Three-dimensional Cell Culture Model for Measuring the Effects of Interstitial Fluid Flow on Tumor Cell Invasion
Summary
Interstitial fluid flow is elevated in solid tumors and can modulate tumor cell invasion. Here we describe a technique to apply interstitial fluid flow to cells embedded in a matrix and then measure its effects on cell invasion. This technique can be easily adapted to study other systems.
Abstract
The growth and progression of most solid tumors depend on the initial transformation of the cancer cells and their response to stroma-associated signaling in the tumor microenvironment 1. Previously, research on the tumor microenvironment has focused primarily on tumor-stromal interactions 1-2. However, the tumor microenvironment also includes a variety of biophysical forces, whose effects remain poorly understood. These forces are biomechanical consequences of tumor growth that lead to changes in gene expression, cell division, differentiation and invasion3. Matrix density 4, stiffness 5-6, and structure 6-7, interstitial fluid pressure 8, and interstitial fluid flow 8 are all altered during cancer progression.
Interstitial fluid flow in particular is higher in tumors compared to normal tissues 8-10. The estimated interstitial fluid flow velocities were measured and found to be in the range of 0.1-3 μm s-1, depending on tumor size and differentiation 9, 11. This is due to elevated interstitial fluid pressure caused by tumor-induced angiogenesis and increased vascular permeability 12. Interstitial fluid flow has been shown to increase invasion of cancer cells 13-14, vascular fibroblasts and smooth muscle cells 15. This invasion may be due to autologous chemotactic gradients created around cells in 3-D 16 or increased matrix metalloproteinase (MMP) expression 15, chemokine secretion and cell adhesion molecule expression 17. However, the mechanism by which cells sense fluid flow is not well understood. In addition to altering tumor cell behavior, interstitial fluid flow modulates the activity of other cells in the tumor microenvironment. It is associated with (a) driving differentiation of fibroblasts into tumor-promoting myofibroblasts 18, (b) transporting of antigens and other soluble factors to lymph nodes 19, and (c) modulating lymphatic endothelial cell morphogenesis 20.
The technique presented here imposes interstitial fluid flow on cells in vitro and quantifies its effects on invasion (Figure 1). This method has been published in multiple studies to measure the effects of fluid flow on stromal and cancer cell invasion 13-15, 17. By changing the matrix composition, cell type, and cell concentration, this method can be applied to other diseases and physiological systems to study the effects of interstitial flow on cellular processes such as invasion, differentiation, proliferation, and gene expression.
Protocol
1. Assay Set-up
- Thaw a small aliquot (<500 μl) of Matrigel on ice at 4 °C (approximately 2 hr).
- Prepare gel recipe (see example volumes in table below): 10x PBS (1x in total volume), 1N sodium hydroxide (equivalent to 0.023 volumes of added collagen, or per the collagen manufacturer’s recommendations, as appropriate), Matrigel and collagen type I to final concentrations of 1 mg/ml and 1.3 mg/ml respectively (other matrix formulations may be used depending on cell type and experiment).
Example gel recipe
| Components | Stock Concentration | Final Concentration | Add volume |
| 10X PBS | 10X | 1X | 0.090 ml |
| Sterile water | 0.346 ml | ||
| 1N NaOH | 0.008 ml | ||
| Matrigel | 9.90 mg/ml | 1 mg/ml | 0.101 ml |
| Rat tail type I collagen | 3.66 mg/ml | 1.3 mg/ml | 0.355 ml |
- Mix components of the gel on ice in the same sequence as above and incubate final solution for 1 hr at 4 °C. In our experience, a 1 hr incubation prior to cell seeding results in a more uniform gelation of the collagen. Make sure to keep Matrigel and collagen on ice at all times and to work as fast as possible to prevent gelation.
- Place 12 mm diameter 8 μm pore cell culture inserts into a 12-well plate using sterilized forceps.
- Count cells and resuspend in serum-free media at 5 x 106 cells/ml (total volume should be 10% of the gel solution).
- Add 100 μl of cell suspension to 900 μl of gel solution (final cell concentration 5 x 105 cells/ml) and mix thoroughly by pipetting gently up and down.
- Add 150 μl of the final mixture to each insert and transfer to a 37 °C, 5% CO2 incubator for 30 minutes until gel polymerizes.
- Using serum-free media,
- Add 100 μl on top of the gel and 1200 μl under the insert for the STATIC condition. The fluid levels inside the insert and outside in the well should be approximately equal, resulting in minimal pressure difference across the gel and no interstitial flow.
- Add 100 μl under the insert and 650 μl above gel for the FLOW condition. Be careful to avoid any air bubbles beneath the insert, as they will prevent cells from migrating through the membrane at these locations. The pressure difference generated by these volumes is approximately 1.3 cm H2O (or 1 mm Hg).
- Place plate in a 37 °C, 5% CO2 incubator for 24 hr.
2. Cell Staining and Counting
- Add 500 μl of 1X PBS per well into new 24-well plate; this will be used to wash the inserts.
- Remove the medium remaining in the upper portion of the flow transwells and determine the volume. Calculate total eluted volume by subtracting the remaining volume from the total volume originally added, 650 μl (this will be used for flow rate calculations, see below). Use cotton swabs to remove the gel from the inserts and to wipe the top surface of the membrane to remove non-invaded cells. Place the inserts into the 24-well plate containing 1X PBS for 15 s to wash inserts.
- Remove PBS and add 500 μl of 4% paraformaldehyde (PFA) underneath each insert and incubate for 30 minutes at room temperature to fix the transmigrated cells.
- Remove the PFA and rinse once with 500 μl 1X PBS to remove residual fixative.
- Add 500 μl of 0.5% Triton X-100 solution beneath the inserts and incubate for 10 minutes at room temperature to permeabilize the cells.
- Cut membranes out of the insert using a razor blade and place into 100 μl of 2 μg/ml DAPI in 1X PBS solution, being careful to place the underside of the membrane face down (transmigrated cells face down).
- Wrap plate in aluminum foil and place on a shaker at 150 rpm for 30 minutes at room temperature.
- Wash membranes in 500 μl 1X PBS on shaker (repeat 3 times for 10 minutes each) to remove free DAPI.
- Place membranes on glass slides with transmigrated cells facing up, add mounting solution and cover slip.
3. Data Analysis
- Count DAPI-stained nuclei on 5 randomly selected locations of each membrane (stay away from the edges and use a 10X or 20X objective lens).
- Calculate the average cell count and obtain the percent invasion by using the following formula:
Percent invasion = 100% x (average cell count x membrane surface area) / (surface area of the microscope image x number of cells seeded)
The percent invasion can be normalized to some control condition (typically the static condition) to allow for comparison between independent experiments. - Calculate the average flow rate: divide the total eluted volume in the flow condition by the incubation time (e.g., 24 hr).
- Calculate the average flow velocity: divide the average flow rate by the cross-sectional area of the gel/membrane (in this case 60 mm2).
4. Representative Results
To measure tumor cell invasion under interstitial flow, we performed our 3-D flow invasion assay using MDA-MB-435S metastatic melanoma cells. These cells have previously been shown to invade in response to interstitial fluid flow 13-14. The cells were embedded in a matrix composed of 1.3 mg/ml rat tail tendon collagen type I and 1 mg/ml Matrigel basement membrane matrix at a final cell concentration of 5 x 105 cells/ml. Two different conditions were compared: (1) average interstitial flow = 0.1 μm s-1 and (2) static condition = no measurable flow rate (Figure 1).
After 24 hr, the cells that invaded through the pores of the membrane were stained with DAPI to facilitate cell counting. Figure 2 shows a representative image of the invaded cells. Under brightfield, only the pores of the membrane are visible. Using fluorescence, the DAPI-stained nuclei were used for cell counting and the phalloidin stained F-actin structures were used to visualize the cell body (optional). Using a Student’s t-test assuming equal variances, we showed that interstitial flow significantly increases MDA-MB-435S cell invasion by 2.3-fold over static conditions (p = 0.003) (Figure 3). This corroborates similar findings (but with different matrix conditions and therefore flow velocities) using this cell line 13-14.
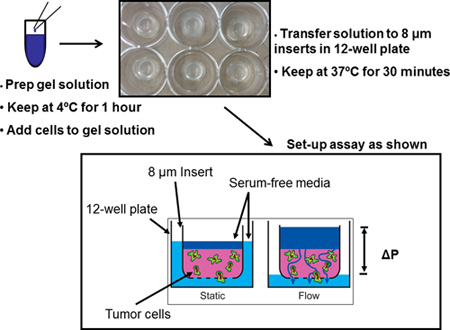
Figure 1. Schematic of the 3-D interstitial fluid flow invasion assay. First prepare gel solution using appropriate concentrations and volumes. Then add cells to gel solution and transfer to cell culture inserts. Finally add appropriate volume of media to each condition and incubate. Interstitial fluid flow is driven by a fluid pressure head.
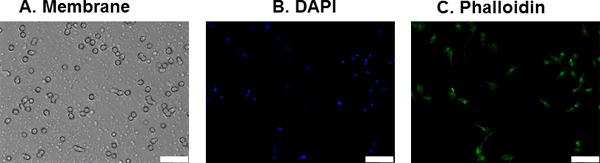
Figure 2. Transmigrated MDA-MB-435S cells on membrane. Invaded cells were fixed after our interstitial fluid flow invasion assay and stained with DAPI and Alexa Fluor 488-conjugated phalloidin to facilitate counting of invaded cells; A) Picture of the membrane under bright field; B) DAPI stained nuclei (in blue); C) Alexa Fluor 488-phalloidin stained F-actin (in green). Scale bar represents 50 μm.
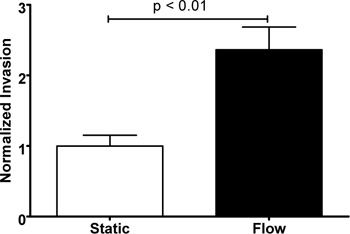
Figure 3. Increased invasion of MDA-MB-435S cells under flow. Cell invasion after 24 hr significantly enhanced by interstitial flow (p = 0.003). The results are normalized to the average static condition and values represent mean ± SEM of 6 cell culture inserts.
Discussion
Here we have described a methodology for quantifying the effect of interstitial flow on tumor cell invasion, using cells embedded in a 3-D matrix within a cell culture insert. This and similar methods have been used to study the effect of interstitial flow on a variety of cell types 13-15, 17. Our approach partially mimics the matrix microenvironment of the tumor by using type I collagen and Matrigel which contain proteins found in the basement membrane of epithelial tissue and the surrounding stroma 21-22. This system is relatively easy to set-up, straightforward, and more cost effective than most microfluidic devices that are used to study interstitial fluid flow in vitro 23-24. It does not require pumps or specialized equipments and allows for multiple conditions to be tested simultaneously. Furthermore, the measurements are comparable to those of existing Boyden chamber assays commonly used to test invasion and migration of cancer cells.
By changing the cell type and the composition of the matrix, different biological systems can be modeled, such as breast cancer 13, blood vessels 15, and dendritic cell trafficking 17. Altering the matrix properties and composition and modulating the pressure head will also change the interstitial fluid flow velocity, thus allowing for flexibility in the assay parameters dependent on the biological system of interest. This system can also be used in co-culture assays.14, 17
In addition to measuring invasion, the interstitial fluid flow assay described here could be expanded to measure changes in other cell behaviors such as protein expression, cell proliferation and differences in cell signaling events. The cells can easily be isolated directly from the gel for RNA, DNA, and protein extraction and used for subsequent molecular biology assays, such as PCR and western blot. This is a highly flexible assay that can be easily set up and adapted to investigate the effects of interstitial flow on a range of cellular processes in a number of cell systems.
One of the main drawbacks of this assay is the fact that flow velocities are very dependent of matrix concentration. Flow velocity increases as Matrigel concentration decreases. This means that a matrix consisting solely of collagen, which can be used in the study of stroma associated cells, will have higher flow velocities (> 1 μm s-1) than one consisting mainly of Matrigel (< 0.05 μm s-1) for a given pressure differential. If a specific flow velocity is required, one can first perform an initial test to identify the matrix composition that provides the desired flow velocity. The thickness of the gel can also be varied to adjust the flow velocity. The assay also does not allow for live cell imaging, where the behavior of individual cells and changes in their speed and directionality can be measured. Instead, this assay provides an endpoint measurement of changes in invasion across a population of cells.
Declarações
The authors have nothing to disclose.
Materials
| Name of the reagent | Company | Catalogue number | Comments |
| Collagen (Rat Tail) | BD | 354236 | Keep sterile |
| Millicell cell culture insert | Millipore | PI8P01250 | 8 μm pore diameter, polycarbonate membrane |
| Matrigel | BD | 354234 | Keep sterile |
| PBS | Sigma Aldrich | 100M-8202 | 10x for preparing gel solution, 1x for washing steps |
| Sodium Hydroxide, 1.0N Solution | Sigma Aldrich | S2770 | Keep sterile |
| DMEM 1X | CellGro | 10-013-CV | Keep sterile |
| Fetal Bovine Serum | Atlanta Biologicals | 511150 | Keep sterile |
| Penicillin Streptomycin | CellGro | 30002CI | Keep sterile |
| Triton X-100 | Sigma Aldrich | X100-500 ml | 0.5% in PBS |
| Paraformaldehyde | Fisher Scientific | 04042-500 | 4% in PBS |
| Deionized Water | Keep sterile | ||
| 4′,6-diaminido-2-phenylindole (DAPI) | MP Biomedicals | 0215757401 | 1 mg/ml stock solution |
| Mounting Solution | Thermo Scientific | TA-030-FM | |
| Trypsin-EDTA | CellGro | 25-052-CI | Keep sterile |
Referências
- Cichon, M. A. Microenvironmental influences that drive progression from benign breast disease to invasive breast cancer. J. Mammary Gland. Biol. Neoplasia. 15, 389-3897 (2010).
- Proia, D. A., Kuperwasser, C. Stroma: tumor agonist or antagonist. Cell Cycle. 4, 1022-1025 (2005).
- Dvorak, H. F. Tumor microenvironment and progression. J .Surg. Oncol. 103, 468-474 (2011).
- Provenzano, P. P. Collagen density promotes mammary tumor initiation and progression. BMC Med. 6, 11 (2008).
- Engler, A. J. Matrix elasticity directs stem cell lineage specification. Cell. 126, 677-689 (2006).
- Paszek, M. J. Tensional homeostasis and the malignant phenotype. Cancer Cell. 8, 241-254 (2005).
- Levental, K. R. Matrix crosslinking forces tumor progression by enhancing integrin signaling. Cell. 139, 891-906 (2009).
- Butler, T. P., Grantham, F. H., Gullino, P. M. Bulk transfer of fluid in the interstitial compartment of mammary tumors. Cancer Res. 35, 3084-3088 (1975).
- Dafni, H. Overexpression of vascular endothelial growth factor 165 drives peritumor interstitial convection and induces lymphatic drain: magnetic resonance imaging, confocal microscopy, and histological tracking of triple-labeled albumin. Cancer Res. 62, 6731-6739 (2002).
- Chary, S. R., Jain, R. K. Direct measurement of interstitial convection and diffusion of albumin in normal and neoplastic tissues by fluorescence photobleaching. Proc. Natl. Acad. Sci. U.S.A. 86, 5385-5389 (1989).
- Heldin, C. H. High interstitial fluid pressure – an obstacle in cancer therapy. Nat. Rev. Cancer. 4, 806-813 (2004).
- Fukumura, D. Tumor microvasculature and microenvironment: novel insights through intravital imaging in pre-clinical models. Microcirculation. 17, 206-225 (2010).
- Shields, J. D. Autologous chemotaxis as a mechanism of tumor cell homing to lymphatics via interstitial flow and autocrine CCR7 signaling. Cancer Cell. 11, 526-538 (2007).
- Shieh, A. C. Tumor cell invasion is promoted by interstitial flow-induced matrix priming by stromal fibroblasts. Cancer Res. 71, 790-800 (2011).
- Shi, Z. D., Wang, H., Tarbell, J. M. Heparan sulfate proteoglycans mediate interstitial flow mechanotransduction regulating MMP-13 expression and cell motility via FAK-ERK in 3D collagen. PLoS One. 6, e15956 (2011).
- Fleury, M. E., Boardman, K. C., Swartz, M. A. Autologous morphogen gradients by subtle interstitial flow and matrix interactions. Biophys J. 91, 113-121 (2006).
- Miteva, D. O. Transmural flow modulates cell and fluid transport functions of lymphatic endothelium. Circ. Res. 106, 920-931 (2010).
- Ng, C. P., Hinz, B., Swartz, M. A. Interstitial fluid flow induces myofibroblast differentiation and collagen alignment in vitro. J. Cell. Sci. 118, 4731-4739 (2005).
- Kunder, C. A. Mast cell-derived particles deliver peripheral signals to remote lymph nodes. J. Exp. Med. 206, 2455-2467 (2009).
- Helm, C. L. Synergy between interstitial flow and VEGF directs capillary morphogenesis in vitro through a gradient amplification mechanism. Proc. Natl. Acad. Sci. U.S.A. 102, 15779-15784 (2005).
- McGuire, P. G., Seeds, N. W. The interaction of plasminogen activator with a reconstituted basement membrane matrix and extracellular macromolecules produced by cultured epithelial cells. J Cell Biochem. 40, 215-227 (1989).
- Kleinman, H. K. Isolation and characterization of type IV procollagen, laminin, and heparan sulfate proteoglycan from the EHS sarcoma. Bioquímica. 21, 6188-6193 (1982).
- Haessler, U. Migration dynamics of breast cancer cells in a tunable 3D interstitial flow chamber. Integr. Biol. (Camb). , (2011).
- Polacheck, W. J., Charest, J. L., Kamm, R. D. Interstitial flow influences direction of tumor cell migration through competing mechanisms. Proc. Natl. Acad. Sci. U.S.A. 108, 11115-11120 (2011).

