Isolation of Myoepithelial Cells from Adult Murine Lacrimal and Submandibular Glands
Summary
The lacrimal gland (LG) has two cell types expressing α-smooth muscle actin (αSMA): myoepithelial cells (MECs) and pericytes. MECs are of ectodermal origin, found in many glandular tissues, while pericytes are vascular smooth muscle cells of endodermal origin. This protocol isolates MECs and pericytes from murine LGs.
Abstract
The lacrimal gland (LG) is an exocrine tubuloacinar gland that secretes an aqueous layer of tear film. The LG epithelial tree is comprised of acinar, ductal epithelial, and myoepithelial cells (MECs). MECs express alpha smooth muscle actin (αSMA) and have a contractile function. They are found in multiple glandular organs and are of ectodermal origin. In addition, the LG contains SMA+ vascular smooth muscle cells of endodermal origin called pericytes: contractile cells that envelop the surface of vascular tubes. A new protocol allows us to isolate both MECs and pericytes from adult murine LGs and submandibular glands (SMGs). The protocol is based on the genetic labeling of MECs and pericytes using the SMACreErt2/+:Rosa26-TdTomatofl/fl mouse strain, followed by preparation of the LG single-cell suspension for fluorescence activated cell sorting (FACS). The protocol allows for the separation of these two cell populations of different origins based on the expression of the epithelial cell adhesion molecule (EpCAM) by MECs, whereas pericytes do not express EpCAM. Isolated cells could be used for cell cultivation or gene expression analysis.
Introduction
Myoepithelial cells (MECs) are present in many exocrine glands including lacrimal, salivary, harderian, sweat, prostate, and mammary. MECs are a unique cell type that combines an epithelial and a smooth muscle phenotype. MECs express α-smooth muscle actin (SMA) and have a contractile function1,2. In addition to MECs, the lacrimal gland (LG) and the submandibular gland (SMG) contains SMA+ vascular cells called pericytes, which are cells of endodermal origin that envelop the surface of vascular tubes3. Although MECs and pericytes express many markers, SMA is the only marker that is not expressed in other LG and SMG cells1,3.
Within the last 40 years, several laboratories reported assays for dissociation of different exocrine gland tissues, in which non-enzymatic and enzymatic approaches were applied. In one of the first reports published in 1980, Fritz and coauthors described a protocol to isolate feline parotid acini using sequential digestion in a collagenase/trypsin solution4. In 1989, Hann and coauthors adjusted this protocol for acini isolation from rat LGs using a mixture of collagenase, hyaluronidase and DNase5. In 1990, Cripps and colleagues published the method of non-enzymatic dissociation of lacrimal gland acini6. Later, in 1998, Zoukhri and coauthors returned to an enzymatic dissociation protocol for following up Ca2+-imaging on LG and SMG isolated acini7. Within the last decade, researchers have turned their focus on isolation of stem/progenitor cells from exocrine glands. Pringle and coauthors described a protocol in 2011 for isolation of mouse SMG stem cells8. This method was based on isolation of stem cell-containing salispheres, which were maintained in culture. The authors claimed that proliferating cells expressing stem cell-associated markers could be isolated from these salispheres8. Shatos and coauthors published the protocol for progenitor cell isolation from uninjured adult rat LGs using enzymatic digestion and collecting “liberated” cells9. Later, in 2015, Ackermann and coauthors adjusted this procedure to isolate presumptive "murine lacrimal gland stem cells" ("mLGSCs") that could be propagated as a mono-layer culture over multiple passages10. However, none of the before mentioned procedures allowed for distinguishing cellular subtypes and individual populations of isolated epithelial cells. In 2016, Gromova and coauthors published a procedure for isolation of LG stem/progenitor cells from adult murine LGs using FACS11. However, this protocol was not intended to isolate MECs.
Recently, we have shown that we are able to isolate SMA+ cells from 3 week-old SMA-GFP mice12. However, at this time we have not separated different populations of SMA+ cells. Here we established a new procedure for the direct isolation of differentiated MECs and pericytes from adult LGs and SMGs.
Protocol
All animal work was conducted according to the National Institute of Health (NIH) guidelines and was approved by Institutional Animal Care and Use Committee of the Scripps Research Institute. All efforts were made to minimize the number of mice and their suffering. All experimental animals received a standard diet with free access to tap water.
NOTE: The main steps for MEC and pericyte isolation are outlined schematically in Figure 1A-F. All reagents and equipment used for this procedure are described in Table 1.
1. Mice and Labelling the SMA cells
- Use adult (2-4 months old) tamoxifen-inducible, αSMA driven reporter mice SMACreErt2/+:Rosa26-TdTomatofl/fl.
NOTE: The SMACreErt2 strain was kindly provided by Dr. Ivo Kalajzic13. Rosa26-TdTomatofl/fl (B6.Cg-Gt(ROSA)26Sortm9(CAG-tdTomato)Hze/J, also known as Ai9) strain (# 007909) were purchased from Jackson Laboratory (Sacramento, CA). SMA+ cells were labeled by intraperitoneal tamoxifen (TM) administration. - Preparation of tamoxifen solution
- Prepare filtered corn oil. Use 0.22 µm vacuum filter since corn oil is viscous.
- Transfer 1 g of TM powder from the bottle into a 50 mL tube. Add 1 mL of ethanol to the bottle, cap and shake it to rinse then add to a 50 mL tube. Repeat once more with another 1 mL of ethanol.
- Add filtered corn oil to make 50 mL of a 20 mg/mL TM solution. Vortex the tube, wrap it in foil, and put it in a shaking water bath or shaking incubator at 45 °C.
- It may take about 12-24 h to dissolve the TM. From time to time, remove the tube and check for any remaining crystals. Once the TM is completely dissolved, aliquot and store at -80 °C. A thawed aliquot can be reused.
- To label SMA+ cells, inject mice intraperitoneally (IP) with TM on two sequential days.
- Inject 3-4 weeks old SMACreErt2/+:Rosa26-TdTomatofl/f any gender mice with TM at 100 µL/20 g (or 2 mg/20 g) body weight (Figure 1A). Mice are ready to be used for cell isolation in 2-3 days after the last TM injection. If needed, injected mice can be sacrificed at longer periods of time after TM injection.
NOTE: As controls for proper compensation during FACS, one wild type (C57Bl/6) mouse and one SMACreErt2/+:Rosa26-TdTomatofl/fl mouse not injected with TM (with “unstained” MECs) of the same age would be required. Use the same calculations provided for 2 SMACreErt2/+:Rosa26-TdTomatofl/fl mice. Not injected SMACreErt2/+:Rosa26-TdTomatofl/fl will allow evaluation of the DSRed background. The C57Bl/6 mouse will serve as a negative control of unstained cells.
- Inject 3-4 weeks old SMACreErt2/+:Rosa26-TdTomatofl/f any gender mice with TM at 100 µL/20 g (or 2 mg/20 g) body weight (Figure 1A). Mice are ready to be used for cell isolation in 2-3 days after the last TM injection. If needed, injected mice can be sacrificed at longer periods of time after TM injection.
2. Solutions and Buffers
NOTE: The LG is an epithelial origin gland that contains an extracellular matrix that makes dissociation of cells difficult. Therefore, using a special combination of enzymes and a multistep digestion process described below is recommended.
- Dispase type II stock solution (25x)
- Dissolve 120 mg of dispase type II powder in 2 mL of 50 mM HEPES/150 mM NaCl to prepare a 25x stock solution (final concentration of dispase should be 30 Units/mL). Units per milligram may vary depending on the number of mice and concentration of the dispase should be adjusted accordingly.
- Prepare 200 µL aliquots and store them at -70 °C for up to 6 months or 4 °C for several days. Do not freeze/thaw the aliquot of dispase more than once to prevent enzyme degradation.
- DNase type I stock solution
- Dissolve 5 mg of DNase type I powder in a 5 mL solution of 50% glycerol, 20 mM Tris buffer (pH 7.5), and 1 mM MgCl2 (stock concentration should be approximately 2000 Units/mL). Units per milligram may vary depending on number of mice and thus concentration of DNase should be adjusted accordingly.
- Filter the stock solution using a 0.22 µm filter and a 10 mL syringe.
- Prepare 200 µL aliquots and store them at -70 °C for up to 6 months or 4 °C for several days. Do not freeze/thaw more than once to prevent enzyme degradation.
- Digestion medium
- To 10 mL of DMEM low glucose without glutamine, add 100 µL of cell culture supplement (e.g., Glutamax, see Table of Materials) for a dilution of 1:100.
- To 2 mL of DMEM low glucose with cell culture supplement, add 6 mg of Collagenase type I and mix thoroughly by pipetting (enzyme on wet ice), 160 µL of dispase stock solution (2.4 U/mL final concentration), 16 µL of DNase type I stock solution (8 U/mL final concentration), and 12 µL of 1 M CaCl2 (6 mM final concentration).
NOTE: Calcium is required to increase enzymatic activity14,15. All calculations are provided for isolation of cells from four lacrimal glands from two adult mice. The volume of digestion medium may vary depending on the amount of tissue and number of replicates. Do not use more than 4 lacrimal glands from 2-4 months old mice per 2 mL medium.
- Blocking medium I
- To 25 mL of DMEM/F-12, add FBS (15% final concentration), 250 µL of cell culture supplement (see Table of Materials) for a dilution of 1:100, and 50 µL of 0.5 M EDTA pH 8.0 (1 mM final concentration).
NOTE: Of the different types of medium that were compared for this protocol DMEM/F-12 gave the best results. This medium has also been used by other researchers to isolate/culture epithelial cells16,17.
- To 25 mL of DMEM/F-12, add FBS (15% final concentration), 250 µL of cell culture supplement (see Table of Materials) for a dilution of 1:100, and 50 µL of 0.5 M EDTA pH 8.0 (1 mM final concentration).
- Blocking medium II
- To 25 mL of PBS, add 50 µL of 0.5 M EDTA pH 8.0 (1 mM final concentration).
- Recovery medium
- To 2 mL of HBSS supplemented with 5 mM MgCl2, add 100 µL of DNase type I stock solution to 100 U per 2 mL final concentration. Relatively high concentrations of DNase-type I is required to reduce aggregation of epithelial cells.
- Fluorescence activated cell sorting (FACS) buffer
- To 486.5 mL of PBS, add 12.5 mL of serum (2.5% final concentration) and 1 mL of 0.5 M EDTA pH 8.0 (1 mM final concentration).
NOTE: The buffer can be stored at 4 °C for a maximum of 6 weeks.
- To 486.5 mL of PBS, add 12.5 mL of serum (2.5% final concentration) and 1 mL of 0.5 M EDTA pH 8.0 (1 mM final concentration).
3. Adult Mouse Lacrimal Gland Harvesting and Microdissection
- Anesthetize the mouse by isoflurane inhalation (adjust the isoflurane flow rate or concentration to 5% or greater) and sacrifice by cervical dislocation. Perform anesthesia and euthanasia according to institutional IACUCs recommendations.
- Using fine forceps and scissors, remove skin between the eye and ear (Figure 2A).
- To dissect a LG, gently pull LG using tweezers and at the same time scratch the connective tissue around the LG using the sharp tip of small scissors to free it (Figure 2B).
- Avoid cutting with scissors, as the LG and parotid salivary glands are located very close to each other and must be separated prior to dissection. When LG and parotid glands are separated, cut the LG out using scissors. Place glands into a 35 mm dish with 2 mL of cold PBS (keep on ice) (Figure 1B).
- As the LG is covered by a connective tissue capsule/envelope, trim any surrounding fat and connective tissue under a dissecting microscope and remove the LG capsule with two forceps.
- Repeat this step for all glands.
- Check a small piece of tissue under fluorescent microscope to ensure cell labeling (Figure 1C).
4. Preparation of LG Single-cell Suspension
- Transfer all LGs into a 35 mm dish with 0.5 mL of room temperature (RT) digestion media and mince LGs using small scissors into very small pieces (approximately 0.2-1 µm2). Normally, it takes about 3 min to mince 4 LGs (Figure 2C).
- Transfer minced tissue into a 2 mL round bottom tube using a wide-bore pipette filter tip. Use a normal sized pipette tip with the tip cut off (Figure 2D).
- Add up to 2 mL digestion medium and mix by inverting the tube.
- Place tube in a shaking incubator (or shaking water bath), at 37 °C, 100-120 rpm for 90 min.
- Every 30 min slowly pipette gland pieces 20-30 times using a 1,000 µL filter tip with the decreasing bore size (Figure 2D). After incubation/trituration, take a 10 µL aliquot and inspect under a microscope for clusters. If clusters persist, continue digestion.
- After 90 min, pass the sample 2-3 times through an insulin syringe needle (31G) to further release cells into suspension.
NOTE: No visible lacrimal gland pieces should remain in the solution once digestion is completed (Figure 1D). - Transfer cell suspension to a 15 mL tube and add blocking media type I to a total of 5 mL. Invert the tube 2-3 times to mix.
- Pass the cell suspension through a 70 μm cell strainer placed on a 50 mL tube. Wash the strainer with 1 mL of blocking media type I. Repeat step 4.8 again.
- Centrifuge samples at 0.4 x g for 5 min at RT.
- Aspirate the supernatant. Re-suspend cells in 2 mL of blocking medium type II using a 1 mL pipette tip and transfer the cell suspension into a 2 mL microcentrifuge tube.
- Centrifuge the cells at 0.4 x g (24 x 1.5/2.0 mL rotor; see Table of Materials) for 3 min at RT.
- Aspirate supernatant and re-suspend cells in 1 mL of cell detachment solution (see Table of Materials).
NOTE: Here, the cell detachment solution is Accutase, a marine-origin enzyme with proteolytic and collagenolytic activity that detaches/dissociates cells for analysis of cell surface markers. - Incubate cells at 37 °C, at 100-120 rpm for 2-3 min. Over-digestion with cell detachment solution may damage cellular membranes.
- Transfer cell suspension into a 50 mL tube and add up 10 mL of blocking medium type I. Centrifuge tube at 0.4 x g (24 x 1.5/2.0 mL rotor; see Table of Materials) for 5 min.
- Discard supernatant and re-suspend cells in 6 mL of recovery media and incubate cells for 30 min at RT.
- Check 10 µL of cell suspension under the microscope to ensure complete cell dissociation (Figure 3).
- Count cells using a cell counter and Trypan blue. Normally, we expect 4 x 105-6 x 106 cells from four LGs (one sample).
- Centrifuge cells at 0.4 x g (24 x 1.5/2.0 mL rotor; see Table of Materials) for 3 min at RT and proceed to antibody staining.
5. Antibody Staining
- Add up to 5 x105 cells to a 2 ml tube containing 400 µL of FACS buffer. Add 5 µL of Brilliant Violet 421 anti-mouse CD326 (EpCAM) and 0.5 µL of Ghost Red 780 (Viability Dye).
- In parallel, prepare controls to adjust FACS compensation:
Negative control-1 (cells from wild type mouse)
Background control unstained cells from SMACreErt2/+:Rosa26-TdTomatofl/fl
Cy7-780 stained cells (cells from the wild type mouse stained with the Ghost Red 780 Viability Dye)
EpCAM-Brilliant Violet 421 (cells from wild type mouse stained with the EpCAM-Brilliant Violet 421 antibody).
NOTE: For each control sample use a minimum of 1 x 105 cells per 400 µL of FACS buffer. - After adding each reagent mix cells thoroughly by pipetting.
- Wrap tube(s) with foil and rotate tubes for 45 min at 4 °C.
- Centrifuge samples at 0.4 x g (24 x 1.5/2.0 mL rotor; see Table of Materials) for 3 min at 4°C.
- Re-suspend cells in 1 mL of FACS buffer. It is important to wash cells to decrease the background during compensation.
6. Fluorescence Activated Cell Sorting
- Transfer cell suspension into 5 mL FACS tubes and proceed with FACS analysis. Keep cells on ice.
- Adjust compensation using single color controls.
- Sort cells at 20 psi through a 100 μm nozzle using appropriate flow cytometer (see Table of Materials). Gating strategy18 is shown in Figure 1E and Figure 4.
- Collect sorted cells into medium, RNA-later, FACS or lysis buffers depending on downstream procedures (Figure 1E,F).
Representative Results
Mouse model to isolate SMA+ MECs and pericytes
The established protocol allows for the isolation of two pure populations: MECs and pericytes from LGs and SMGs (see Table 1). These two types of cells have a different size and appearance. Microvascular pericytes, develop around the walls of capillaries (Figure 5A) and have a squared shape (Figure 5B), while MECs surround the LG secretory acini, have long processes and occupy a relatively large area (Figure 5A,B). The described procedure is based on genetic cell labeling of SMA+ in the TM-inducible SMACreErt2/+:Rosa26-TdTomatofl/fl mouse strain. Additonally, EpCAM antibodies allow the researchers to distinguish epithelial SMA+:EpCAM+ cells of ectodermal origin (MECs) and SMA+EpCAM– cells of endodermal origin (pericytes).
Preparation of single-cell suspension
The LG contains a filamentous extracellular matrix that must be digested thoroughly. The provided protocol allows preparation of a single cell solution for FACS analysis and further applications. The example of dissociated cells is shown in Figure 3.
MEC and pericyte isolation by FACS
To distinguish MECs from pericytes, single cells were stained with antibody to EpCAM, which detects only epithelial cells. The main population of cells was determined by forward and side scatter area gating (Figure 4A). Doublets were excluded by plotting the forward scatter area versus width and with the side scatter area versus width (Figure 4B). Dead cell exclusion was done via Ghost Red 780 (Viability Dye) (Figure 4C). An unlabeled control (Figure 4E), background control and antibody control labeled with a single primary antibody were used to determine the background noise (nonspecific antibody binding) and to establish proper compensation for optimum separation between signals (Figure 4D). Data analyses were performed by using FlowJo software.
MECs and pericytes were gated by DsRed labeling. DsRed+ dim (not shown) and DsRed+ bright cells within both MEC and pericyte cell populations were detected (Figure 4D). The brightness of labeled cells may depend on level of SMA expression or degree of reporter activation upon TM injection19. Only DS Red+ bright cell population were collected since only fully differentiated cells were required. The DS Red+ dim cell populations require further investigation.
Downstream applications
It is well known that MECs play an important contractile function in exocrine glands. Moreover, they are very plastic cells and have features of stem cells. Therefore, isolated MECs could be used in multiple applications. For example, cells can be cultured, used for RNA isolation or transplantation (Figure 1F)12,20,21,22.
| Parameter | Lacrimal gland | Submandibular gland |
| Number of mice per sample | 2 | 1 |
| Number of glands per sample | 4 | 2 |
| Dissection glands | Separate from parotid gland | Separate from sublingual gland |
| Concentration of collagenase per sample | 6 mg/2 mL | 9 mg/2 mL |
| Approximate cell number after enzymatic dissociation | 4×105-6×105 | 9×105-1.5×106 |
| Recovery step (see the section “Adult Mouse Lacrimal Gland Single-cell Dissociation”, point 15) | Re-suspend cells in 6 ml of recovery media | Re-suspend cells in 12 mL of recovery media |
| Volume of FACS buffer during antibody staining | 400 µL | 2 tubes by 400 µL; it is better to split the cells into two or three tubes that each tube has not more than 6×105 |
Table 1: Modifications of the protocol for isolation of cells from the submandibular gland (SMG). The table describes major modifications required to isolate MECs and pericytes from murine SMG in comparison with the procedure for murine LG.
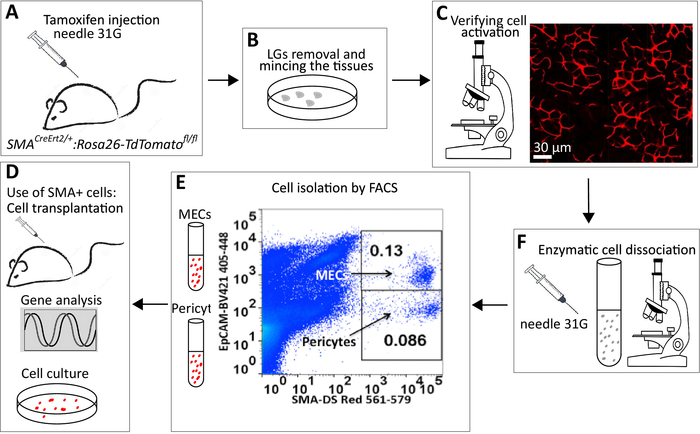
Figure 1: A schematic representation of the experiment. (A) IP injections of the SMACreErt2/+:Rosa26-TdTomatofl/flmice with TM. (B) Isolation and mincing of the LG or SMG. (C) Analysis of cell labeling using fluorescence microscope. (D) Multi-step enzymatic digestion to prepare a single-cell solution. It is critical to check digestion steps under a light microscope to ensure that cells are released from clusters. (E) Example of gating showing SMA+ bright DS Red+/EpCAM+ (MECs) and DS Red+bright/EpCAM- (pericytes). (F) Collected MECs and pericytes could be subjected to different downstream procedures including cell cultivation, RNA isolation and gene expression analysis and cell transplantation. Please click here to view a larger version of this figure.
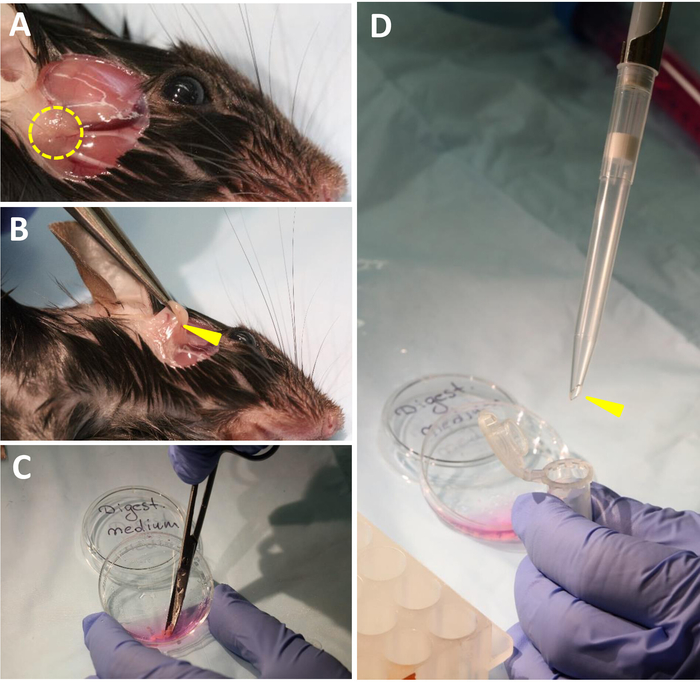
Figure 2: Critical steps of LG isolation/mincing. (A) Removal of skin between the eye and ear to dissect LG. Dashed yellow circle indicates LG location. (B) LG dissection. Yellow arrowhead points out area between lacrimal and parotid glands. (C) LG mincing in digestion medium using scissors with curved, blunt ends. (D) Transfer of minced tissue into 2 mL tube. Yellow arrowhead depicts wide bore size 1 ml tip required for transferring. Please click here to view a larger version of this figure.
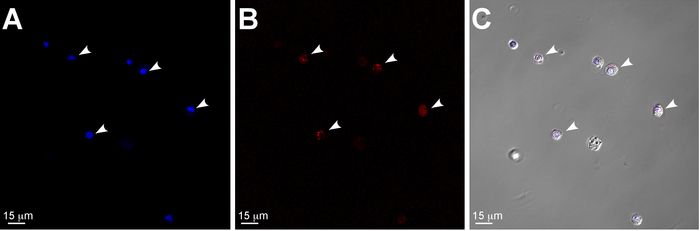
Figure 3: Confocal and differential interference contrast (DIC) images illustrating dissociated single cells from murine LG. (A-C) single cells isolated from two LGs of one 4 month old SMACreErt2/+:Rosa26-TdTomatofl/fl mouse. Nuclei are stained with DAPI (blue). White arrowheads denote SMA+ (DS Red) cells: MECs or pericytes. Scale bar = 15 μm. Please click here to view a larger version of this figure.
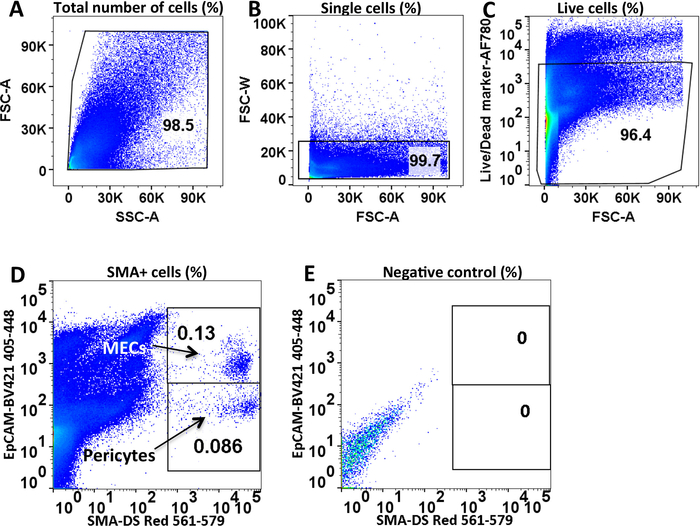
Figure 4: Identification of murine LG MECs and pericytes using FACS. (A) Determination of the main population of LG cells by forward and side scatter area gating. (B) Exclusion of doublets via forward scatter area versus width. (C) Dead cell exclusion via Ghost Red 780 (Viability Dye). (D) MEC (SMA+ bright/EpCAM+) and pericyte (SMA+ bright/EpCAM–) populations distinguished based on staining with EpCAM antibody. (E) Unlabeled control (cells from wild type mouse). On each plot the % of gated cells is provided. Please click here to view a larger version of this figure.
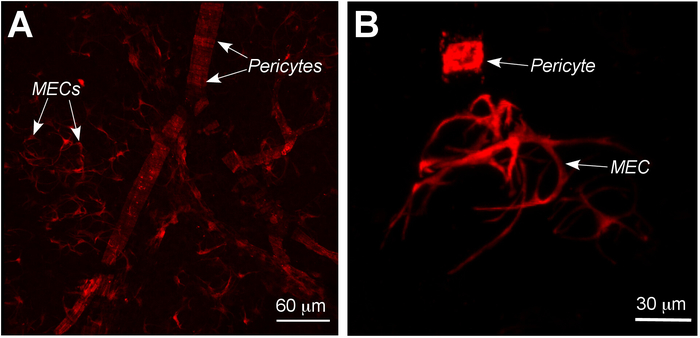
Figure 5: Confocal images showing difference in distribution and shape between MECs and pericytes isolated from murine LG. (A) Whole mount preparation of LG with labeled cells showing difference in distribution between MEC and pericytes. (B) The shape of MEC and pericyte is different. Pericyte is relatively small and has squired shape, whereas MEC is large and has irregular shape and several long processes. Arrowheads = MECs and pericytes. Please click here to view a larger version of this figure.
Discussion
This manuscript described a protocol of MEC and pericyte isolation from LG and SMG. This procedure was based on genetic labeling of SMA, the only reliable biomarker of MECs and pericytes.
The urgency to develop this protocol was motivated by the almost total absence of literature highlighting the isolation of MECs from murine LGs and SMGs. Although genetic labeling was previously used, using SMA-GFP mice to isolate SMA+ cells from young three-week-old LGs12, it did not allow the use these older mice for cell isolation due to partial loss of signal in adult mice. In addition, GFP-labeled cells give a relatively high background in FACS applications23 and require additional compensations. In contrast, the SMACreErt2/+:Rosa26-TdTomatofl/fl mouse line shows no or a low background and high levels of SMA labeling activation throughout mouse postnatal development, adulthood and aging. Usage of the SMACreErt2/+:Rosa26-TdTomatofl/fl mouse is especially important for studies focused on disease progression or aging, since SMA+ cells in these mice could be labeled prior to disease development and studied later when disease/aging progresses. Another critical step of the protocol is thorough LG mincing and the following digestion. This step may reduce the cell number obtained during FACS analysis. Overall, isolation and immediate analysis of primary cells is also important due to significant changes in the transcriptional profiles of cells maintained in culture24.
Additionally, the described protocol for MEC and pericyte isolation from murine LGs enables different downstream procedures. Protein, RNA and DNA extractions are possible from both single-cell populations, although several mice/samples need to be processed in parallel or sequentially to increase the number of cells. Taken together, the obtained results demonstrate an effective and relatively straightforward way for MEC and pericyte isolation from different murine glandular tissue.
Declarações
The authors have nothing to disclose.
Acknowledgements
We thank Dr. Ivo Kalajzic for providing us with the SMACreErt2 mouse strain, Takeshi Umazume for mouse tailing and genotyping, Mark Shelley for acquiring professional pictures for Figure 2. We also thank Scripps Council of Scientific Editors and Mark Shelley for Scientific English editing. We are grateful to The Scripps Research Institute Flow Cytometry core for assistance with cell sorting and to Dr. Robin Willenbring for multiple discussions/advice on FACS data analysis.
This work was supported by the National Institutes of Health, National Eye Institute Grants 5 R01 EY026202 and 1 R01 EY028983 to H.P.M.
Materials
| Biosafety Cabinet | SterilCard Baker | 19669.1 | Class II type A/B3 |
| 10 ml Disposable serological pipets | VWR | 89130-910 | Manufactured from polystyrene and are supplied sterile and plugged |
| 10 mL Disposable serological pipets | VWR | 89130-908 | Manufactured from polystyrene and are supplied sterile and plugged |
| 15 mL High-clarity polypropylene conical tubes | Falcon | 352196 | |
| 25 mL Disposable serological pipets | VWR | 89130-900 | Manufactured from polystyrene and are supplied sterile and plugged |
| 5 mL FACS round-bottom tubes | Fisher Scientific, Falcon | 14-959-11A | |
| 50 mL High-clarity polypropylene conical tubes | Falcon | 352070 | |
| Antibiotic-antimycotic | Invitrogen | 15240-062 | |
| Appropriate filter and non-filter tips | Any available | Any available | |
| BD Insulin Syringes | Becton Dickinson | 328468 | with BD Ultra-Fine needle ½ mL 8 mm 31G |
| BD Syringes 10 mL | Becton Dickinson | 309604 | Sterile |
| Brilliant Violet 421 anti mouse CD326 (EpCAM) | Biolegend | 118225 | Monoclonal Antibody (G8.8) |
| CaCl2 1M solution | BioVision | B1010 | sterile |
| Cell culture dishes 35 mm | Corning | 430165 | Non-pyrogenic, sterile |
| Collagenase Type I | Wortington | LS004194 | |
| Corn oil | Any avaliable | Any avaliable | From grocery store |
| Corning cell strainer size 70 μm | Sigma-Aldrich | CLS431751-50EA | |
| Digital Stirrer PC-410D | Corning | Item# UX-84302-50 | |
| Dispase II | Sigma-Aldrich | D4693-1G | |
| Dissecting scissors, curved blunt | McKesson Argent | 487350 | Metzenbaum 5-1/2 Inch surgical grade stainless steel non-sterile finger ring handle |
| DNase I | Akron Biotech, catalog number | AK37778-0050 | |
| Dulbecco’s Modified Eagle’s Medium – low glucose (DMEM) | Sigma-Aldrich | D5546-500ML | with 1000mg/L glucose and sodium bicarbonate, without L-glutamine |
| Dulbecco’s Modified Eagle’s Medium/F12 (DMEM/F12) | Millipore | DF-042-B | without HEPES, L-glutamine |
| Easypet 3 pipette controller | Eppendorf | 4430000018 | with 2 membrane filters 0.45 µm, 0.1 – 100 mL |
| Ethanol | Sigma-Aldrich | E7023-500ML | |
| Ethylenediaminetetraacetic acid (EDTA) | Sigma-Aldrich | E6758 | |
| Fisher Vortex Genie 2 | Fisher Scientific | 12-812 | |
| FlowJo version 10 | Any available | Any available | |
| Fluorescence binocular microscope Axioplan2 | Carl Zeiss | ID# 094207 | |
| Ghost Red 780 Viability Dye | Tonbo Biosciences | 13-0865-T100 | |
| GlutaMAX Supplement | ThermoFisher Scientific, Gibco | 35050061 | |
| Glycerol 99% | Sigma-Aldrich | G-5516 | |
| Hand tally counter | Heathrow Scientific | HEA6594 | |
| Hank's Balanced Salt Solution (HBSS) | Sigma Millipore | H6648-500ML | Modified, with sodium bicarbonate, without calcium chloride, magnesium sulphate, phenol red. |
| Hank's Balanced Salt Solution (HBSS) | ThermoFisher Scientific | 14025092 | With calcium, magnesium, no phenol red. |
| Hausser Bright-Line Phase Hemocytometer | Fisher Scientific | 02-671-51B | 02-671-51B |
| HEPES 1M solution | ThermoFisher Scientific, Gibco | 15630-080 | Dilute 1/10 in ddH20 |
| HyClone Fetal Bovine Serum (FBS) | Fisher Scientific | SH3007002E | |
| Hydrochloric Acid (HCl), 5N Volumetric Solution | JT Baker | 5618-03 | To adjust Tris buffer pH |
| Innova 4230 Refrigerated Benchtop Incubator | New Brunswick Scientific | SKU#: | Shaker; 37 °C, 5% CO2 in air |
| Iris scissors | Aurora Surgical | AS12-021 | Pointed tips, delicate, curved, 9 cm, ring handle |
| Isoflurane Inhalation Anesthetic | Southern Anesthesia Surgical (SAS) | PIR001325-EA | |
| MgCl2 1M solution | Sigma-Aldrich | 63069-100ML | |
| Microcentrifuge tubes 1.5 mL | ThermoFisher Scientific | 3451 | Clear, graduated, sterile |
| Microsoft Power Point | Any available | Any available | |
| NaCl powder | Sigma-Aldrich | S-3014 | |
| Nalgene 25 mm Syringe Filters | Fisher Scientific | 724-2020 | |
| Pen Strep | Gibco | 15140-122 | |
| pH 510 series Benchtop Meter | Oakton | SKU: BZA630092 | |
| Phosphate buffered saline (PBS) | ThermoFisher Scientific | 10010023 | pH 7.4 |
| Pure Ethanol 200 Proof | Pharmco-Aaper | 111000200 | |
| Red blood cell lysis buffer 10x | BioVision | 5831-100 | |
| Roto-torque Heavy Duty Rotator | Cole Parmer | MPN: 7637-01 | |
| Safe-lock round bottom Eppendorf tubes 2 mL | Eppendorf Biopur | 22600044 | PCR inhibitor, pyrogen and RNAse-free |
| Scissors | Office Depot | 375667 | |
| Sorting flow cytometer MoFlo Astrios EQ | Beckman Coulter | B25982 | With Summit 6.3 software |
| Sorvall Legend Micro 17R Microcentrifuge | Thermo Scientific | 75002441 | All centrifugation performed at RT |
| Sorvall RT7 Plus Benchtop Refrigerated Centrifuge | Thermo Scientific | ID# 21550 | RTH-750 Rotor. All centrifugation performed at RT |
| Stemi SV6 stereo dissecting microscope | Carl Zeiss | 455054SV6 | With transmitted light base |
| Tamoxifen | Millipore Sigma | T5648-1G | |
| Trizma base powder | Sigma-Aldrich | T1503 | |
| Trypan blue solution | Millipore Sigma | T8154 | |
| Two Dumont tweezers #5 | World Precision Instruments | 500342 | 11 cm, Straight, 0.1 x 0.06 mm tips |
| Upright microscope | Any available | Any available | With transmitted light base |
| Vacuum filtration systems, standard line | VWR | 10040-436 | |
| Variable volume micropipettes | Any available | Any available |
Referências
- Makarenkova, H. P., Dartt, D. A. Myoepithelial Cells: Their Origin and Function in Lacrimal Gland Morphogenesis, Homeostasis, and Repair. Current Molecular Biology Reports. 1 (3), 115-123 (2015).
- Haaksma, C. J., Schwartz, R. J., Tomasek, J. J. Myoepithelial cell contraction and milk ejection are impaired in mammary glands of mice lacking smooth muscle alpha-actin. Biology Of Reproduction. 85 (1), 13-21 (2011).
- Siedlecki, J., et al. Combined VEGF/PDGF inhibition using axitinib induces alphaSMA expression and a pro-fibrotic phenotype in human pericytes. Graefe’s Archive for Clinical and Experimental Ophthalmology. , (2018).
- Fritz, M. E., LaVeau, P., Nahmias, A. J., Weigel, R. J., Lee, F. Primary cultures of feline acinar cells: dissociation, culturing, and viral infection. American Journal of Physiology. 239 (4), G288-G294 (1980).
- Hann, L. E., Tatro, J. B., Sullivan, D. A. Morphology and function of lacrimal gland acinar cells in primary culture. Investigative Ophthalmology & Visual Science. 30 (1), 145-158 (1989).
- Cripps, M. M., Bromberg, B. B., Bennett, D. J., Welch, M. H. Structure and function of non-enzymatically dissociated lacrimal gland acini. Current Eye Research. 10 (11), 1075-1080 (1991).
- Zoukhri, D., Hodges, R. R., Rawe, I. M., Dartt, D. A. Ca2+ signaling by cholinergic and alpha1-adrenergic agonists is up-regulated in lacrimal and submandibular glands in a murine model of Sjogren’s syndrome. Clinical Immunology and Immunopathology. 89 (2), 134-140 (1998).
- Pringle, S., Nanduri, L. S., van der Zwaag, M., van Os, R., Coppes, R. P. Isolation of mouse salivary gland stem cells. Journal of Visualized Experiments. (48), (2011).
- Shatos, M. A., Haugaard-Kedstrom, L., Hodges, R. R., Dartt, D. A. Isolation and characterization of progenitor cells in uninjured, adult rat lacrimal gland. Investigative Ophthalmology & Visual Science. 53 (6), 2749-2759 (2012).
- Ackermann, P., et al. Isolation and Investigation of Presumptive Murine Lacrimal Gland Stem Cells. Investigative Ophthalmology & Visual Science. 56 (8), 4350-4363 (2015).
- Gromova, A., et al. Lacrimal Gland Repair Using Progenitor Cells. Stem Cells Translational Medicine. 6 (1), 88-98 (2017).
- Hawley, D., et al. Myoepithelial cell-driven acini contraction in response to oxytocin receptor stimulation is impaired in lacrimal glands of Sjogren’s syndrome animal models. Scientific Reports. 8 (1), 9919 (2018).
- Matic, I., et al. Quiescent Bone Lining Cells Are a Major Source of Osteoblasts During Adulthood. Stem Cells. 34 (12), 2930-2942 (2016).
- Bond, M. D., Van Wart, H. E. Characterization of the individual collagenases from Clostridium histolyticum. Bioquímica. 23 (13), 3085-3091 (1984).
- Eckhard, U., Schonauer, E., Brandstetter, H. Structural basis for activity regulation and substrate preference of clostridial collagenases. G, H, and T. Journal of Biological Chemistry. 288 (28), 20184-20194 (2013).
- Breggia, A. C., Himmelfarb, J. Primary mouse renal tubular epithelial cells have variable injury tolerance to ischemic and chemical mediators of oxidative stress. Oxidative Medicine and Cellular Longevity. 1 (1), 33-38 (2008).
- Mueller, S. O., Clark, J. A., Myers, P. H., Korach, K. S. Mammary gland development in adult mice requires epithelial and stromal estrogen receptor alpha. Endocrinology. 143 (6), 2357-2365 (2002).
- Guthmiller, J. J., Zander, R. A., Butler, N. S. Measurement of the T Cell Response to Preerythrocytic Vaccination in Mice. Methods in Molecular Biology. 1325, 19-37 (2015).
- Seime, T., et al. Inducible cell labeling and lineage tracking during fracture repair. Development, Growth & Differentiation. 57 (1), 10-23 (2015).
- Hawley, D., et al. RNA-Seq and CyTOF immuno-profiling of regenerating lacrimal glands identifies a novel subset of cells expressing muscle-related proteins. PLoS One. 12 (6), e0179385 (2017).
- Tata, A., et al. Myoepithelial Cells of Submucosal Glands Can Function as Reserve Stem Cells to Regenerate Airways after Injury. Cell Stem Cell. 22 (5), 668-683 (2018).
- Song, E. C., et al. Genetic and scRNA-seq Analysis Reveals Distinct Cell Populations that Contribute to Salivary Gland Development and Maintenance. Scientific Reports. 8 (1), 14043 (2018).
- Knight, A. n. d. r. e. w. W., B, N. Distinguishing GFP from cellular autofluorescence. Biophotonics International. 8 (7), 7 (2001).
- Januszyk, M., et al. Evaluating the Effect of Cell Culture on Gene Expression in Primary Tissue Samples Using Microfluidic-Based Single Cell Transcriptional Analysis. Microarrays (Basel). 4 (4), 540-550 (2015).

