Severe Burn Injury in a Swine Model for Clinical Dressing Assessment
Summary
To closely mimic the mode of burn injuries requires the interplay between clinical observation and studies in animal models. In this study, a swine model of severe burn injury was established to assess an experimental dressing in physiological and pathophysiological settings.
Abstract
Wound healing is a dynamic repair process and is the most complex biological process in human life. In response to burn injury, alterations in biological pathways impair the inflammation response, resulting in delayed wound healing. Impaired wound healing frequently occurs in patients with diabetes leading to unfavorable outcomes such as amputation. Hence, dressings having beneficial effect in promoting burn wound repair are needed. However, studies on burn wound treatment are limited due to lack of proper animal models. Our previous study demonstrated wound-healing performance in rat and swine models using a minimally invasive surgical technique. This study aimed to demonstrate a swine model of severe burn injury that eliminates wound contraction and more closely approximates the human processes of re-epithelialization and new tissue formation. This protocol provides a detailed procedure for creating consistent burn wounds and examining the wound-healing performance under the treatment of an experimental dressing in a swine model. Six burn wounds were created symmetrically on the dorsum, which were covered with a clinical dressing composed of four layers: an inner contact layer of experimental materials, an inner intermediate layer of waterproof film, an outer intermediate layer of gauze, and an outer layer of adhesive plaster. Upon the completion of experiments, wound closure, wound area, and Vancouver Scar Scale score were examined. The samples of skin resected from each animal post-sacrifice were histologically prepared and stained using hematoxylin and eosin staining. Antibacterial activity of each dressing in the context of wound healing was also examined. The application of the clinical dressing to the wounds in swine model mimics the biological processes of human wound healing with respect to the processes of epithelialization, cellular proliferation, and angiogenesis. Therefore, this swine model provides an easy-to-learn, cost-effective, and robust method to assess the effect of clinical dressings in severe burn injury.
Introduction
A burn injury initiates the inflammatory process and induces complex pathological effects, which influence numerous body functions immediately after an accident, resulting in negative impact on patients' quality of life. Impaired wound healing causes significant morbidity and mortality among patients with diabetes mellitus1,2. Most patients with burn injuries experience pain during burn wound debridement, which is known as an excruciating process despite the use of powerful opioid analgesics3.
In contrast to other mammals, swine share several anatomic and physiologic characteristics with humans regarding the process of epithelialization, cellular proliferation, and angiogenesis. This makes swine a potentially better model for certain procedures and studies, and they are often used in subsequent studies that demonstrated promising results in mice. These features have led to the increasing use of swine as a major species in preclinical testing. Recently, a rapid increase in biomedical research with respect to cardiovascular, urinary, integumentary, and digestive systems has been observed4,5,6. This study aims to demonstrate a swine model of severe burn injury that eliminates wound contraction and provides a closer approximation of the human wound healing processes and the formation of new tissue. Six burn wounds were created symmetrically on the dorsum, three on each side of the spine of the swine. Next,anexperimental dressing was examined in a swine model of severe burn injury, which can be adapted to replicate human wound healing (Figure 1). The wounds were covered with a clinical dressing, which is composed of four layers: an inner contact layer of experimental materials, an inner intermediate layer of waterproof film, an outer intermediate layer of gauze, and an outer layer of adhesive plaster. A waterproof film keeps the wound environment moist while preventing bacterial infection and allowing gases to permeate the dressing. The outer intermediate layer of gauze was applied on the waterproof films and secured by an outer layer of adhesive plaster. At the completion of experiments, wound closure, wound area, and Vancouver Scar Scale (VSS) score were examined. The samples of skin resected from each animal post-sacrifice were histologically prepared and stained using hematoxylin and eosin (HE) staining. Antibacterial activity of each dressing in the context of wound healing was also examined in this model. Our previous study has demonstrated wound-healing performance in rat and swine model using a minimally invasive surgical technique7. Since there were six burn wounds on the dorsum within each swine, each experimental dressing was tested and evaluated in all positions to minimize bias related to wound healing process in different spots on the swine dorsum. Therefore, the swine model of severe burn injury established in this study provides a new approach for the evaluation of clinical dressings and facilitates the development of a novel treatment for burn injury. This study provides crucial tools to uncover the pathophysiology of burn wound healing.
Protocol
Procedures involving animal subjects have been approved by the Animal Care Committee at National Defense Medical Center, Taiwan (R.O.C). This study was conducted in the Laboratory Animal Center at the National Defense Medical Center. Swine weighing between 20 and 25 kg has been successfully instrumented using this protocol.
1. Adaptation of the Animals to Human Handling
- After arrival in the facility, house the animals solitarily but let them interact with each other.
- Provide the animals ad libitum access to food and water.
- Acclimate swine to human handling and transportation from the animal facility to the experimental laboratory by handling the animal at least once a day for one week.
- Fast the animal for at least 12 hours before surgery to prevent nausea, vomiting, and aspiration of stomach fluids.
2. Sedation
- Before the burn wound creation, sedate animals via an intravenous injection with Zoletil 50 (25 mg/kg).
3. Intubation and Ventilation
- Place the animal on a table and/or trolley in sternal position.
- Open the mouth of the animal with an oral spreader.
- In case of insufficient relaxation of the jaws or presence of swallowing reflexes, which hinder intubation, mask the swine with isoflurane to induce sedation.
- Monitor blood pressure, heart rate, and body temperature by physiological signal monitor during the surgery to prevent potential complications.
4. Anesthesia
- Induce and maintain anesthesia; preferably anesthetize the animal via an intramuscular injection of tiletamine and zolazepam (25 mg/kg + 25 mg/kg).
- Intubate the animal with the endotracheal tube when muscle relaxation, characterized by loss of jaw tone and palpebral reflexed, was observed.
- Maintain all pigs in an anesthetic state at a vaporizer setting of 0.5–2.5% (v/v) isoflurane until the end of the surgery.
- Examine the depth of anesthesia by testing pain reflexes with a hind leg toe pinch before surgery. When necessary, add additional anesthesia or wait for a few minutes. Check pain reflexes regularly throughout the surgery.
5. Sterilization of the Surgical Site
- Shave and clean the skin of the animal over an area of approximately 25 cm width from the vertebral column all the way to the axilla on both sides.
- Scrub the moisturized skin with povidone-iodine scrub (75 mg/mL) for approximately 5 min.
- Remove the povidone-iodine soap from the skin using wet sterile gauzes.
- Sterilize the skin with povidone-iodine lotion (100 mg/mL).
- Cover the animal with sterile surgical drapes to reduce bacterial transfer and subsequent contamination of the surgical site.
6. Burn Wound Creation
- Use a surgical marking pen to mark the center of six burn wounds symmetrically on the dorsum of the pig. Ensure that the distance between each burn wound is at least greater than the radius of the wound (Figure 1A).
- Fill a modified soldering iron with 50 mL of glycerin and insert an electronic thermometer into it to monitor the temperature. The hot iron possesses a flat area of approximately 9 cm2 (Figure 1B).
- Heat up the iron to 137–139 °C with a hot plate (Figure 1C).
- Create six uniform burn wounds by placing the iron on the marked area without applying any force for 30 seconds (Figure 1C).
- Wash the burn wounds with 0.9% saline solution (Figure 1D).
- Measure wound dimensions and record the wound by photomicrography (Figure 1E).
7. Preparation of Dressings
- Cover each wound with the inner contact layer of a four-layer clinical dressing through direct contact. For this layer, use CAPS-containing dressing or alternative materials (Figure 1F).
- Apply a waterproof film onto the clinical dressing to serve as a barrier against bacterial penetration (Figure 1F).
- Cover each wound with a gauze (0.5 cm thick) and fix with paper tape to serve as the mid-layer of the dressing (Figure 1G).
- Secure the gauze with an outer layer of adhesive plaster. Extend this layer to the torso to avoid the displacement of the dressing (Figure 1H-1J).
8. Post-burn Care and Measurement
- Inject the swine with buprenorphine (0.1 mg/kg, IM) for pain management for every 8-12 h, starting before recovery from anesthesia, for one week to reduce potential pain.
- Allow the pig free access to feed and water.
- Change the clinical dressings every 2 days for the first 10 days and then twice a week for the 6-week study.
- Clean and measure wounds before reapplying clinical dressings. Administer anesthetics during dressing changes.
- Record the wound by photomicrography for comparison of wound healing rate every 2 days for the first 10 days and then twice a week for the 6-week study.
- Calculate the wound re-epithelialization or contraction as the percentage of the original wound size according to a previously described method. The analysis of wound closure was conducted in a double-blinded manner.
- Measure the burn scar using VSS, which consists of four variables: vascularity, height (thickness), pliability, and pigmentation on post-burn days 0, 7, 21, and 42. Each variable has four to six possible scores. The total score ranges from 0 to 14, whereby a score of 0 reflects normal skin.
9. Bacterial Growth Experiments of Post-burn Tissues
- Swab the wound for antibacterial testing on post-burn days 0, 7, 21, and 42.
- Place the swab into 100 mL of 0.9% sterile saline solution and gently vortex to achieve a homogenous suspension.
- Serially dilute (10−1–10−5) the homogenate and plate 100 μL of each dilution in selective and nonselective media, respectively.
- Incubate all dilutions under aerobic conditions at 37 °C for 24–72 hours.
- Plate triplicate aliquots of 10 μL each from all dilutions onto blood agar plate supplemented with 5% sheep blood to isolate aerobic Gram-positive organisms.
- Incubate the sample by spreading or pouring the sample uniformly on the surface of an agar plate overnight for determining the number of colony-forming units (CFUs).
- Read the plates after overnight incubation. Invert the sheep blood agar plate and divide the bottom of the dish into four equal quadrants using a marker and small ruler.
- Place the plate onto the stage of a dissection microscope and count the colonies on each plate. By definition, a colony must have a minimum of 300 CFU to be enumerated.
- Count the bacterial colonies in each of the three replicates. Calculate the average value of the three replicates. Determine the CFU per plate by multiplying the average value by the final dilution factor.
10. Euthanasia and Tissue Fixation
- Intravenously inject an overdose of sodium pentobarbital euthanasia solution (80–120 mg/kg).
- Perform en bloc excision of burn wound tissue to include the underlying musculature and surrounding unwounded tissue.
- Fix tissues with 10% neutral buffered formalin.
- Mix 10 mL of formaldehyde (37%) in 90 mL of phosphate buffered solution (PBS) and store in 4 °C.
- Transfer tissues to fixative and swirl the container to ensure all tissues are completely immersed in fixative. The volume of fixative must be 30 times the tissue volume.
- Fix tissues overnight at 4 ˚C.
- Dehydrate tissues with ethanol and embed into paraffin blocks. Perform the following steps at 4 ˚C on a shaker.
- Wash twice with PBS for 30 minutes.
- Dehydrate tissues with 70% ethanol for 8 hours, 80% ethanol overnight, 95% ethanol for 8 hours, and then in 100% ethanol overnight.
- Incubate tissues in 100% ethanol for a further 8 hours.
- Incubate tissues in three changes of xylene each for 30 minutes.
- Replace the xylene with freshly melted (52 °C) wax, and incubate at 52 °C in an oven for 1 hour.
- Replace the wax with fresh wax and incubate at 52 °C in an oven for 3 hours, and then replace once more and incubate at 52 °C overnight.
- Incubate tissues with two more changes of wax each for 1 hour, and then embed the tissue and store at 4 °C.
- Cut and stain the paraffin-embedded sections with HE, and visualize via a light microscope with 100× magnification.
- Create paraffin sections using a rotary microtome.
- Dewax sections with three changes of xylene each for 3 minutes.
- Rehydrate tissues with 100%, 95%, 80%, and 70% ethanol each for 3 minutes, and then immerse in distilled water.
- Stain with hematoxylin for 10 minutes, and then rinse in running tap water.
- Differentiate with 0.1% hydrochloric acid ethanol for 5 minutes, and rinse in tap water.
- Stain with 0.5% eosin for 1 minute.
- Dehydrate tissues with 70%, 80%, 95%, and 100% ethanol each for 2 minutes.
- Clear the staining with xylene, and dry in fume hood.
Representative Results
Burn duration of 30 seconds by hot iron resulted in wounds that were circular with a well-defined margin and uniformly pale with a rim of erythema (Figure 1D). Within each animal, there were six burn wounds on the dorsum. The arrangement of burn wounds was depicted in Figure 1K. Burn wounds were completely covered with CAPS-containing dressing and used to evaluate the depth of scar formation on post-burn days 0, 7, 21, and 42 and re-epithelialization, as determined by gross inspection. Burn wounds were re-epithelialized based on gross inspection on post-burn day 42. Wound size in these animals was evaluated to determine the wound healing rate (Figure 2A). Wound areas were 9, 10, 8, and 3 cm2 on post-burn days 0, 7, 21, and 42, respectively. A significant reduction in wound area was observed on post-burn day 42 compared with day 0. The healing rate was defined as the greatest average wound margin distance from the wound center divided by the time to complete wound closure. Wounds treated with CAPS-containing dressing showed a 73.43±6.33% wound closure on post-burn day 42 (Figure 2B).
Figure 3 shows the VSS scores with respect to scar vascularity, pliability, pigmentation, and height on post-burn days 0, 7, 21, and 42. The VSS score peaked at 7.4±0.5 on post-burn day 21 and decreased to 3.33±0.58 on post-burn day 42.
All animals were sacrificed on post-burn day 42. Samples of skin resected from each animal post-sacrifice were histologically prepared and stained by H&E. Histologic examination of the samples confirmed that full-thickness burns were achieved, and the wounds appeared fully healed (Figure 4). Necrosis resulting from burns could be observed in the epidermis, dermis, and dermal components of the wound without significantly affecting the underlying muscle (Figure 4). The dermis thickness beneath the experimental dressing was 5.4 mm (Figure 4A).In addition, the sloughing of the dermis and lymphocytic infiltration are observed in the H&E staining, as indicated by the red arrow in Figure 4B.
The antibacterial property of the experimental dressing was determined using a CFU assay on post-burn days 0, 7, 21, and 42. The result showed a slight increase in bacterial cell numbers observed between post-burn days 0 and 21 followed by a significant increase on post-burn day 42 (Figure 5). This result suggests that the swine model of severe burn injury established in this study could be used to monitor the clinical performance of experimental dressings, including antibacterial property.
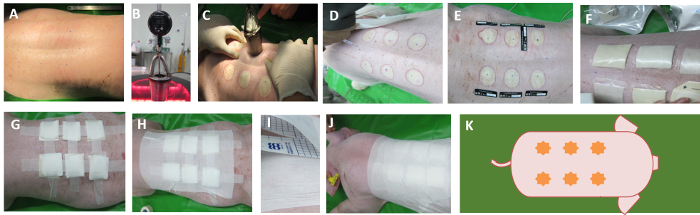
Figure 1. Burn wound creation and application of a CAPS-containing clinical dressing. Following hair removal and preparation of the skin with iodine and alcohol, (A) surgical marking pen was used to outline six circles on the dorsum, either side of the midline. (B) The modified iron was filled with glycerin and an electronic thermometer inserted into glycerin to show the temperature. (C) The iron is heated to 137-139 °C with a hot plate, and six uniform burn wounds were created on the marks of the skin. To create burn wounds, place the hot iron with no external force on the swine's back skin (30 seconds) to create two full-thickness wounds. (D) After all the wounds were created, the wounds were washed with 0.9% saline solution. (E) The scales were placed next to the wounds for taking pictures. (F) The first layer is covered with a test dressing and the second layer with a waterproof film. (G) The third layer is covered with the gauze of about 0.5 cm thickness and fixed with paper tape. (H-J) Secure the gauze with an outer layer of adhesive plaster. This layer extends to the torso to avoid the displacement of the dressing. (K) Schematic diagram of the burn wound distribution. Please click here to view a larger version of this figure.
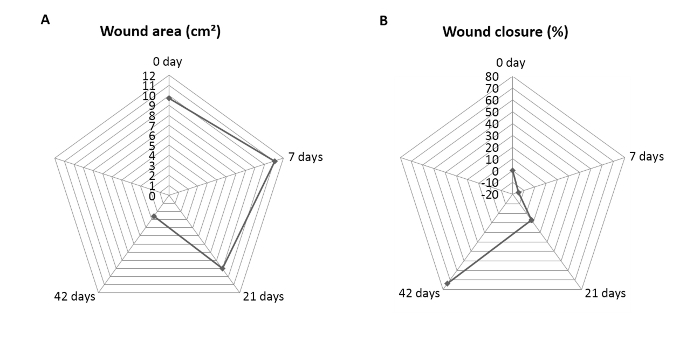
Figure 2. Change of wound size in a swine model. (A) The rate of wound closure was determined as a percentage of the original wound on day 0. Wounds had almost completely contracted on post-burn 42 day, and (B) changes of the wound area were observed between 0 to 42 days of a post-burn animal model. It shows the greatest decrease on post-burn day 42, exhibiting 90 ± 4% wound area closure. Please click here to view a larger version of this figure.
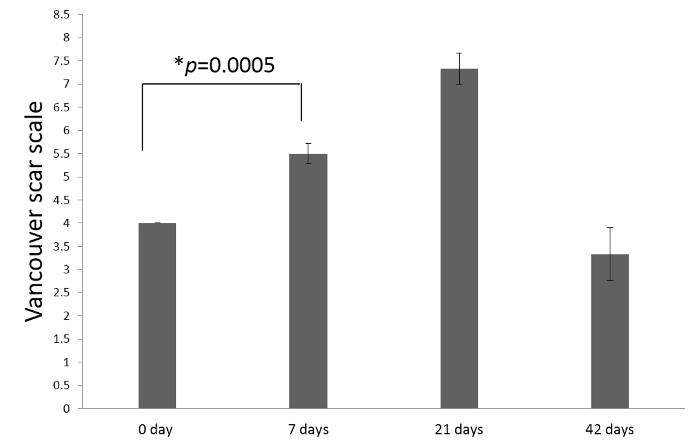
Figure 3. The average total scores of Vancouver Scar Scale (VSS) using a double-blind experimental design on post-burn days in a swine model. Scar assessment includes pigmentation, vascularity, pliability, and height of scar. A lower score indicates the scar on the condition that closely approximates normal skin (P = 0.0005). Please click here to view a larger version of this figure.
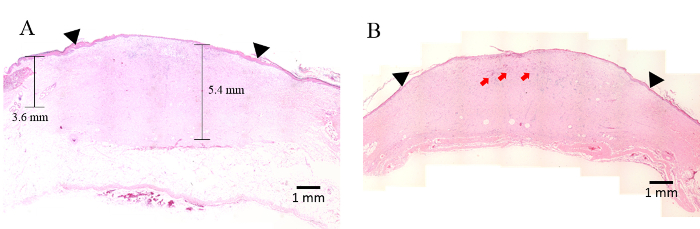
Figure 4. Hematoxylin and eosin staining on post-burn day 42. (A, B) The black inverted triangle indicates the repaired burn tissue. The morphology of the full-thickness wound was smooth and continuous, and the papillary layer resembled features of hypertrophic scarring. Note the presence of neo-epidermis covering the wound surface. (B) The red arrow indicates the dermal connective tissue-infiltrated inflammatory cells in the burn wound eschar above the viable dermis below. Original magnification, ×10. Please click here to view a larger version of this figure.
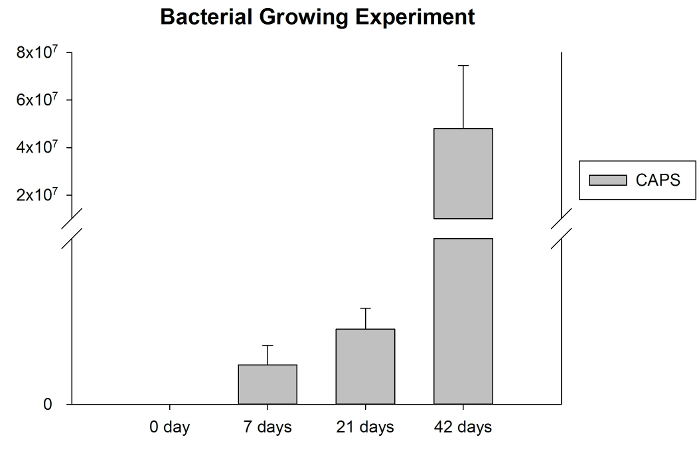
Figure 5. A bar diagram showing bacterial counting of wounds at different time points. Antibacterial activity of post-burn wound animal model at 0, 7, 21, and 42 days. Antibacterial activity was evaluated by colony-forming units (CFU) assay in three independent experiments. Please click here to view a larger version of this figure.
Discussion
The present study established a swine model of severe burn injury and examined the model using a CAPS-containing dressing. Our results suggest that this swine model can be used for monitoring the clinical performance of experimental dressings, including antibacterial property. Wound healing rate, wound closure, and antibacterial activity were also analyzed using VSS, H&E staining, and antibacterial test. The use of animal burn models has been developed as a valuable tool to review the pathophysiology of burn injury. There are certain biological benefits of using rats as experimental subjects, including the high number of rat-specific reagents and the practical aspects of obtaining transgenic rats, to discovering the molecular signaling pathways active in the recovery process. However, the major disadvantage of using rats in an animal burn model is its failure to completely approximate the human wound healing process. Re-epithelialization is the main healing approach observed in humans8, while wound healing in rats strongly depends on wound contraction in a rapid manner9. In addition, the non-vulnerability of rats to hypertrophic or keloid scar formation confirmed the dissimilarity of their wound recovery process from that of humans. On the other hand, swine have recently become the subject of researcher attention because their skin architecture is similar to that of human skin. Additionally, the wound healing process in swine and humans occurs through physiologically similar phases such as inflammation, proliferation, re-epithelialization, and remodeling. Generally, burns in swine heal by 21 days, with re-epithelialization occurring between 7 and 14 days post-wound infliction10,11, which fit to the timelines observed in humans. Swine also show greater morbidity compare to the rats due to their body size that makes more resistant to wound infection. Mice are usually the first model used to evaluate burn wound conditions under different treatments. Additionally, in contrast to other mammals, swine share a number of anatomic and physiologic characteristics with humans regarding the process of epithelialization, cellular proliferation, and angiogenesis. This makes swine potentially a better model, and they are often used in subsequent studies that demonstrated promising results in mice 12,13,14,15. Moreover, the distribution of dermal collagen and elastic content, cutaneous blood supply, the sequence of events in wound healing, and re-epithelialization have made the swine a standard model of wound healing and reconstructive surgical treatments15,16,17,18.Within each animal, there were six burn wounds located on the upper, middle, and lower back of the swine and distributed symmetrically to the spine. Burn wounds can be created on the similar locations but on different swine. Therefore, each experimental dressing can be tested and evaluated in different locations to minimize bias related to wound healing process in different spots on the swine dorsum. In addition, this swine model of severe burn injury established in this study also benefits from ensuring that each swine serves as its own control with one wound receiving novel treatment and the other as vehicle control, leading to a significant reduction in the number of animals.
The management of burn injuries is one of the major complications in wound healing. The human healing process is composed of four biological processes: hemostasis, inflammation, proliferation, and maturation. Once the healing process is disturbed or retarded, it promotes a fibroblastic proliferation that results in a hypertrophic scar, which is visible and tends to be raised above the surrounding skin16. To ensure reproducibility and accurate quantification, adequate adhesion using adhesive plaster is required immediately after the injury to minimize the delay in time. Cutaneous irritation is a common reaction from adhesive plaster that results in a rash around the clinical dressing. Thus, caution is recommended in the use of currently available adhesive plaster to prevent itchiness. Following aseptic techniques and thoroughly disinfecting all equipment (e.g., caliper) to avoid cross-contamination between swines are imperative.
Burn wound healing is an intricate process. A certain number of factors have been identified to have a negative influence on the wound healing process such as pain, itching, and bacterial contamination. Pain has a negative impact on animal's diet. Without sufficient nutrient uptake, delayed wound healing is inevitable. Therefore, each swine was injected with buprenorphine (0.1 mg/kg, IM) daily for 7 consecutive days to reduce pain. Food intake was monitored one week after the burn wound creation. Once a decrease in food intake was observed on day 7, buprenorphine (0.1 mg/kg, IM) administration was continued until day 14. Bacterial infection, which is a potential risk for wound healing, may trigger inflammation and further dampen the healing process. Therefore, clinical dressings were covered with waterproof films to prevent and minimize potential contamination. Itching is an uncomfortable part of the wound healing process. Swine may scratch their own back on the ground to reduce itch, which can be unfavorable for wound healing process. A good dressing possesses the properties of accelerating burn wound healing with an acceptable scar, preventing infection and inflammation, alleviating pain, and permitting easy and early mobilization19,20. The dressing can be removed when required for further topical applications and/or measurement of the wound area21,22. Although applying a four-layer clinical dressing to a swine is a more complicated and time-consuming process than clinical, swine model allows closer approximation to a human biological response than mouse model. The many potential advantages of using swine model make it a better platform for preclinical testing than other species.
The clinical efficacy in terms of wound closure efficiency and wound area reduction was monitored using a CAPS-containing dressing that revealed a 73.43±6.33% wound closure on post-burn day 42. Moreover, the VSS score, a validated subjective scale score, was also used to assess wounds in swine in this study17.It is important to monitor hypertrophic scar formation during treatment with experimental dressings, particularly in clinical trials18. Application of CAPS in the treatment of burn wounds resulted in a significant change in VSS scores on post-burn day 42 compared with the scores on day 21. A lower score indicates the injured area attempts to restore to a condition that closely approximate normal skin color pigmentation, vascularity, pliability, and height. In addition, the results of H&E staining indicated an intact stratum corneum layer in the CAPS-treated area. This result suggests that the swine model of a severe burn injury can be used reliably to evaluate the wound area, wound closure, VSS score, and healing progress.
This protocol represents the numerous detailed steps for demonstrating the use of an experimental dressing on a swine model of severe burn injury and can be applied to the evaluation of any types of clinical dressing. A modified soldering iron filled with 50 mL of glycerin is capable of maintaining temperature within 137 to 139°C during the wound creation process. However, this method has limitations such as inability to absolutely mimic the real burn temperature and the narrowness of the burn created area due to the soldering iron scope. However, the main advantages of the method overcome those limitations. This modified iron can be used to create consistent wounds with accuracy and ease compared to the traditional methods. Moreover, this model provides a sensitive measure of the physiological and pathophysiological response of a thermal burn injury under the treatment of various clinical dressings. The advantage of the proposed model, besides the economic advantages, is that it is easy to be handled by those with relatively little surgical experience. The application of the clinical dressing to the wounds in the swine model mimics the biological processes of human wound healing. Therefore, the swine model of severe burn injury established and tested with clinical dressing in this study facilitates the development of novel treatment for burn injury. This study provides crucial tools to uncover the pathophysiology of burn wound healing. In conclusion, this swine model provides an easy-to-learn, cost-effective, and robust method to assess the effect of clinical dressings in severe burn injury.
Declarações
The authors have nothing to disclose.
Acknowledgements
This study was supported by a grant from the Tri-Service General Hospital; National Defense Medical Center, Taiwan (TSGH-C107-042); Ministry of Science and Technology, Taiwan (MOST 106-2314-B-016 -014); and the National Defense Medical Center (MAB-106-055; MAB-106-010; MAB-107-064).
Materials
| Sedation: | ||||
| Ketamine | Merial | 2528 ESP | 10 mL vial | |
| Azaperone | China chemical & pharmaceutical | 47W406 | 100 mL vial | |
| Atropine | Oriental chemical works | IN120802 | 1 mL vial | |
| Anesthetic: | ||||
| Tiletamine+Zolazepam | Virbac | BC91 | 5 mL vial | |
| Isoflurane | Baxter | N002A225 | 100 mL vial | |
| Surgery: | ||||
| Hair clippers | Moser | – | – | |
| Povidone iodine scrub solution | Ever star | HA161202 | 4 L barrel | |
| Modified iron | – | – | – | |
| Electronic thermometer | Dogger | A9SA-ST9215C | – 50~300℃ | |
| 0.9% saline solution | CHI SHENG | KC130 | 500 mL vial | |
| Gauze | China Surgical Dressings Center | MO15900080 | 10 x10 cm | |
| CAPS | CoreLeader Biotech Co., Ltd, Taipei, Taiwan | – | – | |
| Paper tape | 3M | NDC-8333-1530-01 | 2.5 cm x 9.1m | |
| Waterproof film | 3M | NDC-8333-1600-40 | 10 cm x 10 m | |
| Adhesive plaster | Young chemical | BH1426015 | 10 cm x 10 m | |
| Dissection: | ||||
| Pair of standard sharp/blunt straight scissors (14 cm) | Shinetec instruments | ST-S114 | – | |
| Halstead-Mosquito (12.5 cm) | Shinetec instruments | ST-H012 | – | |
| Handle(# 4) | Shinetec instruments | ST-H004 | – | |
| Surgical Blade(#21) | Shinetec instruments | ST-B021 | – | |
| Post-Fixation & Storage: | ||||
| 50 ml Plastic centrifuge tube | Falcon | 352070 | – | |
| 10% neutral buffered formalin | Leica | 3800604EG | – | |
| Bacterial Growth Experiments | ||||
| Blood agar plate (BAP) (TSA with 5% sheep blood) | CMP | – | 90 mm Mono |
Referências
- Sen, C. K., et al. Human skin wounds: a major and snowballing threat to public health and the economy. Wound Repair Regeneration. 17, 763-771 (2009).
- Sen, C. K. Wound healing essentials: let there be oxygen. Wound Repair Regeneration. 17, 1-18 (2009).
- Verhaegen, P. D., et al. Differences in collagen architecture between keloid, hypertrophic scar, normotrophic scar, and normal skin: an objective histopathological analysis. Wound Repair Regeneration. 17, 649-656 (2009).
- Swindle, M. M., Adams, R. J. . Experimental Surgery and Physiology: Induced Animal Models of Human Disease. , (1988).
- Swindle, M. M., Smith, A. C. . Swine as Models in Biomedical Research. , (1992).
- Tumbleson, M. E., Schook, L. B. . Advances in Swine in Biomedical Research. Vols 1-2. , (1996).
- Wang, C. H., et al. Enhanced wound-healing performance of a phyto-polysaccharide-enriched dressing – a preclinical small and large animal study. International Wound Journal. 14, 1359-1369 (2017).
- Rowan, M. P., Cancio, L. C., Elster, E. A., Burmeister, D. M., Rose, L. F., Natesan, S., Chan, R. K., Christy, R. J., Chung, K. K. Burn wound healing and treatment: review and advancements. Journal of Critical Care. 19, 243 (2015).
- Wong, V. W., Sorkin, M., Glotzbach, J. P., Longaker, M. T., Gurtner, G. C. Surgical approaches to create murine models of human wound healing. Journal of Biomedicine and Biotechnology. 969618, 8 (2011).
- Gaines, C., Poranki, D., Du, W., Clark, R. A., Van Dyke, M. Development of a porcine deep partial thickness burn model. Burns. 39, 311-319 (2013).
- Gnyawali, S. C., Barki, K. G., Mathew-Steiner, S. S., Dixith, S., Vanzant, D., Kim, J., Dickerson, J. L., Datta, S., Powell, H., Roy, S., Bergdall, V., Sen, C. K. High-resolution harmonics ultrasound imaging for non-invasive characterization of wound healing in a pre-clinical swine model. PLoS One. 10 (3), e0122327 (2015).
- Eaglstein, W. H., Mertz, P. M. New methods for assessing epidermal wound healing: the effects of triamcinolone acetonide and polyethelene film occlusion. Journal of Investigative Dermatology. 71, 332-384 (1978).
- Swindle, M. M., Smith, A. C. Comparative anatomy and physiology of the pig. Scandinavian Journal of Laboratory Animal Sciences. 25, 1-10 (1998).
- Swindle, M. M. . Swine in the Laboratory: Surgery, Anesthesia, Imaging and Experimental Techniques. , (2007).
- Mertz, P. M., et al. IL-1 as a potent inducer of wound re-epithelization. Progress in Clinical and Biological Research. 365, 473-480 (1991).
- Eming, S. A., Martin, P., Tomic-Canic, M. Wound repair and regeneration: mechanisms, signaling, and translation. Science Translation Medicine. 6, 265-266 (2014).
- Aryza, M. J., Baryza, G. A. The Vancouver Scar Scale: an administration tool and its interrater reliability. The Journal of Burn & Rehabilitation. 16, 535-538 (1995).
- Bystrom, A., Claesson, R., Sundqvist, G. The antibacterial effect of camphorated paramonochlorophenol, camphorated phenol and calcium hydroxide in the treatment of infected root canals. Endodontics & Dental Traumatology. 1, 170-175 (1985).
- Reig, A., Tejerina, C., Codina, J., Hidalgo, J., Mirabet, V. Application of a new cicatrization dressing in treating second-degree burns and donor sites. Annals of Burn and Fire Disasters. 4, 174 (1991).
- Hindy, A. Comparative study between sodium carboxymethyl-cellulose silver, moist exposed burn ointment, and saline-soaked dressing for treatment of facial burns. Annals of Burn and Fire Disasters. 22, 131-137 (2009).
- Galiano, R. D., et al. Topical vascular endothelial growth factor accelerates diabetic wound healing through increased angiogenesis and by mobilizing and recruiting bone marrow-derived cells. The American Journal of Pathology. 164, 1935-1947 (2004).
- Thangarajah, H., et al. The molecular basis for impaired hypoxia-induced VEGF expression in diabetic tissues. Proceedings of the National Academy of Sciences of the United States of America. , 13505-13510 (2009).

