Full-Circle Cauterization of Limbal Vascular Plexus for Surgically Induced Glaucoma in Rodents
Summary
The goal of this protocol is to characterize a novel model of glaucomatous neurodegeneration based on 360° thermic cauterization of limbal vascular plexus, inducing subacute ocular hypertension.
Abstract
Glaucoma, the second leading cause of blindness worldwide, is a heterogeneous group of ocular disorders characterized by structural damage to the optic nerve and retinal ganglion cell (RGC) degeneration, resulting in visual dysfunction by interrupting the transmission of visual information from the eye to the brain. Elevated intraocular pressure is the most important risk factor; thus, several models of ocular hypertension have been developed in rodents by either genetic or experimental approaches to investigate the causes and effects of the disease. Among those, some limitations have been reported such as surgical invasiveness, inadequate functional assessment, requirement of extensive training, and highly variable extension of retinal damage. The present work characterizes a simple, low-cost, and efficient method to induce ocular hypertension in rodents, based on low-temperature, full-circle cauterization of the limbal vascular plexus, a major component of aqueous humor drainage. The new model provides a technically easy, noninvasive, and reproducible subacute ocular hypertension, associated with progressive RGC and optic nerve degeneration, and a unique post-operative clinical recovery rate that allows in vivo functional studies by both electrophysiological and behavioral methods.
Introduction
Medical literature understands glaucoma as a heterogeneous group of optic neuropathies characterized by progressive degeneration of retinal ganglion cells (RGCs), dendrites, soma, and axons, resulting in structural cupping (excavation) of the optic disc and functional deterioration of the optic nerve, leading to amaurosis in uncontrolled cases by interrupting the transmission of visual information from the eye to the brain1. Glaucoma is currently the most common cause of irreversible blindness worldwide, predicted to reach approximately 111.8 million people in 20402, thus deeply affecting patients' quality of life (QoL) and leading to significant socioeconomic concerns3.
Elevated intraocular pressure (IOP) is one of the most important and the only modifiable risk factor for the development and progression of glaucoma. Among the multiple types of glaucoma, all, except for normal tension glaucoma (NTG), are associated with elevated IOP at some time in the clinical history of the disease. Despite remarkable clinical and surgical advances to target IOP and slow down or stop disease progression, patients still lose sight due to glaucoma4,5. Therefore, a thorough understanding of the complex and multifactorial pathophysiology of this disease is imperative for the development of more effective treatments, especially to provide neuroprotection to RGCs.
Among a variety of experimental approaches for the understanding of disease mechanisms, animal models based on ocular hypertension (OHT) most closely resemble human glaucoma. Rodent models are particularly useful as they are low-cost, are easy to handle, can be genetically manipulated, have a short lifespan, and present ocular anatomical and physiological features comparable to humans, such as aqueous humor production and drainage6,7,8,9,10,11,12,13. Currently used models include sclerosis of the trabecular meshwork following injection of hypertonic saline into episcleral veins14, intracameral injection of microbeads15 or viscoelastic substances16, cauterization of vortex veins17, photocoagulation of the trabecular meshwork with argon laser18, circumlimbal suture19, and use of a transgenic model of age-related OHT (DBA/2J mice)8. However, invasiveness, post-operative opacification of the cornea, anterior segment disruption, extensive learning curves, expensive equipment, and highly variable postoperative IOPs, are among few of the reported pitfalls associated with the current models, making the development of an alternative model of OHT a demand to overcome these problems20,21,22.
The present protocol formalizes a novel surgical procedure to induce OHT as a proxy to glaucoma, based on limbal plexus cauterization (LPC) in rodents23. This is an easy, reproducible, accessible, and non-invasive model that provides high efficiency and low variability of IOP elevation, associated with a uniquely high rate of full clinical recovery, therefore providing in vivo functional evaluation in a reduced number of animals used in each experiment. The surgery technique induces subacute OHT with a gradual return to baseline levels in a few days, which models the hypertensive attack seen in acute angle-closure glaucoma. Moreover, the IOP recovery in the model is followed by continuous glaucomatous neurodegeneration, which is useful for future mechanistic studies of the secondary degeneration of RGCs, which occurs in several cases of human glaucoma despite adequate control of IOP.
Protocol
All procedures were performed in compliance with the Statement for the Use of Animals in Ophthalmic and Visual Research from the Association for Research in Vision and Ophthalmology (ARVO) and approved by the Ethics Committee on the Use of Animals in Scientific Experimentation from the Health Sciences Center, Federal University of Rio de Janeiro (protocol 083/17). In the present work, Lister Hooded rats of both genders were used, aged 2-3 months and weighing 180-320 g. However, the procedure can be adapted in different rat strains of various age ranges.
1. Ocular hypertension surgery and clinical follow-up
- House animals in a controlled temperature environment and 12 h light/dark cycle (6 am: light on/ 6 pm: light off) with standard pelletized gamma-irradiated food (Nuvilab® CR-1, Quimtia S/A, Brazil) and water (triple filter, with non-toxic activated charcoal) available ad libitum.
- Prepare a stock anesthetic cocktail mixture composed of three parts of 10% ketamine hydrochloride and one part of 2% xylazine hydrochloride (diluent: sterile water).
- Gently restrain the animal by holding the dorsal skin and induce anesthesia by intraperitoneal administration of 1 µL/g of body weight of the mixture (ketamine: 75 mg/kg; xylazine: 5 mg/kg). Check for proper anesthetization after approximately 5 min by performing a toe pinch response.
- Topically anesthetize both eye surfaces by instilling proxymetacaine hydrochloride 0.5% eyedrop. Wait for 30-60 s and remove the remaining solution from the anterior aspect of the globe, by gently touching the nasal or lateral bulbar conjunctiva with a small sterile cotton swab.
- Measure baseline IOP of both experimental and contralateral control eyes by placing the animal in a ventral decubitus position on the benchtop such that the corneal surface is easily accessible to the tip of the tonometer.
- Use either applanation or rebound handheld tonometer (Figure 1A). Position the tonometer tip such that it slightly touches the central corneal zone perpendicularly. Acquire and average 3-5 reliable machine-generated averages. Each mean is automatically calculated by the device after six successful individual measurements.
- Load the rebound tonometer with a probe and press the measurement button once to turn it on, while placing the tip of the device upwards, in order to avoid the probe to fall. After turning on, the display will show 00 indicating that the equipment is ready to measure.
- Position the device with the tip of the probe at a distance of 1-4 mm from the cornea and acquire measurements by rapidly and carefully pressing the measurement button, without moving the equipment. Each successful measure is identified by a short beep and after six times, the mean is displayed on the device's screen.
NOTE: Despite a long experience with applanation tonometer, to optimize the entire procedure the authors recommend the use of a rebound tonometer, which provides easier acquisition of reliable IOP measurement.
- Place the animal on a slight lateral decubitus position under a stereo microscope, and carefully plan the surgery by inspecting the experimental eye at 40x magnification. Do not forget to maintain the contralateral control eye lubricated during the procedure by instilling a drop of carmellose sodium or sodium hyaluronate.
- With the aid of curved forceps, gently push forward the experimental eyeball so as to expose the vasculature that surrounds 360° of the limbus13. With the other hand, gently cauterize the vessels all around the cornea with a low-temperature ophthalmic cautery (1,300 °F; Bovie Medical, USA) (Figure 1B-E).
NOTE: The mentioned cautery has a round tip that should touch limbal vasculature longitudinally. - Be careful not to cauterize the corneal periphery, as this may result in post-operative corneal opacification, which precludes in vivo assessment of retinal function.
- Observe the emergence of small circular marks of cauterization on the scleral limbus, the obliteration of limbal vasculature, and pupil dilation in the operated eye, which are signs of a successful surgical procedure.
NOTE: As the limbal vasculature of rats and mice is anatomically similar13,24 and the cautery used has a gentle small tip, this protocol can work for the mouse eye as well without any adaptation. - After surgery, check the immediate post-operative IOP in both eyes such as described in step 1.5.
- Apply a drop of ophthalmic prednisolone acetate (1.2 mg/mL) and maintain this in contact with the anterior surface of the experimental eye for around 40 s, then replace it with an ophthalmic ointment of antibiotics (oxytetracycline hydrochloride 30 mg/g plus polymyxin B 10,000 U/g; or ciprofloxacin 3.5 mg/g). Perform intramuscular injection of tramadol hydrochloride (single dose; 2 mg/kg) to prevent pain after the procedure.
- Follow closely the animal recovery from anesthesia, preferentially in a warm environment such as a heating pad or inside its housing cage with proper bedding. Pay attention to the respiratory pattern.
- Perform a daily clinical follow-up of the experimental eye with topical medications composed of a non-steroidal anti-inflammatory drug (NSAID, e.g., ketorolac trometamol 0.5% eye drop) and antibiotic ointment (preferably the same one used immediately after surgery).
NOTE: The use of NSAID rather than steroidal anti-inflammatory eye drops is recommended because the latter may induce OHT and is regularly applied for glucocorticoid-induced glaucoma models in rodents.- Under slight sedation (ketamine: 18.75 mg/kg; xylazine: 1.25 mg/kg), measure IOP preferentially at the same period of the day, to avoid bias due to physiological circadian IOP fluctuation.
- During the ocular examination, note rare clinical intercurrences, such as hyphema, corneal fibrosis, or scleral thinning associated with uveal prolapse. Corneal edema, chemosis, and conjunctival hyperemia are common but temporary.
- Notice full clinical recovery at around postoperative day 7. Limbal revascularization usually begins in the first two days after surgery, in line with the expected gradual IOP return to baseline.
2. Optomotor response (OMR) analysis
NOTE: For this procedure, a specific system was used25.
- Arrange four computer monitors in a quadrangle, that delimit an arena with a platform in the middle. On the monitors', display the image of a virtual cylinder with vertically oriented sine-wave gratings (alternate black and white stripes), rotating around the platform at a fixed speed (12 degree/s) and contrast (100%).
- Position a video camera above the platform, allowing the experimenter to watch the animal's movements. Perform the test in photopic conditions and set the software preferentially on manual/separate mode in order to evaluate each eye separately.
- Allow animals to habituate for approximately 2 min on the platform. Maintain the red crosshair cursor on the video frame between the eyes of the freely moving animal, as it indicates the center of the virtual cylinder (Figure 1G)25.
- Observe the OMR, consisting of a reflexive tracking of the animal's head and neck elicited by the rotating gratings. Test the left and the right visual pathways by rotating the directions of the sine-wave gratings clockwise and counterclockwise, respectively.
- Initially present a stimulus with low spatial frequency (0.042 cycles/degree). Then, increment the frequency progressively until tracking movement is not noticed anymore. The highest spatial frequency in which a clear OMR is elicited corresponds to the threshold spatial frequency of the evaluated eye.
- If the animal eventually drops off the platform during the exam, immediately return it to the platform and resume the test.
3. Recording of pattern-electroretinogram (PERG)
NOTE: The electroretinogram was recorded using a specific system for signal processing and related software for storage and analysis of the waveforms.
- Deeply anesthetize animals by intra-muscular injection of ketamine hydrochloride and xylazine hydrochloride (75 mg/kg and 5 mg/kg, respectively). Deep anesthesia (surgical plane) decreases the chances of artifacts by involuntary muscle movements or alternative sources of noise during the exam. Check for proper anesthetization by performing a toe pinch response.
NOTE: The mentioned anesthetic agents do not affect the amplitude of the response26. As a small needle inserted into the cornea was used as the active electrode, intra-muscular anesthesia is preferred instead of intraperitoneal, as it is easier to reinject in case the rat needs a booster dose (1/2 of the initial dose), with lower chances to modify the electrode position and thus leading to more reproducible records during the whole experiment, which may last up to 60 min. - Topically anesthetize the cornea with a drop of proxymetacaine hydrochloride 0.5%, and maintain the fellow eye moistened with ophthalmic lubricants.
- Carefully insert the active electrode (stainless steel needle 0.25 mm × 15 mm) at the temporal periphery of the cornea. Additionally, insert the reference and ground electrodes (stainless steel needle 0.4 mm × 37 mm) into the subcutaneous tissue of the ipsilateral temporal canthus and in one of the hind limbs, respectively (Figure 1I).
- For PERG, set the stimulus to a black and white reserving checkerboard, alternating at 15 reversals/s with constant average luminance (250 cd/m2). Set the band-pass filter to 1 Hz-100 Hz.
NOTE: The rapid reversing stimulus generates the steady-state PERG, a stable and reproducible sinusoid that gathers the wave component most likely associated with RGC bioelectrical response: NII deflection of the transient-state PERG. - Position the animal at 20 cm from the stimulus screen (LCD monitor 0.58 m; Figure 1I), monitor signal baseline, and start PERG acquisition by pressing Analysis button on the acquisition system.
- During the procedure, keep the animals adapted to environmental light (white light of ~140 lux). Here, six distinct spatial frequencies (in cycles per degree: 0.018, 0.037, 0.073, 0.146, 0.292, 0.585) were presented in random sequence.
NOTE: The software used for signal processing automatically performs averaging. The average of 200-300 individual waves was considered adequate to stand out from the noise and analyze wave amplitude.
4. Quantification of retinal ganglion cells somas
NOTE: The following procedure is for quantification of RGC somas, based on immunohistochemical staining of retinal flat-mounts with an antibody against the brain-specific homeobox/POU domain protein 3A (Brn3a).
- Subject experimental animals to cervical dislocation euthanasia, preceded by carbon dioxide inhalation to induce unconsciousness.
- Immediately after euthanasia, dissect both eyes under a stereo microscope, using toothed forceps and curved scissors.
- Perform careful dissection such that both the distal portion of the extraocular muscles and the caruncle remain attached to the globe, as they are important landmarks for topographic orientation of the retina (described in step 4.5). Try to save a stretch of the optic nerve attached to the globe as long as possible for future analysis.
- After enucleation, place the eyes in a 1 mL solution of 4% paraformaldehyde (4% PFA) in 0.1 M phosphate buffer (PBS) and keep it for 24 h for proper chemical fixation.
- Separate the retina from the rest of the ocular tissues.
- Under a dissecting microscope place the eyeball in a Petri dish covered with 1x PBS, and pay attention to important landmarks such as nasal caruncle, choroid fissures, and scleral imprints from vortex veins for proper topographic orientation27.
- Penetrate the anterior chamber from the central cornea by using toothed forceps and curved scissors (westcott). Make two radial cuts to the superior (dorsal) aspect of the sclera, toward the scleral foramen of the optic nerve, so as to delimit the dorsal quadrant of the eyeball.
- Separate the cornea from the rest of the globe through a longitudinal 360° cut at the scleral limbus. Remove the lens and the iris and delimit the dorsal retinal quadrant using the same scleral radial demarcations described above (step 4.5.2).
- Carefully detach the retinal tissue from the choroid and sclera, avoiding both random lacerations throughout the tissue and eventual topographic loss of orientation.
- Separate the ciliary body from retinal ora serrata. Carefully remove the remaining vitreous body from the retinal cup by using both curved non-toothed forceps plus curved scissors (pull vitreous and dissect it close to the internal limiting membrane) and a small brush.
NOTE: Removing vitreous humor is an important step to obtain a strong and clean immunohistochemical signal.
- Transfer isolated retinas into a 24-well culture plate (one retina per well) containing 1 mL of 1x PBS and keep the inner retina facing up.
- Permeabilize tissue by washing 3x for 10 min with a non-ionic surfactant 0.5% diluted in 1x PBS (0.3 mL). Then keep the tissue gently shaking in 5% bovine serum albumin (BSA) in non-ionic surfactant 2% and 1x PBS (blocking solution; 0.25 mL) for 60 min at room temperature.
- During step 4.7, prepare the Brn3a primary antibody solution by diluting 1:200 in non-ionic surfactant 0.5% and 1x PBS plus 5% BSA, and store it at 4 °C.
- After 60 min of tissue blocking, incubate retinas in 0.2 mL of primary antibody solution at 4 °C for 72 h with gentle shaking.
- Wash the tissue 3x for 10 min with 1x PBS, then incubate the tissue for 2 h at room temperature in 0.2 mL of the secondary antibody solution diluted 1:750 in 1x PBS plus 5% BSA.
- Further, incubate the tissue for 10 min in nuclear counterstain solution for fluorescent nuclei staining. Conclude immunohistochemistry with a final washing step with 1x PBS (0.3 mL) repeated 3x.
- Transfer retinas with the aid of two small brushes onto glass microscope slides, maintaining the vitreous side up. Position the dorsal retinal quadrant up on the microscope slides (previously delimited in step 4.5.3). Make two more radial cuts towards the optic nerve head to delimit the other 3 quadrants (nasal, ventral, and temporal).
NOTE: The cut dimensions are not fixed. They should not be too short that impairs an efficient flattening of the retina on the slide and should not be too long so that it reaches the optic nerve foramen and completely separates the delimited retinal quadrant from the rest of the tissue. - Finally, apply 0.2 mL of antifade mounting medium on a glass coverslip and place this onto the flat-mounted retina for tissue microscopic analysis. To estimate RGCs density, examine the flat mounts under a confocal epifluorescence microscope using a 40x/1.3 objective.
- For each quadrant of the retina, take eight photos: two from the central retina (~0.9 mm from optic disc), three from mid-retina (~2.0 mm from optic disc), and three from the peripheral retina (~3.7 mm from optic disc), totaling to 32 photos per retina. Use the FIJI software to count Brn3a-positive cells and estimate the mean cell density.
5. Examination of the optic nerve
- After euthanasia and eyeball enucleation (steps 4.1-4.3), remove the proximal segment of the intraorbital portion of the optic nerve (1-2 mm), including part of the intraocular portion, and immediately place the samples into vials/tubes containing 0.2-0.3 mL of cold fixative (2.5% glutaraldehyde solution in 0.1 M sodium cacodylate buffer (pH 7.4)) for 2 h.
NOTE: The following steps for optic nerve processing are performed in the same vials/tubes from step 5.1 - Wash the material 3x for 5 min with cold 0.1 M sodium cacodylate buffer. Post fixate tissue for 1 h under gentle shaking in a solution of 1.0% osmium tetroxide in 0.8% potassium ferrocyanide and 5 nM calcium chloride diluted in 0.1 M sodium cacodylate buffer at 4 °C (0.1-0.2 mL).
- Wash optic nerve fragments 3x for 5 min with cold 0.1 M sodium cacodylate buffer and subsequently with cold distilled water, 3x for1 min. Keep material overnight under gentle shaking in a solution of 1.0% uranyl acetate in distilled water for staining at 4 °C (0.1-0.2 mL). Wash fragments 3x with cold distilled water.
- Progressively dehydrate the tissue with a graded acetone series (0.5 mL each), with subsequent replacements of the following dilutions in distilled water: 2x 7 min incubation in 15% ice-cold acetone; 2x 7 min incubation in 30% ice-cold acetone; 2x 7 min incubation in 50% ice-cold acetone; 2x 7 min incubation in 70% ice-cold acetone; 2x 7 min incubation in 80% ice-cold acetone; 2x 7 min incubation in 90% ice-cold acetone; 2x 15 min incubation in 100% acetone at room temperature (RT).
- Perform 3 infiltration/ embedding steps, with subsequent replacement of the following solutions: 1 part epoxy resin: 2 parts acetone (total volume: 0.5 mL), at RT for 12 h; 1 part epoxy resin: 1 part acetone (total volume: 0.5 mL), at RT for 12 h; 2 parts epoxy resin: 1 part acetone (total volume: 0.5 mL), at RT for 12 h. Finally, infiltrate tissue in pure epoxy resin at RT for 24 h.
- Remove samples from the specimen carrier, transfer them to embedding molds, and let it polymerize for 48 h at 60 °C. Cut transversal semi-thin sections (300-400 nm) of the optic nerve fragments using an ultramicrotome, collect and transfer them onto a microscope glass slide.Stain sections with toluidine blue and image the using optic microscope at 100x magnification.
- For ultrastructural analysis, perform ultrathin cross-sections (70 nm), collect them on copper grids, and stain them with uranyl acetate and lead citrate. Examine the sections in a transmission electron microscope.
Representative Results
The quantitative variables are expressed as mean ± standard error of the mean (SEM). Except for the comparison of IOP dynamics between OHT and control groups (Figure 1F), statistical analysis was performed using two-way ANOVA followed by Sidak's multiple comparisons test. A p-value < 0.05 was considered statistically significant.
Figure 1 illustrates surgical steps of the full-circle limbal plexus cauterization (LPC) model, with important landmarks such as 360° thermal-induced disappearance of limbal vessels, as well as the mild to moderate mydriasis in the operated eye at the end of the procedure.
In the present series of 131 rats, full-circle limbal plexus cauterization (LPC) induced IOP elevation immediately after surgery from 13.0 ± 0.2 mmHg at baseline to 22.7 ± 0.4 mmHg. Peak postoperative IOP was observed on the first day after surgery (25.3 ± 0.6 mmHg), followed by a gradual return to baseline levels at the 6th postoperative day (statistical analysis: multiple t-test corrected for multiple comparisons using the Holm-Sidak method; Figure 1F). Corneal fibrosis or edema were clinical intercurrences that could potentially compromise accurate IOP measurement. The first, on one hand, was rare, affecting 3.92% of animals and noticed late during the postoperative follow-up, thus sparing the first 5 days of ocular hypertension and preserving the subacute OHT profile described23. Corneal edema, on the other hand, was a more common complication seen during the first few days after surgery (1-3 days), but mostly mild and temporary, thus did not robustly affect IOP23.
Retinal function was evaluated both behaviorally and electrophysiologically using the optomotor reflex and pattern-ERG, respectively (Figure 1G–J). Both parameters showed two phases of impairment: an ocular hypertension acute phase at 3rd day post-surgery, and a secondary degeneration phase at the 30th day post-surgery (Figure 1H, Figure J, and Table 1). In between, a period of functional recovery was detected, as previously discussed elsewhere23.
Compared with control fellow optic nerves, axonal counts in semi-thin transversal optic nerve sections showed progressive decrease after surgery (3rd day: 68.3% ± 0.9%; 7th day: 59.2% ± 2.6%; 14th day: 45.4% ± 2.2%; 30th day: 28.2 % ± 3.0%; two-way ANOVA: p < 0.0001; Figure 2A). Ultra-structurally, optic nerves from control eyes presented densely packed myelinated fibers, separated by thin glial cells processes, and evident axonal microtubules and neurofilaments (Figure 2B). In contrast, at 3 days after OHT, we found focal disruption of axon bundles, a few degenerated fibers, cytoplasmic vacuolation in glial cells processes, and condensed chromatin in glial cell nuclei. After 7 days of OHT induction, there was an increase in degenerated axonal fibers, hypertrophic glial cell processes, and swelling and voids in individual axonal fibers. At 14 days, one of the most prominent changes was a greater disarrangement of the optic nerve fibers, associated with the invasion of glial cell processes among the axons. Filament bundles filled these processes and dark degenerated fibers, and myelin breakdown was more common, associated with detached and vacuolized lamellae (Figure 2B).
The density of Brn3a+ profiles decreased along time (Figure 2C), mainly in the dorsal and temporal retinal quadrants, down to 32.4% ± 9.6% and 35.7% ± 9.1% after 30 days, respectively (Figures 2D–G and Table 2).
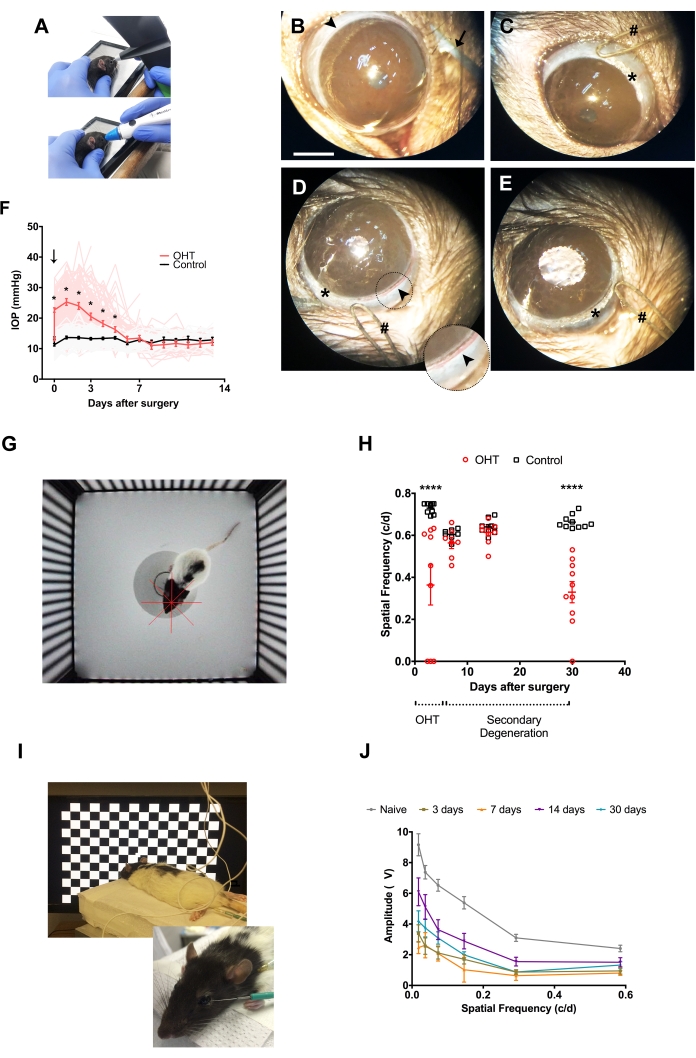
Figure 1: Thermal cauterization of limbal vascular plexus and consequences to retinal function in vivo. (A) Alternative methods to measure IOP in rats: rebound tonometry (superior), and applanation tonometry (inferior). (B–E) Surgical procedure; arrowhead: limbal vascular plexus; arrow: curved forceps used to expose anterior surface of the eyeball and optimize surgical assessment to limbal vessels; hash: the round tip of the low-temperature ophthalmic cautery; asterisk: cauterization mark. Scale bar: 2 mm. Inset in (D) shows in higher magnification the limbal vasculature to be cauterized. (F) Time course of IOP measurements in OHT (red) and control (black) eyes (n = 131). Vertical downward arrow: LPC surgery. * = p < 0.05 (G) Arena for optomotor response analysis, comprised of four computer monitors arranged in a quadrangle, with a platform in the middle. The monitors display the image of a virtual cylinder composed of vertical alternate black and white stripes moving around the animal with constant rotational speed and variable spatial frequencies. The red crosshair corresponds to the center of the virtual cylinder. (H) Optomotor responses. Two distinct phases are distinguished upon surgical follow-up: the OHT phase (0-5 days) and the secondary degeneration phase (6-30 days). **** = p < 0.0001. (I) Electrodes and animal positioning for pattern-ERG (PERG) acquisition: the active electrode at the temporal periphery of the cornea, and the reference and ground electrodes into the subcutaneous tissue of the ipsilateral temporal canthus and one of the hind limbs, respectively. The animal is positioned at 20 cm from the stimulus screen. (J) PERG amplitude upon stimuli with different spatial frequencies. Similar to optomotor response, PERG evaluation also shows two distinct phases of responses after surgery: ocular hypertensive at 3 days post-surgery, followed by recovery at day 7 and 14, although still statistically lower than naïve responses, and a subsequent decrease at day 30 after surgery. c/d = cycles per degree. Naïve group: eyes of animals unexposed to any previous experimental manipulation. (H) and (J) show mean ± SEM, plus individual replicates in (H). Please click here to view a larger version of this figure.
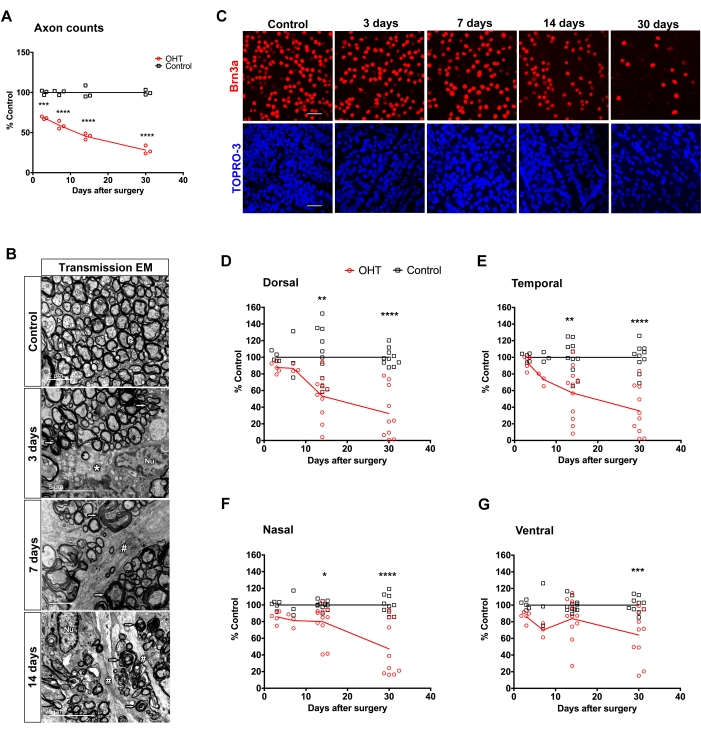
Figure 2: Structural assessment of retina and optic nerve after limbal vascular plexus cauterization (LPC) with low-temperature ophthalmic cautery. (A) Axon counts at distinct times after OHT (n = 3). *** = p = 0.0005, **** = p < 0.0001. (B) Electron micrographs of optic nerve degeneration following OHT. The left image shows the control optic nerve, and the following images illustrate the progressive degeneration after 3, 7, and 14 days of OHT. Arrowhead: normal myelinated fibers; thin arrows: degenerated fibers; asterisk: cytoplasmic vacuolation; hashes: glial cells process; and Nu: glial cell nucleus. (C) Photomicrographs of representative counting fields of RGCs labeled with an antibody to Brn3a (red) and TO-PRO3 labeled nuclei (blue); scale bar: 50µm. (D–G) Distribution of average Brn3a+ cell density in the four quadrants of the retina after 3-30 days of surgery. The graphs show individual averages of RGC densities for 3-11 animals per time after the procedure. * = p < 0.05; ** = p < 0.01; *** = p < 0.001; **** = p < 0.0001. Quantitative data is mean ± SEM. Please click here to view a larger version of this figure.
Table 1: Statistical analysis of PERG data. Two-way ANOVA followed by Sidak's multiple comparisons test. P-value less than 0.05 was considered statistically significant. c/d = cycles per degree. Please click here to download this Table.
Table 2: Regional loss of RGC after LPC. SEM: standard error of the mean. Control: fellow eye. P-values less than 0.05 were considered statistically significant (*). Please click here to download this Table.
Discussion
Limbal plexus cauterization (LPC) is a novel post-trabecular model with the advantage that it targets easily accessible vascular structures not requiring conjunctival or tenon dissection17,28. Differently from the vortex veins cauterization model, a renowned OHT model based on the surgical impairment to choroid venous drainage, venous congestion is not expected to influence IOP rise in the LPC model, as limbal veins are situated upstream in aqueous humor outflow. Also, it is technically easy to learn and low-cost, requiring mostly a low-temperature thermic cautery. Moreover, it is associated with a unique rate of OHT induction and full clinical recovery (> 90%, as previously reported)23, which reduces the number of animals necessary for experiments and allows for both electrophysiological and behavioral analysis. Finally, glaucomatous degeneration is present in both the RGC layer and optic nerve in a relatively short time frame after surgery, enabling either short-term or mid-term experimental designs23. Future studies are necessary to further elucidate the impact of full-circle limbal vasculature cauterization on individual retinal layers.
Critical steps in the surgical protocol are: (1) the cauterization tip must gently touch the scleral limbus parallel to the vessel axis; (2) corneal tissue must be spared, not only during cauterization but also during animal manipulation. Regularly perform eyedrop instillation and removal of excess solution from eye surface with a cotton swab (be careful not to scrub the cotton swab on the corneal surface while removing any liquid, as it leads to corneal epithelial abrasion and eventual post-surgery complications); (3) a full-circle of contiguous cauterization marks should be visualized; (4) do not neglect postoperative clinical follow-up with a non-steroidal anti-inflammatory drug and antibiotic ointment, at least until the 5th day after surgery.
The subacute IOP elevation seen in the described model differs from the pressure dynamics of open-angle glaucoma but is akin to acute angle-closure glaucoma, neovascular glaucoma, or multiple types of post-trabecular glaucoma with elevated episcleral venous pressure29. This is a major limitation of this method, as open-angle glaucoma is the most prevalent phenotype of the disease, characterized by chronic OHT and slowly progressive RGC degeneration. Nevertheless, the association of progressive normalization of IOP with continuous RGC degeneration represents a unique opportunity to study, in the same animal model, both the biological mechanisms that link ocular hypertension with the development and progression of glaucoma, as well as the secondary degenerative process mostly observed in open-angle glaucoma cases, whereupon patients present with glaucoma progress despite clinical or surgical success in reaching target IOP30. Thus, this model represents an opportunity to better elucidate this phenomenon through short-term or mid-term experimental designs, and eventually develop pressure-independent neuroprotective therapies to benefit patients that still lose sight despite best IOP control treatment.
Declarações
The authors have nothing to disclose.
Acknowledgements
We acknowledge our laboratory technicians José; Nilson dos Santos, Daianne Mandarino Torres, José Francisco Tibúrcio, Gildo Brito de Souza, and Luciano Cavalcante Ferreira. This research was funded by FAPERJ, CNPq, and CAPES.
Materials
| Acetone | Isofar | 201 | Used for electron microscopy tissue preparation (step 5) |
| Active electrode for electroretinography | Hansol Medical Co | – | Stainless steel needle 0.25 mm × 15 mm |
| Anestalcon | Novartis Biociências S/A | MS-1.0068.1087 | Proxymetacaine hydrochloride 0.5% |
| Calcium chloride | Vetec | 560 | Used for electron microscopy tissue preparation (step 5) |
| Cautery Low Temp Fine Tip 10/bx | Bovie Medical Corporation | AA00 | Low-temperature ophthalmic cautery |
| Cetamin | Syntec do Brasil Ltda | 000200-3-000003 | Ketamine hydrochloride 10% |
| DAKO | Dako North America | S3023 | Antifade mounting medium |
| DAPI | Thermo Fisher Scientific | 28718-90-3 | diamidino-2-phenylindole; blue fluorescent nuclear counterstain; emission at 452±3 nm |
| Ecofilm | Cristália Produtos Químicos Farmacêuticos Ltda | MS-1.0298.0487 | Carmellose sodium 0.5% |
| EPON Resin | Polysciences, Inc. | – | Epoxy resin used for electron microscopy, composed of a mixture of four reagents: Poly/Bed 812 Resin (CAT#08791); DDSA – Dodecenylsuccinic Anhydride (CAT#00563); NMA – Nadic Methyl Anhydride (CAT#00886); DMP-30 – 2,4,6-tris(dimethylaminomethyl)phenol (CAT#00553) |
| Glutaraldehyde | Electron Microscopy Sciences | 16110 | Used for electron microscopy tissue preparation (step 5) |
| Hyabak | União Química Farmacêutica Nacional S/A | MS-8042140002 | Sodium hyaluronate 0.15% |
| Icare Tonolab | Icare Finland Oy | TV02 (model number) | Rebound handheld tonometer |
| IgG donkey anti-mouse antibody + Alexa Fluor 555 | Thermo Fisher Scientific | A31570 | Secondary antibody solution |
| LCD monitor 23 inches | Samsung Electronics Co. Ltd. | S23B550 | Model LS23B550, for electroretinogram recording |
| LSM 510 Meta | Carl Zeiss | – | Confocal epifluorescence microscope |
| Maxiflox | Cristália Produtos Químicos Farmacêuticos Ltda | MS-1.0298.0489 | Ciprofloxacin 3.5 mg/g |
| MEB-9400K | Nihon Kohden Corporation | – | System for electroretinogram recording |
| monoclonal IgG1 mouse anti-Brn3a | MilliporeSigma | MAB-1585 | Brn3a primary antibody solution |
| Neuropack Manager v08.33 | Nihon Kohden Corporation | – | Software for electroretinogram signal processing |
| Optomotry | CerebralMechanics | – | System for optomotor response analysis |
| Osmium tetroxide | Electron Microscopy Sciences | 19100 | Used for electron microscopy tissue preparation (step 5) |
| Potassium ferrocyanide | Electron Microscopy Sciences | 20150 | Used for electron microscopy tissue preparation (step 5) |
| Reference and ground electrodes for electroretinography | Chalgren Enterprises | 110-63 | Stainless steel needles 0.4 mm × 37 mm |
| Sodium cacodylate buffer | Electron Microscopy Sciences | 12300 | Used for electron microscopy tissue preparation (step 5) |
| Ster MD | União Química Farmacêutica Nacional S/A | MS-1.0497.1287 | Prednisolone acetate 0.12% |
| Terolac | Cristália Produtos Químicos Farmacêuticos Ltda | MS-1.0497.1286 | Ketorolac trometamol 0.5% |
| Terramicina | Laboratórios Pfizer Ltda | MS-1.0216.0024 | Oxytetracycline hydrochloride 30 mg/g + polymyxin B 10,000 U/g |
| Tono-Pen XL | Reichert Technologies | 230635 | Digital applanation handheld tonometer |
| TO-PRO-3 | Thermo Fisher Scientific | T3605 | Far red-fluorescent nuclear counterstain; emission at 661 nm |
| Triton X-100 | Sigma-Aldrich | 9036-19-5 | Non-ionic surfactant |
| Uranyl acetate | Electron Microscopy Sciences | 22400 | Used for electron microscopy tissue preparation (step 5) |
| Xilazin | Syntec do Brasil Ltda | 7899 | Xylazine hydrochloride 2% |
| Carl Zeiss | – | Stereo microscope for surgery and retinal dissection |
Referências
- Weinreb, R. N., Aung, T., Medeiros, F. A. The Pathophysiology and Treatment of Glaucoma A Review. JAMA. 311 (8), 1901-1911 (2014).
- Tham, Y. C., et al. Global prevalence of glaucoma and projections of glaucoma burden through 2040: A systematic review and meta-analysis. Ophthalmology. 121 (11), 2081-2090 (2014).
- Quaranta, L., et al. Quality of Life in Glaucoma: A Review of the Literature. Advances in Therapy. 33 (6), 959-981 (2016).
- Heijl, A., et al. Reduction of intraocular pressure and glaucoma progression: results from the Early Manifest Glaucoma Trial. Archives of Ophthalmology. 120 (10), 1268-1279 (2002).
- Susanna, R., De Moraes, C. G., Cioffi, G. A., Ritch, R. Why Do People (Still) Go Blind from Glaucoma. Translational Vision Science & Technology. 4 (2), 1 (2015).
- Fujikawa, K., et al. VAV2 and VAV3 as candidate disease genes for spontaneous glaucoma in mice and humans. PLoS One. 5 (2), 9050 (2010).
- Mao, M., Hedberg-Buenz, A., Koehn, D., John, S. W., Anderson, M. G. Anterior segment dysgenesis and early-onset glaucoma in nee mice with mutation of Sh3pxd2b. Investigative Ophthalmology & Visual Science. 52 (5), 2679-2688 (2011).
- John, S. W., et al. Essential iris atrophy, pigment dispersion, and glaucoma in DBA/2J mice. Investigative Ophthalmology & Visual Science. 39 (6), 951-962 (1998).
- Aihara, M., Lindsey, J. D., Weinreb, R. N. Ocular hypertension in mice with a targeted type I collagen mutation. Investigative Ophthalmology & Visual Science. 44 (4), 1581-1585 (2003).
- Chou, T. H., Tomarev, S., Porciatti, V. Transgenic mice expressing mutated Tyr437His human myocilin develop progressive loss of retinal ganglion cell electrical responsiveness and axonopathy with normal iop. Investigative Ophthalmology & Visual Science. 55 (9), 5602-5609 (2014).
- vander Zypen, E. Experimental morphological study on structure and function of the filtration angel of the rat eye. Ophthalmologica. 174 (5), 285-298 (1977).
- Aihara, M., Lindsey, J. D., Weinreb, R. N. Aqueous humor dynamics in mice. Investigative Ophthalmology & Visual Science. 44 (12), 5168-5173 (2003).
- Morrison, J. C., Fraunfelder, F. W., Milne, S. T., Moore, C. G. Limbal microvasculature of the rat eye. Investigative Ophthalmology & Visual Science. 36 (3), 751-756 (1995).
- Morrison, J. C., et al. A rat model of chronic pressure-induced optic nerve damage. Experimental Eye Research. 64 (1), 85-96 (1997).
- Sappington, R. M., Carlson, B. J., Crish, S. D., Calkins, D. J. The microbead occlusion model: A paradigm for induced ocular hypertension in rats and mice. Investigative Ophthalmology & Visual Science. 51 (1), 207-216 (2010).
- Zhu, M. D., Cai, F. Y. Development of experimental chronic intraocular hypertension in the rabbit. Australian and New Zealand Journal of Ophthalmology. 20 (3), 225-234 (1992).
- Shareef, S. R., Garcia-Valenzuela, E., Salierno, A., Walsh, J., Sharma, S. C. Chronic ocular hypertension following episcleral venous occlusion in rats. Experimental Eye Research. 61 (3), 379-382 (1995).
- Levkovitch-Verbin, H., et al. Translimbal laser photocoagulation to the trabecular meshwork as a model of glaucoma in rats. Investigative Ophthalmology & Visual Science. 43 (2), 402-410 (2002).
- Zhao, D., et al. Characterization of the Circumlimbal Suture Model of Chronic IOP Elevation in Mice and Assessment of Changes in Gene Expression of Stretch Sensitive Channels. Frontiers in Neuroscience. 11, (2017).
- Biswas, S., Wan, K. H. Review of rodent hypertensive glaucoma models. Acta Ophthalmologica. 97 (3), 331-340 (2019).
- Pitha, I., et al. Sustained Dorzolamide Release Prevents Axonal and Retinal Ganglion Cell Loss in a Rat Model of IOP-Glaucoma. Translational Vision Science & Technology. 7 (2), 13 (2018).
- Grozdanic, S. D., et al. Temporary elevation of the intraocular pressure by cauterization of vortex and episcleral veins in rats causes functional deficits in the retina and optic nerve. Experimental Eye Research. 77 (1), 27-33 (2003).
- Lani, R., et al. A subacute model of glaucoma based on limbal plexus cautery in pigmented rats. Scientific Reports. 9 (1), 16286 (2019).
- vander Merwe, E. L., Kidson, S. H. The three-dimensional organisation of the post-trabecular aqueous outflow pathway and limbal vasculature in the mouse. Experimental Eye Research. 125, 226-235 (2014).
- Prusky, G. T., Alam, N. M., Beekman, S., Douglas, R. M. Rapid quantification of adult and developing mouse spatial vision using a virtual optomotor system. Investigative Ophthalmology & Visual Science. 45 (12), 4611-4616 (2004).
- Sasovetz, D. Ketamine hydrochloride: an effective general anesthetic for use in electroretinography. Annals of Ophthalmology. 10 (11), 1510-1514 (1978).
- Stabio, M. E., et al. A novel map of the mouse eye for orienting retinal topography in anatomical space. The Journal of Comparative Neurology. 526 (11), 1749-1759 (2018).
- Blanco, R., et al. A Chronic Ocular-Hypertensive Rat Model induced by Injection of the Sclerosant Agent Polidocanol in the Aqueous Humor Outflow Pathway. International Journal of Molecular Sciences. 20 (13), 3209 (2019).
- Paranhos, A., Prata, J. A., de Mello, P. A., da Silva, F. A. Post-Trabecular Glaucomas with Elevated Episcleral Venous Pressure. Mechanisms of the Glaucomas. , 139-157 (2008).
- Ou, Y., Jo, R. E., Ullian, E. M., Wong, R. O. L., Della Santina, L. Selective Vulnerability of Specific Retinal Ganglion Cell Types and Synapses after Transient Ocular Hypertension. The Journal of Neuroscience: The Official Journal of The Society for Neuroscience. 36 (35), 9240-9252 (2016).

