Isolation of Monocyte-Macrophage Lineage Cells from Rat Bones by Secondary Adherence Method
Summary
Here we present a protocol for the isolation of BMMs from SD rats, called the secondary adherence method.
Abstract
With a decrease of bone mineral density, bones are more likely to fracture, thus negatively affecting a patient’s quality of life. The growth and development of bones are mainly regulated by osteoblasts and osteoclasts. It has been widely accepted that osteoclasts are derived from bone marrow monocyte-macrophage cells (BMMs). BMMs and other hematopoietic stem cells are located in the bone marrow cavity. Therefore, isolating single stable BMMs from different and heterogeneous cell populations is a huge challenge. Here we present a protocol for the isolation of BMMs from SD rats, called the secondary adherence method. Adherent cells cultured for 24-48 h in primary culture were collected. Flow cytometric analysis showed that approximately 37.94% of the cells were CD11b/c+ (monocyte-macrophage surface antigen). Tartrate resistant acid phosphatase (TRAP) staining and western blot analysis demonstrated that BMMs could differentiate into osteoclasts in vitro. The above findings suggested that the secondary adherence cells could be considered as a suitable cellular model for osteoclast differentiation research.
Introduction
It has been reported that monocyte-macrophage lineage cells existing in the bone marrow can differentiate into blood monocytes and tissue macrophages1,2. The above cells, which can differentiate into osteoclasts to balance bone growth and development, are commonly used as a cell model to induce osteoclasts in vivo3,4. Bone marrow is a special tissue containing several different types of cells, which include but are not only limited to bone marrow mesenchymal stem cells, bone marrow monocyte-macrophage cells (BMMs), hematopoietic stem cells, endothelial cells, and immune cells. In fact, several previous studies suggested that adherent cells rushed out of the bone marrow cavity of the long bone could differentiate into osteoblasts, osteoclasts, chondrocytes, or adipocytes5,6,7,8. Although, different isolation and culture methods have been used to produce different homogeneous cell populations, there are still great challenges in isolating and culturing BMMs from a variety of different cell types.
Several methods have been developed to extract bone marrow mononuclear macrophages (BMSCs). However, the majority of these methods are complex9,10,11. For example, density gradient centrifugation requires a specialized kit and the operation is time-consuming and cumbersome. This method is suitable for the isolation of BMMs from high-volume blood samples, but not from bone marrow samples9,12,13. In addition, extracting tissue samples using collagenase digestion is a complex and time-consuming procedure; this method is not recommended for the isolation of BMMs from bone marrow samples14,15. In addition, although flow separation can result in highly purified monocyte/macrophage populations, it requires very large sample sizes and high instrument and equipment requirements10,16. Additionally, the microbead enrichment method is extremely expensive and is not feasible in a general laboratory17.
Therefore, in the current study a convenient, fast, and cheap method was proposed for the isolation of mononuclear macrophages from the bone marrow. Bone marrow cells adhered for different time points were used to isolate BMMs using a secondary adherence method. BMMs extracted with the above method could induce the formation of osteoclasts in vitro, thus providing a simple and convenient cell model for the future study of osteoporosis in vitro.
Protocol
All experiments in this study were conducted in accordance with the animal experiment guidelines of the Zhejiang Chinese Medical University Laboratory Animal Research Center (Approval No: IACUC-20181029-11).
1. Cell extraction
- Put the Sprague-Dawley rats (SD rats, 1-10 days old, male or female) in the euthanasia cages filled with CO2 at a balanced rate of 30%-70% container volume/minute. After the rats lose consciousness (20-60 min), euthanize the rat by cervical dislocation to ensure a painless death.
- Immerse the rats in 75% alcohol for 10 min for disinfection.
- Carefully remove all the limbs of the rat with scissors and forceps, aspirate with PBS using a pipette and flush the blood adhering to the limbs.
- Add 5 mL of penicillin/streptomycin solution into 500 mL of DMEM and mix well. Take a 50 mL sterile centrifuge tube, add 5 mL of FBS and 45 mL of DMEM medium, and mix thoroughly to obtain a 10% FBS DMEM containing 1% penicillin/streptomycin solution. Add 2 mL of the above culture medium into a 5 mL tube.
- Transfer the limb bones into the 5 mL tube. Use scissors to cut the limb bones in the tube into small pieces (1-3 mm) and mix the homogenate to re-suspend the bone marrow cells into the culture medium. Stand for 5 min until the tissue fragments settled to the bottom of the tube.
- Add 10 mL of 10% FBS DMEM into a 100 mm culture dish, and then transfer the supernatant into the culture dish. When aspirating the supernatant, avoid transferring the tissue debris into the culture dish.
- Incubate at 37 °C and 5% CO2 for approximately 24 h. After this incubation time, the majority of mesenchymal stem cells will adhere to the culture dish wall and grow slowly, while most BMMs will still be suspended in the culture medium.
- Transfer the cell suspension in the 100 mm culture dish to a new 25 cm2 flask and continue to cultivate the cells at 37 °C and 5% CO2 for an additional 24 h. BMMs will adhere to the flask wall at 24-48 h of culture. Remove the old medium carefully and replace with fresh medium after BMMs have adhered to the flask wall.
- Sub-culture when the cells in the flask reached 80%-90% confluency.
NOTE: All the cells were cultured at 37 °C and 5% CO2. During culture, the cell morphology was gradually unified. Cells were large, irregularly shaped, radially growing and attached to the flask in a form of disc, where multiple nuclei could be observed.
2. FACS staining of the cell
- Trypsin digest using 1-2 mL of commercial trypsin at 37 °C and 5% CO2 for 5 min and count secondary adherent cells to ensure a final cell count of 100,000/sample (count cells with Neubauer hemocytometer).
- Divide the cells into three groups (500 µL of PBS contains 100,000 cells): (1) the blank control group containing unstained cells; (2) the isotype control group; and (3) the experimental group (CD11b/c staining).
- Incubate primary antibodies (no antibody for the blank control group; anti-CD11 isotype control for the isotype control group, 0.6 µL of antibody/sample, 1 µg/sample; anti-CD11b/c for the experimental group, 1 µL of antibody/sample, 1 µg/sample) on ice for 30 min.
- Centrifuge at 300-350 x g for 5 min, discard the supernatant, and resuspend the cells with 500 µL of PBS. Repeat the above procedure once to ensure that the unbound primary antibodies are washed away.
- Incubate the corresponding secondary antibody (no antibody for the blank control group; goat anti-rabbit IgG for the isotype control and the experimental groups, 0.25 µL antibody/sample, 1:2,000) on ice in the dark for 30 min.
- Centrifuge at 300-350 x g for 5 min, discard the supernatant, and resuspend the cells with 500 µL of PBS. Repeat the above procedure once to ensure that the unbound second antibodies are washed away.
- Detect the CD11b/c positive cells by flow cytometry (10,000 cells/sample) and analyze the data by software (e.g., FlowJo 7.5).
NOTE: To obtain the final percentage of CD11b/c+ cells, the following formula was used: Percentage of CD11b / c + cells in the experimental group – the percentage of CD11 + cells in the isotype control group.
3. Wright-Giemsa staining
- Seed the adherent secondary cells that have been passaged three times onto 35 mm2 cell climbing sheets (1 × 106 cells/well) and culture at 37 °C and 5% CO2 for 24 h.
- Discard the culture medium and wash thrice with PBS.
- Add Wright-Giemsa dye solution (0.5 mL-0.8 mL) to the cell climbing sheet for 1 min.
- Mix the dye with distilled water (0.5 mL-0.8 mL) with a cotton swab, and stand for 10 min.
- Wash the dye solution with distilled water, and then dry for 1-3 min. Observe under a microscope.
4. TRAP staining
- Seed the adherent secondary cells that have been passaged three times onto 35 mm2 cell climbing sheets (1 × 106 cells/well) and culture at 37 °C and 5% CO2 for 24 h.
- Replace the old medium with the 10% FBS DMEM or osteoclast induction medium supplemented with 50 ng/mL receptor activator of nuclear factor-κB ligand (RANKL) and 30 ng/mL of macrophage colony-stimulating factor (M-CSF), and culture at 37 °C and 5% CO2 for an additional 7 days.
- Stain the cells with the TRAP staining kit according to manufacturer's protocol and observe under a microscope.
NOTE: TRAP-positive cells were defined as osteoclasts, which were purple under a light microscope. The number of TRAP-positive cells was measured using ImageJ software.
5. Western blot
- Seed the adherent secondary cells that have been passaged three times on 35 mm2 cell climbing sheets (1 × 106 cells/well) and culture for 24 h.
- Replace the old medium with fresh 10% FBS DMEM or osteoclast induction medium (50 ng/mL of RANKL and 30 ng/mL of M-CSF) and culture for an additional 7 days at 37 °C and 5% CO2.
- Extract total cellular proteins with RIPA buffer, separate by 10% SDS-PAGE, and transfer to polyvinylidene fluoride membranes18,19.
- Block with 5% skimmilk powder (25 mL) for 2 h and wash three times, for 10 min each time, with TBS-Tween 20 (TBST).
- Incubate primary antibodies at 4 °C overnight (anti-β-actin, anti-TRAP, and anti-cathepsin K; all primary antibodies were diluted 1:1,000, 10 mL diluted antibody/band), and wash thrice with TBST.
- Incubate the secondary antibody (goat anti-rabbit IgG, 1:2,000 dilution, 10 mL diluted antibody/band) at room temperature for 2 h, and wash thrice with TBST. Visualize the protein bands using a developing solution.
NOTE: The expression levels of the above proteins were normalized to β-actin.
Representative Results
The secondary adherent cell population was stable and uniform. With the continuous cell proliferation, the majority of cells became larger, with irregular shape and grew into a radial adherent disk (Figure 2C,D). Flow cytometry showed that the percentage of cells expressing CD11b/c, a molecular marker on the surface of monocyte-macrophage lineage cells, was approximately 37.94% (Figure 2A,B). To further verify that the CD11b/c positive cells were monocyte-macrophage lineage cells, the differentiation of secondary adherent cells into osteoclasts was induced following cell treatment with RANKL and M-CSF. The TRAP staining results showed that compared with the control group, the number of intracellular purple-red granules was significantly increased in cells induced with RANKL and M-CSF (Figure 2E,F). Furthermore, the expression levels of the osteoclast-specific proteins TRAP and cathepsin K were significantly increased (Figure 2G,H).
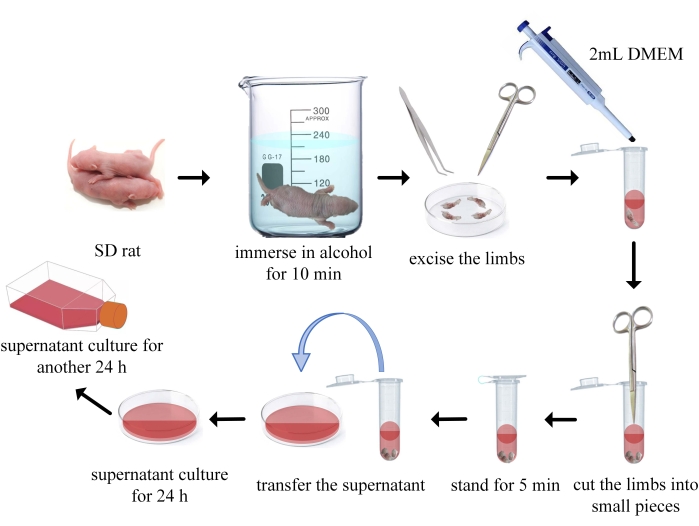
Figure 1: Flow chart of the secondary adherent cell extraction method. Please click here to view a larger version of this figure.
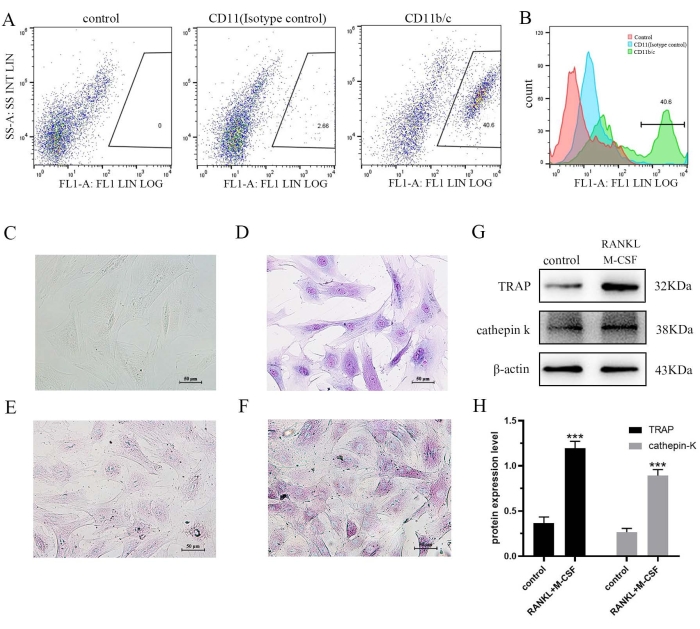
Figure 2: (A–B) After three generations of stable culture of secondary adherent cells, CD11b/c positive cells were detected by flow cytometry. (C–D) The morphology of secondary adherent cells after three generations is shown (C for white light, D for Wright-Giemsa staining). (E–F) TRAP staining was used to detect the effect of RANKL and M-CSF on osteoclast formation (E for control; F for RANKL and M-CSF). (G–H) The cells were treated with RANKL and M-CSF for 7 days and the expression levels of TRAP and cathepsin K were determined by western blot analysis. Data are expressed as the mean ± SD of three independent experiments. ***P < 0.001 vs. the control group. TRAP, tartrate resistant acid phosphatase; RANKL, receptor activator of nuclear factor-κB ligand; M-CSF, macrophage colony-stimulating factor. Please click here to view a larger version of this figure.
Table 1: Method for extracting macrophages Please click here to download this Table.
Discussion
Osteoclasts are one of the most significant cell types involved in the occurrence and development of bone diseases, as well as one of the primary objects of bone disease research20. Monocyte/macrophages can differentiate into osteoclasts. Since mononuclear macrophages (RAW264.7 cells) are too expensive to buy and are easily activated during culture, it is difficult to perform in vitro differentiation experiments using this cell line. Although several methods have been developed for extracting monocytes/macrophages from bone marrow, including density gradient centrifugation, collagenase digestion, microsphere enrichment and flow cytometry (Table 1), these methods are time-consuming, while they require high sample volumes and costly equipment. Therefore, the current study aimed to propose a simple and rapid method to extract monocytec/macrophages from bone marrow samples.
There are many cell populations in bone marrow, including BMSCs, endothelial cells and immune cells in addition to BMMs. As we all know that BMSCs can differentiate into osteoblasts, and this process will affect the differentiation of BMMs into osteoclasts21. Uniform and stable BMMs are significant factors for inducing osteoclast differentiation in vitro, so it is particularly important to consider these limiting factors when isolating and extracting BMMs. At the same time, the ability of BMMs to differentiate into osteoclasts will also weaken during the continuous passage. Therefore, it is challenging to isolate BMMs from bone marrow and successfully induce them into osteoclasts.
We found that there are differences in the adherence time of adherent cells in the bone marrow. More specifically, BMSCs basically adhered to the culture dish wall during 0-24 h of culture, while BMMs begin to adhere to the culture dish wall after 24 h. Based on the above finding, the secondary adherence method was selected to isolate BMMs. Suckling SD rats (age, 1-10 days old) were selected to extract BMMs, since the BMMs of young rats exhibit a stronger differentiation ability. The bone marrow cells of SD rats were collected, and after culturing for 24 h, the cell suspension was transferred and cultured for another 24 h. The collected secondary adherent cells contained a large number of BMMs. Following culture, the secondary adherent cells were gradually purified and became stable and uniform. The flow cytometry results demonstrated that the percentage of CD11b/c positive cells reached approximately 37.94%. Furthermore, TRAP staining and western blot analysis showed that the extracted BMMs could differentiate into osteoclasts. This finding was consistent with a previous study also suggesting that BMMs extracted by the secondary adherence method could be successfully differentiated into osteoclasts22.
The secondary adherence method can be used to simply and quickly extract mononuclear macrophages from the bone marrow, without requiring high-tech lab equipment. At the same time, this method is suitable for isolating cells from a small bone marrow sample, thus overcoming the cumbersome and time-consuming difficulties observed in the previous methods commonly used to extract mononuclear macrophages. Overall, BMMs extracted by the secondary adherence method could be successfully differentiated into osteoclasts in vitro, thus providing a stable cell model for the in vitro study of osteoclasts.
Declarações
The authors have nothing to disclose.
Acknowledgements
This work was supported by the Natural Science Foundation of Zhejiang Province (grant no. LY19H060001) and the Zhejiang Traditional Chinese Medicine Science and Technology Plan Project (no. 2022ZB093).
Materials
| 35 mm2 cell climbing slices | NEST Biotechnology | 80102 | |
| Anti-cathepsin K | Abcam | ab19027 | 1:1,000 |
| Anti-CD11 isotype control | Abcam | ab172730 | 1 μg/test,1.675 mg/Ml |
| Anti-CD11b/c | Absin | abs124232 | 1μg/test, 1 mg/mL |
| Anti-TRAP | Abcam | ab191406 | 1:1,000 |
| Anti-β-actin | Beyotime | AF5003 | 1:1,000 |
| Cell climbing slices | NEST Biotechnology | 80102 | |
| Cell culture dish | corning | 430167 | |
| Cell culture flask | corning | 430168 | |
| Dulbecco's modified eagle medium (DMEM) | Gibco | C11995500BT | |
| Fetal bovine serum (FBS) | Gibco | 10099141C | |
| Goat anti-rabbit IgG | Abcam | ab150077 | for IF, 1:2,000 |
| goat anti-rabbit IgG | Abcam | ab6721 | for WB, 1:2,000 |
| M-CSF | Pepro tech | 400-28 | |
| PBS | Biosharp | BL302A | |
| RANKL | Pepro tech | 400-30 | |
| SD rat | Shanghai SLAC Laboratory Animal Co, Ltd | 1-10 days old | |
| SDS-PAGE gel preparation kit | Solarbio | P1200 | |
| TRAP/ALP Staining Kit | Wako | 294-67001 | |
| Trypsin-EDTA solution | Biosharp | BL512A | |
| Wright-Giemsa solution | Keygen Biotech | KGA225-1 |
Referências
- Jakubzick, C. V., Randolph, G. J., Henson, P. M. Monocyte differentiation and antigen-presenting functions. Nature Reviews. Immunology. 17 (6), 349-362 (2017).
- Locati, M., Curtale, G., Mantovani, A. Diversity, mechanisms, and significance of macrophage plasticity. Annual Review of Pathology. 15 (1), 123-147 (2020).
- Boyle, W. J., Simonet, W. S., Lacey, D. L. Osteoclast differentiation and activation. Nature. 423 (6937), 337-342 (2003).
- Ono, T., Nakashima, T. Recent advances in osteoclast biology. Histochemistry and Cell Biology. 149 (4), 325-341 (2018).
- Zhou, X., et al. Wnt/ß-catenin-mediated p53 suppression is indispensable for osteogenesis of mesenchymal progenitor cells. Cell Death & Disease. 12 (6), 521-534 (2021).
- Yu, Q., Zhao, B., He, Q., Zhang, Y., Peng, X. B. microRNA-206 is required for osteoarthritis development through its effect on apoptosis and autophagy of articular chondrocytes via modulating the phosphoinositide 3-kinase/protein kinase B-mTOR pathway by targeting insulin-like growth factor-1. Journal of Cellular Biochemistry. 120 (4), 5287-5303 (2019).
- Li, Z., MacDougald, O. A. Preclinical models for investigating how bone marrow adipocytes influence bone and hematopoietic cellularity. Best Practice & Research. Clinical Endocrinology & Metabolism. 35 (4), 101547-101560 (2021).
- Horowitz, M. C., et al. marrow adipocytes. Adipocyte. 6 (3), 193-204 (2017).
- Maridas, D. E., Rendina-Ruedy, E., Le, P. T., Rosen, C. J. Isolation, culture, and differentiation of bone marrow stromal cells and osteoclast progenitors frommice. Journal of Visualized Experiments. (131), e56750 (2018).
- Schyns, J., et al. Non-classical tissue monocytes and two functionally distinct populations of interstitial macrophages populate the mouse lung. Nature Communications. 10 (1), 3964-3980 (2019).
- Atif, S. M., Gibbings, S. L., Jakubzick, C. V. Isolation and identification of interstitial macrophages from the lungs using different digestion enzymes and staining strategies. Methods in Molecular Biology. 1784, 69-76 (2018).
- Scheven, B. A., Milne, J. S., Robins, S. P. A sequential culture approach to study osteoclast differentiation from nonadherent porcine bone marrow cells. In Vitro Cellular & Developmental Biology. Animal. 34 (7), 568-577 (1998).
- Bradley, E. W., Oursler, M. J. Osteoclast culture and resorption assays. Methods in Molecular Biology. , 19-35 (2008).
- Yu, Y. R., et al. Flow cytometric analysis of myeloid cells in human blood, bronchoalveolar lavage, and lung tissues. American Journal of Respiratory Cell and Molecular Biology. 54 (1), 13-24 (2016).
- Gibbings, S. L., Jakubzick, C. V. A consistent method to identify and isolate mononuclear phagocytes from human lung and lymph nodes. Methods in Molecular Biology. 1799, 381-395 (2018).
- Jacquin, C., Gran, D. E., Lee, S. K., Lorenzo, J. A., Aguila, H. L. Identification of multiple osteoclast precursor populations in murine bone marrow. Journal of Bone and Mineral Research. 21 (1), 67-77 (2006).
- Gibbings, S. L., et al. Three unique interstitial macrophages in the murine lung at steady state. American Journal of Respiratory Cell and Molecular Biology. 57 (1), 66-76 (2017).
- Higashi, S. L., et al. Ultra-high-speed western blot using immunoreaction enhancing technology. Journal of Visualized Experiments. (163), e61657 (2020).
- Gallagher, S., Chakavarti, D. Immunoblot analysis. Journal of Visualized Experiments. 20 (16), 759 (2008).
- Yin, Z., et al. Glycyrrhizic acid suppresses osteoclast differentiation and postmenopausal osteoporosis by modulating the NF-κB, ERK, and JNK signaling pathways. European Journal of Pharmocology. 859, 172550 (2019).
- Liu, F., et al. LRRc17 controls BMSC senescence via mitophagy and inhibits the therapeutic effect of BMSCs on ovariectomy-induced bone loss. Redox Biology. , (2021).
- Jin, X., et al. Thioacetamide promotes osteoclast transformation of bone marrow macrophages by influencing PI3K/AKT pathways. Journal of Orthopedic Surgery and Research. 17 (1), 53-63 (2022).

