Development and Evaluation of a Rat Model of Full-Thickness Cartilage Defects
Summary
This protocol establishes a full-thickness cartilage defects (FTCD) model by drilling holes in the femoral trochlear groove of rats and measuring the subsequent pain behavior and histopathological changes.
Abstract
Cartilage defects of the knee joint caused by trauma are a common sports joint injury in the clinic, and these defects result in joint pain, impaired movement, and eventually, knee osteoarthritis (kOA). However, there is little effective treatment for cartilage defects or even kOA. Animal models are important for developing therapeutic drugs, but the existing models for cartilage defects are unsatisfactory. This work established a full-thickness cartilage defects (FTCD) model by drilling holes in the femoral trochlear groove of rats, and the subsequent pain behavior and histopathological changes were used as readout experiments. After surgery, the mechanical withdrawal threshold was decreased, chondrocytes at the injured site were lost, matrix metalloproteinase MMP13 expression was increased, and type II collagen expression decreased, consistent with the pathological changes observed in human cartilage defects. This methodology is easy and simple to perform and enables gross observation immediately after the injury. Furthermore, this model can successfully mimic clinical cartilage defects, thus providing a platform for studying the pathological process of cartilage defects and developing corresponding therapeutic drugs.
Introduction
Articular cartilage is a highly differentiated and dense tissue consisting of chondrocytes and extracellular matrix1. The surface layer of articular cartilage is a form of hyaline cartilage, which has a smooth surface, low friction, good strength and elasticity, and excellent mechanical stress tolerance2. The extracellular matrix comprises collagen proteoglycan and water, and type II collagen is the main structural component of the collagen, as it accounts for about 90% of the total collagen3. As no blood vessels or nerves exist in cartilage tissue, it lacks the ability to self-repair after injury4. Therefore, cartilage defects caused by trauma have always been an intractable joint disease in clinics; additionally, this joint disease tends to strike young people, and the global incidence is on the rise5,6. The knee joint is the most common site of cartilage defects, and defects here are accompanied by joint pain, joint dysfunction, and articular cartilage degeneration, eventually leading to knee osteoarthritis (kOA)7. Cartilage defects of the knee joint bring economic and physiological burdens to patients and seriously affect the patients’ quality of life8. This disease poses a major and urgent clinical challenge with no imminent solutions. Currently, surgery is the mainstay of treatment for cartilage defects, but its long-term outcome remains unsatisfactory9.
Clinical cartilage defects eventually lead to kOA, and, thus, kOA animal models are commonly used for the pathological study of cartilage defects and drug development. The establishment of animal models is important for understanding the pathophysiological process of cartilage defect repair, which can be used to observe the cartilage regeneration and the alteration between fibrocartilage and hyaline cartilage10. However, commonly used kOA animal models, such as surgical models of anterior cruciate ligament transection (ACLT), destabilization of the medial meniscus (DMM), ovariectomy (OVX), and Hulth, usually need long-term modeling and only allow for pathological and pain evaluations, which poses limitations to the efficiency of drug development11. Besides the surgical models, chemical models, such as monoiodoacetate (MIA) and papain injection, also result in cartilage defects, but the degree of the defect cannot be well managed, and the conditions are far from the clinical reality11. Collision is another approach to model cartilage defects in larger animals, but this method depends on the use of specific instruments and is rarely applied12.
In summary, the existing kOA models are not ideal for studying the pathogenesis of cartilage defects or developing new drugs, and a specific and standardized model for cartilage defects is needed. This study established a full-thickness cartilage defects (FTCD) model by drilling holes in the femoral trochlear groove in rats. Gross observation, pain behavior tests, and histopathological analysis were conducted for model evaluation. Unlike other animal models of kOA, this model has little effect on the rats’ general condition. This modeling approach is accessible, can be well managed, and supports the understanding of progression from cartilage defects to kOA and the development of effective therapeutics. This model can also be used for testing therapies that prevent kOA by healing defects in pre-osteoarthritic joints.
Protocol
The animal experiments were approved by the Medical Standards and Ethics Committee of Zhejiang University of Traditional Chinese Medicine, which conforms to the China legislation on the use and care of laboratory animals. In the present study, 6 week old male Sprague-Dawley (SD) rats weighing 150-180 g were used. The animals were obtained from a commercial source (see the Table of Materials).
1. Establishment of a full-thickness cartilage defects model in rats
- After 1 week of acclimatization to the new environment, randomly and equally divide the rats into two groups (n = 8 rats/group). The rats in the sham group will have the sham surgery, while the rats in the model group will have the experimental surgery involving drilling holes in the femoral trochlear groove.
NOTE: Each cage must be covered with sterile corncob paddings (see the Table of Materials) to protect the rats' toes. - Anesthetize the rats by an intraperitoneal injection (i.p.) of pentobarbital sodium (40 mg/kg). Then, gently press the toes of the rats to confirm adequate anesthesia. Use a vet ointment on the rats' eyes to prevent dryness while under anesthesia.
NOTE: The animal surgery needs to be performed in a dedicated operating room using autoclaved surgical instruments. The operators must wear clean laboratory coats, facemasks, head covers, and sterile gloves during the surgery. Place sterile pads over the surgical area and sterilize all equipment before use. Provide thermal support throughout the procedure. - Place the rat on the operating table in a supine position, shave the left and right hind limbs, and clean the knee joint area with surgical soap, followed by alternating antiseptic povidone-iodine solution and alcohol three times under sterile conditions. Place a sterile drape over the rat, and expose the disinfected knee joint only.
- Make a 1 cm incision with a scalpel blade (number 11) in the middle of the rat knee joint from top to bottom, and cut the joint capsule and quadriceps femoris tendon along the medial edge of the patella after the superficial dissection.
NOTE: The quadriceps femoris tendon is attached to the patella and to the femoral condyle during the flexion of the knee joint13. The groove seen in the joint capsule is the femoral trochlear groove, and the distal femoral condyle forms the medial and lateral condyles. - Turn the patella to the outside, and flex the tibia and fibula at a 90° angle to fully expose the trochlea of the femoral condyle. Use a 1.6 mm diameter circular drill bit (see the Table of Materials) vertical to the cartilage surface at 4,000 rpm for 10 s to make one full-thickness cartilage defect in the femoral trochlear groove with a depth of 0.1 mm.
NOTE: Use saline intermittently to minimize thermal trauma to the surrounding bone tissue during the drilling procedure. - Wipe the surgical site with cotton balls soaked in a 0.9% saline solution, replace the patella, keep the knee in an extension position, and suture the incision layer by layer with nonabsorbable 4-0 sutures (see Table of Materials).
- Place the animals on heating pads with sternal recumbency, monitor them until they wake up, and then return them to their cages. Inject buprenorphine (0.05 mg/kg) subcutaneously every 8 h three times after the operation for pain relief.
- Test the pain-related behavior of all the rats at 3 days, 10 days, and 17 days after the surgery, as described in section 2.
2. Mechanical withdrawal threshold (MWT)
NOTE: The MWT of the bilateral posterior plantar of rats was measured by the classical von Frey filament pain measurement method14.
- Place the rat in a single plastic chamber (17 cm x 11 cm x 13 cm) on a wire mesh platform (see the Table of Materials), and place the wire mesh base 50 cm above a table. Measure the MWT after 30 min of adaptation.
- Press the von Frey filament (see the Table of Materials) perpendicularly on the plantar surface of each rat's hind paw, and bend the brush for about 2 s, avoiding the thickest part of the center of the hind paw.
- Gradually increase the stimulus weight from the lowest 4 g until a positive response (paw withdrawal or paw licking) occurs.
NOTE: The interval between each stimulation should be more than 1 min. The MWT is defined as three positive responses in five stimulations; record the stimulus weight in grams. - Calculate the mean values for the sham and model groups according to the recorded minimal stimulus weight in grams.
3. Histopathological and immunohistochemical analysis
- At 17 and 56 days after surgery, anesthetize the rats with a 40 mg/kg pentobarbital sodium (i.p.), and sacrifice all the rats by withdrawing blood from the heart. Isolate the knees by cutting the bone at the mid-femur and mid-tibia, and dissect the surrounding muscle tissue. Remove the knee joints for histological analysis.
- Fix the knee joints in 20 mL of 10% paraformaldehyde solution for 48 h at room temperature, and then decalcify them with 20 mL of 10% EDTA solution in an orbital shaker for 8 weeks at 4 °C. Change the EDTA solution every day.
- Trim the knee joint to fit the size of the embedding box. Embed the dehydrated knee joints in 100% paraffin15.
- Place the paraffin-embedded knee joints on the holder of a microtome, adjust the angle, and trim the paraffin sample with a blade until the surface is flat.
- Set the thickness of the paraffin slices to 3 µm, and flatten the slices in a water bath at 40 °C.
- Stick the slices on glass slides, place them in a 45 °C baking machine (see the Table of Materials) until they are dry, and store them at room temperature.
- Dewaxing and rehydration: Dewax the slices in an oven at 60 °C for 4 h, and then place the slices successively in 100% xylene (three times), 100% ethanol (two times), 95% ethanol, 80% ethanol, and 75% ethanol for 5 min each time.
- Hematoxylin and eosin (H&E) staining
- Dewax, rehydrate, and wash the slices with double-distilled water for 2 min.
- Stain the slices with 0.5% hematoxylin (see Table of Materials) for 3 min, and wash the slices with double-distilled water until there is no hematoxylin residue on the slice surfaces.
- Immerse the slices in 1% hydrochloric acid alcohol for 3 s, and wash the slices with double-distilled water for 2 min.
- Soak the slices in 1% ammonia water for 10 s, and wash the slices with double-distilled water for 2 min.
- Stain the slices with eosin (see Table of Materials) for 1 min, and wash the slices with double-distilled water until there is no eosin residue on the slice surfaces.
- Immerse the slices in 95% ethanol, 100% ethanol, and 100% xylene (three times) successively for 1 min each time.
- Add a drop of neutral resin (see Table of Materials) to each slice, and seal it with a coverslip.
- Safranin O/Fast Freen (SO) staining
- Dewax, rehydrate, and wash the slices with double-distilled water for 2 min.
- Stain the slices with 0.05% Fast Green (see the Table of Materials) for 3 min, and wash the slices with double-distilled water until there is no Fast Green residue on the surface of the slices.
- Immerse the slices in 1% acetic acid solution for 10 s, and wash the slices with double-distilled water for 2 min.
- Stain the slices with 2.5% SO (see Table of Materials) for 2 min, and then wash the slices with double-distilled water until there is no SO residue on the slice surfaces.
- Immerse the slices in 95% ethanol, 100% ethanol, and 100% xylene (three times) successively for 1 min each time.
- Add a drop of neutral resin to each slice, and seal it with a coverslip.
- Toluidine blue (TB) staining
- Dewax, rehydrate, and wash the slices with double-distilled water for 2 min.
- Immerse the slices in 1% TB solution (see the Table of Materials) for 2 min, and wash the slices with double-distilled water until there is no toluidine blue residue on the surface of the slices.
- Immerse the slices in 95% ethanol, 100% ethanol, and 100% xylene (three times) successively for 1 min each time.
- Add a drop of neutral resin to each slice, and seal it with a coverslip.
- Masson staining
- Dewax, rehydrate, and wash the slices with double-distilled water for 2 min.
- Add Bouin solution (see the Table of Materials) dropwise to the slices, stain them at 37 °C for 2 h, and wash them with double-distilled water until the yellow color on the surface of the slices disappears.
- Stain the slices with celestite blue (see the Table of Materials) for 3 min, and wash the slices with double-distilled water until there is no celestite blue residue on the slice surfaces.
- Stain the slices with hematoxylin for 3 min, and wash the slices with double-distilled water until there is no hematoxylin residue on the slice surfaces.
- Immerse the slices in acidic ethanol for 5 s, and wash the slices with double-distilled water for 2 min.
- Stain the slices with Ponceau fuchsin (see the Table of Materials) for 10 min, and wash the slices with double-distilled water until there is no Ponceau fuchsin residue on the slice surfaces.
- Immerse the slices in phosphomolybdic acid (see the Table of Materials) for 10 min, then immerse them in TB solution for 5 min, and wash the slices with double-distilled water until there is no TB residue on the surface of the slices.
- Immerse the slices in a weak acid solution for 2 min, and wash the slices with double-distilled water for 2 min.
- Immerse the slices in 95% ethanol, 100% ethanol,and 100% xylene (three times) successively for 1 min each time.
- Add a drop of neutral resin to each slice, and seal it with a coverslip.
- Observe all the slices under a microscope in a double-blind setting to determine the degree of articular cartilage degeneration according to Mankin's scoring system16.
- Immunohistochemistry
- Dewax and rehydrate the slices routinely, and wash the slices with PBS for 2 min.
- Immerse the slices in sodium citrate solution, and place the slices in an oven at 60 °C for 4 h to repair the antigen. Wash the slices with PBS three times for 3 min each.
- Immerse the slices in a 0.3% Triton X-100 solution for 10 min, and wash the slices twice with PBS for 3 min each time.
- Block the endogenous peroxidase activity by adding a 3% H2O2 solution in methanol at room temperature for 30 min. Wash the slices twice with PBS for 3 min each time.
- Incubate the sections with 5% goat serum in PBS for 30 min at room temperature to block any non-specific binding. Wash the slices twice with PBS for 3 min each time.
- Add 100 µL of PBS-diluted primary antibodies (anti-col1, 1:50; anti-col3, 1:50; anti-col2, 1:100; and anti-MMP13, 1:100; see the Table of Materials) to each slice, and incubate them overnight at 4 °C. Wash the slices twice with PBS for 3 min each time.
- Incubate each slice with 100 µL of PBS-diluted (1:100) secondary antibody (goat anti-rabbit or goat anti-mouse, see the Table of Materials) at room temperature for 20 min. Wash the slices twice with PBS for 3 min each time.
- Add 100 µL of 3, 3 '-diaminobenzidine (DAB, see the Table of Materials) working solution to each slice.
- Observe and record the appearance time of a brown color under a microscope; the chromogenic reaction turns the epitope sites brown17. Treat the rest of the samples with the same recorded reaction time.
- After the slices turn brown, wash the slices twice with double-distilled water for 3 min each time.
- Re-stain the slices with hematoxylin for 1 min, and wash the slices with double-distilled water for 2 min.
- Immerse the slices in hydrochloric acid for 3 s, and wash the slices with double-distilled water for 2 min.
- Soak the slices in 1% ammonia water for 10 s, and wash the slices with double-distilled water for 2 min.
- Immerse the slices in 95% ethanol, 100% ethanol, and 100% xylene (three times) successively for 1 min each time.
- Add a drop of neutral resin to each slice, and seal it with a coverslip.
Representative Results
In this work, a rat model of FTCD was established by drilling holes in the femoral trochlear groove and detecting the subsequent pain behavior and histopathological changes. As shown in Figure 1, 3 days after modeling, compared with the sham group, the MWT of rats in the model group was significantly reduced, suggesting hyperalgesia caused by the FTCD. At 17 days after modeling, the mechanical withdrawal threshold of the rats in the model group remained at a low level, indicating that the pain sensitization could last at least 17 days. The histopathological staining results showed that, in the sham group, the structure of the articular cartilage was clear, the cartilage surface was intact, the chondrocytes were evenly distributed, and type II collagen was highly expressed. On the contrary, in the model group, the cartilage surface formed a depression, the chondrocytes were lost, the expression of matrix metalloproteinase MMP13 increased, and the expression of type II collagen decreased (Figure 2 and Figure 3).

Figure 1: Development of MWT after cartilage defects. Mechanical withdrawal thresholds of the hind paws were assessed after cartilage defects were induced. n = 8 rats/group. Values are presented as mean ± SEM. **P < 0.01 versus sham group, ***P < 0.001 versus sham group. A Student's t-test was performed. Please click here to view a larger version of this figure.
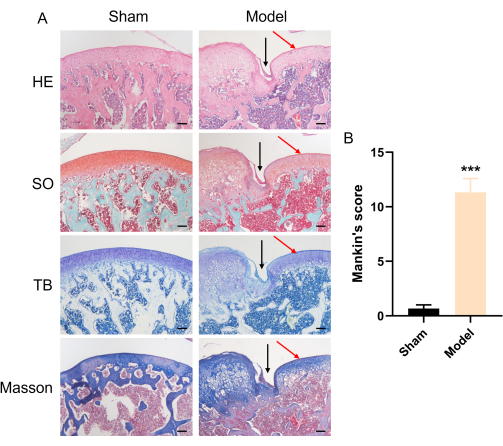
Figure 2: Histopathological observation (HE, SO, TB, and Masson staining) and Mankinʹs scoring of the rat knee joints on day 17 after the cartilage defects treatment. (A) Representative histological pictures of an FTCD rat. The black arrows indicate the cartilage defects. Scale bar = 200 µm. (B) Statistical analysis of the osteoarthritis scorings in the sham and model groups. n = 6 rats/group. Values are presented as mean ± SEM. ***P < 0.001 versus sham group. A Student's t-test was performed. Please click here to view a larger version of this figure.
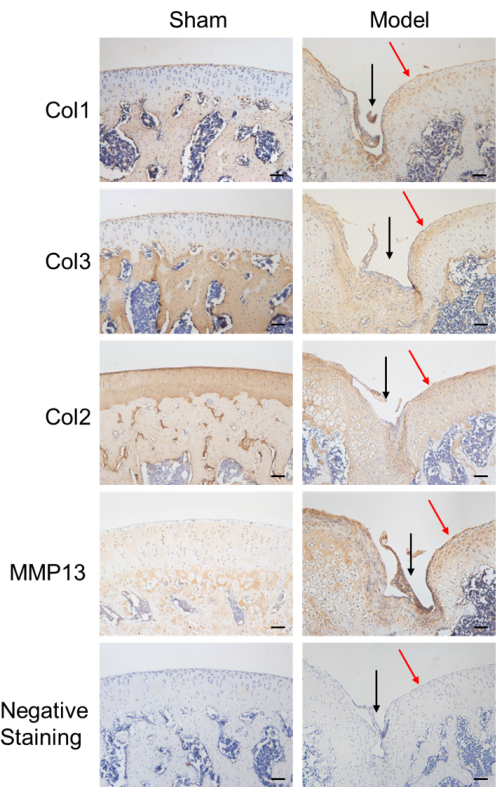
Figure 3: Immunohistochemical observation of the expression of Col1, Col3, Col2, and MMP13 and negative staining in the rat cartilage on day 17. Representative histological pictures of an FTCD rat. The black arrows indicate the cartilage defects. Scale bar = 100 µm. Please click here to view a larger version of this figure.
Supplementary Figure 1: Representative pictures of inducing full-thickness cartilage defects by drilling in the rat femoral trochlear groove. (A) Sham rat. (B) Model rat. Please click here to download this File.
Supplementary Figure 2: Histological evaluation showing complete filling of the full-thickness cartilage defects in rats. (A) A representative image on day 17. (B) A representative image on day 56. Scale bar = 200 µm. Please click here to download this File.
Discussion
This study describes an animal model for mimicking clinical cartilage defects by drilling holes in the femoral trochlear groove of rats (Supplementary Figure 1). After cartilage injury, the excitability or responsiveness of peripheral nociceptors is enhanced, which can result in a decrease in the pain threshold and the enhancement of responsiveness to stimulation18. In preclinical studies, the modeling of cartilage defects in different species of animals has always caused pain19. Clinical research has also shown that the pain visual analog scale (VAS) scores of patients with cartilage injuries are significantly lower than those of healthy individuals20. We used the FTCD model to test the effect of the FTCD treatment, and the results showed that the decrease in the MWT was not transient, and the MWT did not recover quickly within a short period of time. After a period of treatment, the MWT in model group was still significant, while the treatment group was relieved (data not shown). Clinical efficacy is generally assessed based on a 1 month course of treatment, so even if recovery occurs after a few months, it does not affect the experimental application of this model. Moreover, pathological staining and immunohistochemistry were applied to observe cartilage surface defects and demonstrate the establishment of FTCD.
This method to model FTCD has the following advantages: (1) the easy and simple operation; (2) the short modeling time; (3) the high success ratio; and (4) the presence of visible progression via gross observation. Unlike other animal models, this model can be standardized. The drilling depth and diameter of the FTCD model are easy to control, which is beneficial for standardizing the FTCD model and increases its repeatability. Secondly, the diameter of the drilling hole is a key factor that determines the repair efficiency. Osteochondral defects with a diameter of 1.4 mm can self-recover spontaneously, leading to failure in the appropriate evaluation of therapeutic treatments21. To overcome these shortcomings and achieve standardization, preliminary experiments were conducted, and it was determined that the cartilage defects would not spontaneously repair up to 17 days after surgery if the FTCD surgery was performed on the articular cartilage surface with drill holes of 1.6 mm in diameter. Over time, the FTCD caused by drilling shows cartilage repair, and the defective cartilage is largely repaired by 8 weeks post-surgery (Supplementary Figure 2). In terms of applications, this model could not only be used for studying the cartilage defects caused by kOA but also for studying traumatic cartilage defects, namely post-traumatic osteoarthritis22. Self-repaired cartilage always forms fibrocartilage rather than hyaline cartilage at the injured site, and this model might also be suitable for studying the pathogenesis and treatment of cartilage fibrosis23.
In terms of the limitations of this model, immature rats were chosen, as cartilage defects caused by trauma in clinical practice tend to occur in young people. However, in immature rats at the skeletal developmental stage, the cartilage is thinner than that of mature rats, which may affect the results of the experiment24. Previous research has shown that the ability of stem cells to regenerate after cartilage damage is reduced in adult mice compared to juvenile mice25. We selected 6 week old rats for the experiment, and these rats could also be used to observe the mechanisms of stem cell repair; additionally, the therapeutic effects in 6 week old rats are more pronounced than in adult rats (data not shown). We also need to model FTCD in older rats, and it could be speculated that repair may be slower in aged rats due to a decreased stem cell regenerative capacity. Research has shown that the articular cartilage surrounding osteochondral defects possesses catabolic activity, and the expression of IL-1β and FGF2 and a disturbance in the FGFr1/FGFr3 balance are important in initiating the process of early osteoarthritic disease21. However, the FTCD model still has limitations in evaluating pre-osteoarthritic defect repair. Another limitation of this study was the lack of measurement of MWTs after 17 days of modeling.
In conclusion, this model would be an ideal and standardized animal model for mimicking cartilage defects by drilling holes in the femoral trochlear groove of rats. This model not only mimics the occurrence and development of clinical FTCD but also provides a reliable animal model for evaluating therapeutic treatments against FTCD.
Declarações
The authors have nothing to disclose.
Acknowledgements
This study was supported by the Zhejiang Natural Science Foundation (grant number LQ20H270009), the Natural Science Foundation of China (grant numbers 82074464 and 82104890), the Zhejiang Traditional Chinese Medical Science Foundation (grant numbers 2020ZA039, 2020ZA096, and 2022ZB137) and the Medical Health Science and Technology Project of Zhejiang Provincial Health Commission (grant number 2016KYA196).
Materials
| 3, 3 '-diaminobenzidine | Hangzhou Zhengbo Biotechnology Co., Ltd. | ZLI-9019 | The dye for IHC staining |
| Anti-Collagen III antibody | Novus | NB600-594 | Primary antibody for IHC |
| Anti-Collagen II antibody | Abcam (UK) | 34712 | Primary antibody for IHC |
| Anti-Collagen I antibody | Novus | NB600-408 | Primary antibody for IHC |
| Bouin solution | Shanghai Yuanye Technology Co., Ltd. | R20381 | The dye for Masson staining |
| Celestite blue | Shanghai Yuanye Technology Co., Ltd. | R20381 | The dye for Masson staining |
| Corncob paddings | Xiaohe Technology Co., Ltd | Bedding for animal | |
| Eosin | Sigma-Aldrich | 861006 | The dye for HE staining |
| Fast Green FCF | Sigma-Aldrich | F7252 | The dye for SO staining |
| Goat anti-mouse antibody | ZSGQ-BIO (Beijing, China) | PV-9002 | Secondary antibody for IHC |
| Goat anti-rabbit antibody | ZSGQ-BIO (Beijing, China) | PV-9001 | Secondary antibody for IHC |
| Hematoxylin | Sigma-Aldrich | H3163 | The dye for HE staining |
| Masson | Shanghai Yuanye Technology Co., Ltd. | R20381 | The dye for Masson staining |
| Microdrill | Rwd Life Science Co., Ltd | 78001 | Equipment for surgery |
| MMP13 | Cell Signaling Technology, Inc. (Danvers, MA, USA) | 69926 | Primary antibody for IHC |
| Modular tissue embedding center | Thermo Fisher Scientific (USA) | EC 350 | Produce paraffin blocks |
| Neutral resin | Hangzhou Zhengbo Biotechnology Co., Ltd. | ZLI-9555 | Seal for IHC |
| Nonabsorbable suture | Hangzhou Huawei Medical Supplies Co.,Ltd. | 4-0 | Equipment for surgery |
| Pentobarbital sodium | Hangzhou Zhengbo Biotechnology Co., Ltd. | WBBTN5G | Anesthetized animal |
| phosphomolybdic acid | Shanghai Yuanye Technology Co., Ltd. | R20381 | The dye for Masson staining |
| Ponceau fuchsin | Shanghai Yuanye Technology Co., Ltd. | R20381 | The dye for Masson staining |
| Rotary and Sliding Microtomes | Thermo Fisher Scientific (USA) | HM325 | Precise paraffin sections |
| Safranin-O | Sigma-Aldrich | S2255 | The dye for SO staining |
| Scalpel blade | Shanghai Lianhui Medical Supplies Co., Ltd. | 11 | Equipment for surgery |
| Sodium citrate solution (20x) | Hangzhou Haoke Biotechnology Co., Ltd. | HK1222 | Antigen retrieval for IHC |
| Sprague Dawley (SD) rats | Shanghai Slake Experimental Animal Co., Ltd. | SD | Experimental animal |
| Tissue-Tek VIP 5 Jr | Sakura (Japan) | Vacuum Infiltration Processor | |
| Toluidine Blue | Sigma-Aldrich | 89640 | The dye for TB staining |
| Von Frey filament | UGO Basile (Italy) | 37450-275 | Equipment for MWT assay |
| Wire mesh platform | Shanghai Yuyan Instruments Co.,Ltd. | Equipment for MWT assay |
Referências
- Zhang, Z. Chondrons and the pericellular matrix of chondrocytes. Tissue Engineering. Part B, Reviews. 21 (3), 267-277 (2015).
- Correa, D., Lietman, S. A. Articular cartilage repair: Current needs, methods and research directions. Seminars in Cell & Developmental Biology. 62, 67-77 (2017).
- Kuo, S. M., Wang, Y. J., Weng, C. L., Lu, H. E., Chang, S. J. Influence of alginate on type II collagen fibrillogenesis. Journal of Materials Science. Materials in Medicine. 16 (6), 525-531 (2005).
- Li, M., et al. The immune microenvironment in cartilage injury and repair. Acta Biomaterialia. 140, 23-42 (2022).
- Epanomeritakis, I. E., Lee, E., Lu, V., Khan, W. The use of autologous chondrocyte and mesenchymal stem cell implants for the treatment of focal chondral defects in human knee joints-A systematic review and meta-analysis. International Journal of Molecular Sciences. 23 (7), 4065 (2022).
- Jiang, Y. H., et al. Cross-linking methods of type I collagen-based scaffolds for cartilage tissue engineering. American Journal of Translational Research. 14 (2), 1146-1159 (2022).
- Southworth, T. M., Naveen, N. B., Nwachukwu, B. U., Cole, B. J., Frank, R. M. Orthobiologics for focal articular cartilage defects. Clinics in Sports Medicine. 38 (1), 109-122 (2019).
- Chen, Z., et al. Kindlin-2 promotes chondrogenesis and ameliorates IL-1beta-induced inflammation in chondrocytes cocultured with BMSCs in the direct contact coculture system. Oxidative Medicine and Cellular Longevity. 2022, 3156245 (2022).
- Richter, D. L., Schenck, R. C., Wascher, D. C., Treme, G. Knee articular cartilage repair and restoration techniques: A review of the literature. Sports Health. 8 (2), 153-160 (2016).
- Tessaro, I., et al. Animal models for cartilage repair. Journal of Biological Regulators and Homeostatic Agents. 32 (6), 105-116 (2018).
- Kim, J. E., Song, D. H., Kim, S. H., Jung, Y., Kim, S. J. Development and characterization of various osteoarthritis models for tissue engineering. PLoS One. 13 (3), e0194288 (2018).
- Mrosek, E. H., et al. Subchondral bone trauma causes cartilage matrix degeneration: An immunohistochemical analysis in a canine model. Osteoarthritis and Cartilage. 14 (2), 171-178 (2006).
- Ralphs, J. R., Benjamin, M., Thornett, A. Cell and matrix biology of the suprapatella in the rat: A structural and immunocytochemical study of fibrocartilage in a tendon subject to compression. Anatomical Record. 231 (2), 167-177 (1991).
- Jin, Y., et al. A somatosensory cortex input to the caudal dorsolateral striatum controls comorbid anxiety in persistent pain. Pain. 161 (2), 416-428 (2020).
- Zhanmu, O., Yang, X., Gong, H., Li, X. Paraffin-embedding for large volume bio-tissue. Scientific Reports. 10 (1), 12639 (2020).
- Mankin, H. J., Dorfman, H., Lippiello, L., Zarins, A. Biochemical and metabolic abnormalities in articular cartilage from osteo-arthritic human hips. II. Correlation of morphology with biochemical and metabolic data. Journal of Bone and Joint Surgery. American Volume. 53 (3), 523-537 (1971).
- Levey, A. I., et al. A light and electron microscopic procedure for sequential double antigen localization using diaminobenzidine and benzidine dihydrochloride. Journal of Histochemistry and Cytochemistry. 34 (11), 1449-1457 (1986).
- Pace, M. C., et al. Neurobiology of pain. Journal of Cellular Physiology. 209 (1), 8-12 (2006).
- Zhang, X., et al. Magnetic nanocarriers as a therapeutic drug delivery strategy for promoting pain-related motor functions in a rat model of cartilage transplantation. Journal of Materials Science. Materials in Medicine. 32 (4), 37 (2021).
- Siebold, R., Suezer, F., Schmitt, B., Trattnig, S., Essig, M. Good clinical and MRI outcome after arthroscopic autologous chondrocyte implantation for cartilage repair in the knee. Knee Surgery, Sports Traumatology, Arthroscopy. 26 (3), 831-839 (2018).
- Katagiri, H., Mendes, L. F., Luyten, F. P. Definition of a critical size osteochondral knee defect and its negative effect on the surrounding articular cartilage in the rat. Osteoarthritis and Cartilage. 25 (9), 1531-1540 (2017).
- Farnham, M. S., Larson, R. E., Burris, D. L., Price, C. Effects of mechanical injury on the tribological rehydration and lubrication of articular cartilage. Journal of the Mechanical Behavior of Biomedical Materials. 101, 103422 (2020).
- Wu, L., et al. Lysophosphatidic acid mediates fibrosis in injured joints by regulating collagen type I biosynthesis. Osteoarthritis and Cartilage. 23 (2), 308-318 (2015).
- Chu, C. R., Szczodry, M., Bruno, S. Animal models for cartilage regeneration and repair. Tissue Engineering. Part B, Reviews. 16 (1), 105-115 (2010).
- Murphy, M. P., et al. Articular cartilage regeneration by activated skeletal stem cells. Natural Medicines. 26 (10), 1583-1592 (2020).

