Generation of Ventricular-Like HiPSC-Derived Cardiomyocytes and High-Quality Cell Preparations for Calcium Handling Characterization
Summary
Here we describe and validate a method to consistently generate robust human induced pluripotent stem cell-derived cardiomyocytes and characterize their function. These techniques may help in developing mechanistic insight into signaling pathways, provide a platform for large-scale drug screening, and reliably model cardiac diseases.
Abstract
Human induced pluripotent stem cell-derived cardiomyocytes (iPSC-CMs) provide a valuable human source for studying the basic science of calcium (Ca2+) handling and signaling pathways as well as high-throughput drug screening and toxicity assays. Herein, we provide a detailed description of the methodologies used to generate high-quality iPSC-CMs that can consistently reproduce molecular and functional characteristics across different cell lines. Additionally, a method is described to reliably assess their functional characterization through the evaluation of Ca2+ handling properties. Low oxygen (O2) conditions, lactate selection, and prolonged time in culture produce high-purity and high-quality ventricular-like cardiomyocytes. Similar to isolated adult rat cardiomyocytes (ARCMs), 3-month-old iPSC-CMs exhibit higher Ca2+ amplitude, faster rate of Ca2+ reuptake (decay-tau), and a positive lusitropic response to β-adrenergic stimulation compared to day 30 iPSC-CMs. The strategy is technically simple, cost-effective, and reproducible. It provides a robust platform to model cardiac disease and for the large-scale drug screening to target Ca2+ handling proteins.
Introduction
Human induced pluripotent stem cell-derived cardiomyocytes (iPSC-CMs) are an attractive human-based platform to model a large variety of cardiac diseases in vitro1,2,3,4,5,6,7,8. Moreover, iPSC-CMs can be used for the prediction of patient responses to novel or existing drugs, to screen hit compounds, and develop new personalized drugs9,10. However, despite significant progress, several limitations and challenges need to be considered when using iPSC-CMs11. Consequently, methods to improve cardiac differentiation protocols, to enhance iPSC-CMs efficiency and maturation, and to generate specific cardiomyocyte subtypes (ventricular, atrial, and nodal) have been intensely studied and already led to numerous culture strategies to overcome these hurdles12,13,14,15.
Notwithstanding the robustness of these protocols, a major concern for the use of iPSC-CMs is the reproducibility of long and complex procedures to obtain high-quality cardiomyocytes that can ensure the same performance and reproducible results. Reproducibility is critical not only when comparing cell lines with different genetic backgrounds, but also when repeating cellular and molecular comparisons of the same cell line. Cell variability, such as well-to-well differences in iPSCs density, may affect cardiac differentiation, generating a low yield and poor-quality cardiomyocytes. These cells could still be used to perform experiments that do not require a pure population of CMs (e.g., when performing Ca2+ transient measurements). Indeed, when performing electrophysiological analysis, the non-CMs will not beat, neither spontaneously nor under electrical stimulation, so it will be easy to exclude them from the analysis. However, because of the poor quality, iPSC-CMs can show altered electrophysiological characteristics (e.g., irregular Ca2+ transient, low Ca2+ amplitude) which are not due to their genetic makeup. Therefore, especially when using iPSC-CMs to model cardiac disease, it is important not to confuse results from a poor-quality CM with the disease phenotype. Careful screening and exclusion processes are required prior to proceeding to electrophysiological studies.
This method includes optimized protocols to generate high-purity and high-quality cardiomyocytes and to assess their function by performing Ca2+ transient measurements using a calcium and contractility acquisition and analysis system. This technique is a simple, yet powerful, way to distinguish between high efficiency and low efficiency iPSC-CM preparations and provide a more physiologically relevant characterization of human iPSC-CMs.
Protocol
The experiments using adult rat cardiomyocytes in this study were conducted with approved Institutional Animal Care and Use Committee (IACUC) protocols of Icahn School of Medicine at Mount Sinai. The adult rat cardiomyocytes were isolated from Sprague Dawley rat hearts by the Langendorff-based method as previously described16.
1. Preparation of Media
- Prepare hiPSC media.
- Equilibrate the supplement and the basal medium to room temperature (RT). Ensure that the supplement has thawed completely. Mix 400 mL of the basal medium and 100 mL of the supplement and filter using a 0.22 μm vacuum-driven filter. Store at 4 °C and equilibrate to RT before use.
- Prepare RPMI + B27.
- Equilibrate the B27 supplement and the basal medium (RPMI 1640) to RT. Ensure that the supplement has thawed completely. Mix 490 mL of the basal medium and 10 mL of the 50x supplement and filter using a 0.22 μm vacuum driven filter. Store at 4 °C and equilibrate to RT before use.
- Prepare RPMI + B27 (-) insulin.
- Equilibrate the B27 (-) insulin supplement and the basal medium (RPMI 1640) to RT. Ensure that the supplement has thawed completely. Mix 490 mL of the basal medium and 10 mL of the 50x supplement and filter using a 0.22 μm vacuum driven filter. Store at 4 °C and equilibrate to RT before use.
- Prepare selection media (RPMI (-) glucose + B27 + lactate).
- Equilibrate the B27 supplement and the basal medium (RPMI 1640 (-) glucose) to RT. Ensure that the supplement has thawed completely. Mix 490 mL of the basal medium and 10 mL of the 50x supplement, add 4 mM sodium lactate constituted in sterile water, and filter using a 0.22 μm vacuum driven filter. Store at 4 °C and equilibrate to RT before use.
- Prepare RPMI 20.
- Equilibrate the basal medium (RPMI 1640) to RT. Mix fetal bovine serum (FBS) (20% final concentration) and RPMI. Filter using a 0.22 μm vacuum driven filter and store at 4 °C. Equilibrate to RT before use.
- Prepare passaging media by adding Rho-associated, coiled-coil containing protein kinase (ROCK) inhibitor (2 μM final concentration) to hiPSC media.
- Prepare D0 media by adding GSK-3 inhibitor, CHIR 99021 to RPMI + B27 (-) insulin media (10 μM final).
- Prepare D3 and D4 media by mixing RPMI + B27 (-) insulin media with IWR-1 (5 μM final).
NOTE: D1 and D5 media is constituted of RPMI + B27 (-) insulin. D7 media is constituted of RPMI + B27. - Prepare blocking buffer (2% bovine serum albumin [BSA], 2% FBS, 0.05% NP-40 in phosphate-buffered saline [PBS]): In a 50 mL conical tube, add 1 g of BSA, 1 mL of FBS, 49 mL of PBS, and 250 μL of NP-40. Mix until fully dissolved.
2. Preparation of Human Embryonic Stem Cell (hESC)-qualified Matrix Coated Plates and Coverslips
NOTE: Perform all the steps under a sterilized tissue culture hood.
- Thaw hESC-qualified matrix stock solution overnight on ice at 4 °C. Refer to the product specification sheet to determine appropriate aliquot volumes as this may vary depending upon the stock. Store these aliquots at -20 °C in 1.5 mL microcentrifuge tubes.
- In order to use the matrix for coating plates or glass coverslips, first thaw an aliquot of hESC-qualified matrix at 4 °C for 30 min.
- Aliquot 24 mL of cold Dulbecco's modified Eagle medium (DMEM): nutrient mixture F-12 (DMEM/F:12) media into a 50 mL conical tube.
- Mix the cold DMEM/F:12 media with a 2 mL glass pipette in order to cool down the surface of the pipette.
- Using the same pipette, take up approximately 500−700 μL of cold DMEM/F:12 and mix with the hESC-qualified matrix aliquot within the microcentrifuge tube itself.
- Once properly mixed, transfer the solution to the 50 mL conical tube containing the cold DMEM/F:12 and mix again.
- For a standard 6 well plate, add 1 mL of this mixture to each well. Ensure that the well is entirely covered. Leave the plates at RT underneath the tissue culture hood for at least 30 min. If desired, store at 4 °C immediately after plating for up to 1 week and equilibrate to RT for 30 min prior to use.
- In order to use the plates, aspirate the matrix and replace with appropriate media. Use immediately.
- Store the glass coverslips in a sterile environment (e.g., inside a sterile tissue culture hood).
- Before coating, wipe each individual coverslip with 70% ethanol. Once the coverslip is dry, place it inside a well of a sterile 6 well plate.
- Take 250−300 μL of the hESC-qualified matrix solution and carefully dispense it directly onto the center of the glass coverslip. Leave the coverslips at RT underneath the tissue culture hood for at least 30 min before use.
3. Preparation of Small Molecules
NOTE: Reconstitute all small molecules and Wnt modulators in DMSO unless otherwise stated.
- Prepare 10 mM aliquots of 25 µL each of IWR-1 and CHIR 99021 and store at -20 °C.
- Reconstitute 10 mM aliquots of 50 µL each of Thiazovivin (ROCK inhibitor) and store at -20 °C.
4. Maintenance and Passaging of iPSCs
NOTE: Perform all of the following steps under a sterile tissue culture hood.
- Maintain the iPSCs in standard 6 well plates and perform all steps under sterile conditions. Maintain the cells with 2 mL of hiPSC media per well. Change the media every other day. Keep the cells at 37 °C, 6% O2, 5% CO2. Passage the cells when they are between 70−80% confluent.
- Equilibrate PBS without Ca2+ and Mg2+ to RT.
- To start the passaging process, add 1 mL of PBS without Ca2+ and Mg2+ to the well that needs to be passaged. Incubate at RT for 7−10 min. Check the cells underneath the microscope to ensure that PBS treatment has not resulted in complete dissociation of the monolayer.
- Remove PBS and replace with 1 mL of passaging media. Use a cell lifter to gently scrape and lift the cells from the surface of the well.
- Mechanically dissociate the cells using a sterile 2 mL glass pipette. Repeat until the cells are well dissociated evenly into small colonies when observed under a microscope.
- Once the cells have been sufficiently dissociated, add 5 mL of passaging media to split the cells 1:6. Adjust the amount of passaging media to be added to match the preferred split ratio.
- Aspirate the hESC-qualified matrix from the hESC-qualified matrix-coated plate and replace with 1 mL of passaging media per well. Add 1 mL of dissociated cells per well.
5. Cardiomyocyte Differentiation
- Use hiPSC lines that are well-established (more than 20 passages) and exhibit a homogeneous morphology before starting cardiac differentiation.
- Ensure that the iPSCs are around 70−80% confluent.
- Wash the cells 1x in PBS without Ca2+ and Mg2+.
- Add 2 mL of D0 media (step 1.7) per well and transfer the cells back to the incubator.
- After 24 h, replace with 3 mL of D1 media per well for 48 h.
- On day 3, replace the media with 2 mL of D3 media per well. Repeat with D4 media on day 4.
- On day 5, replace the media with 3 mL of D5 media per well.
- On day 7, replace the media with 3 mL of D7 media per well and transfer to an incubator with 37 °C, 5% CO2, and normal O2 concentration. Replace the D7 (RPMI + B27) media every 2 days.
6. Selection Procedure and iPSC-CM Dissociation
- Ten days before performing the Ca2+ transient measurements or any functional analysis, replace the RPMI + B27 media with 3 mL of selection media per well for 48 h.
- Replace the media with 3 mL of selection media for another 48 h.
- Replace the media with 2 mL of RPMI + B27 media per well for 24 h.
- Coat standard 6 well plates as described in section 2.
- Add 1 mL of sterile 0.25% trypsin with EDTA to each well. Incubate the plate at 37 °C for 5 min.
- Using a 1,000 μL pipette, mechanically dissociate the cells so that single cells can be seen when observed under a microscope.
- Transfer the cells to a sterile 15 mL conical tube and add 2 mL of RPMI 20 media per well. Centrifuge for 5 min at 800 x g.
- Aspirate the supernatant and resuspend the cells in RPMI + B27 media. Aspirate the hESC-qualified matrix from the plates and replace with 1 mL of RPMI + B27 media.
- Using a 1,000 mL pipette tip, mechanically dissociate the cell pellet until the solution appears homogeneous.
- Transfer roughly 500,000 cells to each well. Transfer to incubator for 24 h.
- Replace the media with 3 mL of selection media per well for 48 h.
- Replace the media with 3 mL of RPMI + B27 per well. Maintain cells in D7 media (RPMI + B2 changing media every 2 days until ready for functional analysis.
7. Preparation of iPSC-CMs for Flow Cytometry
- Once the cells are of the desired age and have undergone metabolic selection (section 6), wash the cells with PBS without Ca2+ and Mg2+.
- Add 1 mL per well of trypsin 0.25% and incubate for 5−7 min at 37 °C.
- Pipette the mixture 5−10x with a P1000 tip to singularize the cells, and transfer to a 15 mL tube containing 2 mL of RPMI 20.
- Spin the cells at 600 x g for 5 min.
- Add 100 µL of fixation solution (4% PFA) to the cell pellet. Add the solution dropwise with continuous, gentle vortexing and then set on ice for 15 min.
- Add 1.5 mL of PBS. Collect the cells by centrifugation and aspirate the supernatant.
- For every experiment, include one unstained control per fixation/permeabilization condition.
- Resuspend cells in 500 µL of blocking solution (2% FBS/2% BSA in PBS with 0.1% NP-40) for 30 min at RT.
- Without removing the blocking solution, add the primary antibody MLC2V/MLC2A (5 mg/mL), and incubate 45 min at RT.
- Wash with blocking buffer. Collect cells by centrifugation and aspirate solution.
- Add secondary antibody Alexa Fluor 555/488(1:750) diluted in the blocking buffer for 45 min at RT or overnight at 4 °C.
- Add 1.5 mL of PBS. Collect cells by centrifugation and aspirate solution.
- Resuspend cells in 250−300 µL of PBS. Use a P1000 pipette to disaggregate the cells.
- Prepare round bottom tubes with a 35 µm nylon mesh cell strainer cap. Pre-wet the cell strainer with 50 µL of PBS and set the tube on ice.
- Transfer the solution with the disaggregated cells to the round bottom tubes with the cell strainer caps. Allow the cell solution to drain naturally or tap the bottom of the tube against a flat surface, as necessary, to ensure complete drainage and collection of the cells into the tube. Make sure to set the tube back on ice as soon as possible.
- Rinse the cell strainer with 250 µL of PBS to recover any residual cells.
- Maintain the tubes on ice and cover with aluminum foil until flow cytometry analysis.
8. Plating Cardiomyocytes onto Glass Coverslips
NOTE: Perform all steps in a sterile environment.
- Prepare the glass coverslips as described in steps 2.10 and 2.11.
- Once the iPSC-CMs have been selected and are of the desired age, follow steps 6.5−6.7 to dissociate the iPSC-CMs.
- Aspirate the supernatant and resuspend the cells in a sufficient amount of RPMI 20 media to have roughly 300,000 cells per 250 μL.
- Using a 2 mL glass pipette, mechanically dissociate the cell pellet until the solution appears homogeneous.
- Aspirate the hESC-qualified matrix from the coverslips.
- Using a 1000 μL pipette, mix and pull 250 μL of the solution from the 15 mL conical tube.
- Slowly dispense the 250 μL of the solution onto the glass coverslips, taking extra care to only add to the area where the hESC-qualified matrix coating is present.
- Transfer carefully to the incubator and leave overnight, taking care not to shake or spread the cells on the coverslips. The next morning, gently add 2 mL of D7 (RPMI + B27) media to each well with the coverslip. Change the media after 24 h and every 2 days after that.
9. Fixing Cells
- Ensure that an adequate amount of PBS with Ca2+ and Mg2+ is equilibrated to 4 °C.
- Prepare a 4% paraformaldehyde (PFA) solution diluted in PBS with Ca2+ and Mg2+ and equilibrate to 4 °C.
- Once the iPSC-CMs are of appropriate age, have undergone metabolic selection (section 6), and have been plated on coverslips (section 8), wash the cells 3x with 1 mL of cold PBS with Ca2+ and Mg2+ per well.
- Add 1 mL of cold 4% PFA and leave the cells at RT underneath the hood for 15 min.
- Wash the cells with cold PBS to remove excess PFA.
- Add 2 mL of PBS with Ca2+ and Mg2+ and store at 4 °C.
10. Immunofluorescence Staining
- Remove PBS from the fixed cells and add 1 mL of blocking buffer. Incubate for 1 h at RT.
- Remove blocking buffer and add the primary antibody (5 mg/mL) diluted in blocking buffer. Incubate overnight at 4 °C.
- Wash 3x in PBS with Ca2+ and Mg2+ for 5 min each.
- Add secondary antibody diluted in blocking buffer 1:1,000.
- Cover the plate with aluminum foil to protect it from light and incubate for 45 min at RT. Keep the aluminum foil for the following steps.
- Wash 3x in PBS with Ca2+ and Mg2+, 5 min each.
- Add a sufficient amount of DAPI working solution to completely cover the cells and incubate at RT for 10 min.
- Wash the sample thoroughly with PBS with Ca2+ and Mg2+ to remove excess DAPI.
- Take new glass slides and add a drop of mounting media in the middle. Use the dropper to evenly spread the mounting media. Put the coverslips with the cells face down on the slides.
NOTE: Cells can be stored for 30 days if protected from light.
11. Assessment of Intracellular Ca2+ Transients
- Once the iPSC-CMs are at least 3 months old, have undergone metabolic selection (section 6), and have been plated on coverslips, treat them with 2 μL of Fura-2, AM (final concentration: 1 μM) and incubate at 37 °C for 10 min.
NOTE: Fura-2 is light-sensitive. Perform all loading procedures and experiments in the dark. - Prepare the calcium and contractility acquisition and analysis system.
- Power the system ensuring that the arc lamp is initiated (Figure 1B).
- Place the chamber on the system and connect the tubes from the pump to the appropriate inlet and outlets and the electric wire from the stimulator to the chamber as shown in Figure 1C.
- Fill the perfusion tube that runs through the fluidic inline heater with 37 °C prewarmed Tyrode's solution.
- Adjust the camera and framing aperture dimensions to minimize background area.
- Mount a glass coverslip with iPSC-CMs into the chamber and fasten.
- Add 500 μL of Tyrode's solution directly on top of the fastened glass coverslip gently and start perfusing the chamber (1.5 mL/min) with Tyrode's solution.
- Pace iPSC-CMs with 1 Hz field stimulation using the electrical stimulator (10 V, 4 ms).
- Incubate iPSC-CMs in the chamber with stimulation for at least 3−5 min for the cells to wash the Fura-2 dye and adapt to the environment and to wash out the fluorescent dye.
- Adjust the viewing window to the left upper area of the glass coverslip.
- Begin recording.
- After collecting a consistent stream of 5−10 peaks, click Pause to temporarily stop recording.
- Ensuring that neither the focus nor dimensions of the viewing window are altered, move the microscope's stage to the adjacent area, moving toward the opposite end, and resume recording.
- Repeat steps 11.9 and 11.10 to scan across the coverslip, initially moving to the left, then downwards in a zig-zag fashion to cover the whole coverslip area. This consists of 80−100 measurements per coverslip.
NOTE: Restrict the total measurement time to 10 min as secondary factors cause a decrease in Ca2+ transient. - Once the Ca2+ transients are acquired, analyze the data with the fluorescence traces analysis software according to the manufacturer's instructions.
Representative Results
The protocol described in Figure 1A generated highly pure cardiomyocytes that acquire a ventricular/adult-like phenotype with time in culture. As assessed by immunofluorescence staining for the atrial and ventricular myosin regulatory light chain 2 isoforms (MLC2A and MLC2V, respectively), the majority of the cells generated by this protocol were MLC2A-positive at day 30 after induction of cardiac differentiation, while MLC2V was expressed in much lower amounts at the same time point (Figure 2A, top panels). As the time in culture increased (day 60 and 90), a complete switch of MLC2 isoforms (MLC2A to MLC2V) was observed (Figure 2A, bottom panels). In order to quantify the MLC2A and MLC2V-positive cells, flow cytometry analysis was performed. In accordance with the immunofluorescence results, the flow cytometry data demonstrated early stage (day 30) iPSC-CMs mostly expressing MLC2A (29.8% MLC2A + vs. 1.9% MLC2V +) (see Figure 2B, left panel), as compared to late stage (day 90) iPSC-CMs, which mostly expressed MLC2V (41.3% MLC2V + vs. 16.7% MLC2A +) (see Figure 2B, right panel). Because the expression pattern of MLC2A and MLC2V is known to be a hallmark of cardiac differentiation and maturation, these results suggest that prolonged culture time increases maturation of iPSC-CMs and that the majority of cells appear to be committed to the ventricular phenotype. We then assessed the time dependence of Ca2+ handling maturation. Ca2+ transient was measured in iPSC-CMs at the three differentiation times (day 30, 60, and 90), and compared to isolated adult rat cardiomyocytes (ARCMs). The Ca2+ amplitude was significantly increased in the iPSC-CMs at day 90 and was similar to the ARCMs (Figure 3A,B). The rate of Ca2+ reuptake (decay-tau) at day 90 was significantly faster compared to day 30 iPSC-CMs, and closer to ARCMs (Figure 3C). The effect of β-adrenergic stimulation on Ca2+ transients was further evaluated by treating the cells with 10 nM of isoproterenol (ISO) for 10 min at 37 °C. As observed in ARCMs, ISO significantly increased Ca2+ transient and accelerated the rate of Ca2+ reuptake in day 90 iPSC-CMs. No changes were observed in day 30 iPSC-CMs (Figure 3D-F). Interestingly, day 90 iPSC-CMs were able to follow increasing electrical stimulation (from 0.5−3 Hz), in a similar manner to that observed in ARCMs (Figure 4B). Taken together, these results show that iPSC-CMs derived from this method have similar characteristics to those seen in native CMs, specifically ventricular-like phenotypes, mature Ca2+ handling properties, and positive β-adrenergic responses.
Contaminating noncardiac cells may influence the functional maturation of human iPSC-CMs during differentiation17. Figure 4A shows a comparison between 3 month-old cells obtained from high efficiency differentiations (top panels, Video 1), robustly expressing the specific ventricular marker (MLC2V), and 3-month-old cells obtained from low efficiency differentiations (bottom panels, Video 2), showing iPSC-CMs mixed with alpha-smooth muscle actin (αSMA)-positive cells. Interestingly, functional analysis demonstrated altered Ca2+ transients in the mixed iPSC-CMs/non-CMs preparation compared to the iPSC-CMs from the high efficiency differentiation. In particular, the cells obtained from low efficiency differentiations exhibited very small Ca2+ amplitude and automaticity (Figure 4B, middle panel) compared to pure iPSC-CMs (Figure 4B, top panel) and ARVCs (Figure 4B, bottom panel). In addition, cells from low efficiency differentiations presented arrhythmic patterns (Figure 4C).
It is important to note that the iPSC-CMs shown in the top panel of Figure 4A also underwent metabolic selection, which is an important step to further enrich a population of iPSC-CMs that is already derived from a highly efficient differentiation. These data indicate that high efficiency cardiac differentiation protocols that generate high-quality CMs are necessary to accurately recapitulate a cardiac phenotype in vitro.
Figure 5 shows the Ca2+ amplitude variability throughout different areas of the CMs monolayer, plotted over a time course of 1,200 s. The Ca2+ amplitudes measured at the beginning of a stimulation frequency of 1 Hz (200 s) were highly variable and became more consistent as the stimulation continued (200−900 s). However, after a time period of 900 s the Ca2+ amplitude was considerably reduced. These data indicate that, when recording Ca2+ transients in iPSC-CMs, it is necessary to let the cells stabilize at 37 °C under constant stimulation for at least 200 s. Additionally, recordings have to be restricted to a specific time window (200−900 s) to ensure reproducible results.
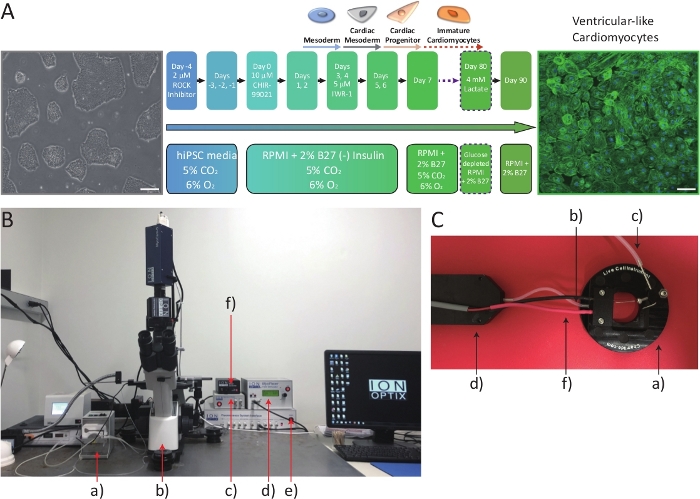
Figure 1: Overall schematic of iPCS-CMs preparation and Ca2+ transient instruments. (A) Schematic of cardiac differentiation protocol showing the developmental stages of differentiating iPSCs. Scale bar = 50 μm. (B) The calcium and contractility acquisition and analysis system. a) Peristalitic pump b) Inverted microscope c) Power source for the digital camera d) Electrical stimulator e) Fluorescence system interface f) Filter wheel controller (C) System chamber. a) System chamber body. b) Fluid inlet. c) Fluid outlet. d) Fluidic inline solution heater. f) Electrodes connecting the system chamber to the electrical stimulator. Please click here to view a larger version of this figure.
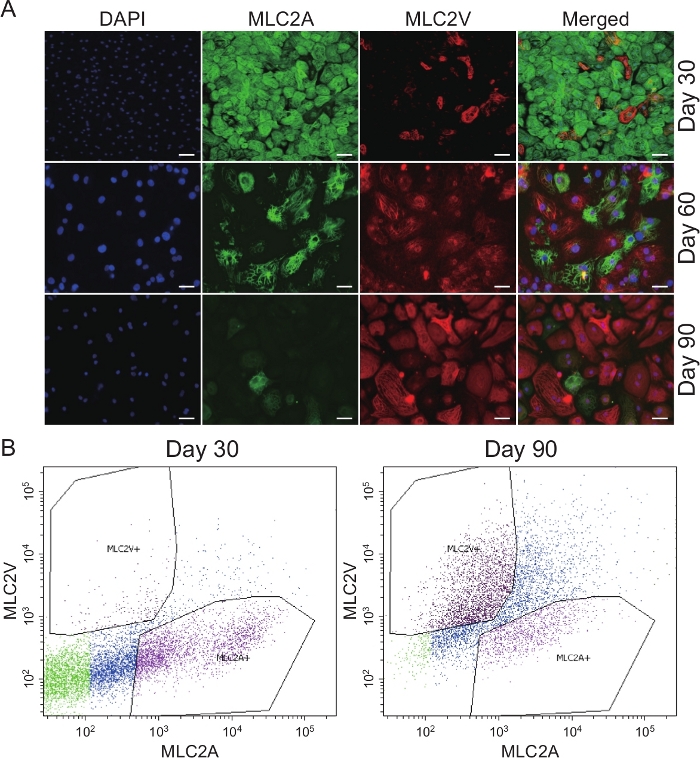
Figure 2: Comparison of MLC2A and MLC2V expression in iPSC-CMs at different days of differentiation. (A) Immunofluorescence staining of iPSC-CMs at day 30, 60, and 90 after differentiation induction for MLC2A and MLC2V. Scale bar = 20 μm. (B) Flow cytometry analysis of iPSC-CMs for MLC2V and MLC2A at 1 month and 3 months post differentiation. Please click here to view a larger version of this figure.
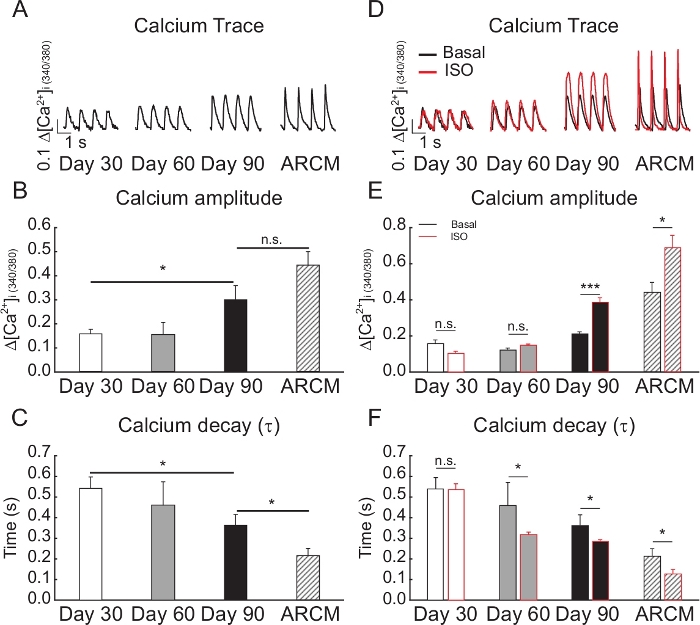
Figure 3: Comparison of iPSC-CMs properties at different days of differentiation. (A) Representative Ca2+ transient traces at different days of differentiation. (B–C) Average Ca2+ amplitude and Ca2+ decay elicited during stimulation at 1 Hz. (D–F) Effect of isoproterenol (ISO, 10 nM) treatment in iPSC-CMs at different days of differentiation. ARCMs indicate isolated rat adult cardiomyocytes. N = 70−100 areas; *p < 0.05; ***p < 0.001, as determined by Student's t-test. Data are represented as Mean ± S.E.M. Please click here to view a larger version of this figure.
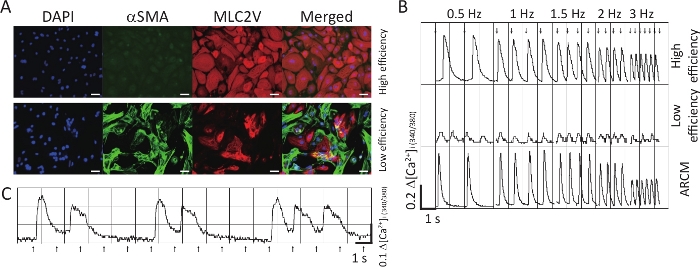
Figure 4: Comparison between high efficiency and low efficiency differentiations of iPSC-CMs. (A) Immunofluorescence staining of iPSC-CMs for αSMA and MLC2V. Scale bar = 20 μm. (B) Representative traces showing Ca2+ transients from high efficiency (above) and low efficiency (middle) differentiations, and isolated rat adult cardiomyocytes (below). The myocytes were stimulated at 0.5 Hz, 1 Hz, 1.5 Hz, 2 Hz, and 3 Hz. (C) Representative trace showing an arrhythmic pattern from a bad differentiation. Arrows indicate point pacing. ARCMs indicates isolated rat adult cardiomyocytes. Please click here to view a larger version of this figure.
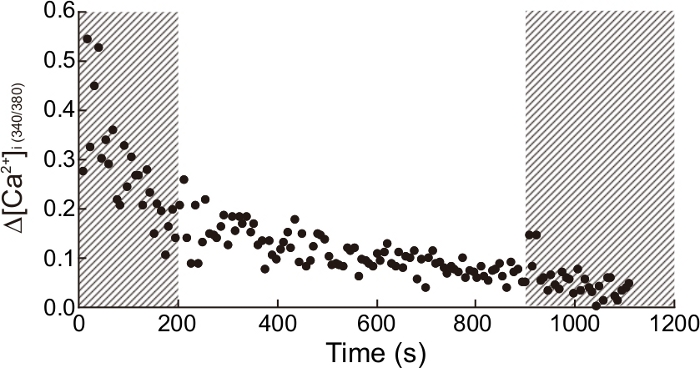
Figure 5: Time-dependent Ca2+ amplitude from a single coverslip. Ca2+ amplitudes from different areas in a coverslip were plotted in a time-dependent manner. Non-recommended time periods to measure Ca2+ transient are indicated in the dashed areas (<200 s or >900 s). Please click here to view a larger version of this figure.

Video 1: Representative video showing an example of a high efficiency differentiation. Please click here to download this video.
Video 2: Representative video showing an example of a low efficiency differentiation. Please click here to download this video.
Video 3: Representative video showing an example of a homogeneous monolayer distribution of iPSC-CMs on a glass coverslip. This video shows the recommended cell density to be used for functional analysis. Please click here to download this video.
Video 4: Representative video showing an example of cells from a low efficiency differentiation plated onto a glass coverslip. The cells in the video were obtained from a low efficiency differentiation. Cells from such differentiations are usually not distributed homogeneously across the coverslip and it is difficult to consistently find beating cells. Please click here to download this video.
Discussion
Critical steps for using human iPSC-CMs as experimental models are: 1) generating high-quality cardiomyocytes (CMs) that can ensure the consistent performance and reproducible results; 2) allowing the cells to mature in culture for at least 90 days to adequately assess their phenotype; 3) performing electrophysiological studies, e.g. calcium (Ca2+) transient measurements, to provide a physiologically relevant functional characterization of human iPSC-CMs. We developed a monolayer-based differentiation method that produces high-quality ventricular-like iPSC-CMs. Our method relies on several crucial factors and is a variant of existing protocols14,18. Unlike other protocols, this one uses low oxygen conditions (5% O2) for iPSCs maintenance as well as during the first week of their differentiation into CMs. Low levels of oxygen mimic the environmental condition during embryonic and fetal heart development19. Hypoxic conditions have also been shown to increase iPSCs proliferation and their subsequent differentiation to CMs20. Seeding density and proper iPSCs passaging are also critical factors for a successful cardiac differentiation. High differentiation efficiency is observed when 70−80% confluent iPSCs are dissociated into small cell aggregates using an enzyme-free solution, and seeded at a split ratio of 1:6. Cardiac differentiation can be started when the cells reach a confluency of 70−80%, expected about 4 days after splitting. CHIR99021 (10 μM) treatment at the beginning of the differentiation will cause significant cell death. However, cell proliferation is observed when CHIR99021 is removed from culture the following day. A correct balance between cell death and proliferation is essential for preserving a monolayer culture throughout the differentiation, which is another important factor for a successful differentiation. If the iPSC density is either too low or too high, the subsequent differentiation will be a low efficiency cardiac differentiation (Figure 4A, Video 2). Figure 4A shows such an example of a low efficiency differentiation, where the cardiomyocytes derived from a healthy donor are mixed with other cell types, such as α-smooth muscle actin-positive cells. Importantly, Ca2+ transients recorded from such preparations showed altered characteristics, such as low Ca2+ amplitude and arrhythmic patterns, which could easily be misinterpreted as biological variation. A successful and high efficiency differentiation from the same cell line, instead, generated a pure population of ventricular-like cardiomyocytes (Figure 4A, Video 1) along with regular Ca2+ transients (Figure 4B, top panel). Thus, the quality of the differentiation plays an important role in the overall magnitude, shape, regularity, and frequency of a Ca2+ transient.
Another important aspect of a successful and efficient differentiation is obtaining an enriched population of cardiomyocytes. While undifferentiated stem cells and other iPSC-derived cell types use glucose as an energy source, CMs can use lactate efficiently for ATP and glutamate production21. Thus, in order to obtain an even purer population of CMs, the cells are cultured in lactate-supplemented and glucose-depleted culture medium. Culturing iPSC-CMs in this selecting media for a long time, however, may lead to functional impairment. Therefore, to purify iPSC-CMs and still preserve their function, metabolic selection is performed only for 10 days, from days 80−90 since differentiation induction. After this period, the selection media is replaced, and the cells are maintained in RPMI/B27 media. While the selection protocol can be implemented much earlier22 (e.g., around day 15), it is advisable to wait until day 80, as doing so can prevent residual iPSCs or other cell types from proliferating by the time the cells are ready for functional studies (day 90). It is vital therefore, that for a successful, efficient, and robust cardiac differentiation, all of the above-mentioned factors are taken into consideration.
In addition to the robustness and efficiency of cardiac differentiation protocols, maturation and cell type specification (e.g., atrial and ventricular cell types) also pose a major challenge for using iPSC-CMs to model cardiac disease. Over the last few years, many promising maturation strategies have been reported, including electrical stimulation, mechanical stretch, and substrate stiffness23. However, prolonged in vitro culture of iPSC-CMs continues to be a simple and practical approach to generate adult-like cardiomyocytes24,25.
Consistent with other published reports12, iPSC-CMs generated by this in vitro differentiation protocol show robust expression of ventricle-specific MLC2V and much lower levels of MLC2A at day 90 (Figure 2A,B). While fetal CMs contain both MLC2A and MLC2V isoforms, adult ventricular CMs only contain MLC2V. This switch in MLC isoform prevalence occurs during the neonatal stage26.
Cardiomyocytes generated through this protocol express ventricular specific marker, such as MLC2V, which progressively increases in abundance as the cells are kept in culture longer (Figure 2A,B). This indicates that a prolonged in vitro culture enhances the maturation of CMs that appear to be committed to the ventricular phenotype.
Time in culture also greatly affects Ca2+ handling maturation24,25. Previous reports described that after 20 days of the differentiation induction, there were no major changes in the calc Ca2+ transients as the cells grew older25. However, the cardiomyocytes generated from the protocol detailed here show progressive changes in Ca2+ amplitude and Ca2+ decay over the three time points evaluated (30, 60, and 90 days after induction of cardiac differentiation) (Figure 3A-C). Interestingly, these data show that Ca2+ transient parameters, such as Ca2+ amplitude and Ca2+ decay, measured in day 90 iPSC-CMs were similar to those measured in isolated rat adult cardiomyocytes (ARCMs). Moreover, the 90 days old iPSC-CMs were also able to follow various electrical stimulations ranging from 0.5 Hz to 3 Hz consistently, similar to ARCMs (Figure 4B). In future work, it would be interesting to see how an even longer time in culture affects the functional characteristics of these iPSC-CMs as compared to ARCMs.
Additionally, since β-adrenergic receptor signaling shows a distinct time-dependent maturational pattern after cardiac induction27, the effect of isoproterenol on Ca2+ transient in iPSC-CMs generated through this protocol was tested. Stimulation with isoproterenol led to a positive lusitropic response in day 90 iPSC-CMs, similar to what is observed in ARCMs (Figure 3D-F).
The functional assessments of iPSC-CMs performed in this study have some differences and limitations as compared to isolated adult CMs. The adult CMs have well-developed and well-arranged sarcomeres. This allows for contractility measurements through detection of sarcomere movement. Because none of the current protocols generate fully matured, adult-like iPSC-CMs, contractility measurements through sarcomere detection are not feasible. While it is possible to detect the movement of the cell edges when the cell contracts, it is significantly more challenging to track those movements in a precise manner in iPSC-CMs. As of now, technological advancements are needed to allow for accurate assessments of contractility in these cells. On the other hand, it is possible to measure Ca2+ transients of iPSC-CMs using a Ca2+ indicator like Fura-2. Unlike adult CMs, however, iPSC-CMs are clustered and do not have a well-defined border. Therefore, the recorded Ca2+ transients usually do not represent a single cell, but rather a group of cells in a specific area. While it is possible to adjust the area size, recording Ca2+ transients from a single cell is incredibly challenging. It is critical that when CMs are plated onto the coverslips, the density is such that they achieve a homogenous monolayer distribution (Video 3), which allows for consistently finding areas with cells. If the cells are not distributed as a monolayer on the coverslip (Video 4), there will be areas without cells, and if the coverslip is screened specifically for areas with beating cells to perform the measurements, this can lead to a "selection bias". Instead of selecting a specific area of beating cells, it is recommended to collect data from multiple areas throughout the coverslip, which is only possible when the cells are distributed as a homogenous monolayer. Measurements from 100 such different areas, which take about 10 min, are sufficient to provide reliable functional properties of the cells. Lastly, as shown in Figure 5, iPSC-CMs need time to adjust to the measurement conditions. For reliable measurements, it is recommended that the cells should be stabilized at the measurement conditions for at least 3 min.
Declarações
The authors have nothing to disclose.
Acknowledgements
This research was supported by AHA Scientist Development Grant 17SDG33700093 (F.S.); Mount Sinai KL2 Scholars Award for Clinical and Translational Research Career Development KL2TR001435 (F.S.); NIH R00 HL116645 and AHA 18TPA34170460 (C.K.).
Materials
| Anti-Actin, α-Smooth Muscle antibody, Mouse monoclonal | Sigma Aldrich | A5228 | |
| Alexa Fluor 488 goat anti mouse | Invitrogen | A11001 | |
| Alexa Fluor 555 goat anti rabbit | Invitrogen | A21428 | |
| B27 Supplement | Gibco | 17504-044 | |
| B27(-) insulin Supplement | Gibco | A18956-01 | |
| CHIR-99021 | Selleckchem | S2924 | |
| DAPI nuclear stain | ThermoFisher | D1306 | |
| DMEM/F12 (1:1) (1X) + L- Glutamine + 15mM Hepes | Gibco | 11330-032 | |
| Double Ended Cell lifter, Flat blade and J-Hook | Celltreat | 229306 | |
| Falcon Multiwell Tissue Culture Plate, 6 well | Corning | 353046 | |
| Fluidic inline heater | Live Cell Instrument | IL-H-10 | |
| Fura-2, AM | Invitrogen | F1221 | |
| hESC-qualified matrix | Corning | 354277 | Matrigel Matrix |
| hPSC media | Gibco | A33493-01 | StemFlex Basal Medium |
| IWR-1 | Sigma Aldrich | I0161 | |
| Live cell imaging chamber | Live Cell Instrument | EC-B25 | |
| MLC-2A, Monoclonal Mouse Antibody | Synaptic Systems | 311011 | |
| Myocyte calcium and contractility system | Ionoptix | ISW-400 | |
| Myosin Light Chain 2 Antibody, Rabbit Polyclonal (MLC2V) | Proteintech | 10906-1-AP | |
| Nalgene Rapid Flow Sterile Disposable Filter units with PES Membrane | ThermoFisher | 124-0045 | |
| PBS with Calcium and Magnesium | Corning | 21-030-CV | |
| PBS without Calcium and Magensium | Corning | 21-031-CV | |
| Premium Glass Cover Slips | Lab Scientific | 7807 | |
| RPMI medium 1640 (-) D-glucose (1X) | Gibco | 11879-020 | |
| RPMI medium 1640 (1X) | Gibco | 11875-093 | |
| Sodium L-lactate | Sigma Aldrich | L7022 | |
| StemFlex Supplement | Gibco | A33492-01 | |
| Thiazovivin | Tocris | 3845 | |
| Trypsin-EDTA (0.25%) | ThermoFisher | 25200056 | |
| Tyrode's solution | Boston Bioproducts | BSS-355w | Adjust pH at 7.2. Add 1.2mM Calcium Chloride |
Referências
- Karakikes, I., et al. Correction of human phospholamban R14del mutation associated with cardiomyopathy using targeted nucleases and combination therapy. Nature Communications. 6, 6955 (2015).
- Moretti, A., et al. Patient-specific induced pluripotent stem-cell models for long-QT syndrome. The New England Journal of Medicine. 363 (15), 1397-1409 (2010).
- Davis, R. P., et al. Cardiomyocytes derived from pluripotent stem cells recapitulate electrophysiological characteristics of an overlap syndrome of cardiac sodium channel disease. Circulation. 125 (25), 3079-3091 (2012).
- Fatima, A., et al. In vitro modeling of ryanodine receptor 2 dysfunction using human induced pluripotent stem cells. Cellular Physiology and Biochemistry: International Journal of Experimental Cellular Physiology, Biochemistry, and Pharmacology. 28 (4), 579-592 (2011).
- Novak, A., et al. Cardiomyocytes generated from CPVTD307H patients are arrhythmogenic in response to beta-adrenergic stimulation. Journal of Cellular and Molecular Medicine. 16 (3), 468-482 (2012).
- Lan, F., et al. Abnormal calcium handling properties underlie familial hypertrophic cardiomyopathy pathology in patient-specific induced pluripotent stem cells. Cell Stem Cell. 12 (1), 101-113 (2013).
- Sun, N., et al. Patient-specific induced pluripotent stem cells as a model for familial dilated cardiomyopathy. Science Translational Medicine. 4 (130), (2012).
- Stillitano, F., et al. Modeling susceptibility to drug-induced long QT with a panel of subject-specific induced pluripotent stem cells. eLife. 6, (2017).
- Matsa, E., Burridge, P. W., Wu, J. C. Human stem cells for modeling heart disease and for drug discovery. Science Translational Medicine. 6 (239), (2014).
- Mordwinkin, N. M., Lee, A. S., Wu, J. C. Patient-specific stem cells and cardiovascular drug discovery. Journal of the American Medical Association. 310 (19), 2039-2040 (2013).
- Youssef, A. A., et al. The Promise and Challenge of Induced Pluripotent Stem Cells for Cardiovascular Applications. Journal of the American College of Cardiology: Basic to Translational Science. 1 (6), 510-523 (2016).
- Cyganek, L., et al. Deep phenotyping of human induced pluripotent stem cell-derived atrial and ventricular cardiomyocytes. Journal of Clinical Investigation: Insight. 3, 12 (2018).
- Keung, W., Boheler, K. R., Li, R. A. Developmental cues for the maturation of metabolic, electrophysiological and calcium handling properties of human pluripotent stem cell-derived cardiomyocytes. Stem Cell Research, Therapy. 5 (1), 17 (2014).
- Bhattacharya, S., et al. High efficiency differentiation of human pluripotent stem cells to cardiomyocytes and characterization by flow cytometry. Journal of Visualized Experiments: JoVE. (91), e52010 (2014).
- Ronaldson-Bouchard, K., et al. Advanced maturation of human cardiac tissue grown from pluripotent stem cells. Nature. 556 (7700), 239-243 (2018).
- Gorski, P. A., et al. Measuring Cardiomyocyte Contractility and Calcium Handling In Vitro. Methods in Molecular Biology. 1816, 93-104 (2018).
- Kim, C., et al. Non-cardiomyocytes influence the electrophysiological maturation of human embryonic stem cell-derived cardiomyocytes during differentiation. Stem Cells and Development. 19 (6), 783-795 (2010).
- Lian, X., et al. Directed cardiomyocyte differentiation from human pluripotent stem cells by modulating Wnt/beta-catenin signaling under fully defined conditions. Nature Protocols. 8 (1), 162-175 (2013).
- Patterson, A. J., Zhang, L. Hypoxia and fetal heart development. Current Molecular Medicine. 10 (7), 653-666 (2010).
- Correia, C., et al. Combining hypoxia and bioreactor hydrodynamics boosts induced pluripotent stem cell differentiation towards cardiomyocytes. Stem Cell Reviews. 10 (6), 786-801 (2014).
- Tohyama, S., et al. Glutamine Oxidation Is Indispensable for Survival of Human Pluripotent Stem Cells. Cell Metabolism. 23 (4), 663-674 (2016).
- Tohyama, S., et al. Distinct metabolic flow enables large-scale purification of mouse and human pluripotent stem cell-derived cardiomyocytes. Cell Stem Cell. 12 (1), 127-137 (2013).
- Tu, C., Chao, B. S., Wu, J. C. Strategies for Improving the Maturity of Human Induced Pluripotent Stem Cell-Derived Cardiomyocytes. Circulation Research. 123 (5), 512-514 (2018).
- Lundy, S. D., Zhu, W. Z., Regnier, M., Laflamme, M. A. Structural and functional maturation of cardiomyocytes derived from human pluripotent stem cells. Stem Cells and Development. 22 (14), 1991-2002 (2013).
- Hwang, H. S., et al. Comparable calcium handling of human iPSC-derived cardiomyocytes generated by multiple laboratories. Journal of Molecular and Cellular Cardiology. 85, 79-88 (2015).
- Scuderi, G. J., Butcher, J. Naturally Engineered Maturation of Cardiomyocytes. Frontiers in Cell and Developmental Biology. 5, 50 (2017).
- Jung, G., et al. Time-dependent evolution of functional vs. remodeling signaling in induced pluripotent stem cell-derived cardiomyocytes and induced maturation with biomechanical stimulation. FASEB Journal : Official Publication of the Federation of American Societies for Experimental Biology. 30 (4), 1464-1479 (2016).

