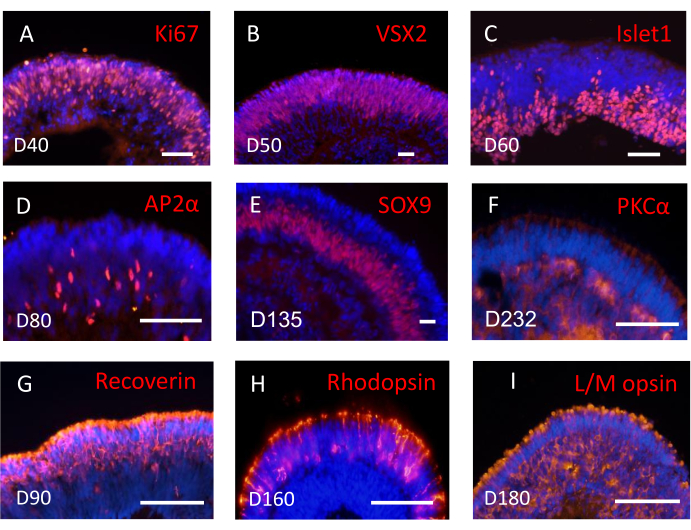
Figure 2: Subtypes of retinal cells were sequentially detected in three-dimensional retinal tissues. Example images of major retinal cell types expressing specific markers by immunofluorescent staining. (A–B) Retinal progenitor cells expressed Ki67 (A) and VSX2 (B). (C) Islet1 positive retinal ganglion cells located in the basal side of the neural retina. (D) Amacrine cells positive for AP2α. (E) Muller glial cells positive for SOX9. (F) PKCα positive bipolar cells. (G) Recoverin positive photoreceptor cells. (H) Rhodopsin positive rod photoreceptors. (I) L/M-opsin positive cone photoreceptors. Scale bar = 50 µm. Please click here to view a larger version of this figure.
Discussion
In this multi-step retinal induction protocol, hPSCs were guided step by step to gain the retinal fate, and self-organized into retinal organoids containing laminated NR and RPE. During the differentiation, hPSCs recapitulated all major steps of human retinal development in vivo, from EF, OV, and RPE, to retinal lamination, generating all subtypes of retinal cells, including retinal ganglion cells, amacrine cells, bipolar cells, rod, and cone photoreceptors, and muller glial cells in a spatial and temporal order. The recapitulation of retinal development would benefit downstream applications, such as retinal disease modeling.
A couple of protocols have been established to generate retinal organoids from hPSCs3,4,5,6,7,8,9,10,13,14,15,16,17,18,19,20. According to the culture conditions, the protocols can be classified into 2D, 3D, and the combination of 2D and 3D approaches9,13. The 2D approaches6,10,22 mean all the induction process occurs in the adherent culture conditions, generating retinal cells without architecture from hPSCs. In contrast, the 3D approaches7,11,23 mean all the induction process is under the suspension culture conditions, yielding organized retinal tissues. For example, Sasai, Y. et al.7,24 reported a SFEBq method (serum-free floating culture of embryoid-body-like aggregates with quick re-aggregation) to guide ESCs to differentiate into optic cups in suspension culture. Using the multi-step 3D approaches8,11,18,20,25 including this protocol, hPSCs have been induced toward retinal fates and organoids under both adherent and suspension culture conditions.
To induce hPSCs to neural retinal fate, a series of exogenous factors have been added to the media in many protocols. For example, Lamba, et al.26 added a combination of noggin (an inhibitor of the BMP pathway) and Dickkopf-1 (dkk1, an antagonist of the Wnt/β-catenin signaling pathway) and insulin-like growth factor-1 (IGF-1) to direct ESCs to an anterior neural fate. Osakada et al.6 added DAPT (a Notch signaling pathways inhibitor) and Left-Right Determination Factor A (a WNT signaling pathways inhibitor) to obtain rod and cone photoreceptor precursors. Kuwahara et al.27 and Capowski et al.3 added BMP4 for brief, early exposure of hPSCs culture to improve OV production. By contrast, this optimized retinal induction protocol is simple and low cost without requiring extrinsic signaling modulators except the basic supplement of N2 and B27.
Retinoic acid (RA) plays an important role in retinal development and photoreceptor determination28,29,30. Most of the protocols were developed with the supplement of RA (0.5-1 µM) in certain periods. Our studies have demonstrated that too high concentration of RA or too long period of RA treatment result in rod-rich photoreceptors but inhibit cone differentiation8,18. However, in this optimized protocol, RA is not added to the culture media throughout the whole differentiation process18, promoting the production of cone photoreceptors, which is responsible for human day-time vision and color vision and required for cell replacement of RD treatment. Although some studies reveal thyroid hormone signaling directs cone subtypes in mice and human retina31,32, the regulator for cone commitment is still unclear33. In these studies from Kim et al.34 and Lowe et al.35, the long-term culture also without any exogenous retinoic acid generated cone-rich retinal organoids, which is consistent with this optimized protocol.
The key points of this protocol to grasp are to make high quality EBs and to seed EBs appropriately. Cells grow fast during early EB suspension culture. The medium should be changed every day and be enough to provide abundant nutrition. The size of EBs, approximate 200 µm in diameter, is appropriate for the retinal differentiation. The plating density of EBs at 2-3 EBs per cm2 is suitable for most of hPSC lines. The best advantage of this optimized protocol is the quantification of EB size and plating density to significantly enhance the efficiency and repeatability of retinal induction. We have clearly described all the steps in detail, which largely helps the inexperienced researchers to learn and repeat the retinal induction.
In addition, retinal induction efficiency largely depends on the quality and differentiation potency of the hPSCs36,37. Different hPSCs have different efficiencies. Some hPSC lines indeed have poor efficiency, which might be due to the reprogramming methods, somatic cells, and so on. This protocol has been confirmed to be suitable for various hPSCs to obtain 3D retinal organoids and the RPE, including various hESCs and hiPSCs reprogrammed from fibroblasts, blood, and urine cells18,20,21. In general, with this protocol described above, one well of hPSCs (about 80% confluence) in a 6-well plate can generate about 1,000 EBs, yielding roughly 200 ROs. Therefore, this protocol with high efficiency is suitable for large-scale production of retinal organoids and benefits downstream applications including basic and translational study.
In summary, the optimized retinal induction protocol is simple and low cost with high repeatability and efficiency, offers promising personalized models of retinal diseases and provides abundant cell source for cell therapy, drug screening, and gene therapy test.
Declarações
The authors have nothing to disclose.
Acknowledgements
This study was supported by the National Key R&D Program of China (2016YFC1101103, 2017YFA0104101), the Guangzhou Science and Technology Project Fund (201803010078), the Science & Technology Project of Guangdong Province (2017B020230003), the Natural Science Foundation (NSF) of China (81570874, 81970842), Hundred talent program of Sun Yat-sen University (PT1001010), and the Fundamental Research Funds of the State Key Laboratory of Ophthalmology.
Materials
| (−)-Blebbistatin | Sigma | B0560-5mg | ROCK-inhibitor |
| 1 ml tips | Kirgen | KG1313 | 1 ml |
| 10 ml pipette | Sorfa | 3141001 | Pipette |
| 100 mm Tissue culture | BIOFIL | TCD000100 | 100 mm Petri dish |
| 100 mm Tissue culture | Falcon | 353003 | 100 mm Petri dish |
| 15 ml Centrifuge tubes | BIOFIL | CFT011150 | Centrifuge tubes |
| 35 mm Tissue culture dishes | Falcon | 353001 | 35 mm Petri dish |
| 5 ml pipette | Sorfa | 313000 | Pipette |
| 50 ml Centrifuge tubes | BIOFIL | CFT011500 | Centrifuge tubes |
| 6 wells tissue culture plates | Costar | 3516 | Culture plates |
| Anti-AP2α Antibody | DSHB | 3b5 | Primary antibody |
| ANTIBIOTIC ANTIMYCOTIC 100X | Gibco | 15240062 | Antibiotic-Antimycotic |
| Anti-ISL1 Antibody | Boster | BM4446 | Primary antibody |
| Anti-Ki67 Antibody | Abcam | ab15580 | Primary antibody |
| Anti-L/M opsin Antibody | gift from Dr. jeremy | / | Primary antibody |
| Anti-PAX6 Antibody | DSHB | pax6 | Primary antibody |
| Anti-rabbit 555 | Invitrogen | A31572 | Donkey anti-Rabbit IgG (H+L) Secondary Antibody, Alexa Fluor 555 |
| Anti-Recoverin Antibody | Millipore | ab5585 | Primary antibody |
| Anti-Rhodopsin Antibody | Abcam | ab5417 | Primary antibody |
| Anti-sheep 555 | Invitrogen | A21436 | Donkey anti-Sheep IgG (H+L) Secondary Antibody, Alexa Fluor 555 |
| Anti-SOX9 Antibody | Abclonal | A19710 | Primary antibody |
| Anti-VSX2 Antibody | Millipore | ab9016 | Primary antibody |
| B-27 supplement W/O VIT A (50X) | Gibco | 12587010 | Supplement |
| Cryotube vial | Thermo scientific-NUNC | 375418 | 1.8 ml |
| DAPI | DOJINDO | D532 | 4',6-Diamidino-2-phenylindole dihydrochloride; multiple suppliers |
| Dimethyl sulphoxide(DMSO) Hybri-max | Sigma | D2650-100ML | Multiple suppliers |
| DMEM | Gibco | C11995500BT | Medium |
| DMEM /F12 | Gibco | C11330500BT | Medium |
| EDTA | Invitrogen | 15575-020 | 0.5 M PH 8.0 |
| FBS | NATOCOR | SFBE | Serum |
| Filter | Millipore | SLGP033RB | 0.22μm, sterile Millex filter |
| GlutaMax, 100X | Gibco | 35050061 | L-alanyl-L-glutamine |
| Heparin | Sigma | H3149 | 2 mg/ml in PBS to use |
| Matrigel, 100x | Corning | 354277 | Extracellular matrix (ECM) |
| MEM Non-Essential Amino Acids Solution (100X) | Gibco | 11140050 | MEM NEAA |
| mTeSR1 | STEM CELL | 85850 | hPSCs maintenance medium (MM) |
| N2 supplement | Gibco | 17502048 | Supplement |
| Phosphate-buffered saline (PBS) buffer | GNM | GNM10010 | Without Ca+,Mg+,PH7.2±0.1 0.1M |
| Taurine | Sigma | T0625 | Supplement |
| Ultra-low attachment culture dishes 100mm petri dish, low-attachment | Corning | CLS3262-20EA | Petri dish |
Referências
- Flaxman, S. R., et al. Global causes of blindness and distance vision impairment 1990-2020: a systematic review and meta-analysis. The Lancet Global Health. 5 (12), 1221-1234 (2017).
- Sayed, N., Liu, C., Wu, J. C. Translation of human-induced pluripotent stem cells: From clinical trial in a dish to precision medicine. Journal of American College of Cardiology. 67 (18), 2161-2176 (2016).
- Capowski, E. E., et al. Reproducibility and staging of 3D human retinal organoids across multiple pluripotent stem cell lines. Development. 146 (1), (2019).
- Brooks, M. J., et al. Improved retinal organoid differentiation by modulating signaling pathways revealed by comparative transcriptome analyses with development in vivo. Stem Cell Reports. 13 (5), 891-905 (2019).
- Zerti, D., et al. Developing a simple method to enhance the generation of cone and rod photoreceptors in pluripotent stem cell-derived retinal organoids. Stem Cells. 38 (1), 45-51 (2020).
- Osakada, F., et al. Toward the generation of rod and cone photoreceptors from mouse, monkey and human embryonic stem cells. Nature Biotechnology. 26 (2), 215-224 (2008).
- Nakano, T., et al. Self-formation of optic cups and storable stratified neural retina from human ESCs. Cell Stem Cell. 10 (6), 771-785 (2012).
- Zhong, X., et al. Generation of three-dimensional retinal tissue with functional photoreceptors from human iPSCs. Nature Communication. 5, 4047 (2014).
- Liu, C., Oikonomopoulos, A., Sayed, N., Wu, J. C. Modeling human diseases with induced pluripotent stem cells: from 2D to 3D and beyond. Development. 145 (5), (2018).
- Lamba, D. A., Gust, J., Reh, T. A. Transplantation of human embryonic stem cell-derived photoreceptors restores some visual function in Crx-deficient mice. Cell Stem Cell. 4 (1), 73-79 (2009).
- Reichman, S., et al. From confluent human iPS cells to self-forming neural retina and retinal pigmented epithelium. Proceedings of the National Academy of Sciences. 111 (23), 8518-8523 (2014).
- Maeda, A., Mandai, M., Takahashi, M. Gene and Induced Pluripotent Stem Cell Therapy for Retinal Diseases. Annual Review Genomics and Human Genetics. 20, 201-216 (2019).
- Kruczek, K., Swaroop, A. Pluripotent stem cell-derived retinal organoids for disease modeling and development of therapies. Stem Cells. 38 (10), 1206-1215 (2020).
- O’Hara-Wright, M., Gonzalez-Cordero, A. Retinal organoids: a window into human retinal development. Development. 147 (24), (2020).
- Eckert, P., Knickmeyer, M. D., Schutz, L., Wittbrodt, J., Heermann, S. Morphogenesis and axis specification occur in parallel during optic cup and optic fissure formation, differentially modulated by BMP and Wnt. Open Biology. 9 (2), 180179 (2019).
- Patel, A., Sowden, J. C. Genes and pathways in optic fissure closure. Seminals in Cell and Development Biology. 91, 55-65 (2019).
- Chan, B. H. C., Moosajee, M., Rainger, J. Closing the Ggap: Mechanisms of epithelial fusion during optic fissure closure. Frontiers in Cell and Developmental Biology. 8, (2021).
- Li, G., et al. Generation of retinal organoids with mature rods and cones from urine-derived human induced pluripotent stem cells. Stem Cells International. 2018, 4968658 (2018).
- Liu, S., et al. Self-formation of RPE spheroids facilitates enrichment and expansion of hiPSC-derived RPE generated on retinal organoid induction platform. Investigative Ophthalmology and Visual Science. 59 (13), 5659-5669 (2018).
- Luo, Z., et al. An optimized system for effective derivation of three-dimensional retinal tissue via Wnt signaling regulation. Stem Cells. 36 (11), 1709-1722 (2018).
- Li, G., et al. Generation and characterization of induced pluripotent stem cells and retinal organoids from a leber’s congenital amaurosis patient with novel RPE65 mutations. Frontiers in Molecular Neuroscience. 12, 212 (2019).
- Matsa, E., Ahrens, J. H., Wu, J. C. Human induced pluripotent stem cells as a platform for personalized and precision cardiovascular medicine. Physiology Reviews. 96 (3), 1093-1126 (2016).
- Wahlin, K. J., et al. Photoreceptor outer segment-like structures in long-term 3d retinas from human pluripotent stem cells. Science Reports. 7 (1), 766 (2017).
- Eiraku, M., et al. Self-organizing optic-cup morphogenesis in three-dimensional culture. Nature. 472 (7341), 51-56 (2011).
- Reichman, S., et al. Generation of storable retinal organoids and retinal pigmented epithelium from adherent human iPS cells in xeno-free and feeder-free conditions. Stem Cells. 35 (5), 1176-1188 (2017).
- Lamba, D. A., Karl, M. O., Ware, C. B., Reh, T. A. Efficient generation of retinal progenitor cells from human embryonic stem cells. Proceedings of the National Academy of Science U. S. A. 103 (34), 12769-12774 (2006).
- Kuwahara, A., et al. Generation of a ciliary margin-like stem cell niche from self-organizing human retinal tissue. Nature Communication. 6, 6286 (2015).
- da Silva, S., Cepko, C. L. Fgf8 expression and degradation of retinoic acid are required for patterning a high-acuity area in the retina. Developmental Cell. 42 (1), 68-81 (2017).
- Mitchell, D. M., et al. Retinoic acid signaling regulates differential expression of the tandemly-duplicated long wavelength-sensitive cone opsin genes in zebrafish. PLoS Genetics. 11 (8), 1005483 (2015).
- Stevens, C. B., Cameron, D. A., Stenkamp, D. L. Plasticity of photoreceptor-generating retinal progenitors revealed by prolonged retinoic acid exposure. BMC Developmental Biology. 11 (1), (2011).
- Eldred, K. C., et al. Thyroid hormone signaling specifies cone subtypes in human retinal organoids. Science. 362 (6411), (2018).
- Yang, F., Ma, H., Ding, X. Q. Thyroid hormone signaling in retinal development, survival, and disease. Vitamins and Hormones. 106, 333-349 (2018).
- Brzezinski, J. A., Reh, T. A. Photoreceptor cell fate specification in vertebrates. Development. 142 (19), 3263-3273 (2015).
- Kim, S., et al. Generation, transcriptome profiling, and functional validation of cone-rich human retinal organoids. Proceedings of the National Academy of Science U. S. A. 116 (22), 10824-10833 (2019).
- Lowe, A., Harris, R., Bhansali, P., Cvekl, A., Liu, W. Intercellular adhesion-dependent cell survival and ROCK-regulated actomyosin-driven forces mediate self-formation of a retinal organoid. Stem Cell Reports. 6 (5), 743-756 (2016).
- Carcamo-Orive, I., et al. Analysis of transcriptional variability in a large human ipsc library reveals genetic and non-genetic determinants of heterogeneity. Cell Stem Cell. 20 (4), 518-532 (2017).
- DeBoever, C., et al. Large-scale profiling reveals the influence of genetic variation on gene expression in human induced pluripotent stem cells. Cell Stem Cell. 20 (4), 533-546 (2017).

