Stem Cell Transplantation in an in vitro Simulated Ischemia/Reperfusion Model
Summary
We demonstrate how to set up an in vitro ischemia/reperfusion model and how to evaluate the effect of stem cell therapy on postischemic cardiac cells.
Abstract
Stem cell transplantation protocols are finding their way into clinical practice1,2,3. Getting better results, making the protocols more robust, and finding new sources for implantable cells are the focus of recent research4,5. Investigating the effectiveness of cell therapies is not an easy task and new tools are needed to investigate the mechanisms involved in the treatment process6. We designed an experimental protocol of ischemia/reperfusion in order to allow the observation of cellular connections and even subcellular mechanisms during ischemia/reperfusion injury and after stem cell transplantation and to evaluate the efficacy of cell therapy. H9c2 cardiomyoblast cells were placed onto cell culture plates7,8. Ischemia was simulated with 150 minutes in a glucose free medium with oxygen level below 0.5%. Then, normal media and oxygen levels were reintroduced to simulate reperfusion. After oxygen glucose deprivation, the damaged cells were treated with transplantation of labeled human bone marrow derived mesenchymal stem cells by adding them to the culture. Mesenchymal stem cells are preferred in clinical trials because they are easily accessible with minimal invasive surgery, easily expandable and autologous. After 24 hours of co-cultivation, cells were stained with calcein and ethidium-homodimer to differentiate between live and dead cells. This setup allowed us to investigate the intercellular connections using confocal fluorescent microscopy and to quantify the survival rate of postischemic cells by flow cytometry. Confocal microscopy showed the interactions of the two cell populations such as cell fusion and formation of intercellular nanotubes. Flow cytometry analysis revealed 3 clusters of damaged cells which can be plotted on a graph and analyzed statistically. These populations can be investigated separately and conclusions can be drawn on these data on the effectiveness of the simulated therapeutical approach.
Protocol
1. Preparing H9c2 cardiomyoblast cells
H9c2 rat cardiomyoblasts were obtained from ATCC (Wesel, Germany) and expanded in high glucose (4.5 g/L) DMEM containing 10% fetal bovine serum, 4 mM L-glutamine, 100 U/ml penicillin and 100 μg/ml streptomycin. The H9c2 myoblast cell line is derived from embryonic rat heart, it is used as an in vitro model for both skeletal and cardiac muscle 8,9.
Prepare 12 well plate of H9c2 cells:
- Remove culture medium from Petri dish of H9c2 cells.
- Wash cells twice with 5 ml PBS and digest with 2-3 ml 0.05% trypsin/EDTA.
- Place Petri dish for 5 minutes in the 37°C incubator.
- Inhibit trypsin with 10 ml DMEM culture medium.
- Spin down the cells at 1200 RPM for 8 minutes.
- After removing the supernatant, resuspend cells in 1 ml DMEM culture medium and count cells with hemocytometer using Trypan Blue staining.
- Seed 30,000 H9c2 cells in 2 ml DMEM per well in a 12 well plate.
- Place the plate into the 37°C CO2 incubator for 24 hours.
2. Simulated ischemia/reperfusion
Ischemia/reperfusion was simulated in vitro by performing oxygen glucose deprivation (OGD) on H9c2 cell cultures. This procedure was performed on the stage of the Zeiss confocal microscope with the PeCon cell incubation system (Erbach, Germany) allowing the observation of the cells during OGD.
- Remove medium by aspiration from 12 well plate of H9c2 cells.
- Wash cells twice with PBS.
- Add 3 ml glucose free medium per well.
- Place the plate into the cell incubation system.
- Start nitrogen gas to purge the oxygen out of the incubation system.
- Wait until the level of oxygen reaches 0.5% then start the timer. 0.5% oxygen means approximately 3 mmHg partial tension. Subject cells to 150 minutes of simulated ischemia.
- At the end of simulated ischemia, remove the glucose free medium from the cells.
- Pipette 2 ml normal culture medium to the wells.
- Place the 12 well plate for 30 minutes in the 37°C CO2 incubator; this is the “reperfusion period”.
3. Preparing rescue mesenchymal stem cells
Bone marrow of human origin was taken into T75 flasks and diluted with Dulbecco’s Modified Eagle’s Medium (DMEM) culture medium containing 10% fetal calf serum (FCS), 100 U/ml penicillin and 100 μg/ml streptomycin, 4 mM L-glutamine and 1 g/l glucose. The flasks were incubated at 37°C in a fully humidified atmosphere of 5% CO2 for 3 days. After the incubation period the BMSCs adhered to the surface of the flasks and the remaining components of bone marrow were eliminated by washing with PBS. The used BMSCs were between 1 and 5 passages in the experiments. The identity of cells was confirmed by the presence of lineage-specific cell surface markers with flow cytometry. Hematopoietic linage-specific surface markers (CD34, CD45) and mesenchymal surface markers (CD73, CD90, CD105 and CD166) were investigated.
During simulated ischemia, prepare rescue mesenchymal stem cells for transplantation.
- Dilute the fluorescent dye Vybrant DiD in 1:200 ratio in DMEM culture medium.
- Aspirate medium from the rescue cells and add 300 μl DMEM with Vybrant DiD.
- Place Petri dish for 30 minutes in the 37°C CO2 incubator.
- Remove culture medium from Petri dish of mesenchymal stem cells.
- Wash cells twice with 5 ml PBS and digest with 2-3 ml 0.05% trypsin/EDTA.
- Place Petri dish for 5 minutes in the 37°C incubator.
- Inhibit trypsin with 10 ml DMEM culture medium.
- Spin down the cells at 1200 RPM for 8 minutes.
- After removing the supernatant, resuspend cells in 1 ml DMEM culture medium and count cells with hemocytometer using Trypan Blue staining.
- After 30 minutes at the end of the reperfusion add 20,000 mesenchymal stem cells per well to the postischemic H9c2 cardiac myoblasts.
- Place co-culture of cells into the 37°C CO2 incubator for 24 hours.
4. Analysis with confocal microscopy
After the simulated ischemia/reperfusion injury, cells are incubated in normal culture medium for 24 hours.
- After 24 hours wash cells twice with PBS.
- Stain the cells in 500 μl live/dead dye solution for 30 minutes (5 nM calcein and 400 nM Ethidium-homodimer-2 in PBS) (5×104 cell/ml).
The evaluation of confocal images for live and dead cells selected by morphology and fluorescence can be performed with the ImageJ software (National Institutes of Health, USA). In case of co-cultures, MSCs can be distinguished from H9c2 cells due to their Vybrant DiD cell labeling. The ratio of dead cells can be evaluated in 4 independent fields of view (objective 10x) for each culture in a blind fashion10.
H9c2 cardiomyoblasts cultured on 42 mm glass coverslips can also be investigated with time lapse video microscopy during and after ischemia/reperfusion injury using the cell incubation system of the confocal microscope. Cell-to-cell interactions such as cell fusion and formation of intercellular nanotubes can be studied over time.
5. Analysis with flow cytometry
After the simulated ischemia/reperfusion injury, cells are incubated in normal culture medium for 24 hours.
- After 24 hours, harvest the culture media. It is very important to collect the culture medium also because some dead cells could have detached from the culture dish surface during the 24 hour incubation.
- Wash cells twice with PBS and digest with 0.05% trypsin-EDTA.
- Inhibit trypsin with DMEM culture medium.
- Spin down the cells and the harvested culture medium at 1200 RPM (approximately 150-200g) for 8 minutes.
- Discard the supernatant.
- Suspend the cells in 500 μL live/dead dye solution for 30 minutes (5 nM calcein and 400 nM Ethidium-homodimer-2 in PBS) (5×104 cell/ml).
- Transfer cells to flow cytometry tubes.
- Start the CellQuest Pro software.
- To set up the flow cytometer for measurement, prepare control samples of live and dead cells.
- Plate H9c2 cells at a density of 3×104 in 12-well plates as described in point 1.
- Prepare live cell controls with single trypsinisation as described in point 1.
- Prepare dead cell controls by treatment with 10 μM H2O2 for 1 hour.
- After trypsinisation, resuspend live and dead cell controls in 500 μl live/dead dye solution (5 nM calcein-AM and 400 nM Ethidium-homodimer-2 in PBS).
- The instrument settings should be adjusted so the living cells are calcein positive and ethidium homodimer negative, while the dead cells are calcein negative and ethidium homodimer positive.
- The percentage of these populations can be expressed as bars on a graph and statistical analysis can be performed on these values.
- Measure the different experimental groups.
6. Representative results:
Our results showed that added mesenchymal stem cells could improve the survival of postischemic cardiac cells. Confocal microscopy images revealed direct cell-to-cell interactions, such as partial membrane connection or intercellular nanotubes, which probably play an important role in the observed protection10. These nanotubes span over distances of several cell diameters, and their diameters were between 200 and 500 nm (white arrows).
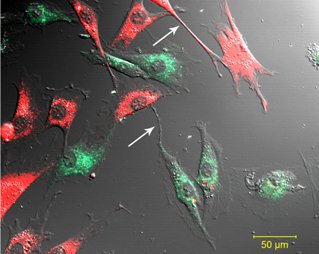
Figure 1. Nanotube formation between cells. Nanotube formation (white arrow) was observed between Vybrant DiO labeled cardiomyoblasts and Vybrant DiD labeled mesenchymal stem cells.
Flow cytometry analysis showed the populations of surviving and necrotic cardiomyoblasts and mesenchymal stem cells. Interestingly, we also observed a third, ‘injured’ cardiomyoblast cell population which contained calcein but also contained the indicator of the dead cells, ethidium homodimer (Figure 2).
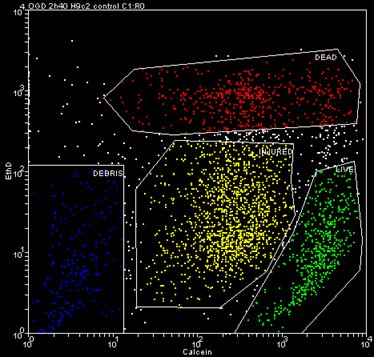
Figure 2. Representative dot-plot of the different cell populations after simulated ischemia.
The evaluation of several experiments showed that addition of mesenchymal stem cell to postischemic H9c2 cardiomyoblast increased significantly the percentage of living cells compared to the cardiac myoblasts cultured alone after ischemia (Figure 3).
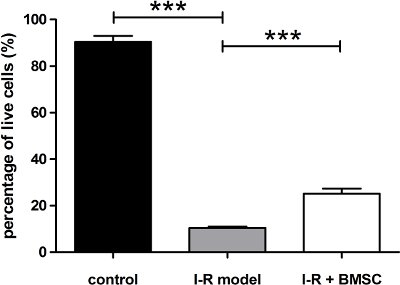
Figure 3. Effect of stem cell treatment on postischemic cardiomyoblasts. Percentage of living cells was significantly reduced after simulated ischemia compared to control (control vs. I-R model: 90.36±2.60 vs. 10.52±0.48, *** p>0.001), which increased after addition of mesenchymal stem cells (I-R model vs. I-R + BMSC: 10.52±0.48 vs. 25.21±2.13, *** p>0.001). Data represent mean±SEM. Statistical analysis of data was carried out using one-way analysis of variance with Tukey’s multiple comparison post hoc test.
Discussion
The demonstrated protocol is an in vitro approach to the much more complex issue of stem cell therapy in myocardial infarct, with all the advantages and disadvantages of such a model. Obviously it cannot reflect the complex (e.g. immunological) events taking place during and after myocardial infarct but can focus on the direct effects of the added cells on the postischemic cells. The effects of simulated ischemia on H9c2 cardiomyoblasts highly depend on the time of OGD, on the passage number of the used cells and on the O2 concentration. One must adjust these parameters accurately and find the optimal conditions for a certain cell type in order to have a standardized protocol. Afterwards, the protocol can be used in many ways (renal, hepatic or cerebral ischemia/reperfusion11,12,13,14) to investigate the effects of different added cell types such as embryonic stem cells, induced pluripotent stem cells or adult stem cells15, the effectiveness of conditioned medium16 or various pretreatments17,18 or any parameter in the focus of interest.
Declarações
The authors have nothing to disclose.
Acknowledgements
This work was supported by OTKA (Hungarian Scientific Research Fund) D45933, T049621, TÉT (Hungarian Science and Technology Foundation) A4/04, Arg-17/2006 and SIN, Bolyai, Öveges Fellowships and TÁMOP 4.2.2-08/1/KMR-2008-0004 and 4.2.1/B 09/1/KMR-2010-0001. OTKA 83803. We would like to thank William Gesztes for providing the voice-over.
Materials
| Name of the reagent | Company | Catalogue number | Comments |
| calcein-AM | Molecular Probes | L3224, C3099 | http://www.invitrogen.com |
| ethidium homodimer-2 | Molecular Probes | L3224, E3599 | http://www.invitrogen.com |
| Vybrant DiD | Molecular Probes | V22887 | http://www.invitrogen.com |
Table 1. Reagents.
| Name of the equipment | Company | Comments (optional) |
| PeCon cell incubation system for Zeiss microscopes | PeCon GmbH | www.pecon.biz/ |
Table 2. Equipment.
Referências
- Dimmeler, S., Burchfield, J., Zeiher, A. M. Cell-Based Therapy of Myocardial Infarction. Arterioscler Thromb Vasc Biol. 28, (2008).
- Kim, S. U., de Vellis, J. Stem cell-based cell therapy in neurological diseases: A review. Journal of Neuroscience Research. 87, 2183-21 (2009).
- Lee, K., Chan, C. K., Patil, N., Goodman, S. B. Cell therapy for bone regeneration-Bench to bedside. Journal of Biomedical Materials Research Part B: Applied Biomaterials. 89, 252-25 (2009).
- Gaipa, G. GMP-based CD133+ cells isolation maintains progenitor angiogenic properties and enhances standardization in cardiovascular cell therapy. Journal of Cellular and Molecular Medicine. 14, 1619-1619 (2010).
- Trounson, A. New perspectives in human stem cell therapeutic research. BMC medicine. 7, 29-29 (2009).
- Mazhari, R., Hare, J. M. Mechanisms of action of mesenchymal stem cells in cardiac repair: potential influences on the cardiac stem cell niche. Nat Clin Pract Cardiovasc Med. 4, S21-S21 (2007).
- Kimes, B. W., Brandt, B. L. Properties of a clonal muscle cell line from rat heart. Experimental Cell Research. 98, 367-367 (1976).
- Sardao, V. A. Vital imaging of H9c2 myoblasts exposed to tert-butylhydroperoxide–characterization of morphological features of cell death. BMC Cell Biol. 8, 11-11 (2007).
- Hescheler, J. Morphological, biochemical, and electrophysiological characterization of a clonal cell (H9c2) line from rat heart. Circulation research. 69, 1476-1476 (1991).
- Cselenyak, A. Mesenchymal stem cells rescue cardiomyoblasts from cell death in an in vitro ischemia model via direct cell-to-cell connections. BMC Cell Biol. 11, 29-29 (2010).
- Sauvant, C. Implementation of an in vitro model system for investigation of reperfusion damage after renal ischemia. Cell Physiol Biochem. 24, 567-567 (2009).
- Namas, R. A. Hypoxia-Induced Overexpression of BNIP3 is Not Dependent on Hypoxia-Inducible Factor 1alpha in Mouse Hepatocytes. Shock. 36, 196-196 (2011).
- Cao, L. Hypoxia/Reoxygenation Up-Regulated the Expression of Death Receptor 5 and Enhanced Apoptosis in Human Hepatocyte Line. Transplantation Proceedings. 38, 2207-2207 (2006).
- Meloni, B. P., Meade, A. J., Kitikomolsuk, D., Knuckey, N. W. Characterisation of neuronal cell death in acute and delayed in vitro ischemia (oxygen-glucose deprivation) models. Journal of Neuroscience Methods. 195, 67-67 (2011).
- Mimeault, M., Hauke, R., Batra, S. K. Stem cells: a revolution in therapeutics–recent advances in stem cell biology and their therapeutic applications in regenerative medicine and cancer therapies. Clinical Pharmacology & Therapeutics. 82, 252-252 (2007).
- Angoulvant, D. Mesenchymal stem cell conditioned media attenuates in vitro and ex vivo myocardial reperfusion injury. J Heart Lung Transplant. 30, 95-95 (2010).
- Lim, Y. J., Zheng, S., Zuo, Z. Morphine Preconditions Purkinje Cells against Cell Death under In Vitro Simulated Ischemia/Reperfusion Conditions. Anesthesiology. 100, 562-562 (2004).
- Guo, J. Estrogen-receptor-mediated protection of cerebral endothelial cell viability and mitochondrial function after ischemic insult in vitro. J Cereb Blood Flow Metab. 30, 545-545 (2010).

