Surgical Approach, Challenges, and Resolutions for Uterus Transplantation in Rats
Summary
The present protocol describes all the essential steps for successful uterine transplantation (UTx) in rats. The rat model has proven suitable to promote the clinical implementation of UTx; however, rat UTx is a highly complex procedure requiring careful instructions.
Abstract
Uterine transplantation (UTx) is a new approach for treating women with absolute uterine factor infertility (AUFI). An estimated 3%-5% of women suffer from AUFI. These women were deprived of the option to have children until the advent of UTx. The clinical application of UTx was driven by experimental studies in animals, and the first successful UTx was achieved in rats. Given their physiological, immunological, genetic, and reproductive characteristics, rats are a suitable model system for such transplants. In particular, their short gestation period is a clear advantage, as the usual endpoint of experimental UTx is successful pregnancy with live birth. The biggest challenge for rat models remains the small anatomy, which requires advanced microsurgical skills and experience. Although UTx has led to pregnancy in the clinic, the procedure is not established and requires continuous experimental optimization. Here, a detailed protocol is presented, including essential troubleshooting for rat UTx, which is expected to make the entire procedure easier to grasp for those without experience in this type of microsurgery.
Introduction
Uterine transplantation (UTx) is a novel treatment for absolute uterine factor infertility (AUFI). AUFI results from an absence (congenital or acquired) or malformation of the uterus and affects 3%-5% of women worldwide1. Ethical, legal, or religious reasons rule out adoption or surrogacy for many women who have a desire for motherhood but suffer from AUFI2. For these women, UTx remains the only option to start their own family. UTx has been applied in the clinic, albeit with mixed success; the procedure is technically challenging and requires steady improvement for its clinical establishment.
In 2014, the first transplantation of a uterus from a live donor (LD)-resulting in successful pregnancy-was performed by the pioneering Swedish group of Brännström3. The first birth following UTx from a deceased donor (DD) was reported in 2016 in Brazil4. By 2021, more than 80 UTxs have been performed worldwide, however with a success rate of about 50% and with grafts coming from LD for the majority1.
Although not life-saving, UTx is an increasingly popular procedure to fulfill the desires for own progeny. As such, the demand for grafts is mounting, placing DD donation into a future focus. However, DD donation is complicated due to considerably longer cold (and in the case of cardiac death, also warm) ischemic exposures, elevating the risks of graft dysfunction and rejection5,6. Surgical technique, demanding compatibility matching, and associated immunosuppression remain critical issues regarding UTx outcomes7.
To manage the above risks in the clinic, appropriate animal models for the exploration of ischemia and immunosuppression are needed. The most clinically relevant endpoint for animal models remains successful birth; to date, pregnancies following experimental UTx have been achieved in mice, rats, sheep, rabbits, and cynomolgus monkeys8. While larger animals are predestined for acquiring and optimizing surgical techniques, rodents come with the distinct advantage of short gestation periods. Therefore, rodent models are superior regarding practical, financial, and ethical considerations9. However, the main challenge of UTx in mice is the small anatomy, with the highly demanding surgery tied to the low reproducibility of murine UTx10. By contrast, rats are surgically more accessible and retain the advantages of short gestation times. As such, the rat has become the model of choice for UTx9. Wranning et al. introduced the rat model of orthotopic UTx in 2008, and using this model, the first live birth following UTx and natural mating has been reported11,12,13. Subsequent studies have had critical contributions to the implementation of UTx in humans9.
Nonetheless, UTx remains challenging in rats, and only a few groups as of yet have mastered this surgical technique. One relevant obstacle to the spread of rat UTx among researchers is the lack of a precise description of the individual microsurgical steps, the pitfalls, and the according measures for troubleshooting14. This protocol aims to provide a detailed guide for this highly complex microsurgical procedure to facilitate the implementation of this animal model into future research.
Protocol
All animal experiments were performed following Swiss Federal Animal Regulations and approved by the Veterinary Office of Zurich (n° 225/2019), assuring human care. Female virgin Lewis rats (body weight of 170-200 g) and female virgin Brown Norway rats (170-200 g) were used as uterus donors/recipients, while male Lewis rats (300-320 g) were used for mating. The rats were aged from 12-15 months. The animals were obtained from commercial sources (see Table of Materials) and were housed in controlled conditions and an enriched environment with free access to water and standard food.
1. Uterus retrieval
NOTE: For details on the procedure, please see the previously published reports12,13,15.
- Induce anesthesia with isoflurane and oxygen within an enclosed Plexiglas container (14 cm x 25 cm x 13 cm) for 1-2 min (5 vol% isoflurane in O2).
- Administer buprenorphine subcutaneously (0.05 mg/kg) and bupivacaine (0.5%, 8 mg/kg) subcutaneously in the region of the planned abdominal incision 30 min before surgery.
- Shave all the abdominal skin of the rat with an electric shaver.
- Use tapes to keep the animal fixed on a heating plate during surgery. Apply eye ointment to both eyes.
- Maintain anesthesia during the procedure with 2-4 vol% isoflurane in oxygen by continuous administration through a small nose cone.
- Monitor the anesthetic depth by clinical parameters without specialized tools (respiratory rate of ~70-120/min-a slow rate drop of 50% is acceptable during anesthesia; checking anesthetic depth with toe pinch; color of mucous membranes should be pink, not blue or grey)16, and adjust the isoflurane concentration accordingly.
NOTE: Optional: frequent respiration monitoring during surgery is feasible with the help of an assistant. - Confirm anesthetic depth by performing a toe pinch.
- Clean the abdominal skin in a circular motion with three alternating swabs of an antiseptic solution and 70% alcohol. Allow to dry.
- Place a sterile drape (see Table of Materials) with an abdominal window over the animal.
- Perform median laparotomy.
- Open the abdomen via a 6-8 cm midline long incision, starting 0.5 cm below the xiphisternum toward the hypogastrium. Use a no. 10 scalpel for the skin incision and small sharp scissors for the linea alba incision. Do not damage the liver or the bladder.
- Move the intestines outside the abdominal cavity using cotton swabs, cover them gently with a gauze moistened with sterile saline, and protect them with a sterile plastic bag for better insulation.
- Insert retractors or clips (see Table of Materials) at the left and right abdominal wall folders to keep the peritoneal muscle aside and the abdomen open, to obtain optimal access and visibility of the uterus and associated vessels. Fix the clips/retractors with tapes.
- Apply prewarmed saline to keep the surgical area and the intestines moist and avoid drying of the viscera.
- Harvest the right uterine horn with the common uterine cavity and cervix plus vascular pedicles, including the right uterine, internal, and common iliac vessels.
- Ligate (4/0 polyglactin; see Table of Materials), cauterize, and sever the left uterine horn adjacent to the branching from the common uterine cavity.
- Remove excess fat surrounding the uterus and vagina.
NOTE: Keep the fat around the uterine-vascular system. - Dissect the bladder at its attachment to the cervix with cauterization of all draining and feeding bladder vessels. During cauterization, maintain an adequate distance between the cervix and the vagina to avoid unncecessary cauterization on these two structures. Otherwise, the risk of graft necrosis increases.
NOTE: Most surgical manipulation should affect the bladder. Retract or pull the bladder caudally with a vascular clamp (see Table of Materials) to obtain a better view of excavatio vesicouterina. - Cauterize and sever the descending uterine vessels at the level of the ureter as distal to the cervix as possible.
NOTE: Maintain microcirculation around the vagina and cervix as much as possible during the division. - Separate the cervical/vaginal portion of the future graft from the rectal attachment and the paravaginal and paracervical ligaments.
NOTE: Avoid any cauterization on the graft vagina. - Carefully dissect the vagina via diathermy around 2-3 mm caudal of the cervix.
NOTE: No villi (cervix) is visible inside the vaginal lumen. - Locate both the uterine artery and vein at their origins. Ligate (8/0 polyamide; see Table of Materials), cauterize, and sever the gluteal vessels and all vessels caudal of the uterine vessels.
NOTE: Direct ligation of the common iliac vena caudal to the uterine vena is usually possible. - By blunt dissection, free the common iliac vessels from each other, from the bifurcation of the aorta and the vena cava down to the division of the uterine vessels.
NOTE: One can gain better surgical access to the area by removing one or two adjacent lymph nodes. - Excise the right uterine horn 3 mm from the Fallopian tube, after cauterizing the utero-ovarian pedicle at the same level. This enables anastomosis of the graft uterine horn to the upper part of the recipient uterine horn.
- Place ligatures (8/0 polyamide) directly around the right common iliac artery and vein, proximal to the aortic and caval bifurcations. Make a small incision (0.5-1 mm) into the right common iliac artery adjacent to the bifurcation, and insert a bent, blunted 30 G needle or a straight, blunted 25 G needle into the lumen (for flushing). Secure it with a ligature (6/0 polyamide).
NOTE: A further option is additional securing with a bulldog clamp to avoid displacement of the needle and/or vessel. - Dissect the common iliac vein caudally of the ligature at the right common iliac vein to enable outflow during flushing.
- Flush the graft following the steps below.
- Flush the uterus manually using 3 mL syringes with approximately 9 mL of cold Ringer solution (RHX: Ringer supplemented with 50 IU/mL heparin and 0.4 mg/mL xylazine) at a flowrate of 6 mL/min. Flush again with 6 mL of organ preservation solution supplemented with heparin (50 IU/mL) and xylazine (0.4 mg/mL) (see Table of Materials).
NOTE: Avoid high flushing pressure and ensure proper needle placement. - Remove the transplant when the uterine tissue has turned pale. Cut the common iliac artery caudally of the ligature at the bifurcation of the abdominal aorta.
- Flush the uterus manually using 3 mL syringes with approximately 9 mL of cold Ringer solution (RHX: Ringer supplemented with 50 IU/mL heparin and 0.4 mg/mL xylazine) at a flowrate of 6 mL/min. Flush again with 6 mL of organ preservation solution supplemented with heparin (50 IU/mL) and xylazine (0.4 mg/mL) (see Table of Materials).
- Place the transplant into chilled organ preservation solution (4 °C) for back table preparation and storage before transplantation.
- After removing the graft, euthanize the animal by first turning the isoflurane setting to maximum and then inducing bilateral pneumothorax followed by exsanguination17.
2. Syngeneic uterus transplantation
NOTE: For details on the procedure, please see the previously published reports12,13,15.
- Induce anesthesia and prepare the animal as mentioned in step 1.1.
- Administer effective analgesia (as described in step 1.1.1) and 200 IU/kg high molecular weight heparin 30 min before surgery.
- Perform median laparotomy.
- Open the abdomen via a 6-8 cm long midline incision starting 0.5 cm below the xiphisternum toward the hypogastrium. Use a no. 10 scalpel for the skin incision and small sharp scissors for the linea alba incision. Do not damage the liver and the bladder.
- Move the small intestines outside the abdominal cavity using cotton swabs, wrap them with a sterile moistened gauze, and cover them with a sterile plastic bag for better insulation.
- Insert retractors or clips at the left and right abdominal wall folders to keep the peritoneal muscle aside and the abdomen open, to obtain optimal access and visibility of the uterus and associated vessels. Fix the clips/retractors with tapes.
- Apply prewarmed saline to keep the surgical area and the intestines moist and avoid drying of the viscera.
- Perform a hysterectomy with dissection and mobilization of the upper third of the vagina from the rectum and the bladder.
- Cauterise the microvasculature around the uterus, cervix, and vagina. Cut and separate the uterus from the surrounding structures close to the organ to protect the microcirculation of the uterine sinister.
- Remove fat tissue from the surroundings.
- Amputate the left horn by cauterization. On the right side, preserve a 7-8 mm segment of the upper part of the uterus for later anastomosis to the uterine graft.
- Perform uterus transplantation.
- Mobilize and separate the right common iliac vessels, from the origin of the uterine vessels up to the aortic/caval bifurcation.
- Position the graft in the abdominal cavity. Wrap the graft in a gauze soaked in cold organ preservation solution.
NOTE: The graft needs to be kept cold during anastomosis. - Place atraumatic vascular clamps onto the right common iliac vein at each side, framing the future anastomosis site.
NOTE: Lower the anesthesia to 1-1.5 vol% isoflurane to adapt to the sudden decrease in cardiac preloading and the resulting hypotension. - Cut a slit slightly larger than the opening of the graft vein into the common iliac vein.
- Position the graft vein.
- Place one stay suture (10/0 polyamide; see Table of Materials) into each corner of the slit on the right common iliac vein.
NOTE: Keep the suture knot at the caudal corner loose for better adjustment and to prevent purse-string effects. - Regularly flush the anastomosis area with cooled RHX during the procedure to prevent thromboses.
- Anastomose one side of the graft vein to the recipient's vein with six to eight loops of a continuous suture (Figure 1).
NOTE: Start with the cranial stay suture (10/0 polyamide) and first anastomose the inbound part of the vessels. - Anastomose the other side of the vessel in the same manner, this time starting from the outside.
- Tie a knot at the cranial stay suture, and then one at the caudal stay suture (10/0 polyamide), after finishing the anastomoses at both sides.
NOTE: Tighten the continuous sutures only as much as necessary to prevent purse-string effects. - Place atraumatic vascular clamps onto the right common iliac artery at each side, framing the future anastomosis site.
- Perform the arterial anastomosis (right common iliac arteria [RCIA] via 8-10 loops using interrupted sutures (10/0 polyamide).
NOTE: Interrupted sutures are easier to control than continuous ones (optional with the "fish-mouth" technique)18. Constant flushing of the anastomosis area with cooled RHX during the procedure helps to prevent thromboses. When using continuous sutures, perform this step analogous to venous anastomosis.
- Perform graft reperfusion.
- When both anastomosis sites appear patent and any bleeding is stopped, release the vascular clamps on the graft vessels (Figure 2).
- Inspect the graft for signs of reperfusion, such as reddening, filling of the vein, or pulsation in the graft artery.
- Connect the vaginal cuff of the transplant to the vaginal vault of the recipient by using six to seven intraluminal (6/0 polyglactin) interrupted sutures.
NOTE: Start with a single suture at the 12 o'clock position first, and place the next ones at the 10 and 1 o'clock positions. The two sutures at the 9 and 3 o'clock positions should be tied after the sutures in the front row19,20. - Anastomose the graft uterine horn end-to-end to the remaining cranial uterine segment of the recipient uterus by using five to seven interrupted sutures (7/0 polyamide).
NOTE: Do not stitch through the lumen.
- Close the abdomen with a continuous suture. Use 4/0 polyglactin for suturing the muscle layer, and 6/0 polyamide or surgical wound clips for the skin.
- Let the animal recover in a warmed cage once the transplant is completed. Remain with the animal until it has regained sternal recumbency ability, and maintain single housing until its full recovery. Provide psotopertaive analgesia treatment by subcutaneously administering buprenorphine (0.05 mg/kg) and suitable NSAID, however not before 4-8 hours after the first dose of anesthesia. Provide continuous with buprenorphine via drinking water (1 mg/kg, oral, 5 mL buprenorphine in 160 mL drinking water (0.3 mg/mL)) for three days after surgery.
- The skin suture is removed 10-14 days post suregery.
Representative Results
Results from two groups of rats are presented. UTx was carried out before (group 1, n = 8) and after (group 2, n = 8) protocol adjustment (Table 1) to demonstrate the effects of our modifications (please see the Discussion for an explanation of our modifications)12,15,21.
The outcome of rat UTx is associated with three key phases. The first phase is successful recovery from UTx. Usually, recipients should recover within the first 2 postoperative days. The second phase relates to the health status of the graft 2 weeks after surgery, which decides the inclusion into the mating process (Table 2). The third phase involves spontaneous mating followed by successful birth as evidence of fertility.
All animals from both groups had an uneventful recovery from surgery. During the second phase, four animals were excluded from group 1 and two from group 2. The exclusion was due to graft thrombosis and abscess (n = 4 for group 1, n = 2 for group 2), and narrowed/malformed utero-uterine anastomosis (additionally for n = 1, group 1) on examination at relaparotomy (Table 2). Relaparotomy (along the scar of the original laparotomy) was performed for all females 2 weeks post transplantation, as the physical appearance of the animals had little value as an indicator of graft health. Overall, the 2 week graft survival rate was 50% and 75% for groups 1 and 2, respectively (Table 3).
During phase 3, four females from group 1 were matched for mating with Lewis males, around 9 weeks after UTx. Two females showed signs of pregnancy (bodyweight increase, Figure 3; nesting behavior); however, no live birth was observed. After two mating cycles of three females, pup body parts (bone and tissue) were found in one female rat (°1). Histological examination of hematoxylin and eosin stained tissue sections by a veterinary pathologist revealed these pups developed up to parturition (Figures 4 and Figure 5).
Six females from group 2 were mated with Lewis males (see Figure 6 for bodyweight changes). Three of the six rats (two Lewis and one Brown Norway rat) gave birth to pups, while another two showed signs of pregnancy. The first litter of the female Lewis rat (#1) consisted of two pups (Figure 7). Shortly after birth, the Lewis female became pregnant again; however, only two of three pups survived postnatally (Figure 8). One likely explanation for the one death is infanticide, as it occurs even with healthy pups under conditions of postpartum stress. Likewise, the Brown Norway female (#2) gave birth twice, each time to four pups per mating cycle (Figure 9). The largest litter number of group 2 was delivered by another Lewis female (#3), with seven pups after the first mating cycle. All surviving pups displayed normal development (Figure 10).
Overall, the adaptation of the protocol increased 2 week graft survival from 50% to 75%. Five of six females became pregnant, compared to two out of four from group 1. Likewise, three of six of females gave birth to live pups compared to zero out of four of the group 1 females. In conclusion, the adapted protocol improved both direct surgical outcomes and the rate of successful live births following UTx (Table 4 and Figure 6).
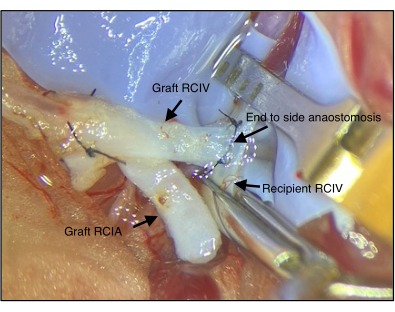
Figure 1: Anastomosis of the graft and the recipient's vein. The right common iliac vein (RCIV) of the graft is connected to the RCIV of the recipient via end-to-side anastomosis (step 2.4). RCIA = right common iliac artery Please click here to view a larger version of this figure.
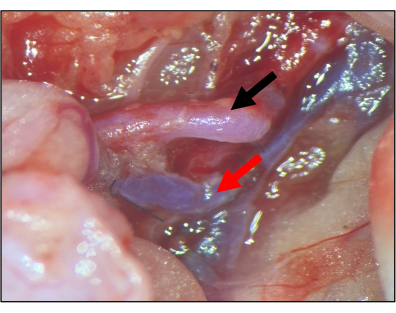
Figure 2: Anastomosis of the graft and the recipient artery. The right common iliac artery (RCIA) is connected to the RCIA of the recipient via end-to-side anastomosis (step 2.4). After opening both vascular clamps (2.5), the artery should be fully perfused in the absence of outer bleeding. Black arrow: graft RCIA; red arrow: graft RCIV. Please click here to view a larger version of this figure.
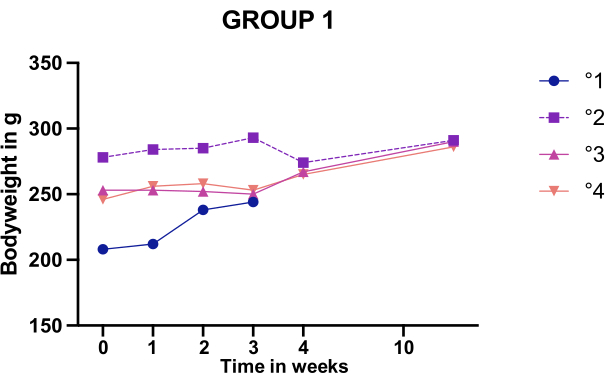
Figure 3: Bodyweight changes after mating of females in group 1. Body weight monitoring of the four group 1 females who displayed an intact graft upon relaparotomy. One animal (°1) was euthanized during Caesarean sectioning to inspect the uterus. Please click here to view a larger version of this figure.
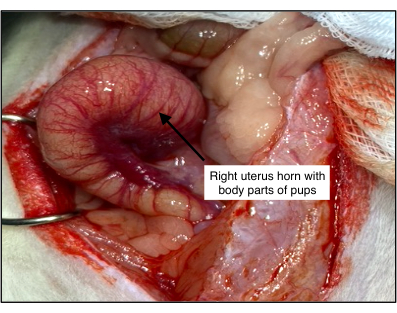
Figure 4: Histological examination of the uterus of the group 1 rat with dead pups. Body parts of pups were found in rat °1. Examination revealed a vital and dilated uterus, suggesting the pups developed normally but could not be delivered. Please click here to view a larger version of this figure.
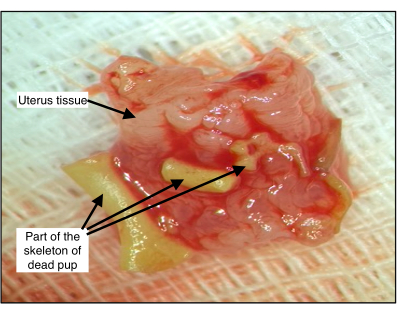
Figure 5: Pup body parts inside the uterus of group 1 rat °1. The developmental stage of the bones was consistent with full-term pups. Please click here to view a larger version of this figure.
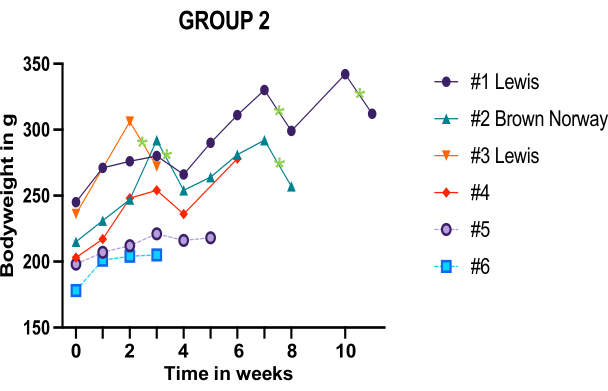
Figure 6: Body weight monitoring of the six group 2 females who displayed an intact graft upon relaparotomy. Green stars mark individual birth events. Hashtag numbers refer to the individual females. Please click here to view a larger version of this figure.

Figure 7: The first live birth after rat UTx following the modified protocol. Two newborn rats and their mother (head to the right; Lewis female #1, group 2). Please click here to view a larger version of this figure.
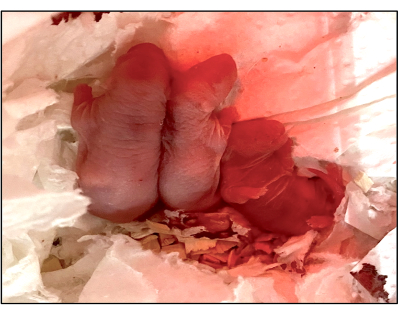
Figure 8: The second litter after the second mating cycle post rat UTx following the modified protocol. Female #1 (group 2) gave birth to three pups, two of which survived. Please click here to view a larger version of this figure.
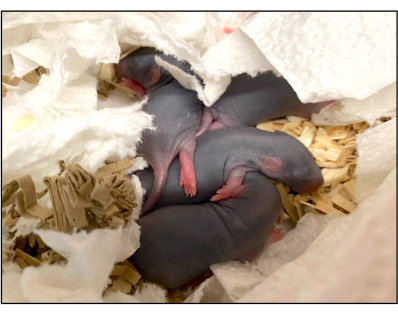
Figure 9: The first litter of the Brown Norway rat after UTx following the modified protocol. After the first mating cycle, the Brown Norway rat (#2, group 2) gave birth to four pups, followed by another four after the second mating cycle. Please click here to view a larger version of this figure.
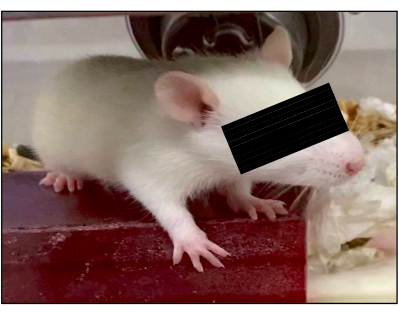
Figure 10: Development of pups. All surviving pups displayed normal development at 3 weeks of age. One representative example is shown. Please click here to view a larger version of this figure.
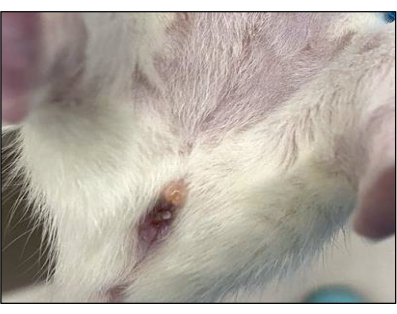
Figure 11: Vaginal plug formation following successful mating. White plugs should form to cover the vagina after fertilization to prevent further mating. Please click here to view a larger version of this figure.
| Group 1 | Group 2 | Syngeneic UTx in rats | Allogeneic UTx in rats | |
| before modification | after modification | see ref. 12 | see ref. 15 | |
| Total number of animals | 8 | 8 | 27 | 14 |
| Two-weeks graft survival | 4/8 (50%) | 6/8 (75%) | 19/27 (70%) | 9/14 (64%) |
| Number of mated females | 4/4 (100%) | 6/6 (100%) | 17/19 (89%) | 9/9 (100%) |
| Full term pregnancy | 1/4 (25%) | 5/6 (83%) | 11/17(65%) | 5/9 (56%) |
| Successfully delivered litter | 0 | 5a/td> | 1 | 5 |
| Total number of living pups | 0 | 20b | 3c | 25d |
| Pregnancy to term w/o live birth | 1 | 1e | 10 | 2 |
Table 1: Outcome comparison of the modified and unmodified protocol for rat UTx. a: Two pregnancies by the same rat; b: Including the infanticide pup; c: Median; d: Sum of the median/delivery; e: Dead pups in resorption stage after three cycles of mating.
| Surgical settings | Group 1 (n = 8) | Group 2 (n = 8) |
| Project stage | initial stage | late stage |
| Cold storage time | 2-3 h | 2-3 h |
| Flushing solution during anastomosis | RH | RHX |
| Vaginal anastomosis | 6/0 Ethilon | 6/0 Vicryl |
| Uterus horn anastomosis | partially continuous suture | interrupted suture |
| Arteria anastomosis | continuous suture | Interrupted and continuous |
| Micro-vascularization around vagina and cervix | cauterization close to the vaginal/cervical tissue | cauterization more distally |
Table 2: Surgical settings group 1 versus group 2.
| Exclusion criteria# |
| Signs of thrombosis (particularly around anastomoses) |
| Major adhesion |
| Constricted uterus |
| Signs of infection |
| Graft necrosis |
Table 3: Exclusion criteria for mating. #Applied at relaparotomy 2 weeks post UTx.
| n (group 1) | n (group 2) | |
| Animals | 8 | 8 |
| Healthy graft after 2 weeks | 4 | 6 |
| Mated | 4 | 6 |
| Full term pregnancy | 1 | 5 |
| Successfully delivered litter | 0 | 5a |
| Total number of living pups | 0 | 20b |
| Pregnancy to term, not live birth | 1 | 0 |
Table 4: Outcomes of group 1 versus group 2. a: Two consecutive pregnancies in the same rat; b: Including the pup killed by infanticide.
Discussion
The protocol presented here offers detailed instructions for the surgical approach behind uterus transplantation in rats. The protocol has been optimized to increase the odds of live births following UTx and subsequent mating. The original protocol has been taken over from the Brännström group12,13, inspired by the mouse work of Akouri et al.10, and modified based on the authors' experiences over the past years. As such, the modifications were driven by true learning curves reflecting the development from failed transplants to reproducible outcomes.
Key modifications that are likely decisive were: (1) the addition of xylazine to the flushing solution during organ retrieval and anastomosis, which stimulated vasodilation leading to reduced thrombotic risks. (2) The use of interrupted sutures for arterial end-to-side anastomosis. Interrupted sutures not only increase surgical control over patency and avoid purse-string effects, but also enable subsequent widening of the anastomotic area to increase the blood flow through the arteria uterina. (3) The application of interrupted and non-absorbable sutures for the uterus horn anastomosis with stitching only within the layer of the perimetrium; this modification prevents constriction of the uterus wall, which may hinder later fertilization. (4) Using absorbable suture material for vaginal anastomosis reduces the risk of vaginal stenosis and thereby increases the chances of live births. (5) Performing any surgical manipulation as far away from the vagina and cervix as possible is a crucial step to avoid vaginal scarring and to increase the chances of live births. (6) The direct ligation of the right common iliac vein and artery instead of ligating the vena cava, abdominal aorta, and the left common iliac vein artery. Direct ligation caudal to the uterine vena is, in most cases, feasible; the direct ligation simplifies organ procurement and shortens surgical times without negatively impacting transplant outcomes. (7) Lowering the isoflurane concentration to 1%-1.5 vol% right after the clamping of the common iliac vena (step 2.4.3) is likewise crucial, as otherwise cardiac death may occur.
The above points are the main elements that distinguish the present protocol from the perhaps most widely used approach described by the Swedish group10,12,13. These measures benefitted the ultimate endpoint of UTx, the live birth, as evinced by the group comparison (original vs. modified protocol; see Table 1). Clearly, the surgical learning curve over time has likewise added to better outcomes; however, its contribution is difficult to estimate.
In addition to a meticulous surgical technique, postoperative recipient care is likewise crucial for the final outcome. Postoperative treatments follow the respective local protocol; the instructions here recommend 0.05 mg/kg buprenorphine after the recipient wakes up from anesthesia, and during the first postoperative day if presenting with pain as defined by our scoring assessment. The scoring criteria are based on behavior (activity, respiration, coat, posture, wound) and body weight. Immediately after surgery, positive signs of recovery are a return of pink coloring of the extremities and ears, as well as a return of red coloring of the eyes. Given the length of the transplant procedure, regular monitoring during the first 3 hours after transplantation is critical. If assessment criteria are not met during these 3 hours, the animal is euthanized to avoid further suffering. Monitoring is continued three times a day for the first 3 postoperative days. If the animal does not fully recover during the first 2 days, it is euthanized. Depending on its condition, the animal remains in a single housing for 2-3 days post-surgery.
Sterile conditions during surgery are yet another aspect relevant to successful outcomes. A strict hygiene routine is mandatory to minimize infection risks. When entering the animal facility, hands must be thoroughly washed before getting dressed in a clean overall, a face mask, gloves, and a cap. The entire surgical area is disinfected with a special swab containing benzyl-C12-18-alkyldimethylammonium chloride, didecyldimethylammonium chloride, and glutaraldehyde. Additional disinfection with 70% ethanol follows. Before each surgical manipulation, gloves are briefly cleaned with 70% ethanol, and the area surrounding the surgical site is covered with a sterile drape. These measures greatly help to minimize infection risks.
One issue with progeny success is the difficulty in monitoring actual pregnancy. Bodyweight increases may be indicative of pregnancy, but can also be due to infanticide and generally do not reliably reflect the state of the female. For example, only modest weight changes overall were apparent for group 1 during monitoring. Animals 3 and 4 displayed similar weight increases over the two mating cycles, yet only animal 4 was pregnant upon relaparotomy (Figure 3 and Figure 4). Moreover, weight loss after the first mating cycle with absent birth may point to pregnancy absorption. Although it remains unclear why tissue absorption results in weight loss, this phenomenon has been repeatedly observed in rat UTx recipients12. Vaginal plug formation post successful mating (Figure 11) likewise was often absent and did not show a correlation to later body weight changes. Given the little value in marking pregnancy, body weight monitoring frequency was reduced for group 2 to minimize animal stress exposure. A further consequence of inconsistent plug presence and weight changes was an unreliable definition of the conception time. Due dates for births have thus been calculated using the 2nd day post male exposure as the conception day. Interestingly, body weight changes in group 2 correlated relatively well with the course of pregnancy, with delivery being marked by a clear drop in weight (Figure 6).
Comparing body weight changes (Figure 6) of animals 1, 2, and 3 (pregnant) with animals 5 and 6 (never pregnant) nicely illustrates the effects of pregnancy and live birth versus unsuccessful mating. The reason behind the different body weight behavior in group 2 compared to group 1 remains unclear. However, weight changes may be more reliable simply due to the higher number of full-term pregnancies in group 2. The low pregnancy rate in group 1 may possibly relate to vaginal stenosis, which was overrepresented relative to group 2 (Table 3) and is a cause of hampered conception. The reduced stenotic incidence in group 2 may be due to the use of non-absorbable instead of absorbable sutures for vaginal anastomosis, likely along with improved surgical manipulation during vaginal dissection in this group.
Unlike the Swedish approach10,12, the protocol described here uses IGL-1 (Institute Georges Lopez) solution for graft storage instead of Ringer or other solutions. While the exact impact of IGL-1 solution on UTx outcomes is unknown, various reports suggest overall benefits in experimental transplantation for this solution22,23,24,25. Lastly, a few protocol steps usually performed for UTx12 did not affect outcomes if skipped. These include the clipping at the left or right uterus horn and the postoperative heparin substitution, which the authors deem optional.
One limitation inherent to experimental UTx is also valid for the presented protocol. The procedure is time-consuming, and organ retrieval followed by transplantation can take up to 6 hours. Therefore, rat UTx requires full focus for several hours in order to perform each step flawlessly. The latter remains the most important aspect of success. Such attention can be accomplished provided the microsurgical skills are fully developed and continuously practiced. Likewise, patience and stamina are key traits to avoid mistakes during the whole procedure. Another consequence of the lengthy procedure is that only one animal can be operated on per day. Careful planning and focus on key questions help to develop a productive research design. In general, it is advised to achieve a graft survival above 70% before committing to the actual experimental series to ensure that findings will be sufficiently robust. Finally, one may wish to reconsider live birth as an endpoint. The high incidence of vaginal stenosis and pregnancy resorption12 markedly increase the number of UTxs required to obtain meaningful results. Pending on the experimental question, other endpoints, such as graft survival, may be more effective.
Despite its increasing application, UTx remains an experimental application also in the clinic. Besides the need for improved surgical approaches and immunosuppressive strategies, the use of grafts from deceased donors, in particular, is an area requiring additional research. Strategies to mitigate ischemic injury are not established, but would be most welcome for the expansion of potential donor pools. Indeed, the impact of ischemia on uteri is ill-researched, with applied knowledge relying on findings from other organs26,27. Rat UTx offers a means to explore ischemic injury in controlled settings while using approaches tailored to the clinic28,29. Of note, live donation is associated with certain risks; donors have a high rate of surgical complications, such as urinary tract and bowel trauma. Accordingly, the demand for dead donations and its research is growing5,30.
Many additional questions exist where rat UTx could be informative. For example, the rat system offers the opportunity to identify and/or validate biological markers that could be used to noninvasively monitor the course of uterus transplantations in the clinic. Recent societal developments may create new questions where the rat model may be of use. UTx is now being advocated also for transgender individuals, with the male anatomy asking for adapted surgical approaches31.
In conclusion, a new protocol of rat UTx based on existing rodent work and modified by hands-on experience is presented. The modified protocol has a high likelihood of rat UTx resulting in live births-provided sufficient microsurgical skills and practice. UTx is perhaps the most complex of the major transplantation procedures. The described instructions shall add to a common protocol, which is currently lacking but needed to establish the demanding procedure of rat UTx within the research community.
Declarações
The authors have nothing to disclose.
Acknowledgements
This study was supported by the Swiss National Science Foundation (project grant no. 310030_192736). We would like to thank Dr. Frauke Seehusen from the Institute of Veterinary Pathology of the University of Zurich for her histopathological support.
Materials
| Angled to Side Scissor 5 mm | F.S.T | 15008-08 | |
| Big Paper Clip | No specific | Used as retractor | |
| Blunt Bend Needle G30 | Unimed S.A. | ||
| Bupivacain 0.5% | Sintetica | ||
| Buprenorphine 0.3 mg/mL | Temgesic | ||
| Dosiernadel G25 | H.SIGRIST& PARTNER AG | ||
| Dumont #5SF Forceps | F.S.T | 11252-00 | |
| Ethilon 10/0 | Ethicon | 2810G | |
| Ethilon 6/0 | Ethicon | 667H | |
| Ethilon 7/0 | Ethicon | EH7446H | |
| Ethilon 8/0 | Ethicon | 2808G | |
| Femal Brown Norway Rats (150-170 g) | Janvier | ||
| Femal Lewis Rats (150-170 g) | Charles River Deutschland | ||
| Fine Scissors – Sharp | F.S.T | 14060-09 | Any other small scissor works too |
| Halsey Micro Needle Holder | F.S.T | 12500-12 | Any other small needholder works too |
| Heparin Natrium 25000 I.E./ 5 mL | B. Braun | ||
| Institute Georges Lopez Perfusion Solution (IGL) | Institute Georges Lopez | Organ preservation solution | |
| Male Lewis Rats (300-320 g) | Charles River Deutschland | ||
| Micro Serrefines 13 mm | F.S.T | 18055-04 | |
| Micro Serrefines 16 mm gebogen | F.S.T | 18055-06 | |
| Micro-Serrefine Clamp Applicator with Lock | F.S.T | 18056-14 | |
| Mölnlyncke Op Towel | Mölnlyncke | 800300 | Sterile drape |
| NaCl 0.9% | B.Braun | ||
| Octenisept | Schülke | ||
| Paper Tape | Tesa | For fixing the animal | |
| Philips Avent Schneller Flaschenwärmer SCF358/02 | Philips | 12824216 | |
| Ringerfundin | B.Braun | ||
| Rompun 2% | Bayer | Xylazine | |
| Round Handled Needle Holders | F.S.T | 12075-12 | |
| Round Handled Needle Holders | F.S.T | 12075-12 | |
| S&T Vessel Dilating Forceps – Angled 45° | F.S.T | 00276-13 | |
| Sacryl Naht | KRUUSE | 152575 | |
| Scapel No 10 | Swann Morton | 201 | |
| Small Histo-Container | Any small histo-container works fine-for coldstorage of the graft | ||
| Small Plastik Bags | Any transparant plastic bags are fine | ||
| Steril Cotton swab | Lohmann-Rauscher | Any steril cotton swab is fine | |
| Sterile Gauze | Lohmann-Rauscher | Any steril gauze is fine | |
| Straight Scissor 8mm | F.S.T | 15024-10 | |
| Surgical microscope – SZX9 | Olympus | OLY-SZX9-B | |
| Sutter Non Stick GLISS 0.4 mm | Sutter | 78 01 69 SLS | |
| Suture Tying Forceps | F.S.T | 00272-13 | |
| ThermoLux warming mat | ThermoLux | ||
| Tissue Forceps for Skin | Any tissue forceps are fine | ||
| Vesseldilatator Forceps | F.S.T | 00125-11 | |
| Vicryl plus 4/0 | Ethicon | VCP292H |
Referências
- Richards, E. G., et al. Uterus transplantation: state of the art in 2021. Journal of Assisted Reproduction and Genetics. 38 (9), 2251-2259 (2021).
- Jones, B. P., et al. Options for acquiring motherhood in absolute uterine factor infertility; adoption, surrogacy and uterine transplantation. The Obstetrician & Gynaecologist. 23 (2), 138-147 (2021).
- Brannstrom, M., et al. The first clinical trial of uterus transplantation: surgical technique and outcome. American Journal of Transplantation. 14, 44 (2014).
- Ejzenberg, D., et al. Livebirth after uterus transplantation from a deceased donor in a recipient with uterine infertility. Lancet. 392 (10165), 2697-2704 (2018).
- Lavoue, V., et al. Which donor for uterus transplants: brain-dead donor or living donor? A systematic review. Transplantation. 101 (2), 267-273 (2017).
- O’Donovan, L., Williams, N. J., Wilkinson, S. Ethical and policy issues raised by uterus transplants. British Medical Bulletin. 131 (1), 19-28 (2019).
- Kisu, I., et al. Long-term outcome and rejection after allogeneic uterus transplantation in cynomolgus macaques. Journal of Clinical Medicine. 8 (10), 1572 (2019).
- Ozkan, O., et al. Uterus transplantation: From animal models through the first heart beating pregnancy to the first human live birth. Womens Health. 12 (4), 442-449 (2016).
- Favre-Inhofer, A., et al. Involving animal models in uterine transplantation. Frontiers in Surgery. 9, 830826 (2022).
- El-Akouri, R. R., Wranning, C. A., Molne, J., Kurlberg, G., Brannstrom, M. Pregnancy in transplanted mouse uterus after long-term cold ischaemic preservation. Human Reproduction. 18 (10), 2024-2030 (2003).
- Sahin, S., Selcuk, S., Eroglu, M., Karateke, A. Uterus transplantation: Experimental animal models and recent experience in humans. Turkish Journal of Obstetrics and Gynecology. 12 (1), 38-42 (2015).
- Wranning, C. A., Akhi, S. N., Diaz-Garcia, C., Brannstrom, M. Pregnancy after syngeneic uterus transplantation and spontaneous mating in the rat. Human Reproduction. 26 (3), 553-558 (2011).
- Wranning, C. A., Akhi, S. N., Kurlberg, G., Brannstrom, M. Uterus transplantation in the rat: Model development, surgical learning and morphological evaluation of healing. Acta Obstetricia et Gynecologica Scandinavica. 87 (11), 1239-1247 (2008).
- Brannstrom, M., Wranning, C. A., Altchek, A. Experimental uterus transplantation. Human Reproduction Update. 16 (3), 329-345 (2010).
- Diaz-Garcia, C., Akhi, S. N., Wallin, A., Pellicer, A., Brannstrom, M. First report on fertility after allogeneic uterus transplantation. Acta Obstetricia et Gynecologica Scandinavica. 89 (11), 1491-1494 (2010).
- R, E., Brown, M. J., Karas, A. Z. . Anesthesia and Analgesia in Laboratory Animals. 2nd edn. , (2008).
- Donovan, J., Brown, P. Euthanasia. Current Protocols. , 8 (2006).
- Rutledge, C., Raper, D. M. S., Abla, A. A. How I do it: superficial temporal artery-middle cerebral artery bypass for flow augmentation and replacement. Acta Neurochirurgica. 162 (8), 1847-1851 (2020).
- Kuo, S. C. -. H., et al. The multiple-U technique: a novel microvascular anastomosis technique that guarantees everted anastomosis sites with solid intima-to-intima contact. Plastic and Reconstructive Surgery. 149 (5), 981 (2022).
- Magee, D. J., Manske, R. C. . Pathology and Intervention in Musculoskeletal Rehabilitation. 2nd edn. , 25-62 (2016).
- Diaz-Garcia, C., Johannesson, L., Shao, R. J., Bilig, H., Brannstrom, M. Pregnancy after allogeneic uterus transplantation in the rat: perinatal outcome and growth trajectory. Fertility and Sterility. 102 (6), 1545-1552 (2014).
- Canovai, E., et al. IGL-1 as a preservation solution in intestinal transplantation: a multicenter experience. Transplant International. 33 (8), 963-965 (2020).
- Habran, M., De Beule, J., Jochmans, I. IGL-1 preservation solution in kidney and pancreas transplantation: A systematic review. PLoS One. 15 (4), 0231019 (2020).
- Mosbah, I. B., et al. IGL-1 solution reduces endoplasmic reticulum stress and apoptosis in rat liver transplantation. Cell Death & Disease. 3 (3), 279 (2012).
- Wiederkehr, J. C., et al. Use of IGL-1 preservation solution in liver transplantation. Transplantation Proceedings. 46 (6), 1809-1811 (2014).
- Tilney, N. L., Guttmann, R. D. Effects of initial ischemia/reperfusion injury on the transplanted kidney. Transplantation. 64 (7), 945-947 (1997).
- de Rougemont, O., Dutkowski, P., Clavien, P. A. Biological modulation of liver ischemia-reperfusion injury. Current Opinion in Organ Transplantation. 15 (2), 183-189 (2010).
- Jakubauskiene, L., et al. Relaxin and erythropoietin significantly reduce uterine tissue damage during experimental ischemia-reperfusion injury. International Journal of Molecular Sciences. 23 (13), 7120 (2022).
- Wang, Y., Wu, Y., Peng, S. Resveratrol inhibits the inflammatory response and oxidative stress induced by uterine ischemia reperfusion injury by activating PI3K-AKT pathway. PLoS One. 17 (6), 0266961 (2022).
- Kisu, I., et al. Risks for donors in uterus transplantation. Reproductive Sciences. 20 (12), 1406-1415 (2013).
- Jones, B. P., et al. Uterine transplantation in transgender women. BJOG: an International Journal of Obstetrics and Gynaecology. 126 (2), 152-156 (2019).

