Culturing Caenorhabditis elegans in Axenic Liquid Media and Creation of Transgenic Worms by Microparticle Bombardment
Summary
C. elegans is usually grown on solid agar plates or in liquid cultures seeded with E. coli. To prevent bacterial byproducts from confounding toxicological and nutritional studies, we utilized an axenic liquid medium, CeHR, to grow and synchronize a large number of worms for a range of downstream applications.
Abstract
In this protocol, we present the required materials, and the procedure for making modified C. elegans Habituation and Reproduction media (mCeHR). Additionally, the steps for exposing and acclimatizing C. elegans grown on E. coli to axenic liquid media are described. Finally, downstream experiments that utilize axenic C. elegans illustrate the benefits of this procedure. The ability to analyze and determine C. elegans nutrient requirement was illustrated by growing N2 wild type worms in axenic liquid media with varying heme concentrations. This procedure can be replicated with other nutrients to determine the optimal concentration for worm growth and development or, to determine the toxicological effects of drug treatments. The effects of varied heme concentrations on the growth of wild type worms were determined through qualitative microscopic observation and by quantitating the number of worms that grew in each heme concentration. In addition, the effect of varied nutrient concentrations can be assayed by utilizing worms that express fluorescent sensors that respond to changes in the nutrient of interest. Furthermore, a large number of worms were easily produced for the generation of transgenic C. elegans using microparticle bombardment.
Introduction
The soil nematode, Caenorhabditis elegans, is a powerful model organism used in numerous studies from genetics to toxicology. As a result of its 1 mm size, rapid generation time of four days, ease of cultivation, and large progeny numbers, these nematodes have been utilized in a number of genetic and pharmacological screens1,2. Researchers take advantage of this worm to identify molecules and pathways conserved in vertebrate systems. These pathways include cell death signals, pathways of aging and metabolism, and the nervous system3-6. Additionally, the transparency of C. elegans allows for the generation of transgenic lines using fluorescent protein reporters, which can be directly visualized to analyze gene expression patterns and protein localization.
In many studies this nematode is cultured on a solid agar-based surface using nematode growth medium (NGM) plates or in liquid cultures seeded with Escherichia coli as a food source7,8. These bacterial food sources can confound biochemical and toxicology studies with interference from bacterial by-products affecting the interpretation of results. In order to avoid these compounding effects, C. elegans can be cultured in an axenic liquid media that is devoid of bacteria as a food source. Using this media, we successfully cultured millions of highly synchronized worms for many standard C. elegans protocols including microarray analysis of differentially regulated genes in C. elegans exposed to different heme concentrations, and production of transgenic worms using gene bombardment. This media is chemically defined and modified from an original recipe formulated by Dr. Eric Clegg9. Using this mCeHR media, we have successfully identified genes involved in heme homeostasis, referred to as heme responsive genes (hrgs)10, which would have not been possible in regular growth conditions which utilize NGM agar plates seeded with E. coli.
In this protocol we describe the procedure for introducing and maintaining C. elegans grown on E. coli to the axenic mCeHR and utilize this method to obtain a large number of worms for producing transgenic C. elegans lines using microparticle bombardment. We also present studies that show the utility of using axenic media for determining the nutritional requirement of C. elegans using heme as an example. These studies show that using mCeHR media allows for rapid growth of a large number of C. elegans for many downstream applications utilized by worm researchers.
Protocol
1. Worm Strains
- Obtain C. elegans wildtype Bristol N2 strains from the Caenorhabditis Genetics Center (CGC) (http://www.cbs.umn.edu/cgc) and maintain them on NGM plates seeded with E. coli strain OP507. Note: Transgenic worm strains IQ6011 (Phrg-1::GFP) utilized were generated as previously described11. IQ6011 can be requested from the corresponding author.
2. Preparation of Modified C. elegans Habitation and Reproduction Medium (mCeHR)
Prepare mCeHR liquid media as described below12. This axenic liquid media allows the worms to grow without any additional food sources. Carry out all manipulations of axenic liquid media and axenic worms using strictly sterile conditions such as a laminar flow hood.
- Obtain ultra pasteurized fat-free milk from a grocery store. Use the refrigerated product and not the room temperature product. Prior to use in mCeHR medium, streak the milk on Brain Heart Infusion (BHI) agar plates and incubate for 3 days at 30 °C and 37 °C to check for sterility. Transfer the milk to 50 ml sterile tubes and store at -80 °C
- Prepare each of the following components and combine in the order and amounts given to prepare 1 L of mCeHR (Table 1A): 10 ml of 2 mM choline diacid citrate, 10 ml of vitamin and growth factor mix (see recipe in Table 1B), 10 ml of 2.4 mM myo-inositol, 10 ml of 2 mM hemin chloride, 250 ml of deionized water. Apply suction and filter through a 0.22 μm filter unit.
- Add 20 ml of nucleic acid mix (recipe in Table 1C), 100 ml of mineral mix (recipe in Table 1D), 20 ml of 170 mg/ml lactalbumin hydrolysate, 20 ml of essential amino acids, 10 ml of non-essential amino acids, 20 ml of 450 mM KH2PO4, 50 ml of 1.45 M D-glucose, 10 ml of 1 M HEPES, sodium salt, 250 ml deionized water.
- Apply suction to filter sterilize the components. Remove the filter and add 1 ml of 5 mg/ml cholesterol. Ensure that the pH of the media is approximately 6-6.5. Note: This solution can be stored at -80 °C for up to one year.
- Add 20% (v/v) ultra pasteurized organic skim milk to the mCeHR medium before use. Use scrupulously sterile technique (for example in a laminar flow hood) when opening and aliquoting the milk. Store the media at 4 °C.
3. Prepare C. elegans for Culture in Axenic mCeHR Liquid Medium
- Grow worms on ten 60 mm NGM plates until there are many gravid worms and a minimal amount of OP50 E. coli on the plates.
- Remove the worms from each plate by rinsing with 5 ml of M9 buffer (recipe in Table 1F), and combine in a 50 ml conical tube.
- Allow the worms to settle by leaving the tube to stand for 5 to 10 min then carefully remove the supernatant containing the E. coli.
- Repeat steps 3.2 and 3.3 2x to remove as much of the residual bacteria as possible before continuing the procedure.
- Resuspend the worms in 0.1 N NaCl in a volume that is a multiple of three, for example 1.5 ml, 3 ml or 6 ml in which individual worms can be seen in the tube.
- Bleach the worms by adding 5 N NaOH and 5% sodium hypochlorite (bleach) solution in a 1:2:6 ratio to the worm suspension. For example, 1 ml 5 N NaOH: 2 ml 5% sodium hypochlorite: 6 ml of the worm suspension. Mix the NaOH and bleach before adding to the worm suspension.
- Vortex the solution vigorously until the gravid worms are dissolved and only eggs are visible within the suspension (about 5 to 10 min). Monitor the process using a phase contrast microscope with a 10X objective.
- After the worms have completely dissolved, immediately pellet the eggs at 800 x g for 45 sec at 4 °C. Remove the supernatant and rinse the pellet twice with 10 ml of sterile water by centrifuging the pellet at 800 x g for 45 sec at 4 °C.
- After bleaching, transfer the egg pellet to a 25 cm2 tissue culture flask containing 10 ml of mCeHR media supplemented with 100 μg/ml tetracycline. Carry out this procedure using sterile techniques. If desired, add additional antibiotics, including 250 μg/ml nalidixic acid, to prevent bacterial growth in the axenic liquid cultures.
- Incubate the liquid cultures at 20 °C on a shaking incubator at approximately 70 rpm.
- Check the worms daily to note the stage of development and the rate of growth.
Note: Worms will initially grow slowly and require 7 to 10 days to become gravid. However, as the worms acclimatize to the liquid medium the generation time will decrease to approximately 4 days for wildtype (N2) worms. - After the worms have developed to the gravid stage in liquid media, transfer the worm culture to a conical tube and pellet the worms at 800 x g for 5 min at 4 °C using a swinging bucket rotor.
- Remove the supernatant and gently resuspend the worm pellet in an equal volume of 0.1 M NaCl. For example, use 10 ml of 0.1 M NaCl for worms cultured in 10 ml of mCeHR. Allow the worms to settle for 5 min on ice.
- Carry out the bleaching procedure as described above in steps 3.5-3.8, and allow the worms to grow for a second generation in the axenic liquid media. If necessary, add the antibiotic again to inhibit bacterial growth.
- Again allow the worms to become gravid then pellet and resuspend the worms in 0.1 M NaCl as described in steps 3.12 and 3.13, then bleach again as described in steps 3.5-3.8, to produce a synchronized population. Grow the L1 larval worms in liquid media without antibiotics from now on.
4. Synchronizing Worms from Liquid Culture
- Pellet and resuspend the worms in 0.1 M NaCl as described above in steps 3.12 and 3.13. Bleach as described above in steps 3.5-3.8.
- Resuspend the egg pellet in 10 ml of M9 buffer and allow the larvae to hatch O/N at 20 °C on a rotating platform shaker. Note: The eggs will hatch into L1 larvae, but will not develop further, synchronizing the worms at the L1 stage.
- To maintain and subculture worms, pellet L1 larvae at 800 x g for 5 min, then resuspend the larvae in 10 ml of mCeHR and transfer to a 25 cm2 flask. Allow the worms to grow at 20 °C on a rotating platform. Maintain a maximum density of 3,000 worms/ml/cm2 to ensure adequate nutrition for the worms.
Note: At this stage, antibiotics are not required to maintain axenic conditions when the procedures are carried out using sterile techniques in a laminar flow hood. Worms should be checked daily to monitor growth.
5. Freezing and Thawing Worms for Axenic Medium Cultures
- Freeze axenic asynchronous worm cultures or synchronized L1 larvae in liquid nitrogen. Pellet worms at 800 x g for 5 min then resuspend in S buffer (6.45 mM KHPO4, 43.55 mM KHPO4, 146.25 mM NaCl) using 0.5 ml for each vial. Add an equal volume of S buffer with 30% v/v glycerol to each vial and transfer the worms to -80 °C before long term storage in liquid N2. Freeze approximately 1 x 106 worms per ml in each vial.
- Thaw worms by incubating the vials at 37 °C for approximately 2 min until almost all of the ice is melted. In a laminar flow hood, transfer the worms to a sterile flask containing mCeHR and place them at 20 °C.
6. Determine the Effect of Hemin Concentration on Growth and Reproduction in mCeHR
- Use axenic mCeHR media to assay nutrient dependent growth of C. elegans. To determine the effect of hemin concentration on worm growth and development, use synchronized axenic N2 worms or other strains of interest. Synchronize L1 larvae as described above in section 4.
- On the following day, pellet and count synchronized L1 larvae and dilute to 1 worm per μl of axenic media. Make 10 mM hemin chloride in 0.3 M NH4OH and adjust to pH 8 using concentrated HCl.
- Add 800 μl of mCeHR media to a 24-well plate. Add 100 worms to each well in 100 μl of media. Add hemin chloride to each well in concentrations ranging from 0 μM, 1.5 μM to 1,000 μM in triplicate. Ensure that all wells contain the same concentration of 0.3 M NH4OH. Gently resuspended the worms before pipetting to ensure that 100 L1 larvae are added to each well.
- Examine the worms daily and note the growth in each concentration in each well. Count the number of worms after 9 days of incubation and plot the number of worms/ml corresponding to each heme concentration (Figure 1).
7. Effect of Hemin Concentration on Heme Sensor Worms
- Incubate synchronized L1 heme sensor worms (Phrg-1::gfp) in mCeHR containing 4 μM, 8 μM, 10 μM and 20 μM hemin.
- Image the worms using fluorescence confocal microscopy, 48 hr post incubation (Figure 2).
8. Utilizing mCeHR to Generate Transgenic C. elegans Using Microparticle Bombardment
Note: The procedure for generating and carrying out microparticle bombardment using unc-119(ed3) worms grown in mCeHR is outlined in Figure 313.
- Worm Preparation
- Transfer approximately 1 x 106 asynchronous unc-119(ed3) worms in 90 ml of mCeHR media in a 175 cm2 flask one week prior to bombardment. Allow the worms to grow at 20 °C on a shaking platform at ~70 rpm (Figure 3-1).
Note: One week later, the worm density should be significantly greater with many adult gravid worms. Approximately 3 x 107 worms will be required for microparticle bombardment. - Chill JM109 E. coli seeded 10 cm NGM plates on ice.
- Transfer the axenic worms to two 50 ml conical tubes and centrifuge the worms at 800 x g for 2 min. Aspirate the supernatant and resuspend the worms in each tube in 50 ml of M9 buffer (Figure 3-2).
- Allow the adult worms to pellet by gravity for 10 to 15 min on ice.
Note: The 2 ml pellet should now contain >90% adult worms. This 2 ml pellet should have approximately 5 x 106 worms and is sufficient for one microparticle bombardment. - Coat the entire surface of the chilled JM109 seeded NGM plate with the 2 ml pellet of worms. Allow the liquid to be completely absorbed then leave the plate for 30 min on ice before proceeding (Figure 3-3).
- Transfer approximately 1 x 106 asynchronous unc-119(ed3) worms in 90 ml of mCeHR media in a 175 cm2 flask one week prior to bombardment. Allow the worms to grow at 20 °C on a shaking platform at ~70 rpm (Figure 3-1).
- Preparation of gold particles
- Weigh 30 mg of gold particles into a siliconized microcentrifuge tube. Add 1 ml 70% ethanol and vortex the tube for 5 min.
- Allow the tube to sit for 15 min, then centrifuge at 6,000 x g for 10 sec to pellet the gold particles. Remove the supernatant using a pipette tip by carefully touching the tip to the side of the tube opposite to the gold particles.
- Wash the gold particles with 1 ml of sterile water, vortex for 1 min and briefly centrifuge the tube at 6,000 x g for 10 sec. Repeat the wash step twice, then resuspend the gold particles in 0.5 ml of sterile 50% glycerol.
Note: The final concentration of the gold particles will be 60 mg/ml and it can be stored for 1 to 2 months at 4 °C.
- Prepare the DNA-gold particle mix
- Combine 10 μg of the desired plasmid DNA with 10 μg of the unc-119 rescue plasmid in a siliconized 1.5 ml microcentrifuge tube. Add 50 μl of DI water and 100 μl of well resuspended gold particle solution then vortex for 1 min.
- Add 150 μl of 2.5 M CaCl2 and vortex the solution again for 1 min. To this solution, add 60 μl of 0.1 M spermidine and vortex for 3 to 5 min.
- Pulse centrifuge the solution at 6,000 x g for 10 sec, remove the supernatant and add 300 μl of 70% ethanol. Vortex well for 1 min, pulse centrifuge at 6,000 x g, remove the supernatant and resuspend in 170 μl of 100% ethanol. Vigorously vortex the solution for 5 to 10 min then pulse centrifuge and remove the supernatant.
- Bombardment (Figure 3-3)
Note: This protocol is carried out with the particle delivery system specified in the Table of Materials/Equipment. Follow the instructions for the particle delivery instrument available.- Place a sheet of tissue paper into a 15 cm plate. Using tweezers, dip seven macrocarriers individually into 100% ethanol and lay them on the tissue to dry.
- Thoroughly clean the biolistic chamber, macrocarrier holder, and target plate with 70% ethanol. Clean and autoclave the stopping screens before each bombardment procedure.
- Load the macrocarrriers into the holder using tweezers and press into the holder using the seating tool.
- Evenly spread 20 μl of the DNA coated gold particles in the center of each macrocarrier and then allow the solution to dry completely.
- Clean the two parts of the pressure divider with ethanol and assemble with ethanol cleaned rupture disks. Screw the assembled pressure divider and rupture disks into the bombardment chamber.
- Assemble an autoclaved stopping screen with the macrocarrier holder and place the apparatus on to the metal shelf required and lock in place.
- Insert the assembled macrocarrier apparatus into the biolistic chamber in the allotted slot below the pressure divider and align with the pressure divider.
- Tape the open NGM plate with the worms onto the target plate shelf and close the door of the chamber.
- To bombard worms, adjust the pressure to that needed to break the rupture disks. Turn on the vacuum to 28 inches of Hg pressure, then press the fire button until the disks rupture.
- Release the vacuum and remove the NGM plate from the apparatus. Wash the worms from the plate and redistribute in twenty 10 cm NGM plates seeded with JM109 E. coli (Figure 3-4).
- Allow the worms to grow for 10 to 14 days at 25 °C and starve the plates. Worms rescued from the unc-119 defect will have normal movement and can be identified at this time. Pick at least 10 “wildtype” worms from separate plates for further analysis as these represent independent transgenic lines (Figure 3-5).
Representative Results
Culturing C. elegans in axenic liquid medium aids in the determination of nutrients that are required by worms, without interference from secondary metabolites produced by E. coli. Wildtype N2 worms acclimatize to mCeHR media within three generations and show growth comparable to worms grown on NGM bacterial plates. Indeed, these worms become gravid within 4 days as compared with 3.5 days for worms grown on OP50 bacteria.
One advantage of using mCeHR was seen in studies that examined the exact nutrient requirement of these worms14. In Figure 1 worms were grown in mCeHR supplemented with increasing amounts of heme, up to 1 mM. Observations of these worms showed a distinct delay in growth at heme concentrations below 4 μM, with worms developing to the L4 larval stage but unable to progress to the gravid stage after nine days in mCeHR. At concentrations of 10 μM and 20 μM heme the worms develop to the gravid stage in 4 days and produce large number of progeny. A maximum number of progeny was seen when worms were grown in mCeHR containing 20 μM heme. Worms continued to develop and produce progeny at 100 μM and 500 μM heme. However, the number of larval progeny significantly declined in comparison to worms grown at the optimal heme concentration of 20 μM heme. Heme concentrations at or above 800 μM resulted in stunted, sickly worms at the L3 larval stage, which indicated that these heme concentrations were toxic to the worms.
In addition to determining the optimal heme concentration and the effect of heme deprivation and heme toxicity on C. elegans growth, heme reporter strains could be utilized to indirectly assess the heme status of the worm within a smaller concentration range. The IQ6011 worm is a transgenic worm that expresses a heme responsive transcriptional reporter, Phrg-1::GFP, that inversely expresses GFP in response to environmental heme concentrations. When this worm is exposed to low environmental heme in mCeHR, GFP is highly expressed. This response is reversed under heme replete conditions, as seen in Figure 2. GFP expression is repressed at 20 μM heme and increases as the heme concentration is decreased. The incremental changes in heme concentration can be accurately correlated with gene response and expression in mCeHR media.
In addition to being able to carefully control nutrient concentrations provided to the worm, mCeHR axenic media allows for the efficient growth of a large number of synchronized worms. This feature can be exploited for microparticle bombardment (Figure 3). Using this procedure at least one integrated line have been developed from every microparticle bombardment carried out (Table 2).
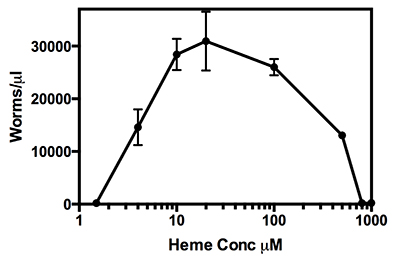
Figure 1. C. elegans heme growth curve. Worms were grown in a range of heme concentrations from 0 μM to 1,000 μM in mCeHR for 9 days. The number of worms in each concentration was counted and plotted. At 1.5 μM heme concentrations worms were L4 and unable to further develop. At 800 μM heme the worms were stunted at L2-L3 stages and showed effects of heme toxicity.

Figure 2. Response of heme sensor (Phrg-1::GFP) strain to different heme concentrations in mCeHR. Synchronized transgenic C. elegans expressing hrg-1::GFP were grown in mCeHR media supplemented with 4, 8, 10, or 20 µM heme for 48 hr. Images were taken with a Zeiss LSM 710 confocal microscope. Scale bar is 100 µm. Please click here to view a larger version of this figure.
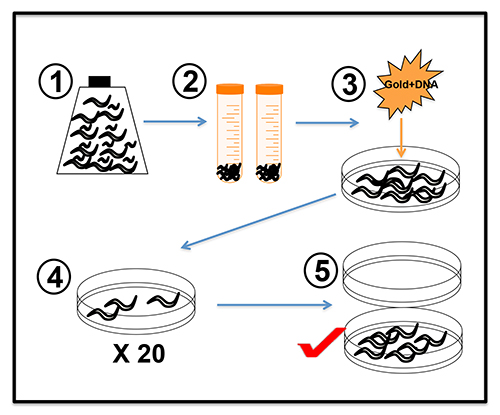
Figure 3. Schematic of microparticle bombardment in C. elegans using unc-119 worms grown in mCeHR. (1) Approximately 3 x 107 unc-119(ed3) worms are grown in 90 ml mCeHR media. (2) Gravids were allowed to settle on ice for 10-15 min in 50 ml conical tubes. (3) A 2 ml pellet of approximately 5 x 106 gravid worms was spread evenly onto an unseeded NGM plate. The worms were bombarded with 12 μg of plasmid of interest and 6 μg of unc-119 rescue plasmid complexed to gold particles. (4) The bombarded worms were split onto twenty 10 cm plates seeded with the E. coli strain JM109. (5) After 2 weeks incubation at 25 °C, plates with wild type worms were selected for analysis of transgene expression strength and segregation rates.
| Table 1A | |
| CeHR, 1 L | |
| Using sterile technique and a 1 L (0.22 μm) vacuum filter unit, filter the following volumes of stock solutions and water in the order described. | |
| Choline diacid citrate | 10 ml |
| Vitamin and growth factor mix | 10 ml |
| myo-Inositol | 10 ml |
| Hemin chloride | 10 ml |
| Deionized water | 250 ml |
| Nucleic acid mix | 20 ml |
| Mineral Mix | 100 ml |
| Lactalbumin hydrolysate | 20 ml |
| Essential Amino Acid Mix | 20 ml |
| Non-essential Amino Acid Mix | 10 ml |
| KH2PO4 | 20 ml |
| D-Glucose | 50 ml |
| HEPES, sodium salt | 10 ml |
| Deionized water | 250 ml |
| Volume will be 800 ml at this point Remove filter unit from vacuum then add: |
|
| Cholesterol | 1 ml |
| Ultra-pasteurized skim milk | 200 ml |
| Table 1B | |
| Vitamin and growth factor mix, 100 ml | |
| Solution 1: To 60 ml of water add: | |
| N-acetyl-α-D-glucosamine | 0.15 g |
| DL-alanine | 0.15 g |
| Nicotinamide | 0.075 g |
| D-pantethine | 0.0375 g |
| DL-pantothenic acid, hemi calcium salt | 0.075 g |
| Folic acid | 0.075 g |
| Pyridoxamine 2HCl | 0.0375 g |
| Pyridoxine HCl | 0.075 g |
| Flavin mononucleotide, sodium salt | 0.075 g |
| Thiamine hydrochloride | 0.075 g |
| Solution 2: Prepare the following chemicals in 5 ml 1 N KOH: | |
| p-aminobenzoic acid | 0.075 g |
| D-biotin | 0.0375 g |
| Cyanocobalamin (B12) | 0.0375 g |
| Folinic acid, calcium salt | 0.0375 g |
| Nicotinic acid | 0.075 g |
| Pyridoxal 5-phosphate | 0.0375 g |
| Solution 3: 0.0375 g (±) α-L-lipoic acid, oxidized form in 1 ml ethanol: | |
| Combine solutions 1, 2, and 3 and bring the final volume to 100 ml. Store in dark at 4 °C or freeze aliquots at -20 °C. Make small volumes of stocks for this mix so it is used quickly. |
|
| Table 1C | |
| Nucleic acid mix, 100 ml | |
| To 60 ml of water add: | |
| Adenosine 5' -monophosphate, sodium salt | 1.74 g |
| Cytidine 5' -phosphate | 1.84 g |
| Guanosine 2' – and 3' -monophosphate | 1.82 g |
| OR | |
| Guanosine 5' -phosphate | 2.04 g |
| Uridine 5' -phosphate, disodium salt | 1.84 g |
| Thymine (add last) | 0.63 g |
| Bring solution to 100 ml and store in the dark at 4 °C or freeze aliquots at -20 °C. Make small volumes of stocks for this mix so it is used quickly. |
|
| Table 1D | |
| Mineral Mix, 1 L | |
| MgCl2•6H2O | 4.1 g |
| Sodium citrate | 2.9 g |
| Potassium citrate monohydrate | 4.9 g |
| CuCl2•2H2O | 0.07 g |
| MnCl2•4H2O | 0.2 g |
| ZnCl2 | 0.1 g |
| Fe(NH4)2(SO4)2•6H2O | 0.6 g |
| CaCl2•2H2O (always add last) | 0.2 g |
| Make small volumes of stocks for this mix so it is used quickly. | |
| Table 1E | |
| Other Components | |
| KH2PO4 | 450 mM |
| Choline di-acid citrate | 2 mM |
| myo-Inositol | 2.4 mM |
| D-Glucose | 1.45 M |
| Hemin chloride | 2 mM in 0.1 N NaOH pH 8.0 |
| HEPES, sodium salt | 1 M stock solution |
| Cholesterol | 5 mg/ml in ethanol |
| Lactalbumin enzymatic hydrolysate | 170 mg/ml |
| Table 1F | |
| M9 Buffer, 1 L | |
| KH2PO4 | 3 g |
| Na2HPO4 | 6 g |
| NaCl | 5 g |
| H2O | 1 L |
| Autoclave 30 min | |
| 1 M MgSO4 (sterile) | 1 ml |
Table 1. Recipes for components of mCeHR and mCeHR.
| Bombardment | Lines with wild type rescue | Lines with rescue / transgene | Stable lines |
| 1 | 2 | 2 | 1 |
| 2 | 8 | 5 | 0 |
| 3 | 4 | 4 | 2 |
| 4 | 5 | 2 | 1 |
| 5 | 5 | 3 | 1 |
| Average | 4.8 | 3.2 | 1 |
Table 2. Average number of transgenic C. elegans generated using microparticle bombardment.
Discussion
In this protocol we present a modified axenic liquid media mCeHR that allows for rapid C. elegans generation with production of a large number of worms. This media shows several advantages as the worms are grown without contaminating E. coli or bacterial byproducts and can be exploited in nutritional and toxicological studies. The use of E. coli or other bacteria in such studies has several drawbacks. For example, the growth of the bacteria can change under various conditions and the bacteria may metabolize molecules that are being assayed, confounding the interpretation of results. Therefore, the development of a defined medium to perform these studies is highly advantageous.
Although C. elegans have been grown in liquid media in previous studies15, worms grown in the C. elegans maintenance medium (CeMM) show a distinct delay in generation times16, unlike what we observe in mCeHR medium. Our main goal was to exploit C. elegans to study nutrient homeostasis with specific emphasis on heme and metal metabolism. With this in mind, mCeHR and modifications generated by our group accomplish this goal and has directly led to the identification of a number of genes required for maintaining heme homeostasis in the worm and ultimately in vertebrate model systems17,18. Reformulated mCeHR-1 media can also be exploited for other nematode species including Panagrellus redivivus, Oscheius myriophila, and C. remanei. In addition low metal formulations mCeHR-2 and mCeHR-3 can be exploited in studies examining heavy metal toxicity and requirements12.
Prior to acclimatization, N2 worms exhibit a longer generation time of approximately 7 to 10 days in mCeHR media. As the worms are subcultured, this generation time decreases to 4 days, similar to that of worms grown on OP50 bacteria. However, the generation time may be longer for certain mutant worms, plausibly because of defects in feeding, movement, or specific nutritional requirements.
When using axenic liquid media it is critical that fastidious sterile techniques are utilized. Usually, the use of antibiotics is minimized and eventually eliminated as the worms acclimatize to the medium and the residual bacteria are eliminated. Antibiotics are initially required to ensure that the cultures are axenic when established as it prevents the growth of residual bacteria that can overwhelm the worm cultures. After two successive rounds of bleaching, antibiotics are omitted from the cultures as continual usage of antibiotics only masks poor aseptic techniques that are essential for growing worms axenically. Using these techniques in a laminar flow cabinet, the authors have been able to grow the worms axenically without contamination. Additionally, proper storage of the media and components is essential for worm maintenance and growth. The worms should be checked for the rate of growth and subcultured to prevent crowding, which can lead to dauer formation as essential nutrients are depleted. This can be prevented by ensuring that the density of worms does not exceed 3,000 worms/ml/cm2. Researchers that are new to growing C. elegans axenically can achieve this by checking the worms daily and counting the worms weekly.
C. elegans researchers take advantage of its transparent properties by generating transgenic worms expressing fluorescently-tagged markers that allow for visualization of gene expression and protein localization. Utilizing biolistic bombardment allowed for generation of low copy integrants that avoided the issues of extrachromosomal arrays and elevated gene expression noted with transgenics generated using injection19. Previously, one drawback of generating transgenics using microparticle bombardment of C. elegans was the requirement for egg plate preparation to generate the large number of unc-119 (ed3) worms necessary for the procedure20. Each transformation required 20 egg plates for the number of worms needed. Using axenic mCeHR liquid culture allows more efficient growth and subsequently more worms for bombardments. Additionally, bombardments can be used in conjunction with drug selection to avoid using unc-119 (ed3) worms21.
Declarações
The authors have nothing to disclose.
Acknowledgements
This work was supported by the National Institutes of HealthGrants DK85035 and DK074797 (I.H).
Materials
| MgCl2.6H2O | Sigma | M-2393 | |
| Sodium citrate | Sigma | S-4641 | |
| Potassium citrate.H2O | Sigma | P-1722 | |
| CuCl2.2H2O | Fisher | C455-500 | |
| MnCl2.4H2O | Fisher | M87-100 | |
| ZnCl2 | Sigma | Z-0152 | |
| Fe(NH4)2(SO4)2.6H2O | Sigma | F-1018 | |
| CaCl2.2H2O | Fisher | C70-500 | |
| Adenosine 5 -monophosphate, sodium salt | Sigma | A-1752 | |
| Cytidine 5 -phosphate | Sigma | C-1006 | |
| Guanosine 2 – and3 -monophosphate | Sigma | G-8002 | |
| Uridine 5 -phosphate, disodium salt | Sigma | U-6375 | |
| Thymine | Sigma | T0376 | |
| N-Acetylglucosamine | Sigma | A3286 | |
| DL-Alanine | Fisher | S25648 | |
| p-Aminobenzoic Acid | Sigma | A-9878 | |
| Biotin | Sigma | B-4639 | |
| Cyanocobalamine (B-12) | Sigma | V-2876 | |
| Folinate (Ca) | Sigma | F-7878 | |
| Niacin | Sigma | N-0761 | |
| Niacinamide | Sigma | N-3376 | |
| Pantetheine | Sigma | P-2125 | |
| Pantothenate (Ca) | Sigma | P-6292 | |
| Pteroylglutamic Acid (Folic Acid) | ACRCS | 21663-0100 | |
| Pyridoxal 5'-phosphate | Sigma | P-3657 | |
| Pyridoxamine.2HCl | Sigma | P-9158 | |
| Pyridoxine.HCl | Sigma | P-6280 | |
| Riboflavin 5-PO4(Na) | Sigma | R-7774 | |
| Thiamine.HCl | Sigma | T-1270 | |
| DL-6,8-Thioctic Acid | Sigma | T-1395 | |
| KH2PO4 | Sigma | P-5379 | |
| Choline di-acid citrate | Sigma | C-2004 | |
| myo-Inositol | Sigma | I-5125 | |
| D-Glucose | Sigma | G-7520 | |
| Lactalbumin enzymatic hydrolysate | Sigma | L-9010 | |
| Brain Heart Infusion | BD | 211065 | |
| Hemin chloride | Frontier Scientific | H651-9 | |
| HEPES, Na salt | Sigma | H-3784 | |
| Cholesterol | J.T. Baker | F676-05 | |
| MEM Non-Essential Amino Acids | Invitrogen | 11140-076 | |
| MEM Amino Acids Solution | Invitrogen | 11130-051 | |
| Nalidixic acid sodium salt | Sigma | N4382 | |
| Tetracycline Hydrochloride | MP Biomedicals | 2194542 | |
| Biolistic Delivery System | BioRad | 165-2257 | |
| Gold particles (Au Powder) | Ferro Electronic Material Systems | 6420 2504, JZP01010KM | |
| or | |||
| Gold Particles 1.0 μm | BioRad | 165-2263 |
Referências
- Kamath, R. S., et al. Systematic functional analysis of the Caenorhabditis elegans genome using RNAi. Nature. 421, 231-237 (2003).
- Nass, R., Blakely, R. D. The Caenorhabditis elegans dopaminergic system: opportunities for insights into dopamine transport and neurodegeneration. Annual review of pharmacology and toxicology. 43, 521-544 (2003).
- Lapierre, L. R., Hansen, M. Lessons from C elegans signaling pathways for longevity. Trends in endocrinology and metabolism TEM. 23, 637-644 (2012).
- Kenyon, C. The plasticity of aging insights from long-lived mutants. Cell. 120, 449-460 (2005).
- Vanfleteren, J. R., Braeckman, B. P. Mechanisms of life span determination in Caenorhabditis elegans. Neurobiology of aging. 20, 487-502 (1999).
- Poole, R. J., Bashllari, E., Cochella, L., Flowers, E. B., Hobert, O. A Genome-Wide RNAi Screen for Factors Involved in Neuronal Specification in Caenorhabditis elegans. PLoS genetics. 7, e1002109 (2011).
- Stiernagle, T. Maintenance of C elegans WormBook the online review of C. elegans biology. , 1-11 (2006).
- Win, M. T., et al. Validated Liquid Culture Monitoring System for Lifespan Extension of Caenorhabditis elegans through Genetic and Dietary Manipulations. Aging and disease. 4, 178-185 (2013).
- Clegg, E. D., LaPenotiere, H. F., French, D. Y., Szilagyi, M. Use of CeHR Axenic Medium for Exposure and Gene Expression Studies. East Coast Worm Meeting. , (2002).
- Severance, S., et al. Genome-wide analysis reveals novel genes essential for heme homeostasis in Caenorhabditis elegans. PLoS genetics. 6, e1001044 (2010).
- Rajagopal, A., et al. Haem homeostasis is regulated by the conserved and concerted functions of HRG-1 proteins. Nature. 453, 1127-1131 (2008).
- Nass, R., Hamza, I. Chapter 1 Unit 1 9. The nematode C. elegans as an animal model to explore toxicology in vivo: solid and axenic growth culture conditions and compound exposure parameters. Current protocols in toxicology editorial board, Mahin D. Maines. , (2007).
- Schweinsberg, P. J., Grant, B. D. C. elegans gene transformation by microparticle bombardment WormBook the online review of C. elegans biology. , 1-10 (2013).
- Rao, A. U., Carta, L. K., Lesuisse, E., Hamza, I. Lack of heme synthesis in a free-living eukaryote. Proceedings of the National Academy of Sciences of the United States of America. 102, 4270-4275 (2005).
- Szewczyk, N. J., Kozak, E., Conley, C. A. Chemically defined medium and Caenorhabditis elegans. BMC biotechnology. 3, 19 (2003).
- Szewczyk, N. J., et al. Delayed development and lifespan extension as features of metabolic lifestyle alteration in C elegans under dietary restriction. The Journal of experimental biology. 209, 4129-4139 (2006).
- White, C., et al. HRG1 is essential for heme transport from the phagolysosome of macrophages during erythrophagocytosis. Cell metabolism. 17, 261-270 (2013).
- Chen, C., Samuel, T. K., Sinclair, J., Dailey, H. A., Hamza, I. An intercellular heme-trafficking protein delivers maternal heme to the embryo during development in C elegans. Cell. 145, 720-731 (2011).
- Praitis, V., Casey, E., Collar, D., Austin, J. Creation of low-copy integrated transgenic lines in Caenorhabditis elegans. Genética. 157, 1217-1226 (2001).
- Berezikov, E., Bargmann, C. I., Plasterk, R. H. Homologous gene targeting in Caenorhabditis elegans by biolistic transformation. Nucleic acids research. 32, e40 (2004).
- Semple, J. I., Biondini, L., Lehner, B. Generating transgenic nematodes by bombardment and antibiotic selection. Nature Methods. 9, 118-119 (2012).

