An Efficient and Flexible Cell Aggregation Method for 3D Spheroid Production
Summary
Here, we describe a rapid and flexible protocol for the formation of 3D cell spheroids through cell aggregation. This is easily adapted to multiple cell types and is suitable for use in a variety of applications including cell migration, invasion, or anoikis assays, and for imaging and quantifying cell-matrix interactions.
Abstract
Monolayer cell culture does not adequately model the in vivo behavior of tissues, which involves complex cell-cell and cell-matrix interactions. Three-dimensional (3D) cell culture techniques are a recent innovation developed to address the shortcomings of adherent cell culture. While several techniques for generating tissue analogues in vitro have been developed, these methods are frequently complex, expensive to establish, require specialized equipment, and are generally limited by compatibility with only certain cell types. Here, we describe a rapid and flexible protocol for aggregating cells into multicellular 3D spheroids of consistent size that is compatible with growth of a variety of tumor and normal cell lines. We utilize varying concentrations of serum and methyl cellulose (MC) to promote anchorage-independent spheroid generation and prevent the formation of cell monolayers in a highly reproducible manner. Optimal conditions for individual cell lines can be achieved by adjusting MC or serum concentrations in the spheroid formation medium. The 3D spheroids generated can be collected for use in a wide range of applications, including cell signaling or gene expression studies, candidate drug screening, or in the study of cellular processes such as tumor cell invasion and migration. The protocol is also readily adapted to generate clonal spheroids from single cells, and can be adapted to assess anchorage-independent growth and anoikis-resistance. Overall, our protocol provides an easily modifiable method for generating and utilizing 3D cell spheroids in order to recapitulate the 3D microenvironment of tissues and model the in vivo growth of normal and tumor cells.
Introduction
Biologically relevant assessment of tumor cell behavior is challenging using traditional two-dimensional (2D) cell culture methodologies, in part because these do not adequately reflect the cell microenvironment found in vivo. Alternative approaches incorporating extracellular matrix components into the culture (e.g., Boyden chamber assays) are more physiologically representative of the in vivo tissue environment. However, they can be limited to assessment of individual cell behavior, and do not recapitulate the complex in vivo combinations of cell-matrix and cell-cell interactions that contribute to tissue or tumor growth1,2,3.
The use of multicellular spheroids is a recent approach that more accurately reproduces the compact architecture of in vivo cell growth1,4. Spheroids can be used to investigate cell-matrix interactions of normal cells, but can also act as tumor analogues to model characteristics of tumor progression, such as metastatic growth or drug resistance4.
Spheroids may be formed by the proliferation of single cells embedded in a matrix5, or more rapidly, by promoting the aggregation of multiple cells to form a single cell cluster (e.g., hanging drop, centrifugation methods)6,7. Existing cell aggregation techniques may require costly materials or specialized equipment. In addition, these spheroids have a wide range of sizes and morphologies and may be difficult to produce in large quantities, making comparisons between growth conditions or treatments difficult. Finally, spheroids generated by these methods can be difficult to isolate from the proteinaceous extracellular matrix in which they are embedded for use in other applications.
Here, we describe a robust and easily modifiable cell aggregation methodology for the rapid formation of consistently sized cell spheroids using commercially available U-bottom cell-repellent plates and an inert adhesion-promoting matrix, methyl cellulose. Once formed, these multicellular spheroids are readily isolated for use in a wide range of applications. The protocol is also easily adapted to generate spheroids through cell proliferation, which may be used to assess other cell processes. Here, we show cell invasion assays, quantified by immunofluorescence staining, and an anoikis assay, as example applications of these two different spheroid formation protocols.
Protocol
NOTE: All reagents and consumables are listed in the Materials List.
1. Spheroid Production by Cell Aggregation
- Methyl cellulose solution: Prepare 100 mL of 100 mg/mL methyl cellulose.
- Heat 50 mL ultrapure H2O to 80 °C. Add 10 g methyl cellulose powder and agitate until particles are evenly dispersed.
- Bring to final volume with cold ultrapure H2O and stir at 4 °C until the solution becomes clear, straw colored, and contains no visible solids.
- Pass through a 0.45 µm filter to remove undissolved solids. Prepared solution may be aliquoted and stored at 4 °C for up to 12 months.
NOTE: Methyl cellulose serves as a non-cytotoxic, inert, suspending agent to enhance cell-cell adhesion and discourage the formation of an adherent cell monolayer. Methyl cellulose is water soluble and can be washed away following spheroid generation.
- Spheroid formation medium
NOTE: Prepare spheroid formation medium immediately before use. Do not store.- Dilute methyl cellulose solution to 1-5 mg/mL in the appropriate culture medium.
- Pass through a 0.22 µm filter to sterilize and remove undissolved solids.
- Dulbecco's phosphate buffered saline (PBS)
- Dilute to 1x and pass through a 0.45 µm filter before use. Prepared solution may be stored indefinitely at room temperature.
- Cell spheroid production (Figure 1)
NOTE: Prepare all reagents in advance. It is important to avoid contamination of solutions by dust, fibers or other insoluble particles. The presence of these contaminants will adversely affect spheroid formation. Culture medium and other solutions should be passed through a 0.45 µm filter to remove these particles when possible. Avoid the use of cotton-filtered pipettes when transferring solutions as these can introduce additional fibers.- Culture cell monolayers to 70% confluence in the appropriate culture medium supplemented with 10% serum. Aspirate culture medium and rinse with 1x PBS to remove debris. Add 3 mL of trypsin-EDTA (0.05% trypsin) dissociation buffer to a 10 cm cell culture dish, and incubate cells at 37 °C and 5% CO2.
- Inspect cells under microscope at 10X magnification to confirm detachment. Neutralize dissociation buffer with 7 mL of serum-supplemented culture medium.
- Centrifuge cell suspension at 500 x g for 10 min to gently pellet cells.
- Remove supernatant. Resuspend cell pellet in 1 mL spheroid formation medium using a p1000 pipette with a filter tip.
NOTE: The cell suspension should be free of clumps.- To triturate cell pellet, press the pipette tip against the bottom of the tube at a slight angle while pipetting to shear cell pellet. Avoid triturating more than 3 – 5 times as this can damage the cells. Pipette slowly and do not expel entire contents of the pipette tip to avoid creating bubbles.
- Count cells using a hemocytometer and dilute cell suspension to 1 x 104 cells/mL.
NOTE: The cell number can be optimized to generate spheroids of varying sizes. Trypan blue staining of damaged cells may be used to confirm viability of resuspended cells. - Transfer cell suspension to a sterile, dust-free multichannel pipette reservoir and use filter tips to dispense 100 µL into each well of a 96-well U-bottom cell-repellent plate.
- Rinse reservoir with filtered ultrapure H2O to remove dust and fibers.
- Mix the cell suspension periodically while seeding to prevent cells from settling to the bottom of the reservoir. To avoid creating bubbles, use a reverse pipetting technique while mixing and dispensing.
- Transfer plates to tissue culture incubator (37 °C and 5% CO2) and inspect daily for spheroid formation. Cells should settle to the bottom of each well within 6 h and generally aggregate into a spheroid within 24-48 h (Figure 2).
- For confirmation of successful spheroid formation, use a p10 tip and gently pipette medium over the spheroid while observing under a microscope at 10X magnification. Properly formed spheroids will loosen from the plate and roll, confirming their 3D structure.
NOTE: Methyl cellulose and serum concentrations should be determined by titration for each cell line to yield formation of a single spheroid per well. Conditions optimized in this way for selected cell lines are shown in Table 1, and examples of the spheroids formed in Figure 2B. Spheroids may be grown for longer than indicated but as they become larger they grow progressively more difficult to handle without fragmentation. If cells form a monolayer, satellite spheroids, or fail to settle to the bottom of the well, the concentration of methyl cellulose and serum may need to be further adjusted for optimal spheroid formation (Figure 3B). Do not use spheroids containing contaminants such as fibers or dust (Figure 3A). Pre-formed spheroids can be treated with compounds of interest such as growth factors, live cell stains, or inhibitors for analysis.
2. Spheroid Invasion Assay (Figure 4)
- Neutralized collagen
- Cool all liquids to 4 °C and hold on ice when working with collagen to prevent unwanted polymerization.
- Prepare fresh 0.1 M and 0.01 M solutions of NaOH and fresh 0.1 M HCl in ultrapure H2O. Pass all solutions through 0.22 µm filters.
- Mix bovine type 1 collagen (3.1 mg/mL stock), 0.01 M NaOH and 10x PBS (8:1:1 ratio by volume) and adjust to pH 7.4 using cold 0.1 M NaOH and HCl. The final concentration of collagen is 2.5 mg/mL.
- Prepare enough neutralized collagen to fill the imaging vessel to a depth of 2-3 mm. A minimum of 100 µL is required for each well of the 8-well tissue culture chamber slide listed in Materials List.
- Embedding spheroids in collagen (Figure 4)
- Coat the bottom of each well of an 8-well tissue culture chamber slide with a minimum of 50 µL neutralized collagen.
- Use reverse pipetting technique to avoid creating bubbles in the collagen. Use the pipette tip to spread the collagen evenly over the well surface.
NOTE: This collagen base layer will hold spheroids in suspension and is typically 1-2 mm thick. The base layer minimizes formation of a meniscus on the upper collagen layer. If the base layer is too thin, spheroids may make contact with the bottom of the chamber slide and form a cell monolayer.
- Use reverse pipetting technique to avoid creating bubbles in the collagen. Use the pipette tip to spread the collagen evenly over the well surface.
- Place the chamber slide, and a 35 mm tissue culture dish filled with ultrapure H2O, in a 10 cm tissue culture dish. Incubate at 37 °C until the collagen is polymerized, typically 1 h. Store remaining neutralized collagen on ice.
NOTE: The water-filled 35 mm dish will provide humidity and prevent drying. The chamber slide should remain in this humidity chamber throughout the assay. The collagen base layer may be left to polymerize overnight. Longer incubations typically result in a more rigid gel. - Using a p1,000 filter tip, collect pre-formed spheroids (step 1.4.7) from the 96-well U-bottom cell-repellent plate in 1.5 mL snap top tubes. Pipette slowly and avoid repeated or vigorous pipetting to minimize fluid-shearing of spheroids. Multiple spheroids may be pooled in a single tube for transfer to one well of the 8-well tissue culture chamber slide.
- Centrifuge the collected spheroids in a tabletop microcentrifuge at 2,000 x g for 2-5 s and remove supernatant using a p200 pipette. Avoid centrifugation for longer than required, as this may cause the spheroids to break apart or clump together.
NOTE: After pipetting off most of the supernatant, the tube may be inverted carefully over a clean paper towel to wick away excess liquid. Do not allow spheroids to dry. - Using a wide bore p200 tip, gently resuspend spheroids in 50 µL neutralized collagen. Do this for each tube of collected spheroids before any spheroids are transferred into the chamber slide. Keep spheroids that have been resuspended in neutralized collagen on ice until ready for transfer to chamber slides.
- Remove chamber slide from incubator and carefully pipette the spheroid-collagen mixture on top of collagen base layer. Incubate at 37 °C and 5% CO2 until collagen is polymerized.
- Do not dispense entire contents of pipette to avoid creating bubbles. Dropwise pipetting reduces the chances of disturbing the collagen base layer.
- Slowly add a minimum of 100 µL warmed culture medium containing desired chemoattractants, inhibitors, or other treatments to each well of the chamber slide. The collagen layer containing spheroids may tear if liquid is pipetted onto it too vigorously. Culture medium can be slowly dispensed down the side of a well to avoid disturbing the collagen.
- Incubate chamber slide at 37 °C and 5% CO2 to allow cell invasion. Cell invasion into the surrounding collagen may be observed by brightfield or fluorescence imaging and quantified by a variety of methods using epifluorescence, confocal or light sheet microscopy.
- Coat the bottom of each well of an 8-well tissue culture chamber slide with a minimum of 50 µL neutralized collagen.
3. Quantification of Invasion: Brightfield Microscopy
- At set time intervals, image collagen-embedded spheroids at 10X magnification using a brightfield microscope (step 2.2.8).
NOTE: For longer-term live-cell experiments, a heated microscope stage and CO2 regulated chamber can be used to maintain spheroids at 37 °C and 5% CO2 while imaging. - Quantify invasiveness using software such as ImageJ to measure the number of cells invading into the surrounding collagen and the average distance invaded at each time point.
4. Quantification of Invasion: Fluorescence Microscopy with Live Cell Stain
- Following step 2.2.8, supplement the medium in the chamber slides with a fluorescent stain such as DAPI, Calcein AM, or other cell-tracking compound appropriate to the cell line as per the manufacturer's instructions.
NOTE: For quantification of invasiveness, homogeneous staining throughout the spheroid is not essential. For applications requiring deeper penetration of stain, longer incubations (>3 h) may be necessary. - Remove the stain and replace with 500 µL warm 1x PBS. Incubate at 37 °C for 10 min to remove excess stain. Repeat a minimum of two times.
- Using a fluorescence microscope, acquire optical sections through the invading spheroids.
NOTE: Prolonged exposure to laser light should be minimized during repetitive imaging to limit phototoxicity and photobleaching. - Use software such as ImageJ to generate a Z-projection, which can be used to quantify the total area of the spheroid, numbers of invading cells, and distances invaded into the collagen matrix.
- As an example, to quantify invasion, adjust the image threshold to exclude background, highlighting only the spheroid and invading cells, and measure their area. The area of the original spheroid can be subtracted from this total to quantify invasiveness.
5. Quantification of Invasion: Immunofluorescence
NOTE: All solutions and buffers should be passed through a 0.45 µm filter before use to remove debris, which can negatively affect staining.
- Prepare PBS+ wash buffer. Prepare 1x PBS and add CaCl2 and MgCl2 to a final concentration of 0.1 mM. Do not autoclave buffer after CaCl2 and MgCl2 are added. Solution may be filter-sterilized and stored at room temperature.
- Prepare neutral buffered formalin (NBF) fixation buffer. Add 5 drops of 1 M NaOH to 0.6 g paraformaldehyde. Bring to 20 mL with PBS+ and incubate at 60 °C until paraformaldehyde is dissolved (approximately 1 h). Cool to room temperature and adjust pH to 7.4 with 1 M HCl.
NOTE: NBF fixation buffer should be prepared immediately before use. - Prepare bovine serum albumin (BSA) blocking buffer. Dissolve BSA in PBS+ at 3% w/v. Solution may be filter sterilized and stored at 4 °C for several days but is best prepared fresh. Do not heat.
- Prepare Permeabilization buffer. Dilute Triton X-100 to 0.2% v/v in PBS+.
NOTE: Permeabilization buffer should be prepared immediately before use. - Optionally, prepare MOWIOL mounting medium. Combine 2.4 g MOWIOL, 6 g glycerol, and 6 mL ultrapure H2O. Mix for 3 h in a rotator at room temperature. Add 12 mL 0.2 M Tris-Cl (pH 8.5). Incubate with mixing at 50 °C for 10 min.
- Centrifuge at 5,000 x g for 15 min to pellet insoluble material. Add anti-bleaching agent, such as 2.5% DABCO. Store in 500 µL aliquots at -20 °C.
- Immunofluorescence staining of spheroids (Figure 5)
NOTE: Use a pipette to slowly add or remove liquids from chamber slide. Avoid the use of an aspirator, as this may disrupt the collagen layer.- Prepare spheroids and embed in collagen-filled chamber slides, as described in sections 1 to 2.
NOTE: Collagen autofluorescence can be minimized by using thin layers or small volumes of collagen. - Remove as much culture medium as possible from each chamber slide.
- Briefly rinse collagen layer with 500 µL PBS+ wash buffer to remove residual medium.
- Add 500 µL fresh PBS+ wash buffer to each well and incubate at 37 °C for 5 min to wash spheroids. Repeat twice.
- Replace PBS+ wash buffer with 500 µL NBF fixation buffer. Incubate at room temperature for 20 – 25 min.
- Remove NBF fixation buffer and rinse with 500 µL PBS+ wash buffer. Wash for 5 min with fresh PBS+ wash buffer a minimum of 3 times.
NOTE: Fixed spheroids can be stored at 4 °C in fresh PBS+ wash buffer for several days. 0.01% sodium azide may be added to the wash buffer to inhibit microbial growth during storage. - Replace PBS+ wash buffer with 500 µL permeabilization buffer. Incubate at room temperature for 10-15 min.
- Replace permeabilization buffer with 500 µL BSA blocking buffer and incubate at room temperature for 1 h.
- Optionally, remove BSA blocking buffer. Using scalpel or scissors, excise spheroids and the surrounding 3 mm x 3 mm area from the collagen layer. Using a wide bore p1000 tip, dislodge the excised block of collagen by rinsing gently with fresh BSA blocking buffer. Transfer collagen block to 1.5 mL tube.
- Centrifuge at 2,000 x g for 2 min and remove supernatant. The tube can be carefully inverted over clean paper towel to wick away excess liquid.
- Add primary antibody to spheroids and incubate at room temperature for 1 h. Add sufficient volume of primary antibody such that the entire collagen layer is evenly submerged. The optional steps above (5.6.8.1-5.6.8.2) typically permit the use of smaller volumes of antibody.
- Submerge chamber slide in a container with at least 10 mL PBS+ wash buffer for 10 min to wash. Repeat a minimum of two times.
- Optional, if spheroids were excised from collagen layer previously (Step 5.6.8.1), add a minimum of 1 mL PBS+ wash buffer to the 1.5 mL tube and gently agitate. Allow to soak for a minimum of 10 min.
- Remove as much PBS+ wash buffer as possible and repeat washing with PBS+ wash buffer a minimum of two times. Centrifuge at 2,000 x g for several min to pellet collagen block.
NOTE: Clean outside surfaces of the chamber slide by wiping with 70% ethanol before submerging in wash buffer.
- Repeat steps 5.6.9-5.6.10 using appropriate secondary antibody.
NOTE: If spheroids were stained directly in the imaging vessel, proceed to step 5.6.13. - Optional, if spheroids were excised from collagen layer previously (Step 5.6.8.1), clean glass slides and confocal microscopy-compatible coverslips with 70% ethanol.
- Remove as much wash buffer as possible from the collagen block and resuspend in ultrapure H2O. Perform this step immediately before mounting. Do not leave stained blocks soaking in water.
- Using fine tipped forceps, grasp edge of collagen block and transfer to glass slide.
- Using the forceps to hold the collagen in place, use a clean cotton swab to wick excess liquid from the collagen block, making sure not to touch the collagen directly.
NOTE: If collagen becomes stuck to cotton swab it can be gently removed either using forceps, or by submerging in water.
- Using reverse pipetting technique, dispense 100 µL MOWIOL mounting medium onto the collagen block and place coverslip over the sample.
- Use a cotton swab or paper towel to wick excess MOWIOL mounting medium and water out from under the coverslip.
- Allow MOWIOL mounting medium to harden overnight at 4 °C. Seal edges of coverslip with nail polish.
- Image stained spheroids using confocal or fluorescence microscopy to observe localization of proteins of interest, formation of invasive structures, etc. (Figures 5B, 5C).
NOTE: Stained spheroids should be protected from light to prevent photobleaching.
- Prepare spheroids and embed in collagen-filled chamber slides, as described in sections 1 to 2.
6. Anoikis Assay
NOTE: The spheroid formation protocol is easily adapted to quantify anchorage-independent growth and anoikis resistance in a variety of cell types.
- Seeding cells for anoikis assay (Figure 6)
- Follow instructions for spheroid production in section 1.4 but use a minimum of 30 mg/mL methyl cellulose to prepare spheroid formation medium.
NOTE: When warmed, 30 mg/mL methyl cellulose should produce a thick gel that holds cells in suspension. - Incubate cells for several days to weeks and inspect regularly at 10X magnification using a microscope. Cells that resist anoikis should proliferate and form colonies.
- Quantify anoikis by measuring the number and size of colonies formed in each well.
NOTE: Once colonies are formed, they may be washed to remove the methyl cellulose and used for spheroid-based assays, as described.
- Follow instructions for spheroid production in section 1.4 but use a minimum of 30 mg/mL methyl cellulose to prepare spheroid formation medium.
Representative Results
We describe a flexible and efficient method to generate discrete spheroids using cell-repellent plates and spheroid formation media supplemented with MC. Under the appropriate conditions of MC and serum, individual cells settle and adhere together at the center of the well to form spheroids with minimal adherence to the well bottom. Using this protocol, spheroids were generated from a variety of cell lines (Figure 2B). Titration of MC and serum concentrations is required for each cell line to identify optimal conditions where only a single spheroid is formed that is robust enough to allow manipulation without fragmenting. Optimally, the spheroids were between 200 to 500 µm in diameter, and consisted of tightly adherent cells with minimal cell debris. Spheroids survived gentle handling without damage, allowing them to be collected and used in a wide variety of assays.
Spheroid formation could be compromised by bacterial or other microbial contaminants (Figure 3A), which resulted in aggregates of dead cells. In the presence of dust or other fiber contamination, multiple or irregularly shaped cell clusters that were only weakly aggregated formed, and the resultant spheroids easily broke apart when handled. Spheroid formation was also affected by suboptimal concentrations of MC or serum in the spheroid formation medium (Figure 3B). In our testing, many cell lines were able to adhere to cell-repellent plates in the absence, or at low concentrations, of MC, and resulted in the formation of a spheroid surrounded by a cell monolayer (Figure 3B-ii). BxPC-3 cell growth in suboptimal MC conditions is shown as an example. In general, higher concentrations of MC prevented cells from adhering to the well, but too high a concentration of MC reduced cell-cell adhesion, and prevented cells from settling to the bottom of the well, resulting in the formation of loose aggregates and numerous satellite spheroids (Figure 3B-i). The concentration of serum also affected cell survival, and cell-cell, and cell-plastic adhesion and needed to be optimized for different cell lines. For cell lines such as HCT-116 and PANC-1, too high a serum concentration resulted in excessive cell proliferation and production of oversized spheroids that were easily damaged by handling, or promoted cell adhesion to the plastic well and the formation of a monolayer (Figure 3B-iii). Interestingly, the effects of insufficient serum differed between cell lines. HCT-116 cell survival was reduced and the spheroids formed were small, containing a large proportion of dead cells in low serum. In contrast, PANC-1 cells were viable in the absence of serum, but became more adherent, and formed multiple aggregates as well as a cell monolayer (Figure 3B-iv, v).
In invasion assays, pre-formed spheroids were resuspended in neutralized collagen to generate a rigid extracellular matrix (Section 2.2, Figure 4). Subsequent addition of a chemoattractant induced individual cells to move outwards from the spheroid and invade into the surrounding matrix (Figure 4B). The number of invading cells and distance invaded can be quantified through brightfield microscopy, or by fluorescence microscopy in the presence of a live cell stain such as DAPI or Calcein AM (Sections 3, 4). In our testing, morphological changes, such as the formation of protrusions, were visible within 6 h of treatment with chemoattractant, and numerous cells could be observed completely detaching from the spheroid and invading into the collagen within 18 to 48 h. For our experiments, we found that 12 to 18 h of invasion was ideal, as longer incubation times resulted in cells moving too far from the spheroid for optimal imaging. Fixed collagen-embedded spheroids could be imaged by brightfield, to quantify the distance and number of cells invading, or immunofluorescence microscopy (Sections 3-5), for visualization of invasive structures formed by the cells (Figures 5B, 5C).
A modification of the described spheroid formation protocol, in which individual cells are suspended in higher concentration MC (30 mg/mL) can be used to monitor anoikis resistance. At these MC concentrations, medium can still be transferred by pipette while cool, but thickened into a solid gel at 37 °C. Anoikis-resistant cells resuspended into this medium remained in suspension and proliferated over a period of 2-4 weeks, forming suspended spheroids of varying sizes (Figure 6B). Anchorage-dependent cells do not form spheroids under these conditions. We were able to quantify anoikis-resistance by counting the number of spheroids formed in each well.
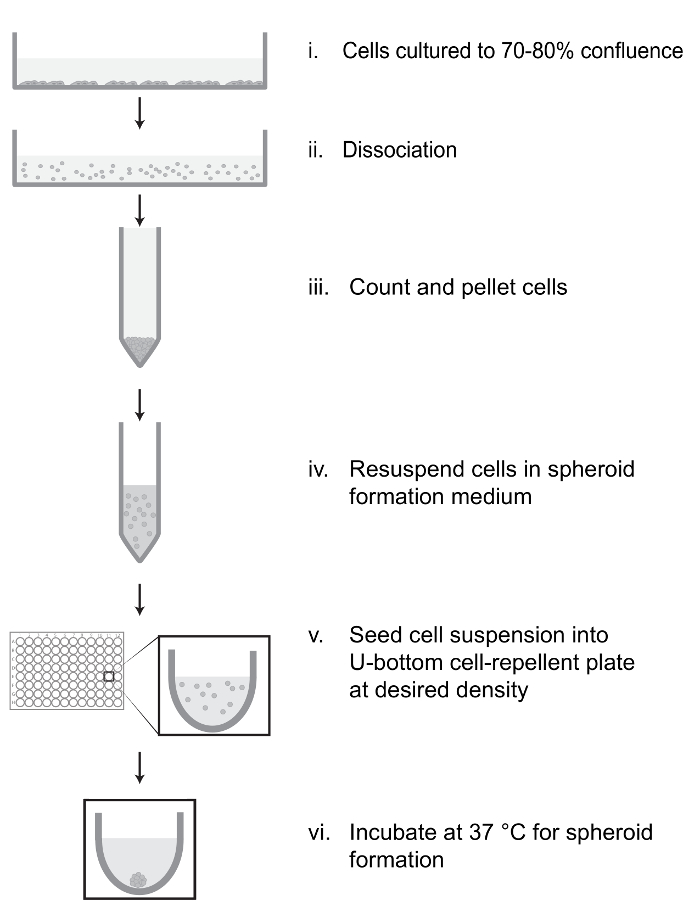
Figure 1: Overview of 3D Spheroid Formation Protocol. Cells grown in monolayer culture are dissociated, counted, pelleted, and resuspended in spheroid formation medium supplemented with methyl cellulose (MC) and serum (i-iv). The suspension is seeded into a U-bottom cell-repellent plate at desired density to allow formation of spheroids of the desired size (v), and the plate is incubated to produce a single, discrete cell spheroid in each well (vi). Please click here to view a larger version of this figure.
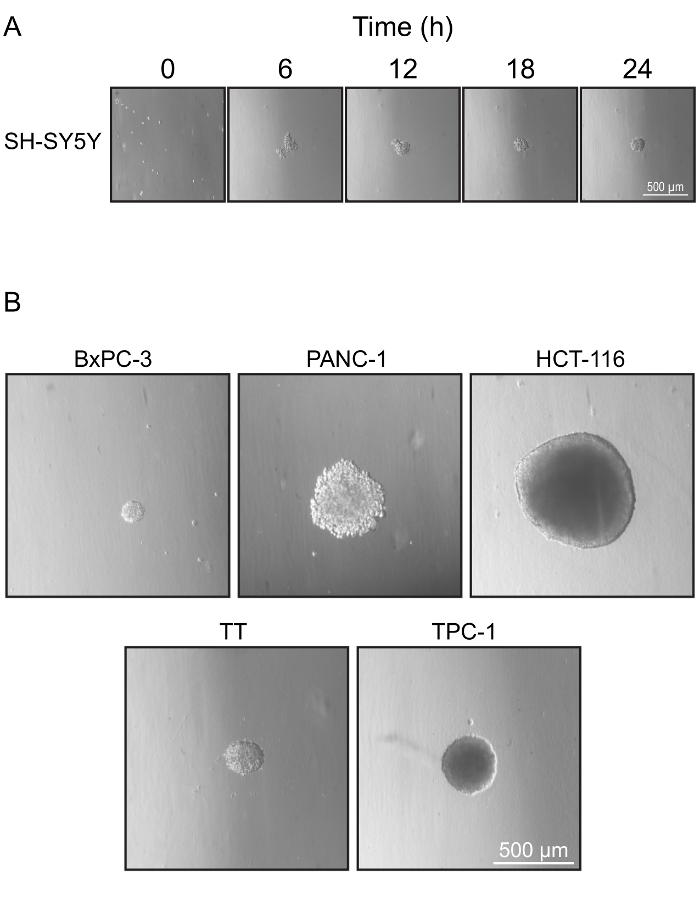
Figure 2: Optimized Spheroid Formation Conditions. (A) Spheroid formation under optimal conditions for SH-SY5Y cells. 1,000 cells seeded into U-bottom cell-repellent plates in media supplemented with 5% serum and 1 mg/mL MC aggregated into compact spheroids within 24 h. (B) Representative images of spheroids formed by various cell lines under optimal conditions shown in Table 1. Please click here to view a larger version of this figure.
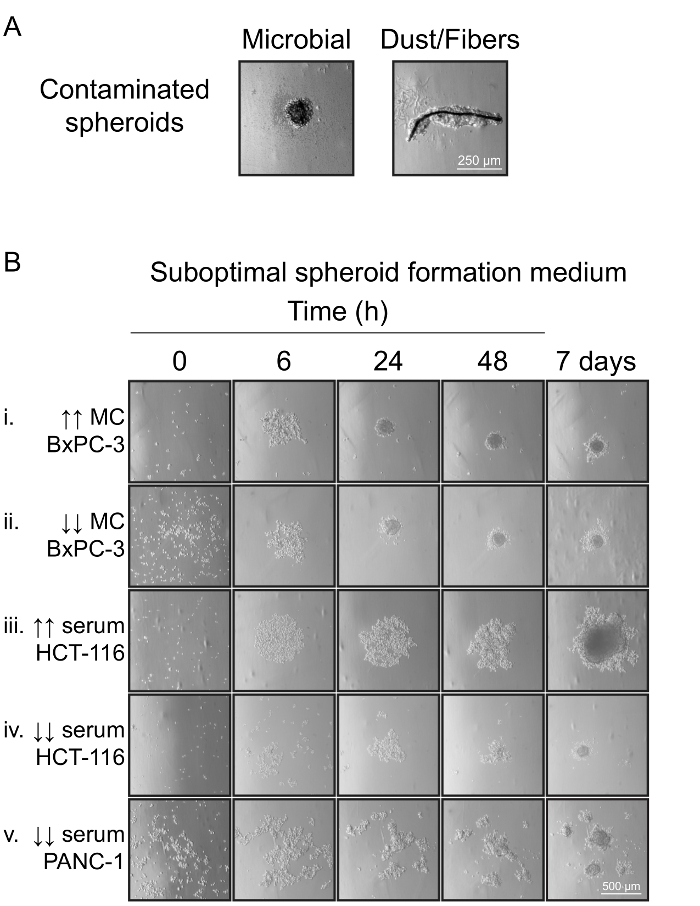
Figure 3: Spheroid Formation Under Suboptimal Conditions. (A) Spheroids formed in the presence of contaminants. Microbial contamination resulted in cell death and loosely aggregated spheroids. In the presence of insoluble contaminants, such as dust or fibers, cells adhered to these materials and did not aggregate. (B) Spheroid formation in suboptimal spheroid formation medium. BxPC-3 cells seeded in the presence of excess MC (10 mg/mL) formed numerous satellite spheroids (i), while insufficient MC (0 mg/mL) resulted in cell aggregates that adhered to the well bottom and formed a monolayer (ii). Similar results were observed for other tested cell lines (not shown). Excess serum (20%) resulted in increased HCT-116 cell proliferation and monolayer formation (iii). Insufficient serum (0%) had cell line-specific effects ranging from cell death in HCT-116 (iv) to the formation of satellite spheroids in PANC-1 (v). Please click here to view a larger version of this figure.
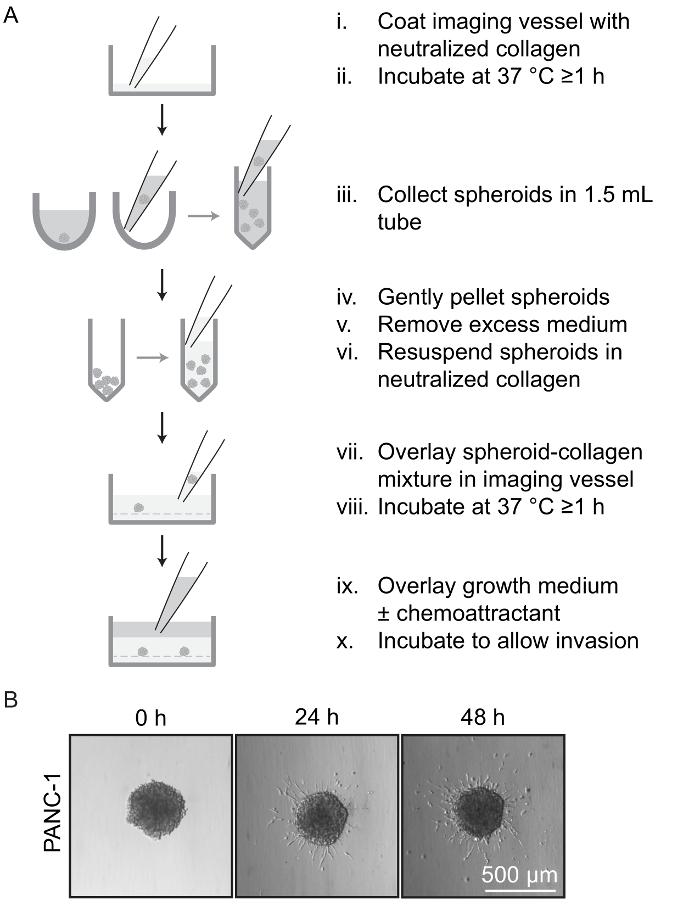
Figure 4: Invasion Assay Using Spheroids. (A) Overview of spheroid invasion assay. The imaging vessel is pre-coated with a layer of neutralized collagen and incubated to polymerize the collagen (i-ii). Pre-formed spheroids are collected in 1.5 mL tubes and resuspended in neutralized collagen (iii-vi). The spheroid-collagen mixture is overlaid on the collagen base-layer and incubated to polymerize the collagen (vii-viii). Growth medium containing desired compounds is then overlaid onto the polymerized spheroid-collagen layer and incubated to allow cell invasion (ix-x). (B) In the presence of a chemoattractant, cells are shown invading into the surrounding collagen matrix from an embedded PANC-1 spheroid at the indicated time points. Each panel shows a phase contrast image acquired using a 10X objective. Please click here to view a larger version of this figure.
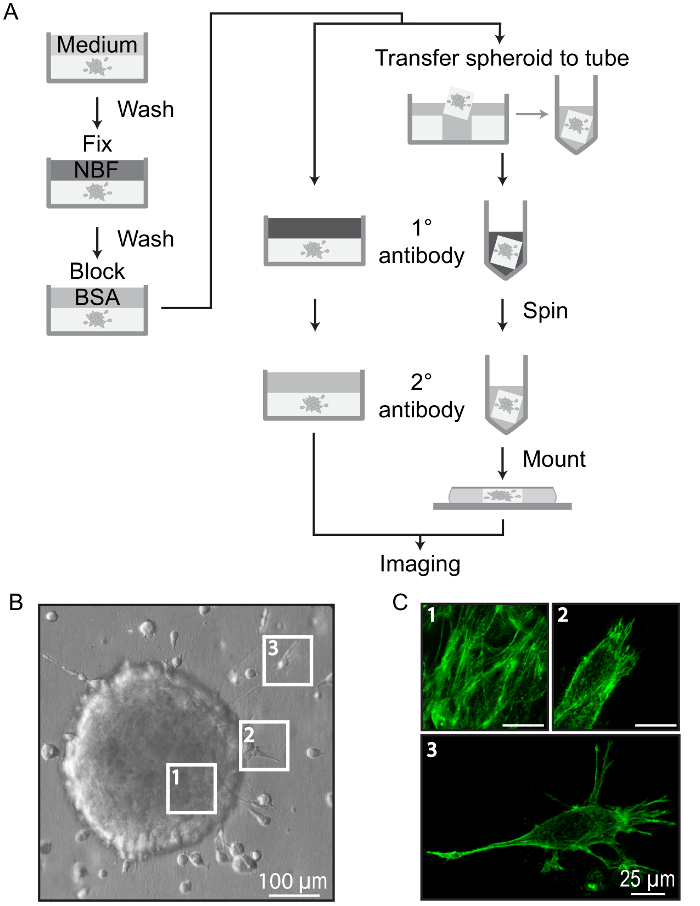
Figure 5: Immunofluorescence Staining of Collagen-embedded Spheroids. (A) Overview of protocol for immunofluorescence staining of spheroids. Embedded spheroids are washed, fixed (NBF), and blocked (BSA) directly in the imaging vessel. Spheroids can be stained directly, or excised and stained in a smaller volume, such as a 1.5 mL tube. Spheroids should be washed with excess buffer following each stain to minimize background fluorescence. Stained spheroids can be imaged directly in the vessel or mounted under a coverslip for imaging and storage. (B) Immunofluorescence images of a TPC-1 cell spheroid embedded in collagen and allowed to invade for 24 h. (C) Images showing phalloidin stained cells typical of regions indicated in B. Each panel shows a 20 µm Z-projection (0.2 µm steps) acquired using a 60X objective: Cells within the spheroid body approximately 20 µm from the surface (1), cells protruding into collagen (2), and cells invading through collagen (3). Scale bars = 25 µm. Please click here to view a larger version of this figure.
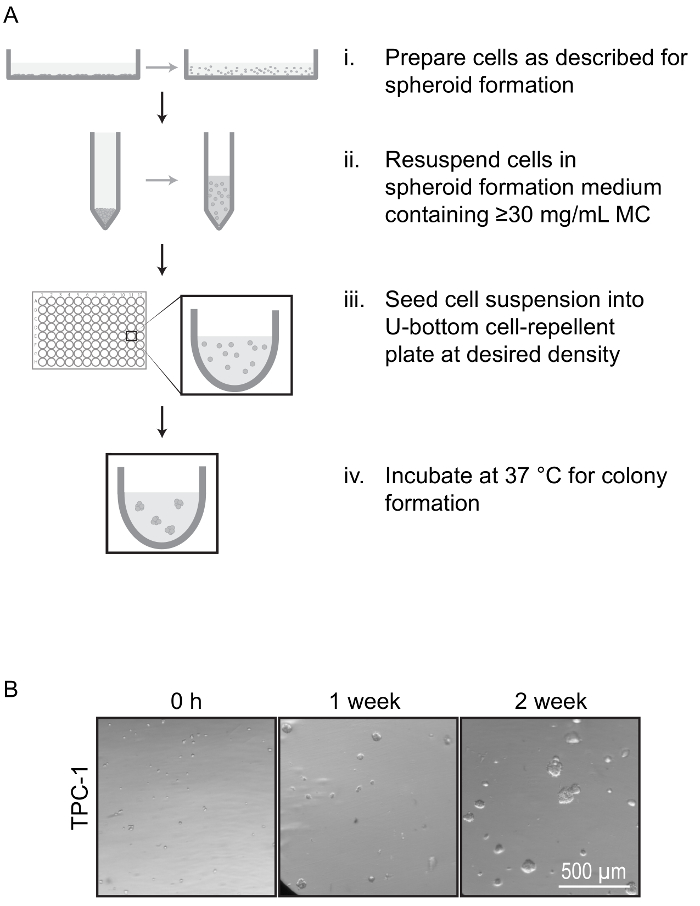
Figure 6: Spheroid Formation Anoikis Assay. (A) Overview of protocol for anoikis assay. Cells are prepared as described in Figure 1, but are resuspended in spheroid formation medium containing a minimum of 30 mg/mL MC which forms a thick layer to hold individual cells in suspension and prevent cell aggregation (i-ii). The cell suspension is seeded into U-bottom cell-repellent plates and incubated (iii-iv). The suspended cells can be monitored for spheroid formation by proliferation, indicating anoikis resistance. (B) Representative images of colony formation during anoikis assay. TPC-1 cells were held in suspension with 30 mg/mL MC and imaged at the indicated time points. Cells that are able to resist anoikis proliferate to form spheroids. Please click here to view a larger version of this figure.
| Cell Line | MC (mg/mL) | Serum (%) | Approximate incubation time |
| SH-SY5Y | 1 | 5 | 24 hours |
| BxPC-3 | 5 | 5 | 3 to 5 days |
| PANC-1 | 5 | 5 | 5 to 7 days |
| HCT-116 | 3 | 10 | 2 to 4 days |
| TT | 1 | 10 | 7 to 10 days |
| TPC-1 | 3 | 5 | 1 to 2 days |
Table 1: Optimal Spheroid Formation Medium Composition and Incubation Times for Validated Cell Lines.
Discussion
We present a rapid and flexible method for producing 3D cell spheroids to model the architecture of in vivo tissues using inexpensive and widely available reagents. Our protocol exploits the non-cytotoxic and adhesion-promoting properties of MC8,9 to mediate cell aggregation and minimize cell monolayer formation. Unlike protein-based matrices isolated from animal sources, MC is inert, contains no growth factors, and is easily removed by washing, allowing isolation of spheroids for use in a variety of applications without the presence of residual matrix. Our protocol uses rapid aggregation of consistent cell numbers to generate homogenously sized spheroids, allowing direct comparisons of spheroids grown under various experimental conditions. This method can be adapted for mixed cultures to model in vivo tumor growth by combining fixed numbers of multiple tumor-associated cell types (e.g. fibroblasts, leukocytes, stromal cells) to more accurately represent the tumor environment.
Modifications of our protocol using cells seeded in high concentrations of MC can be made to monitor anchorage-independent growth of cells able to proliferate in suspension, and may be employed as an anoikis assay (Section 6) to identify or quantify cell transformation. Spheroids generated by our protocol are ideal for applications such as drug screening, allowing comparison of cell growth, metabolism, survival, and invasiveness between treatment conditions. Isolated spheroids may also be embedded into an extracellular matrix, such as collagen, to assess and quantify cell invasiveness in a 3D-microenvironment (Section 2.2, Figure 4) that more accurately models in vivo tumor growth. Further, our protocol can be used to isolate large quantities of spheroids from the MC matrix for protein or RNA preparation, allowing users to evaluate cell signal or gene expression changes in response to treatment or growth conditions.
Our spheroid formation protocol requires optimization of serum and MC concentrations, and the number of cells seeded for each cell line. Cell line-specific characteristics, such as cell viability, cell-cell and cell-plastic adhesiveness, can significantly affect the ease with which spheroids are formed. In general, cell lines with poor viability in monolayer culture required higher serum concentrations to promote spheroid formation, while higher MC concentrations were preferred for highly adhesive cell lines. We therefore recommend that titrations of serum and MC be performed to determine the optimal conditions for cell survival (serum concentration), while minimizing monolayer and satellite spheroid formation (MC concentration). These conditions should result in generation of a single discrete spheroid per well without satellites. Optimal spheroid size for analysis is application-dependent and is affected by the number of cells seeded and serum concentration (Step 1.4.7). Seeding fewer cells produces smaller aggregates that are more easily homogeneously stained, but may not reproduce in vivo cell-cell and cell-microenvironment interactions. Increasing the number of cells seeded generates larger spheroids, which may contain a physiologically representative hypoxic core10 that can affect cell behavior and alter interpretations of growth or survival assays. However, when too large, spheroids may be fragile and are easily broken apart or damaged during isolation. Larger spheroids are also much harder to visualize without specialized imaging platforms for optical sectioning (e.g. light sheet fluorescent microscope) or by embedding or cryosectioning and staining of cross sections through the spheroid11. We recommend a target spheroid size of 200-500 µm for flexibility and ease of handling for most applications. Overall, it is critical to optimize the conditions for spheroid formation in both a cell line-specific and application-specific manner.
Spheroid production protocols require particular attention to avoid contamination of spheroids by dust and other particulates. Cells adhere to dust and other insoluble contaminants, resulting in irregularly shaped, loosely adherent cell aggregates unsuitable for use in analyses (Figure 3A). Sources of contamination can be minimized by passing all solutions through a 0.45 µm filter, rinsing the cell monolayer with filtered medium prior to dissociation (Step 1.4.1), and rinsing multichannel pipette reservoirs with filtered ultrapure water immediately before use (Step 1.4.6.1). Further, cotton-filtered serological pipettes, which can shed fibers into solutions, should be avoided. Although not critical, dust can be further reduced by passing dissociated cells through a 100 µm cell strainer prior to seeding (Step 1.4.6). Evaporative loss of medium is an additional technical consideration, particularly for incubation times exceeding 1 week, such as those required by certain cell lines for spheroid formation (Table 1) or during anoikis assays (Section 6). Evaporation can be minimized by seeding cells in wells in the center region of multi-well plates, filling perimeter wells with ultrapure water, and loosely sealing the edges of the plate or enclosing the plate in a humidified chamber.
3D cell spheroids are a valuable model for the study of both normal and tumor cell behavior and physiology. Our protocol is a rapid and economical method for generation of consistently sized 3D cell spheroids that can be used in an assortment of assays and applications. These spheroids represent valuable tools for characterization of tumor growth and microenvironment interactions as well as models for preclinical evaluation of novel therapies.
Declarações
The authors have nothing to disclose.
Acknowledgements
The authors thank M. Gordon of the Queen’s University Biomedical Imaging Centre for assistance. This work was supported by operating grants from the Cancer Research Society of Canada (19439) and the Canadian Institutes for Health Research (MOP-142303) (LMM), and by Ontario Graduate Scholarships and studentships from the Terry Fox Research Institute Training Program in Transdisciplinary Cancer Research (SMM, EYL), and by a Craig Jury Summer Studentship (SMM).
Materials
| Buffers | |||
| 10x Phosphate buffered saline | Thermo Fisher Scientific | AM9625 | |
| Calcium Chloride Solution | Sigma-Aldrich | 21114 | Used for PBS* wash buffer; Do not autoclave PBS* wash buffer upon addition of calcium chloride |
| Magnesium Chloride Solution | Sigma-Aldrich | M1028 | Used for PBS* wash buffer; Do not autoclave PBS* wash buffer upon addition of magnesium chloride |
| Name | Company | Catalog number | Comments |
| For Spheroid Formation | |||
| 96-well U-bottom Cell-Repellent Plate | Greiner Bio-One | 650970 | |
| Dulbecco's Modified Eagle's Medium | Sigma-Aldrich | D5546 | For culturing SH-SY5Y, PANC-1, TPC-1 cell lines |
| F12K Medium | Thermo Fisher Scientific | 2112722 | For culturing TT cell line |
| Fetal Bovine Serum | Sigma-Aldrich | F1051 | Filter prior to use to remove particulate contaminants |
| Methyl cellulose | Sigma-Aldrich | M7027 | Prepare in water to 100 mg/mL |
| Roswell Park Memorial Institute Medium | Sigma-Aldrich | R8758 | For culturing HCT-116, BxPC-3 cell lines |
| TrypLE Express | Thermo Fisher Scientific | 12605028 | Dissociation buffer |
| Name | Company | Catalog number | Comments |
| For Invasion Assay | |||
| Bovine Type I Collagen | Corning Incorporated | 354231 | Stock 3.1mg/ml; Maintain on ice when in use |
| DMEM Phenol Red Free Low Glucose | Thermo Fisher Scientific | 11054-20 | Less background fluorescence compared to Phenol Red supplemented medium |
| Glial Cell Line Derived Neurotrophic Factor | Peprotech | 450-10 | Chemoattractant |
| Name | Company | Catalog number | Comments |
| For Immunofluorescence Microscopy | |||
| #1.5 Coverglass | Electron Microscopy Sciences | 72225-01 | For mounting excised spheroids |
| Alexa-Fluor 488 Phalloidin | Thermo Fisher Scientific | A12379 | Used to stain actin at 1:200 |
| Bovine Serum Albumin | Bioshop Canada Incorporated | ALB001 | Used in BSA blocking buffer |
| Dabco 33-LV | Sigma-Aldrich | 290734 | Antifade |
| Glycerol | Bioshop Canada Incorporated | GLY001 | Used in MOWIOL mounting medium |
| ImageJ Software | Freeware, NIH | – | Used for image analysis |
| Microslides | VWR International | 48312-024 | For mounting excised spheroids |
| MOWIOL 4-88 | EMD-Millipore | 475904 | Used in MOWIOL mounting medium |
| Paraformaldehyde | EMD-Millipore | PX0055-3 | Used in fixation buffer |
| Triton X-100 | Bioshop Canada Incorporated | TRX777 | Used in permeabilization buffer |
Referências
- Zimmermann, M., Box, C., Eccles, S. A. Two-dimensional vs. three-dimensional in vitro tumor migration and invasion assays. Methods Mol Biol. 986 (986), 227-252 (2013).
- Albini, A., Noonan, D. M. The ‘chemoinvasion’ assay, 25 years and still going strong: the use of reconstituted basement membranes to study cell invasion and angiogenesis. Curr Opin Cell Biol. 22 (5), 677-689 (2010).
- Pampaloni, F., Reynaud, E. G., Stelzer, E. H. K. The third dimension bridges the gap between cell culture and live tissue. Nat. Rev. Mol. Cell Biol. 8 (10), 839-845 (2007).
- Fennema, E., Rivron, N., Rouwkema, J., van Blitterswijk, C., de Boer, J. Spheroid culture as a tool for creating 3D complex tissues. Trends Biotechnol. 31 (2), 108-115 (2013).
- Carletti, E., Motta, A., Migliaresi, C. Scaffolds for tissue engineering and 3D cell culture. Methods Mol Biol. 695, 17-39 (2011).
- Foty, R. A simple hanging drop cell culture protocol for generation of 3D spheroids. J Vis Exp. (51), (2011).
- Handschel, J. G., et al. Prospects of micromass culture technology in tissue engineering. Head Face Med. 3, 4 (2007).
- Stuckhoff, A. P., DelValle, L., Amini, S., White, M. K. . Neuronal Cell Culture Methods and Protocols Vol. 1078 Methods in Molecular Biology. 1078, 119-132 (2013).
- Stewart, G. J., Wang, Y., Niewiarowski, S. Methylcellulose protects the ability of anchorage-dependent cells to adhere following isolation and holding in suspension. Biotechniques. 19 (4), 598-604 (1995).
- Vinci, M., et al. Advances in establishment and analysis of three-dimensional tumor spheroid-based functional assays for target validation and drug evaluation. BMC Biol. 10, 29 (2012).
- Nagelkerke, A., Bussink, J., Sweep, F. C. G. J., Span, P. N. Generation of multicellular tumor spheroids of breast cancer cells: How to go three-dimensional. Anal. Biochem. 437 (1), 17-19 (2013).

