Efficient Differentiation of Mouse Embryonic Stem Cells into Motor Neurons
Summary
We developed a new protocol to improve efficiency of in vitro differentiation of mouse embryonic stem cells into motor neurons. The differentiated ES cells acquired motor neurons features as evidenced by expression of neuronal and motor neuron markers using immunohistochemical techniques.
Abstract
Direct differentiation of embryonic stem (ES) cells into functional motor neurons represents a promising resource to study disease mechanisms, to screen new drug compounds, and to develop new therapies for motor neuron diseases such as spinal muscular atrophy (SMA) and amyotrophic lateral sclerosis (ALS). Many current protocols use a combination of retinoic acid (RA) and sonic hedgehog (Shh) to differentiate mouse embryonic stem (mES) cells into motor neurons1-4. However, the differentiation efficiency of mES cells into motor neurons has only met with moderate success. We have developed a two-step differentiation protocol5 that significantly improves the differentiation efficiency compared with currently established protocols. The first step is to enhance the neuralization process by adding Noggin and fibroblast growth factors (FGFs). Noggin is a bone morphogenetic protein (BMP) antagonist and is implicated in neural induction according to the default model of neurogenesis and results in the formation of anterior neural patterning6. FGF signaling acts synergistically with Noggin in inducing neural tissue formation by promoting a posterior neural identity7-9. In this step, mES cells were primed with Noggin, bFGF, and FGF-8 for two days to promote differentiation towards neural lineages. The second step is to induce motor neuron specification. Noggin/FGFs exposed mES cells were incubated with RA and a Shh agonist, Smoothened agonist (SAG), for another 5 days to facilitate motor neuron generation. To monitor the differentiation of mESs into motor neurons, we used an ES cell line derived from a transgenic mouse expressing eGFP under the control of the motor neuron specific promoter Hb91. Using this robust protocol, we achieved 51±0.8% of differentiation efficiency (n = 3; p < 0.01, Student’s t-test)5. Results from immunofluorescent staining showed that GFP+ cells express the motor neuron specific markers, Islet-1 and choline acetyltransferase (ChAT). Our two-step differentiation protocol provides an efficient way to differentiate mES cells into spinal motor neurons.
Protocol
1. Step 1: Mouse Embryonic Stem (mES) Cell Culture (Timing: 3 days)
1. Plating primary embryonic mouse fibroblasts (PMEF)
- Coat four 100-mm tissue culture dishes with gelatin for PMEF adhesion. Add 8 ml of 0.1% gelatin solution (StemCell Technologies) to each dish and incubate for 30 min at room temperature.
- Remove excess gelatin from dishes. Do not rinse the dishes.
- Dilute one vial of Mitomycin C-inactivated PMEF (Millipore) that contains approx. 5 x 106 cells into 40 ml of PMEF media (see recipe in Table 1) and plate onto four 100-mm tissue culture dishes. The PMEF provide a feeder layer for mES cells.
- PMEF should be plated at least one day before adding mES cell cultures. PMEF can be used up to 1 week after plating.
2. Plating mES Cells
To monitor the efficiency of mES cell differentiation into motor neurons, we used HBG3, an ES cell line derived from a transgenic mouse expressing eGFP under the control of the motor neuron specific promoter Hb9.
- Defrost 1 vial of HBG3 mES cells (approx. 1 x 107, contributed by Dr. Douglas Kerr, Johns Hopkins University) in a 37 °C water bath. Add 5 ml of ES media (see recipe in Table 1) in a 15 ml tube and spin down cells at 800 rpm for 5 min.
- Aspirate supernatant and resuspend mES cells in 20 ml of ES media with freshly added Leukemia inhibitory factor (LIF) at a final concentration of 10 ng/ml. NOTE: LIF should be added on the day of use and is required to maintain the mES cells in an undifferentiated state.
- Remove 10 ml PMEF media from the PMEF in each dish and add 10 ml of the HBG3 ES cell suspension to each dish.
- 24 h after plating, mES cells form small colonies, similar in size to the colony indicated by the arrowhead in Figure 1B. 72 h after plating, colonies increase in size, many being approximately 100 μm in diameter (Figure 1B, indicated by arrows). These cells are ready for differentiation. Media should be replaced daily to maintain proliferation of cells.
2. Step 2: Neural Induction (Timing: 2 days)
To induce differentiation of mES cells into motor neurons, mES cells need to be separated from PMEF and cultivated in a suspension environment. Gelatin coated flasks are used to separate PMEF from mES cells.
- When mES colonies are ready for neural induction (defined as differentiation day 0), coat 2 T150 flasks with 0.1% gelatin (18 ml/flask) for 30 min at room temperature and then remove excess gelatin and wash with 1x PBS three times. NOTE: Each 100-mm dish culture will need one T150-gelatin coated flask to separate PMEF.
- Carefully remove media by aspiration, making sure not to dislodge loosely attached colonies. Add 10 ml PBS briefly and then remove by aspiration to complete wash step.
- To separate mES cells from PMEF, add 0.25% trypsin/EDTA (3 ml/dish, StemCell Technologies) and incubate for 3-5 min at 37 °C until the stem cells and PMEF detach from the plate. Add 15 ml fresh ES media without LIF and pipet cells up and down to break colonies (about 15-20 times). Then, add to a 0.1% gelatin-coated T150 flask and incubate for 30 min at 37 °C. After incubation, PMEF should attach to the gelatinized surface while ES cells should be floating. Collect the floating mES cells into a 50 ml tube.
- Spin down at 800 rpm and resuspend cells in 10 ml of Neural Differentiation Medium (see recipe in Table 1).
- Count the cells using a hemocytometer and then plate approximately 2 x 106 mES cells onto a 100-mm bacterial or suspension culture dish. These plates permit growth of suspension cultures and promote formation of embryoid bodies (EBs).
- On differentiation day 1, mES cells form small floating aggregates (EBs) that are visible under a light microscope. Any carryover PMEF attach to the dish. Transfer the suspension cells and media into a 15 ml tube and centrifuge at low speed (500 rpm) for 3 min. Aspirate media carefully, add 10 ml of fresh Neural Differentiation Medium to the pelleted EBs, and then plate cells in a new bacterial dish. In addition to replenishing the media, this step removes any PMEF that carry over from the prior step.
- On differentiation day 2, EBs are ready to be induced into motor neurons.
3. Step 3: Motor Neuron Specification (Timing: 5 days)
- On differentiation day 2, swirl the culture dish and transfer the media and EBs into a 15 ml tube. Let EBs settle by gravity (~ 10 min) or by centrifugation at low speed (500 rpm) for 3 min.
- Aspirate medium carefully and resuspend EBs in 10 ml of Motor Neuron Differentiation (MND) medium (see recipe in Table 1). Plate EBs in a new bacterial dish and change the media daily for 5 days.
- On differentiation day 7, EBs should be approximately 150 – 200 μm in diameter and should express strong GFP signals (Figure 1C). These EBs are ready to be dissociated and plated onto poly-DL-ornithine/laminin coated dishes or coverslips.
4. Step 4: Preparation of Poly-DL-ornithine/laminin Coated Coverslips (Timing: 2 days)
Two days before dissociating EBs (i.e. on differentiation day 5), prepare poly-DL-ornithine/laminin-coated coverslips.
- Place 12-mm round coverslips (Warner Instruments) on the bottom of a 24-well plate. Dissolve 25 mg poly-DL-ornithine (Sigma-Aldrich) in 25 ml sterile, double distilled water (d2H2O) to obtain a 10X stock solution (1 mg/ml). When ready to use, make a 1/10 dilution of poly-DL-ornithine with d2H2O to obtain concentration of 100 μg/ml. Add 0.5 ml to each well of the 24-well plate and incubate at 4 °C for overnight.
- The following day (differentiation day 6), aspirate poly-DL-ornithine and let the plates and cover slips air dry in a tissue culture hood. Rinse the plate wells with d2H2O three times. Let air dry for another hour.
- Dissolve 1 mg mouse laminin (Millipore) in 5 ml of iced-cold 1X PBS to make 100x stock solution (200 μg/ml). When ready to use, make 1/100 dilution in iced-cold 1X PBS (2 μg laminin/ml). Add 0.5 ml/well in a 24-well plate and incubate in 4 °C for overnight. Before seeding cells, excess laminin should be removed and wells rinsed with 1X PBS two times. Coverslips can be stored in MND medium (0.5 ml/well).
5. Step 5: Axon Elongation (Timing: 2 days)
- On differentiation day 7, collect EBs into a 15 ml tube and allow EBs to settle (approximately 3 min). Aspirate media and re-suspend EBs in PBS. Allow EBs to settle and then aspirate PBS. Repeat this step 3 times. Add 1 ml of Accumax (Millipore) to the EBs, gently mix and incubate for 5 min at room temperature. Then aspirate excess Accumax covering the EBs.
- To dissociate EBs, add 3 ml of MND medium. Pipet up and down 20 times using a 1 ml Eppendorf micropipette (blue tip) to disrupt EBs. Incubate for 2 min at room temperature. Transfer cell suspension to a 12×75 mm tube with cell strainer cap (BD Falcon) to obtain a single cell suspension. Repeat this dissociation step with remaining clumps and cells retained by the filter so as to enrich the yield of single cells. The final single cell suspension will be 6 ml.
- Gently mix this single cell suspension and count cells using a hemocytometer. Dilute cells into MND medium (supplemented with 10 ng/ml of each BDNF, GDNF, CNTF, and NT-3) to a density of 2×105 cells per ml. Add cell suspension (0.5 ml/well) to 24-well plates containing poly-DL-ornithine/laminin coated coverslips (previously prepared as described above). The differentiated cells extend long neurites after 2 days in culture (Figure 1D).
6. Representative Results
Figure 1A shows an outline of the protocol. In step 1, mES cells are cultivated on PMEF and supplemented with LIF to prevent spontaneous differentiation. In general, undifferentiated mES colonies are round and compact, and have clearly defined edges. The colonies are not in contact with one another. Typically, mES cells should be split at a 1:4 ratio every 2~3 days. However, each individual cell line will grow at a different rate and the split ratio must be determined empirically. Arrows in Figure 1B indicate the typical appearance of mES cells after culturing on a PMEF feeder layer for 3 days. These colonies are large (50-100 μm in diameter) but still maintain round and defined edges. At this stage the colonies are ready for differentiation or subculture. An arrowhead in Figure 1B shows a small mES colony that you typically find 24 h after plating. Four days after plating, mES cells become overgrown. They show a flattened appearance and loss of defined boundaries, indicating that they are beginning to differentiate. Such cells are not optimal for differentiation into neuronal cells.
To differentiate mES cells into motor neurons, mES cells need to be cultivated in suspension conditions to allow EB formation. In step 2, ES cells are transferred onto a culture surface without a feeder layer to promote EB formation. In this step, they are incubated in Neural Differentiation medium supplemented with Noggin, bFGF, and FGF-8 for 2 days to direct cells to a neural lineage. Small EBs can be observed under the microscope at day 1 of differentiation. They should be floating in the medium. Carryover PMEF can also be found at day 1 of differentiation and must be removed. These cells adhere to bacterial dishes so are eliminated when EBs are passaged to a new bacterial dish. Thus, by day 2 of differentiation, few or no PMEF should be seen in the culture dish. In step 3, continued transfer of EBs to new dishes should ensure removal of any remaining PMEF. EBs are cultured in Motor Neuron Differentiation (MND) media supplemented with RA and SAG for 5 days to differentiate the cells into motor neurons. During culture, EBs continue to grow in size and can be seen by the naked eye at differentiation day 3~4. Figure 1C shows the microscopic appearance of EBs on differentiation day 7. Unlike the undifferentiated cells, EBs show strong fluorescence due to expression of GFP. These EBs are optimally differentiated and are ready for dissociation. Flow cytometry showed that 51% ± 0.8% of the cells from the dissociated EBs had differentiated into GFP expressing cells5. In step 5, culture of these motor neurons in the presence of GDNF, CNTF, BDNF, and NT3 results in extension of long axonal projections. Figure 1D shows the appearance of differentiated cells 2 days after plating of dissociated EBs. Note long neurites extending from the cell bodies of the plated cells. Immunofluorescent staining shows that the GFP+ cells express the pan-neuronal marker (neurofilament-medium chain) and two motor neuron specific markers, (Islet-1 and choline acetyltransferase, Figure 2).
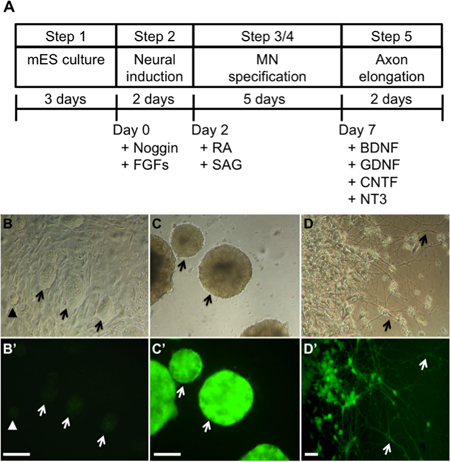
Figure 1. Differentiation of mES cells into motor neurons (picture modified from Wu et al.5). A) Scheme of the differentiation protocol from mES cells to motor neurons. B) Undifferentiated mES cells form round colonies on the top of a fibroblast feeder layer. They have weak GFP expression. C) mES cells were separated from feeder layers and cultured on low-attachment dishes to form EBs. During the first two days of the differentiation process, cells were exposed to 50 ng/ml Noggin, 20 ng/ml bFGF, and 20 ng/ml FGF-8. Subsequently, they were induced to differentiate with 1 μM retinoic acid and 1 μM sonic hedgehog agonist SAG for 5 days. Differentiated mES cells expressed strong GFP fluorescence. D) After 7-day differentiation, EBs were dissociated and plated on poly-DL-ornithine/laminin coated plates. The differentiated cells extended long neural processes after 2 days in culture. Scale bar = 200 μm. MN = motor neuron. B, C and D are bright field images and B’, C’ and D’ are corresponding fluorescence images.
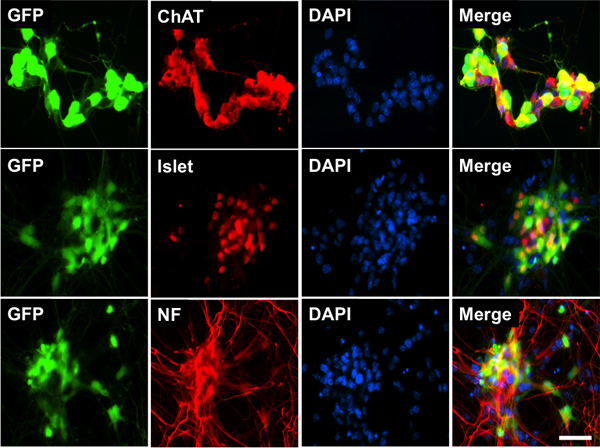
Figure 2. Characterization of mES cell-derived motor neurons. Immunofluorescence staining for neurofilament medium chain (NF-M, red), ChAT (red), and Islet-1 (red) was coincident with GFP (green) expression. DAPI (blue) was used to identify the nuclei. Scale bar = 50 μm.
Discussion
The quality of mES cells is the most critical parameter for efficient generation of motor neurons. mES cells must be cultivated on PMEF to prevent spontaneous differentiation. Addition of LIF helps maintain the mES cells in an undifferentiated state. As mES cells divide every 12-15 h, the culture medium becomes depleted rapidly and must be replaced daily.
Efficient activation of the Shh pathway is a critical parameter needed to induce motor neuron specification. Shh protein (R&D System), and Shh signaling agonists such as Hh agonist HhAg1.3 (Curis, Inc.), purmorphamine (Calbiochem), and SAG (EMD Chemicals) have all been used to promote the formation of motor neurons1-3,10,11. We found SAG to be a more effective molecule than purmorphamine in differentiating cells towards a motor neuronal phenotype5. 1- 2.5 μM purmorphamine with 1 μM RA never gave a differentiation efficiency of more than 20%. However in our hands a combination of 1 μM of RA and 1 μM of SAG gave a differentiation efficiency of approximately 25% in procedures without a neural induction step.
To further enhance differentiation efficiency, we add a 2-day neural induction step prior to the motor neuron specification step. This approach takes advantage of the fact that both Noggin and FGF signaling are crucial for the early stage of neural induction when embryonic cells first enter the neural lineage6-9. In addition to neural induction, FGF signaling maintains cultures as neural precursor cells. Overexpression of Fgf8 in the developing midbrain leads to a dramatic expansion of the neural precursor population in the ventricular zone12. A combination of FGFs and Noggin acts synergistically in inducing neural tissue formation by promoting a posterior neural identity9. Thus the 2-day neural induction step is designed to direct the mES cells towards the neural lineage prior to initiation of the process of motor neuron specification.
The protocol described here is a modification and enhancement of the original established protocol described previously1. Although the timeline of generating motor neurons is the same, we have made a number of modifications to streamline the procedure and improve the yield of motor neurons. By adding a neural induction step to the differentiation protocol, we are able to increase differentiation efficiency from 25% to 50%. Our protocol provides an efficient approach to enrich motor neurons derived from mES cells. Motor neurons cannot be obtained from SMA patients and the poor health of primary motor neurons from SMA mice precludes isolation of cells of sufficient quantity, quality and purity to perform quantitative analyses of proteins affected in this disease. Using this mES cell protocol, we were able to obtain a motor neuron cell population of sufficient quantity and purity for a proteomic study to identify molecular pathways affected in spinal muscular atrophy5.
Declarações
The authors have nothing to disclose.
Acknowledgements
This manuscript is dedicated to the memory of Dr. Wenlan Wang who passed away on May 26, 2011. We thank Dr Douglas A. Kerr for generously providing the HBG3 mES cells used in this study. This work was funded by Nemours, a grant (2 RR016472-10) under the INBRE program of the National Center for Research Resources (NCRR), and a COBRE grant award from the NCRR (5 P20 RR020173-05) to support the Center for Pediatric Research at the Alfred I. duPont Hospital for Children, Wilmington, Delaware, USA.
Materials
| Name of the reagent | Company | Catalogue number | Final concentration |
| PMEF | Millipore | PMEF-H | N.A. |
| PMEF medium | |||
| DMEM | Invitrogen | 11965-118 | N.A. |
| FBS | Invitrogen | 16140-071 | 10% |
| L-glutamine | Invitrogen | 25030-081 | 1% |
| Penicillin/streptomycin | Invitrogen | 15240-063 | 1% |
| mES medium | |||
| DMEM | StemCell Technologies | 36250 | N.A. |
| ES Cell-Qualified FBS | Invitrogen | 16141-079 | 15% |
| GlutaMax-I | Invitrogen | 35050-061 | 1% |
| Non-essential amino acids | Invitrogen | 11140-050 | 1% |
| Nucleosides | Millipore | ES-008-D | 1% |
| β-mercaptoethanol | Millipore | ES-007-E | 0.1 mM |
| Penicillin/streptomycin | Millipore | TMS-AB2-C | 1% |
| LIF | Millipore | LIF2010 | 10 ng/ml |
| Neural Differentiation medium | |||
| DMEM | StemCell Technologies | 36250 | N.A. |
| ES Cult FBS | StemCell Technologies | 06905 | 15% |
| Non-essential amino acids | Invitrogen | 11140-050 | 1% |
| Mono-thio glycerol | Sigma-Aldrich | M-6145 | 1mM |
| Noggin | Invitrogen | PHC1506 | 50 ng/ml |
| FGF-8 | Invitrogen | PHG0274 | 20 ng/ml |
| bFGF | Invitrogen | PHG0024 | 20 ng/ml |
| MND medium (differentiation) | |||
| ES-Cult Basal Medium-A | StemCell Technologies | 5801 | N.A. |
| Knockout serum replacement | Invitrogen | 10828-028 | 10% |
| N-2 supplement | Invitrogen | 17502-048 | 1% |
| ITS Supplement-B | StemCell Technologies | 07155 | 1% |
| Ascorbic acid | StemCell Technologies | 07157 | 1% |
| Penicillin/streptomycin | Millipore | TMS-AB2-C | 1% |
| GlutaMax-I | Invitrogen | 35050-061 | 1% |
| D-glucose | Sigma-Aldrich | G-8270 | 0.15% in d2H2O |
| Fibronectin | StemCell Technologies | 07159 | 5 μg/ml |
| Heparin | Sigma-Aldrich | H3149 | 20 μg/ml in d2H2O |
| β-mercaptoethanol | Millipore | ES-007-E | 0.1 mM |
| Retinoic Acid | Sigma-Aldrich | R-2625 | 1 μM |
| SAG | EMD Chemicals | 566660 | 1 μM |
| MND medium (Motor Neuron culture) | |||
| ES-Cult Basal Medium-A | StemCell Technologies | 5801 | N.A. |
| Knockout serum replacement | Invitrogen | 10828-028 | 10% |
| N-2 supplement | Invitrogen | 17502-048 | 1% |
| ITS Supplement-B | StemCell Technologies | 07155 | 1% |
| Ascorbic acid | StemCell Technologies | 07157 | 1% |
| Penicillin/streptomycin | Millipore | TMS-AB2-C | 1% |
| GlutaMax-I | Invitrogen | 35050-061 | 1% |
| D-glucose | Sigma-Aldrich | G-8270 | 0.15% in d2H2O |
| Fibronectin | StemCell Technologies | 07159 | 5 μg/ml |
| Heparin | Sigma-Aldrich | H3149 | 20 μg/ml in d2H2O |
| β-mercaptoethanol | Millipore | ES-007-E | 0.1 mM |
| BDNF | R&D Systems | 248-BD-005/CF | 10 ng/ml |
| CNTF | R&D Systems | 257-NY-010/CF | 10 ng/ml |
| GDNF | R&D Systems | 212-GD-010/CF | 10 ng/ml |
| NT-3 | R&D Systems | 267-N3-005/CF | 10 ng/ml |
N.A. = Non-applicable.
Table 1. PMEF and Media for mES cell culture or differentiation.
| Name of the reagent | Company | Catalogue number | Final Concentration |
| 0.1% Gelatin | StemCell Technologies | 07903 | N.A. |
| Poly-DL-ornithine | Sigma-Aldrich | P0421 | 0.1 mg/ml in d2H2O |
| Mouse laminin | Millipore | CC095 | 2 μg/ml in PBS |
| 0.25% Trypsin/EDTA | StemCell Technologies | 07901 | N.A. |
| Accumax | Millipore | SCR006 | N.A. |
N.A. = Non-applicable.
Table 2. Reagents for coating and dissociation.
Referências
- Wichterle, H., Lieberam, I., Porter, J. A., Jessell, T. M. Directed differentiation of embryonic stem cells into motor neurons. Cell. 110, 385-397 (2002).
- Miles, G. B. Functional properties of motoneurons derived from mouse embryonic stem cells. J. Neurosci. 24, 7848-7858 (2004).
- Wichterle, H., Peljto, M. Differentiation of mouse embryonic stem cells to spinal motor neurons. Curr. Protoc. Stem Cell. Biol. Chapter 1, Unit 1H.1.1-Unit 1H.1.9 (2008).
- Kiris, E. Embryonic stem cell-derived motoneurons provide a highly sensitive cell culture model for botulinum neurotoxin studies, with implications for high-throughput drug discovery. Stem Cell Res. 6, 195-205 (2011).
- Wu, C. Y. Proteomic assessment of a cell model of spinal muscular atrophy. BMC Neurosci. 12, 25 (2011).
- McMahon, J. A. Noggin-mediated antagonism of BMP signaling is required for growth and patterning of the neural tube and somite. Genes Dev. 12, 1438-1452 (1998).
- Storey, K. G. Early posterior neural tissue is induced by FGF in the chick embryo. Development. 125, 473-484 (1998).
- Launay, C., Fromentoux, V., Shi, D. L., Boucaut, J. C. A truncated FGF receptor blocks neural induction by endogenous Xenopus inducers. Development. 122, 869-880 (1996).
- Sinha, S., Chen, J. K. Purmorphamine activates the Hedgehog pathway by targeting Smoothened. Nat. Chem. Biol. 2, 29-30 (2006).
- Chen, J. K., Taipale, J., Young, K. E., Maiti, T., Beachy, P. A. Small molecule modulation of Smoothened activity. Proc. Natl. Acad. Sci. U.S.A. 99, 14071-14076 (2002).
- Lee, S. M., Danielian, P. S., Fritzsch, B., McMahon, A. P. Evidence that FGF8 signaling from the midbrain-hindbrain junction regulates growth and polarity in the developing midbrain. Development. 124, 959-969 (1997).

