Extraction of Tissue Antigens for Functional Assays
Summary
A simple protocol for preparing extracts of human tissue to be used as a source of antigens in functional T-cell assays is described. This method allows T-cell responses to tissue-derived antigens to be measured in vitro.
Abstract
Many of the antigen targets of adaptive immune response, recognized by B and T cells, have not been defined 1. This is particularly true in autoimmune diseases and cancer2. Our aim is to investigate the antigens recognized by human T cells in the autoimmune disease type 1 diabetes 1,3,4,5. To analyze human T-cell responses against tissue where the antigens recognized by T cells are not identified we developed a method to extract protein antigens from human tissue in a format that is compatible with functional assays 6. Previously, T-cell responses to unpurified tissue extracts could not be measured because the extraction methods yield a lysate that contained detergents that were toxic to human peripheral blood mononuclear cells. Here we describe a protocol for extracting proteins from human tissues in a format that is not toxic to human T cells. The tissue is homogenized in a mixture of butan-1-ol, acetonitrile and water (BAW). The protein concentration in the tissue extract is measured and a known mass of protein is aliquoted into tubes. After extraction, the organic solvents are removed by lyophilization. Lyophilized tissue extracts can be stored until required. For use in assays of immune function, a suspension of immune cells, in appropriate culture media, can be added directly to the lyophilized extract. Cytokine production and proliferation by PBMC, in response to extracts prepared using this method, were readily measured. Hence, our method allows the rapid preparation of human tissue lysates that can be used as a source of antigens in the analysis of T-cell responses. We suggest that this method will facilitate the analysis of adaptive immune responses to tissues in transplantation, cancer and autoimmunity.
Protocol
1. Preparing Spleen Tissue
- Note- all human material should be treated as potentially infectious and all procedures should be conducted in a Class II Laminar Flow Cabinet. Using sterile scissors and forceps, remove fat and fibrous tissue from spleen sections (~1-2 cm in size) and trim off as much of the outer capsule material as possible.
- Cut a small piece (1-2 cm3) of spleen tissue and place each piece into a sterile 50 ml Falcon tube.
- Snap-freeze the pieces of tissue by immersion in liquid N2.
- Store at -80 °C. A similar protocol is suitable for other tissue(s).
2. Preparing Human Islet for Storage
- Culture islets in CMRL media. Collect islets in a 10 ml conical bottom tube and wash twice in PBS by centrifuging at 1,500 rpm for 5 min. Pour off the PBS and drain the residual buffer by placing the inverted tube briefly on a paper towel. Be careful not to dislodge the islets.
- Once drained, re-cap the tube and snap-freeze in liquid nitrogen and store at -80 °C.
3. Preparing Extract
- Prepare BAW mix (10:30:60 %v/v) and store at 4 °C.
- Remove the tube from -80 °C. Thaw at room temperature.
- Add sufficient ice-cold BAW to cover the piece of tissue. For islets use 3-5 ml. For spleen tissue use 10-20 ml depending upon the size of the piece of tissue.
- Assemble the tissue homogenizer. Clean by ‘homogenizing’ 10-20 ml of 70% ethanol/water.
- Homogenize the tissue in multiple bursts, after placing the homogenizer probe into the tube with the tissue and BAW solution. Keep the tube in an ice-bucket thereafter.
- Thoroughly clean the homogenizer between samples, by homogenizing 10-20 ml of 70% ethanol/water and then BAW buffer. This prevents cross-contamination of tissue between samples.Dismantleand clean with 70% ethanol/water after use.
- If an extract containing only soluble material is required, centrifuge homogenized tissue extract at 4,000 rpm at RT for 10 min. If a more crude extract is required, spin at 1,000 rpm at RT for 5 min. The most appropriate technique for extracting the BAW-insoluble proteins depends upon the downstream analysis of the proteins. For use in functional immunological assays we would suggest attempting to dissolve the insoluble fraction in 8 M urea as we have previously found this to be well tolerated6.
- Transfer supernatant to a clean tube and put it on ice.
- Determine the concentration of protein in the extract using a BCA assay or similar.
4. Freeze Drying Extracts
- Depending on the mass of protein required (i.e., 100 μg per tube), dilute the homogenate accordingly and dispense aliquots into labeled 5.0 ml sterile, Falcon (12×75 mm) tubes. We frequently use 100 μg/tube.
- Use an 18-20 gauge sterile syringe needle to make 3 holes in the cap of each tube.
- Freeze tubes either by placing them on dry ice for ~10 min or in a -80 °C freezer for >1 hr. Store at -80 °C until ready to put on the lyophilizer.
- Switch on freeze-drier and allow to equilibrate (-100 °C). This takes about 30 min.
- Depending upon the volume, freeze-drying can be completed within 3 hr but we routinely leave our samples overnight.
- After completion of the drying cycle, switch off vacuum pump and slowly allow pressure into the chamber. Remove rack.
- In a sterile hood, remove the perforated caps from the tubes and replace with new ones (Falcon 352032).
- Store tubes at -20 °C. Samples can be reconstituted in culture media, or other buffers, and used in function or biochemical assays.
5. Representative Results
Figure 1 shows the staining of a protein gel loaded with extract from spleen and islet depleted pancreatic tissue (labeled acinar) and purified human islets (labeled islets). The results show a good representation of proteins of different molecular weight for each tissue.
The capacity of tissue extracts to stimulate human T-cell proliferation was tested using a CFSE-based proliferation assay7 (Figure 2). The PBMC used in this assay were isolated from an individual with type 1 diabetes. The magnitude of the response is expressed as a ratio of the number of CFSEdim cells per 5,000 CD4+, CFSEbright cells without antigen: of CFSEdim cells per 5,000 CD4+, CFSEbright cells with antigen from triplicate samples7. The results show a weak, but detectable, proliferation in response to acinar (CD1=3.5) and a stronger response to islet extract (6.8). Inactivated influenza virus (CDI= 142.6) is included as a positive control.
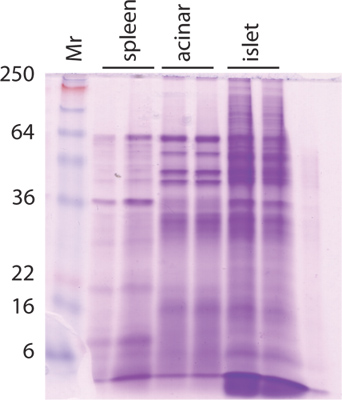
Figure 1. Protein gel.
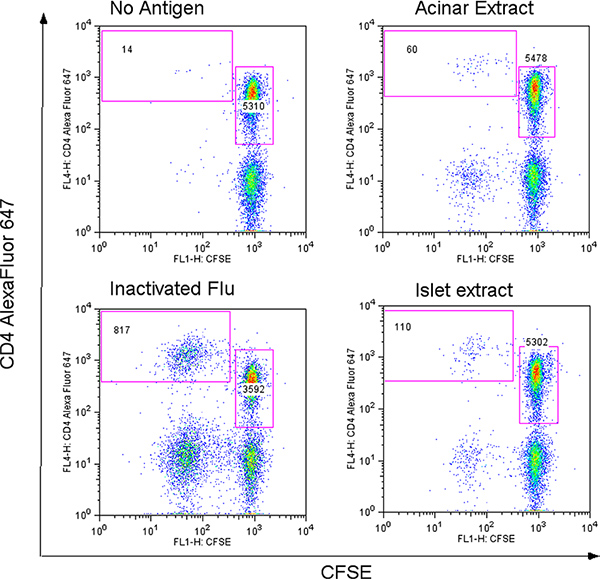
Figure 2. Results from a CFSE-based proliferation assay against acinar and islet extract. Click here to view larger figure.
Discussion
This protocol was developed because we wanted to generate an extract from human tissue that was free from toxic chemicals such as detergents. Specifically we have used it to prepare extracts of human tissue that can be used in assays of human immune function in vitro. Extracts prepared using this protocol can equally be reconstituted in any buffer and used for many biochemical analyses, such as western blotting or liquid chromatography. This makes this technique applicable to many downstream applications.
Using our protocol the response to tissue extracts are not strong. This is expected because we are looking for responses to ‘self’ antigens; in our case T-cell responses against islet antigens are frequently weak 1. Previously we found that human CD4+ T cell responses to recombinant proinsulin and glutamic acid decarboxylase (GAD), autoantigens in type 1 diabetes, could be detected using our CFSE-based proliferation assay 7,8. We have chosen to use tissue extracts to avoid problems associated with using synthetic peptides 9 and recombinant proteins 10.
We do not routinely add protease inhibitors to our extractions. The presence of protease inhibitors may inhibit antigen processing and presentation 11 and consequently inhibit T-cell responses. Instead we perform the extraction on ice in an attempt to prevent protease-mediated degradation. For other applications the inclusion of protease inhibitors may be beneficial if protein degradation is a problem.
Declarações
The authors have nothing to disclose.
Acknowledgements
This work is supported by grants from the Australian National Health and Medical Research Council (NHMRC #559007) and the Juvenile Diabetes Research Foundation (JDRF 4-2006-1025) and the Operational Infrastructure Scheme of the Victorian Government. We thank the members of the Tom Mandel Islet Transplantation Program Islet Isolation Team for providing the human tissues. Human tissues were collected and used with local ethical approval (St. Vincent’s Hospital HREC-A 011/04 and St. Vincent’s Health HREC-A 135/08).
Materials
| Name of the reagent | Company | Catalogue number | Comments |
| 5 ml 12 x 75 mm sterile polystyrene tubes | BD Falcon | 352054 | |
| Caps for tubes polystyrene tubes (above) | BD Falcon | 352032 | |
| 50ml sterile tubes | Becton Dickinson | 352070 | |
| Acetonitrile | Mallinckradt Chemicals | 2856-10 | |
| Butan-1-ol | Sigma Aldrich | 537993-IL | |
| Homogenizer: PRO200 | Bio-strategy | 01-01200 | 10 x 115 mm saw-tooth generator |
| Lyophilizer | Virtis, Benchtop 4K | ||
| Sterile Needle 18-20 gauge | Becton Dickinson | REF 302032 | |
| CMRL-1066 Medium | Sigma | C0422 | |
| PBS | Sigma | D8537 |
Table 1. Specific reagents and equipment.
Referências
- Mannering, S. I. Current approaches to measuring human islet-antigen specific T cell function in type 1 diabetes. Clin. Exp. Immunol. 162, 197-209 (2010).
- Beckhove, P. Rapid T cell-based identification of human tumor tissue antigens by automated two-dimensional protein fractionation. J. Clin. Invest. 120, 2230-2242 (2010).
- Mannering, S. I., Brodnicki, T. C. Recent insights into CD4+ T-cell specificity and function in Type 1 diabetes. Expert Review of Clinical Immunology. 3, 557-564 (2007).
- Mannering, S. I. The A-chain of insulin is a hot-spot for CD4+ T cell epitopes in human type 1 diabetes. Clin. Exp. Immunol. 156, 226-231 (2009).
- Mannering, S. I. The insulin A-chain epitope recognized by human T cells is posttranslationally modified. J. Exp. Med. 202, 1191-1197 (2005).
- Moon, H. C., Joffe, M., Thomas, H. E., Kay, T. W. H., Mannering, S. I. A method for extracting tissue proteins for use in lymphocyte function assays. Journal of Immunological Methods. 359, 56-60 (2010).
- Mannering, S. I. A sensitive method for detecting proliferation of rare autoantigen-specific human T cells. J. Immunol. Methods. 283, 173-183 (2003).
- Mannering, S. I. CD4+ T Cell Proliferation in Response to GAD and Proinsulin in Healthy, Pre-diabetic, and Diabetic Donors. Ann. N.Y. Acad. Sci. 1037, 16-21 (2004).
- Mannering, S. I., Purcell, A. W., Honeyman, M. C., McCluskey, J., Harrison, L. C. Human T-cells recognise N-terminally Fmoc-modified peptide. Vaccine. 21, 3638-3646 (2003).
- Peakman, M. Characterization of preparations of GAD65, proinsulin, and the islet tyrosine phosphatase IA-2 for use in detection of autoreactive T-cells in type 1 diabetes: report of phase II of the Second International Immunology of Diabetes Society Workshop for Standardization of T-cell assays in type 1 diabetes. Diabetes. 50, 1749-1754 (2001).
- Honey, K., Rudensky, A. Y. Lysosomal cysteine proteases regulate antigen presentation. Nat. Rev. Immunol. 3, 472-482 (2003).

