Serial Enrichment of Spermatogonial Stem and Progenitor Cells (SSCs) in Culture for Derivation of Long-term Adult Mouse SSC Lines
Summary
A simple method to derive and maintain spermatogonial stem and progenitor cell lines from adult mice is presented here. The method utilizes feeder cells originating from the somatic cell compartment of the adult mouse testis. This technique is applicable to common mouse strains, including transgenic, knock-out, and knock-in mice.
Abstract
Spermatogonial stem and progenitor cells (SSCs) of the testis represent a classic example of adult mammalian stem cells and preserve fertility for nearly the lifetime of the animal. While the precise mechanisms that govern self-renewal and differentiation in vivo are challenging to study, various systems have been developed previously to propagate murine SSCs in vitro using a combination of specialized culture media and feeder cells1-3.
Most in vitro forays into the biology of SSCs have derived cell lines from neonates, possibly due to the difficulty in obtaining adult cell lines4. However, the testis continues to mature up until ~5 weeks of age in most mouse strains. In the early post-natal period, dramatic changes occur in the architecture of the testis and in the biology of both somatic and spermatogenic cells, including alterations in expression levels of numerous stem cell-related genes. Therefore, neonatally-derived SSC lines may not fully recapitulate the biology of adult SSCs that persist after the adult testis has reached a steady state.
Several factors have hindered the production of adult SSC lines historically. First, the proportion of functional stem cells may decrease during adulthood, either due to intrinsic or extrinsic factors5,6. Furthermore, as with other adult stem cells, it has been difficult to enrich SSCs sufficiently from total adult testicular cells without using a combination of immunoselection or other sorting strategies7. Commonly employed strategies include the use of cryptorchid mice as a source of donor cells due to a higher ratio of stem cells to other cell types8. Based on the hypothesis that removal of somatic cells from the initial culture disrupts interactions with the stem cell niche that are essential for SSC survival, we previously developed methods to derive adult lines that do not require immunoselection or cryptorchid donors but rather employ serial enrichment of SSCs in culture, referred to hereafter as SESC2,3.
The method described below entails a simple procedure for deriving adult SSC lines by dissociating adult donor seminiferous tubules, followed by plating of cells on feeders comprised of a testicular stromal cell line (JK1)3. Through serial passaging, strongly adherent, contaminating non-germ cells are depleted from the culture with concomitant enrichment of SSCs. Cultures produced in this manner contain a mixture of spermatogonia at different stages of differentiation, which contain SSCs, based on long-term self renewal capability. The crux of the SESC method is that it enables SSCs to make the difficult transition from self-renewal in vivo to long-term self-renewal in vitro in a radically different microenvironment, produces long-term SSC lines, free of contaminating somatic cells, and thereby enables subsequent experimental manipulation of SSCs.
Protocol
1. Preparation of Feeder Cells
Note that all reagents described below should be prepared in sterile fashion (see Tables 1 & 2). This protocol employs the JK1 cell line (Cell Biolabs, Inc., catalog #CBA-315) as feeders which is a transformed derivative of adult mouse testicular somatic cells and has been described elsewhere3. Note also that all animal procedures should be performed in accordance with institutional guidelines and regulations.
- JK1 cells can be maintained in culture and used successfully as feeders up to passage 39. Culture JK1 cells in a 100 mm cell culture dish (BD Falcon, catalog #353003) with 10 ml of filter-sterilized feeder growth medium (DMEM supplemented with 10% fetal bovine serum [FBS], 2 mM L-glutamine and antibiotics). When the culture reaches 95% confluence, split the cells 1:6 -1:10 ratio.
- Remove medium from confluent JK1 feeder cell monolayer.
- Wash cell layer once with PBS without calcium and magnesium.
- Add pre-warmed trypsin/EDTA, 1x (0.05% trypsin/0.53 mM EDTA; Corning Cellgro, catalog #25-051-CI), and incubate at 37 °C for 5-10 min.
- Inactivate trypsin with an equal volume of feeder growth medium. Collect cells and centrifuge at 300 x g for 5 min. Re-suspend cells in feeder growth medium.
- Pre-coat multi-well cell culture plates by adding enough 0.4% gelatin solution to cover the well and remove after 5 min at room temperature.
- Plate ~250 cells per mm2 in gelatin-coated cell culture plate (e.g. 48-well or 6-well format). Incubate culture at 37 °C for 16 to 24 hr.
- Remove culture medium from wells. CAUTION: To inactivate cell growth, add a fresh solution of mitomycin-C at 10 μg/ml diluted in DMEM to each well (e.g. 200 μl/well for a 48 well plate). Mitomycin-C is toxic. Handle and dispose of it with care. Incubate at 37 °C for 4 hr.
- Remove mitomycin-C solution from cells. Wash 3 times with DMEM prior to plating stem cells.
- Remove DMEM from feeder cells wells and add enough stem cell medium (100 μl/ well of a 48 well plate; see Table 3) to prevent desiccation of the feeders during passaging. Add SSCs the same day, as described below in sections 3.1 or 3.4, using indicated sizes of wells containing feeders.
2. Dissociation of Mouse Testis Tissue to Obtain Donor Germ Cells
- Harvest adult mouse testes in sterile fashion and temporarily store the testes in a covered Petri dish (without any liquid added). The dish must placed immediately on ice to prevent dessication and maintain cell viability.
- Using sterile fine forceps and a fine scissor in a cell culture hood, make a transverse incision in the tunica albuginea without completely transecting the testis (i.e., cut ~½-¾ distance through) and use forceps to squeeze out seminiferous tubules into a corner of the plate; discard tunica and attached tissue. Keep plate on ice throughout to keep tissue chilled, thereby maintaining its integrity and preventing evaporation.
- Rapidly mince tubules with fine spring scissors at least 3 min per testis, while keeping plate on ice.
- Collect the tubule fragments in a 50 ml conical tube and wash in ~40 ml of chilled PBS/1% bovine serum albumin.
- Centrifuge at 60 x g for 10 min to separate tubule fragments from spermatozoa and debris. Discard supernatant.
- In a 15 ml conical tube, resuspend the pellet in 3 ml per testis of pre-warmed dissociation buffer (DMEM, 0.05% Trypsin, 0.03% Collagenase type I, 80 U/ml of DNAse I and 0.5% bovine serum albumin; See Table 2 for more specific details of dissociation buffer).
- Place the conical tube horizontally in a rack in a shaker set to 150 rpm at 37 °C for 15 min to maximize agitation.
- Spin down at 60 x g for 10 min to separate single cells from undissociated chunks.
- Save the pellet containing chunks until the next step. Collect supernatant from Step 2.8 and add 3 ml DMEM/10% fetal bovine serum to neutralize dissociation buffer. Place tube on ice temporarily.
- Repeat step 2.6 to 2.7 with the saved pellet from step 2.9 to re-digest chunks remaining after step 2.8.
- Add an equal volume of DMEM/10% FBS to neutralize dissociation buffer.
- Recombine suspension from step 2.11 with saved cell suspension from step 2.9. Centrifuge for 5 min at 300 x g.
- Resuspend pellet in stem cell medium in a volume of 16 ml per pair of testes (see Table 3).
3. Plating of Testicular Cell Suspension
- Plate 2 ml of cell suspension per well in a 6-well plate containing feeder cells mitotically-inactivated with mitomycin-C and place in a cell culture incubator at 37 °C in 5% CO2.
- After 48 hr, aspirate medium, add 2 ml fresh medium and return to incubator.
- After 48 hr, add 0.5 ml fresh stem cell medium to each well and return to incubator.
- After 48 hr, subsequently feed cells three times per week thereafter as follows until large, discreet colonies (>50 cells) appear (within 7 to 21 days). For first and second feeding each week (e.g. Monday and Wednesday) remove ~50% of the medium and add back 50% by volume of fresh medium. For third feeding (e.g. Friday), aspirate all medium and replace with 2 ml fresh medium. This approach is based on the rationale that avoidance of complete medium changes minimizes fluctuations in concentrations of paracrine signals that are secreted into the culture medium9.
4. Colony Picking for First Passage
- Remove medium from wells containing colonies to be passaged and add fresh stem cell medium.
- Identify colonies using a 10x objective. With the microscope slightly de-focused, SSC colonies at the edge of the well will appear as homogeneous bright clumps, comprised of many 11-12 μm diameter cells, in which it is difficult or impossible to discern the individual cell borders, since the cells appear fused together (Figure 1A). Non-SSC colonies may appear darker, more granular, or less homogenous, with discernable borders (Figure 1A, inset).
- Using a 200 μl pipette tip, with the pipetteman set to 50 μl, first take up medium from the well and expel to wet the tip. Then, gently nudge the colony with the tip before rapidly withdrawing 50 μl of medium to dislodge the colony by suction into the tip (Figure 1B).
- Expel the medium containing the colony into one well of a 48-well plate containing inactivated feeder cells with 100 μl of stem cell medium (Figure 1C).
- Repeat step 4.3 and add up to 8 colonies to the same well without exceeding 500 μl per well.
- Repeat steps 4.3 to 4.5 to prepare additional wells of the 48 well plate with passage one SSC colonies.
- Wells will be ready to split in 7-14 days or after large clumps of up to 500 cells have emerged (Figure 1D).
5. Subsequent Expansion and Passaging of SSCs by Trituration
- Cells at passage one should be subcultured onto fresh feeders after up to 14 days at a split ratio of 1:2 for the first 3 passages and then 1:4 to 1:6 (every 7 days) thereafter as follows. Gently triturate colonies off a semi-confluent well (e.g. ~300,000 SSCs/well of 6-well plate) with a 1 ml pipette tip by washing medium across the cells. Colonies can be seen detaching. Too much force with the stream of liquid will cause unwanted feeder cells to come off too.
- Collect triturated cells in a conical tube and centrifuge at 300 x g for 5 min. Note: A trypsinization step may be safely added here if desired by the investigator.
- Aspirate supernatant and re-suspend in fresh stem cell medium by thoroughly pipetting up and down to disrupt colonies.
- Plate SSCs on freshly prepared feeder cells inactivated with mitomycin-C as described in section 1.
- Feed cells three times per week as follows. For first and second feeding (e.g. Monday and Wednesday) add 50% by volume of fresh medium. For third feeding (e.g. Friday), aspirate all medium and replace with 2 ml fresh medium. Plates will become confluent in 7-10 days. Cultures should be comprised of >98% germ cells (i.e., <1% contaminating somatic cells) after 5-7 passages, based on immunostaining (e.g. using the murine germ cell marker GCNA) to distinguish germ cells from somatic cells10. Note: These methods were developed in compliance with regulations and requirements set forth by the Institutional Animal Care and Use Committee at Weill Cornell Medical College.
Representative Results
The appearance of passage zero wild type, adult SSC colonies after 7 days is shown in Figure 1. Three-dimensional colonies are comprised of a layer of flat cells attached to the feeders or underling extracellular matrix deposited by the feeders with multiple layers of SSCs growing on top. While healthy SSCs are brightly refractile and uniformly 11-12 μm diameter, the cell borders are difficult to distinguish and the size of the colonies may be highly variable, both within the well and between wells prepared from different mice. Visualization of the colonies is aided by a phase microscope with a digital display and a slightly de-focused focal plane, which enhances the contrast between highly homogeneous SSC colonies and contaminating somatic cells which are present in early passage (0-2) cultures and do not form tightly packed colonies (see Figure 1A inset). Note that the morphology of the feeder cells will change gradually after switching from DMEM/10% to stem cell culture medium after inactivation with mitomycin-C.It is also important to note that the initial cell suspension is not filtered or strained, because residual clumps of somatic cells are thought to enhance survival of SSCs at the time of initial plating and should not be removed until SSC colonies are established. Serial passaging (>5-7 times) is required to purify the germ cells to near homogeneity (>98%) and deplete residual somatic cells. If subsequent experiments will specifically require enrichment of stem cells from the total population of germ cells in the culture, then cell selection using immunomagnetic beads or flow cytometry can be employed for further purification; for example, stem cell enrichment can be obtained using antibodies against Thy1, CD9, α6 integrin and others11. The identity of SSCs should be subsequently confirmed using immunophenotyping, gene expression, and by transplantation, since transplantation analysis is the only means to definitively assess the quantity of authentic, functional stem cells12,13.
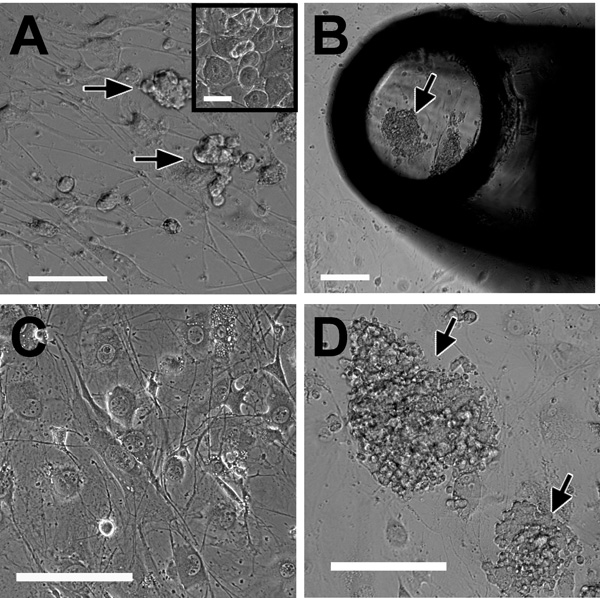
Figure 1. Morphology of early SSC colonies, testicular stromal cells and feeders. A, Appearance of nascent SSC colonies ~5 days after initial plating. Inset shows contaminating monocyte-like somatic cells in a passage zero culture. B, Selection and picking of individual colonies for first passage (~14 days) into 48-well plates. The black structure is the pipette tip. C, Appearance of inactivated JK1 feeder cells. D, Large colonies prior to routine subculturing. Arrows indicate SSC colonies. Scale bar in A is 100 μm (inset, 50 μm) and in B-D is 200 μm.
Discussion
This method for deriving adult SSCs using adult testis-derived feeder cells is robust and has succeeded when common genetic backgrounds (e.g. FVB, C57Bl6 and mixed 129SV/C57Bl/6) and different mutant strains were employed2,3,7,14. In fact, the microenvironment created by the culture system is sufficient to overcome some of the genetic barriers to maintaining adult SSCs in vivo (e.g. in the case of plzf-/- animals)7. While we employ JK1 cells as feeders, non-transformed adult testicular somatic cells can also be used with equal efficacy as JK1 cells2,3.
SESC was designed to efficiently obtain long-term adult SSC lines. Unfortunately, the high efficiency is achieved with a trade-off for the following reason. A notable limitation of the procedure is that the omission of a filtering or straining step at the time of initial plating and the manipulation that the procedure involves, which can affect cell survival, may cause substantial variability in cell numbers plated from well to well, which precludes effective quantitation at the stage of initial colony formation. However, quantitative assays can be performed with slightly later passage cultures at which time a single cell suspension can be easily prepared by standard trypsinization (>passage 5-7).
Because adult SSCs in culture can be expanded and maintained almost indefinitely, this protocol enables the efficient production of cell lines that can be used subsequently in a similar fashion to other established primary cultures or cell lines (i.e. gene and protein expression analyses or ectopic expression of a gene of interest). Nonetheless, it is essential to ultimately confirm results using transplantation analysis as the gold standard or another validated surrogate assay, such as the SSC cluster formation assay12,15.
Declarações
The authors have nothing to disclose.
Acknowledgements
This work was supported by the New York State Department of Health (C026878). M.S. was a New York Stem Cell Foundation-Druckenmiller Fellow. Supported in Part by Research Grant No. 5-FY11-571 from the March of Dimes Foundation.
Materials
| Name of Reagent/ Material | Company | Catalog Number | Comments |
| DMEM | Corning | 10-013 | Diluent for dissociation buffer |
| Trypsin/EDTA | Mediatech | 25-051-CI | |
| Stem cell base medium (StemPro-34) | Life Technologies | 10639-011 | Requires supplementation as per Shinohara et al. (2003)* |
| Stem cell medium supplements | various | see Table 3 | Requires supplementation as per Shinohara et al. (2003)* |
| JK1 cells | Cell Biolabs, Inc. | CBA-315 | Can substitute with adult testicular stromal cells as per Seandel et al. (2007) |
| mitomycin-C (CAUTION) | Sigma-Aldrich | M4287 | Toxic; Handle with care. |
| Gelatin | Sigma-Aldrich | G1890 | 0.4% solution in water |
| EVOS xl digital inverted microscope | Advanced Microscopy Group | – | |
| Table 1. Specific reagents and equipment. *See Table 3 |
|||
| DMEM | Corning | 10-013 | Diluent for dissociation buffer |
| trypsin (1:250) | Life Technologies | 27250-018 | Dissociation buffer: Final 0.05% wt/vol |
| collagenase, type I, 235 U/ml | Worthington | CLS1 235 | Dissociation buffer: Final 0.03% wt/vol |
| DNAse I | Sigma-Aldrich | DN25 | Dissociation buffer: Final 80 U/ml |
| bovine serum albumin | ICP Bio | ABRE-100g | Dissociation buffer: Final 0.5% wt/vol |
| Table 2. Dissociation buffer | |||
| StemPro-34 SFM | Life Technologies | 10639-011 | |
| StemPro-34 Nutrient supplement | Life Technologies | 10639-011 | |
| Additional supplements** | |||
| Non-essential amino acids | Sigma-Aldrich | M7145 | 1X |
| MEM Vitamin solution | Life Technologies | 11120-052 | 1X |
| L-glutamine | Mediatech | 25-005 | 2 mM |
| bovine serum albumin | ICP Bio | ABRE | 0.50% |
| Antibiotic-Antimycotic Solution | Mediatech | 30-004-CI | 1X |
| D(+)glucose | Sigma-Aldrich | G8769 | 6 mg/ml |
| β-estradiol | Sigma-Aldrich | E2758 | 30 ng/ml |
| progesterone | Calbiochem | 5341 | 60 ng/ml |
| fetal bovine serum | variable | n/a | 1% |
| bovine holo-transferrin | Sigma-Aldrich | T1283 | 100 μg/ml |
| insulin | Gemini Bio-Products | 700-112P | 25 μg/ml |
| human GDNF | Life Technologies | PHC7041 | 10 ng/ml |
| human bFGF | Life Technologies | PHG0023 | 10 ng/ml |
| mouse EGF | Life Technologies | PHG0313 | 20 ng/ml |
| putrescine | Research Organics | 0778P | 60 μM |
| sodium Selenite | Sigma-Aldrich | S5261 | 30 nM |
| pyruvic acid | Alfa Aesar | A13875 | 30 μg/ml |
| DL-lactic acid | J.T. Baker | 0196-04 | 1 μg/ml |
| β-mercaptoethanol | Life Technologies | 21985-023 | 50 μM |
| ascorbic acid | Sigma-Aldrich | A4544 | 100 μM |
| D-biotin | Sigma-Aldrich | B4639 | 10 μg/ml |
| Table 3. Stem cell medium | |||
| *Note: Add supplements below before using medium. Filter sterilize and keep it at 4 °C. The medium is stable for at least 2 weeks. | |||
| **We have employed different manufacturers, formulations, and/or lot numbers of these reagents without any apparent deleterious effects. In general, cell culture grade reagents should be employed. |
Referências
- Kanatsu-Shinohara, M., et al. Long-term proliferation in culture and germline transmission of mouse male germline stem cells. Biol. Reprod. 69, 612-616 (2003).
- Seandel, M., et al. Generation of functional multipotent adult stem cells from GPR125+ germline progenitors. Nature. 449, 346-350 (2007).
- Kim, J., Seandel, M., Falciatori, I., Wen, D., Rafii, S. CD34+ testicular stromal cells support long-term expansion of embryonic and adult stem and progenitor cells. Stem Cells. 26, 2516-2522 (2008).
- Ogawa, T., et al. Derivation and morphological characterization of mouse spermatogonial stem cell lines. Arch. Histol. Cytol. 67, 297-306 (2004).
- Schmidt, J. A., et al. In vivo and in vitro aging is detrimental to mouse spermatogonial stem cell function. Biology of Reproduction. 84, 698-706 (2011).
- Zhang, X., Ebata, K. T., Robaire, B., Nagano, M. C. Aging of male germ line stem cells in mice. Biology of Reproduction. 74, 119-124 (2006).
- Hobbs, R. M., Seandel, M., Falciatori, I., Rafii, S., Pandolfi, P. P. Plzf regulates germline progenitor selfrenewal by opposing mTORC1. Cell. 142, 468-479 (2010).
- Nagano, M., Ryu, B. Y., Brinster, C. J., Avarbock, M. R., Brinster, R. L. Maintenance of mouse male germ line stem cells in vitro. Biol. Reprod. 68, 2207-2214 (2003).
- Csaszar, E., et al. Rapid expansion of human hematopoietic stem cells by automated control of inhibitory feedback signaling. Cell Stem Cell. 10, 218-229 (2012).
- Enders, G. C., May, J. J. Developmentally regulated expression of a mouse germ cell nuclear antigen examined from embryonic day 11 to adult in male and female. 163, 331-340 (1994).
- Oatley, J. M., Brinster, R. L. Regulation of spermatogonial stem cell self-renewal in mammals. Annu Rev Cell Dev Biol. 24, 263-286 (2008).
- Brinster, R. L., Zimmermann, J. W. Spermatogenesis following male germ-cell transplantation. Proc Natl Acad Sci U.S.A. 91, 11298-11302 (1994).
- Tang, L., Rodriguez-Sosa, J. R., Dobrinski, I. Germ Cell Transplantation and Testis Tissue Xenografting in Mice. J. Vis. Exp. (60), e3545 (2012).
- Arnold, K., et al. Sox2(+) adult stem and progenitor cells are important for tissue regeneration and survival of mice. Cell Stem Cell. 9, 317-329 (2011).
- Yeh, J. R., Zhang, X., Nagano, M. C. Establishment of a short-term in vitro assay for mouse spermatogonial stem cells. Biology of Reproduction. 77, 897-904 (2007).

