Collecting And Measuring Wound Exudate Biochemical Mediators In Surgical Wounds
Summary
This article provides a detailed and visual description of a methodology for collecting and measuring biochemical inflammatory and nociceptive mediators at the surgical wound site following cesarean delivery. This human bioassay has been used to determine correlations between wound and serum cytokine concentrations and drug-mediated changes in wound cytokines, chemokines and neuropetides.
Abstract
We describe a methodology by which we are able to collect and measure biochemical inflammatory and nociceptive mediators at the surgical wound site. Collecting site-specific biochemical markers is important to understand the relationship between levels in serum and surgical wound, determine any associations between mediator release, pain, analgesic use and other outcomes of interest, and evaluate the effect of systemic and peripheral drug administration on surgical wound biochemistry. This methodology has been applied to healthy women undergoing elective cesarean delivery with spinal anesthesia. We have measured wound exudate and serum mediators at the same time intervals as patient’s pain scores and analgesics consumption for up to 48 hours post-cesarean delivery. Using this methodology we have been able to detect various biochemical mediators including nerve growth factor (NGF), prostaglandin E2 (PG-E2) substance P, IL-1β, IL-2, IL-4, IL-6, IL-7, IL-8, IL-10, IL-12, IL-13, IL-17, TNFα, INFγ, G-CSF, GM-CSF, MCP-1 and MIP-1β. Studies applying this human surgical wound bioassay have found no correlations between wound and serum cytokine concentrations or their time-release profile (J Pain. 2008; 9(7):650-7).1 We also documented the utility of the technique to identify drug-mediated changes in wound cytokine content (Anesth Analg 2010; 111:1452-9).2
Introduction
After a surgical incision, resident and migrating inflammatory cells release pro- and anti-inflammatory cytokines, prostanoids, neuropeptides and chemokines that are important for initiating and maintaining pain.3,4 The ability to collect site-specific biochemical markers is critical to determine the relationship between levels in serum and surgical wound, understand the complex relationship between mediator release and surgical pain, and to be able to evaluate the effect of both systemic and peripheral drug administration on surgical wound biochemistry. Studies have focused on systemic measurements of biochemical mediators and have not considered site-specific release.5,6 Serum measurements of cytokines and chemokines may not be reflective of local surgical wound biochemical events and only indicate a global systemic response to surgical injury. This article provides a detailed and unique visual description of this methodology that was first referenced in 2008 in the Journal of Pain.1 The aim of this JoVE article is to provide a unique and visually enhanced description of the methods.
Protocol
1. Study Protocol
- Obtain Institutional Review Board approval and written informed consent.
- Select an appropriate surgical population for study conduct. This methodology has only been applied to cesarean delivery wounds but most surgical wounds would be appropriate.
- Consider inclusion and exclusion criteria for your study participation. Patients and procedures with high rates of surgical wound infection may not be appropriate.
- Standardize anesthesia and postoperative management for all patients to avoid potential confounders.
2. Nociceptive and Inflammatory Biochemical Mediator Collection
- Insert the ON-Q PainBuster with ON-Q SilverSoaker (I-Flow, Lake Forest, CA) into the subcutaneous layer just prior to wound closure (other tissue layers can be considered). The ON-Q PainBuster Pain Relief System continuously delivers normal saline at a rate of 2 ml/h. The carrier can be combined with or compared to drugs of interest such as a local anesthetic and non-steroidal anti-inflammatories.
- Incorporate a standard three-way stopcock into this system to allow aspiration of wound exudate at specified time points. The continuous infusion prevents the catheter from clotting and improves the reliability of the system to produce exudate samples. If aspiration of exudate is difficult (approximately 5% of cases), consider pushing gently above the wound, changing the subject’s position (e.g., sitting the patient up or lying them flat), using a 0.5-1 ml normal saline flush or withdrawing the catheter 1-2 cm.
- At time points specified by the protocol (e.g., 1, 4, 6, 24, and 48 hr after cesarean delivery), aspirate and withdraw 1 ml of wound exudate. Transfer the exudate into a polyethylene cup containing 30 μl of proteinase inhibitor. We have used Complete Proteinase Inhibitor (Roche Bioscience, Palo Alto, CA) for our purposes.
- At the same time intervals specified by the study protocol, collect 10 ml of blood. We have used a blood collection tube containing lithium heparin; at our institution this is a green-topped glass tube. Add an additional 300 μl of proteinase inhibitor into the blood samples.
- Within 1 hr of collection, put samples on ice and centrifuge at 3,000 rpm for 10 min.
- Remove serum and wound supernate and place in a standard microcentrifuge tube and store at -20 °C.
3. Assay Analysis
- Once all the study exudate and serum samples are collected and stored, thaw and analyze the samples at the same time.
- We have measured the cytokines and chemokines using a multiplex immunoassay. The multiplex immuno-assay technique allows us to assay numerous analytes from small (50 μl) volume samples. The multiplex produce results comparable with those obtained with ELISA. We have used the 17-plex Bio-PlexTM system (BioRad, Hercules, CA) for our analysis. This assay is capable of measuring tumor necrosis factor α (TNFα), interferon α (INFα), interleukin 1β (IL-1β), interleukin 2 (IL-2), interleukin 4 (IL-4), interleukin 5 (IL-5), interleukin 6 (IL-6), interleukin 7 (IL-7), interleukin 8 (IL-8), interleukin (IL-10), interleukin 12 (IL-12), interleukin 13 (IL-13), interleukin 17 (IL-17), granulocyte colony stimulating factor (G-CSF), granulocyte-macrophage colony stimulating factor (GM-CSF), monocyte chemoattractant protein 1 (MCP-1) and macrophage inflammatory protein 1 (MIP-1β). Nerve growth factor was measured with NGF DY256 antibody by adding it to the 17-plex plate with the Bio-Plex amine coupling kit.
- To ensure good quality analysis, each measurement should be made in duplicate. Standard curves for each analyte are generated by using reference analytes supplied by the manufacturer (at concentrations of 0.20, 0.78, 3.13, 12.5, 50, 200, 800, 3,200 pg/ml plus a zero standard for the normal saline). Standard curves are included in each run and sample concentrations are calculated with Bio-Plex Manager software.
- Substance P and prostaglandin E2 were measured using highly sensitivity ELISA Kits. We have used Assay Designs, Inc. (Ann Arbor, Michigan) for this analysis. Perform the analysis in duplicate according to the manufacturer’s specification.
Representative Results
Using this methodology we have been able to detect all biochemical mediators outlined above with the exception of IL-5 in wound exudate and plasma.1,2 Some additional cytokines were not detected in plasma (IL-4, IL-17, TNFα, INFγ, GM-CSF).1 The time course of measured cytokines and chemokines in reported studies were consistent, reaching a plateau within the first few hours that was sustained for the 24 hour observation period..1,2 However, NGF increased steadily over the 24-hours collection period, whereas PGE2 peaked within 1 hour and then steadily decreased.1 Particularly noteworthy results are markedly decreased levels of IL-10 and increased levels of substance P in wounds instilled with the local anesthetic bupivacaine, compatible with an overall pro-inflammatory effect of the drug (Figure 1).2 In a different study we demonstrated that analgesic consumption after cesarean delivery surgery was negatively correlated with wound exudate IL-6, IL-1 β, and G-CSF concentrations (Figure 2).1
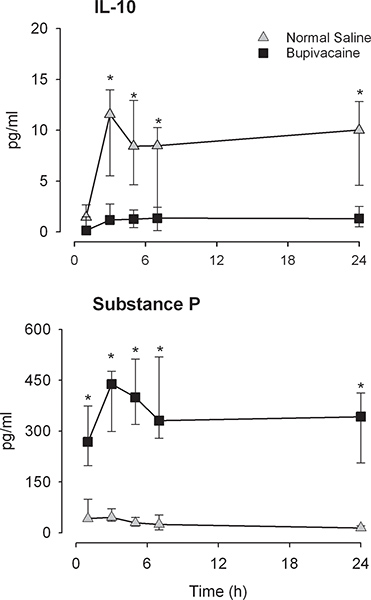
Figure 1. Interleukin-10 (IL-10) and substance P wound exudate levels levels (pg/ml) measured at 1, 3, 5, 7, and 24 hours post-cesarean delivery. The normal saline group is indicated in gray diamonds, and the bupivacaine group is shown with black squares. *=Significantly different between groups (P < 0.005).
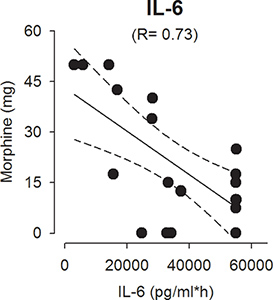
Figure 2. Correlation between interleukin-6 (pg/ml) and morphine consumption in the first 24 hr after cesarean delivery.
Discussion
This methodology outlined above has been found to be sensitive enough to detect biochemical inflammatory and nociceptive mediators in wound exudate. Sampling at various time-points has also allowed us to observe changes of these mediators over time.1,2 We are not aware of any other human surgical wound assay that can serially measure the release profile of inflammatory and nociceptive mediators in surgical wound exudate. Results from our methodology of wound exudate collection from the surgical incision site is different from studies utilizing drains from the hip joint,7,8 which suggests that subcutaneous wound mediator release may be different from intraarticular or other body cavities biochemical mediator release. Additionally, results obtained in drains may be confounded by blood loss into the wound, and therefore they may not accurately reflect local biochemical mediator release.
The poor correlation between wound and serum levels demonstrates the importance of determining site-specific release when studying surgical wounds. Local changes may not be reflected systemically due to many factors including differential systemic and local production in response to surgical trauma and stress. Although we have chosen to measure a particular selection of pro- and anti-inflammatory cytokines and chemokines as well as NGF, SP and PG-E2 from cesarean delivery wound exudate,1,2 this methodology can be applied to other surgical models and many other biochemical mediators.
Surgical wound local anesthetic instillation is a commonly used technique for improving postoperative pain.9 Applying the methodology, we have been able to demonstrate that local anesthetic instillation into surgical wounds does affect wound biochemistry.2 A decrease in IL-10 and increase in substance P in wound exudates with bupivacaine instillation suggests a disruption of anti-inflammatory modulation. Peripherally administered local anesthetics may thereby affect the inflammatory processes,10,11 which may partially account for local anesthetic-induced chondrolysis in joints after surgery.12
ON-Q PainBuster Pain Relief System is a US Food and Drug Administration (FDA) approved device since 1998 that is used for the delivery of local anesthetics to the surgical wound site to reduce pain following surgery. Continuous wound instillation utilizing this and other catheters has not been associated with increased wound infection.9,13 ON-Q PainBuster with ON-Q SilverSoaker is designed to infuse drugs and not to repeatedly sample wound exudate. Although we have had no wound infections with this technique, we stress an aseptic technique while sampling wound exudate from this system. Our proposed modification to facilitate sampling should be reserved for research purposes and not for routine clinical use. We also recommend that the duration the catheter remains in-situ be limited to 48-72 hours.
This surgical site bioassay technique provides a good model to understand the relationship between inflammatory and nociceptive biochemical mediators in postoperative pain. We will use the methodology in future studies to investigate effects of locally administered non-steroidal anti-inflammatory drugs and opioids. The technique may also lend itself to investigations examining wound healing, excessive surgical scarring and wound infection.
Declarações
The authors have nothing to disclose.
Acknowledgements
Dr. Carvalho’s work was supported by a research grant from the Office of Research on Women’s Health and National Institute of Child Health and Human Development of the National Institutes of Health (5K12 HD043452). Dr Angst received supplies (On-Q PainBuster Post-Op Pain Relief System) and funds (for the biochemical assays) from I-Flow (Lake Forest, California).
Referências
- Carvalho, B., Clark, D. J., Angst, M. S. Local and Systemic Release of Cytokines, Nerve Growth Factor, Prostaglandin E2, and Substance P in Incisional Wounds and Serum Following Cesarean Delivery. J. Pain. 9, 650-657 (2008).
- Carvalho, B., Clark, D. J., Yeomans, D. C., Angst, M. S. Continuous subcutaneous instillation of bupivacaine compared to saline reduces interleukin 10 and increases substance P in surgical wounds after cesarean delivery. Anesth. Analg. 111, 1452-1459 (2010).
- Verri, W. A., Cunha, T. M., Parada, C. A., Poole, S., Cunha, F. Q., Ferreira, S. H. Hypernociceptive role of cytokines and chemokines: targets for analgesic drug development. Pharmacol. Ther. 112, 116-138 (2006).
- Watkins, L. R., Maier, S. F. Immune regulation of central nervous system functions: from sickness responses to pathological pain. J. Intern. Med. 257, 139-155 (2005).
- Beilin, B., Bessler, H., Mayburd, E., Smirnov, G., Dekel, A., Yardeni, I., Shavit, Y. Effects of preemptive analgesia on pain and cytokine production in the postoperative period. Anesthesiology. 98, 151-155 (2003).
- Wu, C. T., Jao, S. W., Borel, C. O., Yeh, C. C., Li, C. Y., Lu, C. H., Wong, C. S. The effect of epidural clonidine on perioperative cytokine response, postoperative pain, and bowel function in patients undergoing colorectal surgery. Anesth. Analg. 99, 502-509 (2004).
- Buvanendran, A., Kroin, J. S., Berger, R. A., Hallab, N. J., Saha, C., Negrescu, C., Moric, M., Caicedo, M. S., Tuman, K. J. Upregulation of prostaglandin E2 and interleukins in the central nervous system and peripheral tissue during and after surgery in humans. Anesthesiology. 104, 403-410 (2006).
- Kristiansson, M., Soop, M., Sundqvist, K. G., Soop, A., Suontaka, A. M., Blomback, M. Local vs. systemic immune and haemostatic response to hip arthroplasty. Eur. J. Anaesthesiol. 15, 260-270 (1998).
- Liu, S. S., Richman, J. M., Thirlby, R. C., Wu, C. L. Efficacy of continuous wound catheters delivering local anesthetic for postoperative analgesia: a quantitative and qualitative systematic review of randomized controlled trials. J. Am. Coll. Surg. 203, 914-932 (2006).
- Eriksson, A. S., Sinclair, R., Cassuto, J., Thomsen, P. Influence of lidocaine on leukocyte function in the surgical wound. Anesthesiology. 77, 74-78 (1992).
- Clark, J. D., Shi, X., Li, X., Qiao, Y., Liang, D., Angst, M. S., Yeomans, D. C. Morphine reduces local cytokine expression and neutrophil infiltration after incision. Mol. Pain. 3, 28 (2007).
- Webb, S. T., Ghosh, S. Intra-articular bupivacaine: potentially chondrotoxic. Br. J. Anaesth. 102, 439-441 (2009).
- Scott, N. B. Wound infiltration for surgery. Anaesthesia. 65, 67 (2010).

