Application of Atomic Force Microscopy to Detect Early Osteoarthritis
Summary
We present a method to investigate early osteoarthritic changes at the cellular level in articular cartilage by using atomic force microscopy (AFM).
Abstract
Biomechanical properties of cells and tissues not only regulate their shape and function but are also crucial for maintaining their vitality. Changes in elasticity can propagate or trigger the onset of major diseases like cancer or osteoarthritis (OA). Atomic force microscopy (AFM) has emerged as a strong tool to qualitatively and quantitatively characterize the biomechanical properties of specific biological target structures on a microscopic scale, measuring forces in a range from as small as the piconewton to the micronewton. Biomechanical properties are of special importance in musculoskeletal tissues, which are subjected to high levels of strain. OA as a degenerative disease of the cartilage results in the disruption of the pericellular matrix (PCM) and the spatial rearrangement of the chondrocytes embedded in their extracellular matrix (ECM). Disruption in PCM and ECM has been associated with changes in the biomechanical properties of cartilage. In the present study we used AFM to quantify these changes in relation to the specific spatial pattern changes of the chondrocytes. With each pattern change, significant changes in elasticity were observed for both the PCM and ECM. Measuring the local elasticity thus allows for drawing direct conclusions about the degree of local tissue degeneration in OA.
Introduction
Articular cartilage is an avascular, aneural tissue. Sparsely scattered chondrocytes produce, organize, and maintain an expansive extracellular matrix (ECM) into which they are embedded. As a distinct and specialized part of the ECM, chondrocytes are surrounded by a thin layer of specialized matrix known as the pericellular matrix (PCM). The PCM acts as a mechanosensitive cell-matrix interface1 that protects the chondrocytes2 and modulates their biosynthetic response3. As previously described4, in healthy cartilage, chondrocytes are arranged in specific, distinct spatial patterns that are specific for each tissue layer and joint4,5 and depend on joint-specific mechanical loading mechanisms6. These patterns change from pairs and strings in healthy cartilage to double strings with the onset of osteoarthritis (OA). With further progression of the disease the chondrocytes form small clusters, increasing gradually in size to big clusters in advanced OA. A complete loss of any organizational structure and induction of apoptosis is observed in end stage OA. Thus, chondrocyte cellular arrangement can be used as an image-based biomarker for OA progression4.
Biomechanical properties of cells and tissues not only regulate their shape and function but are also crucial for maintaining their vitality. Changes in elasticity can propagate or trigger the onset of major diseases like cancer or OA. Atomic force microscopy (AFM) has emerged as a powerful tool to qualitatively and quantitatively characterize the biomechanical properties of specific biological target structures on a microscopic scale, measuring a wide range of force, from piconewton to the micronewton. The major application of AFM is to measure the surface topography and mechanical properties of samples at subnanometer resolution7. The measurement device consists of three main components: 1) An AFM probe, which is a sharp tip mounted on a cantilever and is used for the direct interaction with the surface of the sample. When force is applied to the cantilever, deformation of the latter occurs according to the measured tissue's properties. 2) An optical system that projects a laser beam onto the cantilever, which is then reflected to a detector unit. 3) A photodiode detector that catches the light deflected from the cantilever. It converts the received information regarding the laser deflection by the cantilever into a force curve that can be analyzed.
Thus, the main principle of AFM is the detection of the force acting between the AFM probe and the target structure of the sample. The force curves obtained describe the mechanical properties of the target structures on the sample surface like elasticity, charge distribution, magnetization, yield stress, and elastic plastic deformation dynamics8. An important advantage of AFM over other imaging techniques is that AFM can be used to measure the mechanical properties of live cells in medium or tissues in a native state without damaging the tissue. AFM can operate both in liquid or dry conditions. There is no requirement for sample preparation. AFM provides the possibility to image a specimen and measure its mechanical properties simultaneously in specimens that are near physiological conditions. In the present study we describe a novel approach to assess OA progression by measuring the elasticity of the PCM and ECM in native articular cartilage. The correlation of spatial organization of chondrocytes with the degree of local tissue degeneration provides a completely new perspective for early detection of OA. The functional relevance of these patterns has not been evaluated so far, however. Because the major function of articular cartilage is load bearing at low friction, the tissue must possess elastic properties. AFM allows measuring not only the elasticity of the ECM but also of the spatial cellular patterns embedded into their PCM. The observed correlation of elasticity with spatial pattern change of the chondrocytes is so strong that measuring elasticity alone may allow stratification of local tissue degeneration.
Elastic moduli of the PCM and ECM were assessed in 35 µm-thin sections using an AFM system integrated into an inverted phase contrast microscope that allowed simultaneous visualization of the cartilage sample. This protocol is based on a study already published from our laboratory9 and specifically describes how to characterize the spatial arrangement of the chondrocytes and how to measure the elasticity of their associated PCM and ECM. With each pattern change of the chondrocytes, significant changes in elasticity can also be observed for both the PCM and ECM, allowing this technique to be used to directly measure the stage of degeneration of the cartilage.
This validated approach opens up a new way to evaluate OA progression and therapeutic effects at early stages before macroscopic tissue degradation actually starts to appear. Performing AFM measurements consistently is an arduous process. In the following protocol we describe how to prepare the sample to be measured by AFM, how to perform the actual AFM measurements starting with preparation of the cantilever, how to calibrate the AFM, and then how to perform the measurements. Step-by-step instructions give a clear and concise approach to obtain reliable data and provide basic strategies for processing and interpreting it. The discussion section also describes the most common pitfalls of this rigorous method and provides helpful troubleshooting tips.
Protocol
The human cartilage samples were obtained from patients undergoing total knee arthroplasty in the Department of Orthopaedic Surgery of the University Hospital of Tuebingen, Germany, and the Winghofer-hospital, Rottenburg a.N., Germany, for end-stage OA of the knee. Full departmental, institutional, and local ethical committee approval were obtained before commencement of the study (project number 674/2016BO2). Written informed consent was received from all patients before participation. The methods were carried out in accordance with the approved guidelines.
1. Sample preparation
- Preparing the cartilage for cryotome sectioning
- To evaluate degenerative changes in the knee joint, take articular cartilage samples obtained from the load-bearing zone of the femoral condyles after tissue resection from the patients.
NOTE: Samples that are investigated should still contain at least a thin layer of subchondral bone to allow clear identification of the tissue orientation with respect to the inner and outer surface, thus also allowing a standardized tissue harvest of the topmost cartilage layer. The cartilage can be used for AFM measurements until 24 h after surgery. The tissue can be stored in serum-free Dulbecco's modified Eagle's medium (DMEM) with 2% (v/v) penicillin-streptomycin and 1.2% (v/v) amphotericin B prior to use. Storing the samples for more than 24 h can cause artifacts in the AFM measurements due to tissue swelling, however. - Cut the articular cartilage as a whole from the subchondral bone with the help of a scalpel and afterwards embed the cartilage sample in water-soluble embedding medium on the cryotome knob that freezes under low temperature in the cryotome device.
NOTE: The frozen medium stabilizes the tissue it envelops. It also results in the freezing of the cartilage sample along with the embedding medium. When cutting the cartilage from the bone, track the orientation of the tissue. The tissue embedded for the cryosections must be placed in such a way that the top layer (i.e., articular surface) of the cartilage faces the blade. Note that the tissue has to be fully covered by the embedding medium.
- To evaluate degenerative changes in the knee joint, take articular cartilage samples obtained from the load-bearing zone of the femoral condyles after tissue resection from the patients.
- Cryotome sectioning of the cartilage
- Using a standard cryotome, section the tissue at a thickness of 35 µm from the topmost layer (i.e., articular surface) of the articular cartilage that is embedded in the frozen water-soluble embedding medium.
NOTE: In total, up to 300 µm (which corresponds to about nine sections) can be sectioned and used for the analyses. The proposed limit of 300 µm corresponds to the visual penetration depth that can usually be obtained with fluorescence microscopy in hyaline cartilage such as articular cartilage. Thus, it is possible to sort cartilage according to the predominant local spatial pattern and then measure these patterns in the corresponding sections. - Collect the sections on a glass slide and rinse the sections 3x with phosphate-buffered saline (PBS) to remove the water-soluble embedding medium.
- Using a standard cryotome, section the tissue at a thickness of 35 µm from the topmost layer (i.e., articular surface) of the articular cartilage that is embedded in the frozen water-soluble embedding medium.
- Gluing of cartilage sections onto an AFM compatible Petri dish
NOTE: Because the AFM gathers data by mechanical indentation on the sample, the sections need to be fixed in place to allow for precise measurements.- Gently glue the 35 µm thick sections with biocompatible sample glue onto tissue culture dishes that are compatible with the AFM device. To do this, take 2−3 drops of the glue and put them on the tissue culture dish at the location where the edge of the section will end up. Now put the cartilage section on the glue and allow it to stick firmly to the surface of the tissue culture dish.
NOTE: The 35 µm thick sections of hyaline articular cartilage are not very prone to curling. However, if curling occurs when removing the samples from the buffer medium, make sure that as little fluid as possible sticks to the samples. As soon as the excess water has dried up, directly spread the tissue on the dispersed spots of glue so that it is fixed to the surface of the cell culture dish. The glue is applied in a thin layer only at the edges of the sample and not on the entire section. Measure only that portion of the sample that is far from the glued area to eliminate the possibility of the glue interfering with the measurements. - Incubate the sections at room temperature (RT) for 2 min to allow the glue and tissue to bond properly. Then cover the sections with Leibovitz's L-15 medium without L-glutamine and without Sodium bicarbonate so that they are fully submerged.
NOTE: Sudden movements of the sample during fixation or insufficient glue application are common mistakes that result in the tissue’s detachment from the culture dish. Furthermore, applying the Leibowitz medium should be done in a gentle way, so as not to detach the sample from the surface due to "waves" created by the medium. - Place the Petri dish with the glued sections in the standard cell culture incubator at 37 °C until measurements are performed.
NOTE: Make sure that the tissue sections are stable and do not detach from the surface of the tissue culture dish by performing each step slowly.
- Gently glue the 35 µm thick sections with biocompatible sample glue onto tissue culture dishes that are compatible with the AFM device. To do this, take 2−3 drops of the glue and put them on the tissue culture dish at the location where the edge of the section will end up. Now put the cartilage section on the glue and allow it to stick firmly to the surface of the tissue culture dish.
2. Cantilever preparation (gluing the microspheres)
- Preparing the AFM device and diluting the microspheres
- Place the cantilever on the glass block specified for air measurements, fix it with the spring, and mount the glass block on the AFM head.
- Dilute the microspheres in 100% ethanol (100 particles/10 µL) and ultrasonicate for 10 s, so that the microspheres are separated and do not clump together.
- Preparing the setup for gluing
- Clean a glass slide with 70% ethanol and mount it on the sample holder on the AFM device.
NOTE: The cleansing of the slide prevents specks of dust that might interfere with the gluing process from accumulating on the slide's surface. - Place 2 µL of the microsphere suspension in the middle of the slide and 2 µL of glue near the microsphere suspension.
NOTE: Placing the microsphere suspension and glue close to each other is recommended to allow for a quick transition of the cantilever between the glue and the microspheres. If the transition between dipping the cantilever into the glue and making contact with a microsphere is too long, the glue will eventually harden, thereby losing its adhesive properties. - Let the ethanol liquid in the microsphere suspension air-dry, leaving the microspheres in place.
- Clean a glass slide with 70% ethanol and mount it on the sample holder on the AFM device.
- Gluing the microspheres on the cantilever tip
- Dip the cantilever manually into the glue with the help of the stepper motor in 10 µm steps until the tip is covered by glue. Perform this step swiftly, as the glue dries fast.
- Retract the cantilever again by 100 µm, move it towards the microspheres, and position the cantilever tip directly above a single microsphere.
- Run a force spectroscopy measurement to dip the cantilever tip onto the selected microsphere with the parameters shown in Table 1.
NOTE: By executing this step, a measurement of the microsphere's surface is used to establish contact between the cantilever's tip and the microsphere. The relatively long contact time shown in Table 1 allows for ample bonding time between the glue and microsphere. The force-distance curve obtained during this step is generated as a by-product of gluing the microsphere on the cantilever tip and is not processed for further analysis. - Once a microsphere is attached to the cantilever, retract the glass block with the cantilever mounted on top and then remove the AFM head from the microscope. Lastly, unmount the glass block and cantilever from the AFM head.
- Incubate the cantilever at 65 °C for 2 h and let it dry overnight at RT before starting the measurements.
NOTE: Incubation of the cantilever enhances the glue's adhesive properties, rendering the cantilever-microsphere-complex more stable.
3. Preparing the AFM device for measurements
- Adjust a glass block specified for measuring in a liquid environment on the AFM holder so that the upper surface is straight and parallel to the AFM holder.
- With the help of tweezers, carefully mount the selected cantilever on the surface of the glass block, so that the AFM tip with the microsphere protrudes over the polished optical plane.
NOTE: The cantilever's tip needs to protrude over the polished plane in order to be able to reflect the AFM's laser onto the photodetector. Be very careful when placing the cantilever on the AFM so as not to scratch the polished optical surface of the glass block. Scratching the glass block may result in difficulties in adjusting the laser alignment and may interfere with the subsequent measurements. - Because the cantilever will be subject to mechanical pressure, it needs to be fixed in place. Stabilize the cantilever on the glass block by sliding the metallic spring into the groove of the block and clamping the top of the cantilever with the spring with the help of tweezers.
- Carefully place the glass block with the cantilever on the AFM head and secure it with the integrated locking mechanism. Make sure that the spring is facing to the left side so that the cantilever is placed in the correct orientation. Then mount the AFM head with the cantilever on the AFM device.
4. Loading the sample and calibration of the cantilever
NOTE: Here, calibration of the device is performed by running a force curve on the clean surface of the Petri dish filled with Leibovitz’s medium without any sample tissue. Calibration can also be performed by using a separate control AFM dish filled only with the AFM medium without the sample.
- Mounting the sample on the AFM device
- Place the Petri dish prepared in step 1.3.3 onto the AFM sample holder.
- Turn on the Petri dish heater and set it at 37 °C. Allow the tissue culture dish to reach optimal temperature (i.e., 20 min).
- Place a protecting foil on the base of the cantilever assembly to avoid the condensation of medium in the AFM head.
- Calibration of the device
- Open the software (Table of Materials), followed by the laser alignment window, and the window depicting the approach parameter settings. Then turn on the stepper motor, the laser light, and the CCD-camera.
- Use the CCD-camera on the microscope to visualize and identify the cantilever. Using the stepper motor function, lower the cantilever in 100 µm steps until the cantilever is fully submerged in the medium.
NOTE: The submerging of the cantilever in the liquid is visually identifiable via the CCD-camera. When it breaks the water's surface, the cantilever tip creates easily noticeable circular reflections in the water. - Direct the laser on top of the cantilever using the adjustment screws.
NOTE: Do not overwind the screws, as this will damage the laser alignment mechanism. The cantilever is viewed from below and the laser beam comes from above. - Once the laser is positioned on the cantilever, adjust the laser beam with the help of screws in the AFM device so that the reflected beam falls onto the center of the photodetector. Keep monitoring the position of the laser beam using the laser alignment function.
NOTE: The detector picks up the deflection of the laser beam reflected by the cantilever and converts it into an electrical signal. - After laser-cantilever adjustment, the Sum signal has to be 1 V or above while the lateral and vertical deflections have to be close to 0. If these values are not obtained, adjust the photodetector.
- Obtaining the calibration force curve
- In order to reach the surface on the AFM dish to start the measurements, run a scanner Approach with the approach parameters given in Table 2. Once the bottom of the tissue culture dish is reached, retract the cantilever by 100 µm.
NOTE: At this point the probe is exactly 100 µm above from the bottom of the tissue culture dish. - Set up the RUN parameters and adjust the parameters displayed in Table 3. Click the RUN button to start a measurement and obtain a calibration force-distance curve.
NOTE: The force curve obtained here is shown in Figure 1. - On the calibration force-distance curve, select the region for a linear fit of the retracted curve in the software.
NOTE: The linear fit is done to measure the cantilever’s sensitivity. Once the linear fit is in place, the values will be saved by the software. The spring constant measurement or the thermal noise of the cantilever is calculated by the software afterwards. - As the measurements are performed in medium at a temperature of 37 °C, set the temperature variable at 37 °C in the software to mimic physiological conditions as closely as possible.
NOTE: By the end of the calibration, the vertical deflection is saved and displayed in the unit of force, the Newton (N), instead of volts (V), which is the unit of the original registration by the photodiode detector.
- In order to reach the surface on the AFM dish to start the measurements, run a scanner Approach with the approach parameters given in Table 2. Once the bottom of the tissue culture dish is reached, retract the cantilever by 100 µm.
5. Biomechanical characterization of the ECM and PCM by performing elasticity measurements via AFM
- Identifying the chondrocyte patterns in the cartilage section
- Observe the cartilage section under a phase contrast microscope integrated in the AFM system and identify the specific cellular patterns10 of articular cartilage from the osteoarthritic knee: single strings (healthy tissue areas), double strings (beginning tissue degeneration), small and big clusters (both advanced tissue degeneration). See Figure 2A–D.
- Once the specific desired pattern is identified, measure two sites per chosen pattern per matrix type (PCM or ECM) and perform nine measurement repetitions on each measurement site (Figure 2E,F). Ensure a sample size large enough to account for possible inaccuracies.
NOTE: For the PCM measurements, place the cantilever in close proximity to the cells (as shown by the red circles in Figure 2E,F). In order to conduct measurements of the ECM, select a region without any cells (shown by the blue area in Figure 2E,F) and perform the indentation as described in step 5.2.
- Indentation of the targeted site
- Perform an Approach followed by a retraction so that the cantilever is positioned 100 µm above the tissue, which will be the starting position for the subsequent measurements.
- Focus on the PCM or ECM of the pattern to be measured and fix the computer mouse at that point.
NOTE: The mouse will serve as a visual marker for the target site of indentation. Once the focus shifts to the cantilever tip, the tissue structures will become indistinguishable or blurred. - Now focus on the probe and move the tip of the probe to the point previously fixed by the computer arrow. Next, start the measurements with RUN, using the set point parameter obtained by calibration of the cantilever.
NOTE: An exemplary force curve obtained from measuring the PCM of a single string is shown in Supplemental Figure 1.
6. Data processing
NOTE: The data analysis or determination of the elastic modulus is performed using a Hertz model as described previously11,12. The indenter’s shape was spherical due to the usage of microspheres on the tip and the Poisson’s ratio was kept at 0.5 based on previous literature13,14,15.
- Open the data processing software (Table of Materials) compatible with the data obtained from the AFM device.
- Select the Hertz model in the software for processing the force-distance curves. Set the Poisson ratio at 0.5 and select the tip shape as spherical and a tip radius of 12.5 µm.
- Once all the parameters are adjusted, the results are fitted, and the Young’s modulus is calculated and displayed by the software.
Representative Results
Along the physiopathological model from strings to double strings, to small and finally to big clusters, both ECM (Figure 3A) and PCM (Figure 3B) elastic moduli decreased significantly between each pattern change. The only exception was the difference in ECM between strings and double strings (p = 0.072). The results show that the ECM/PCM ratio (Figure 4B) did not change significantly, whereas a marked decrease in the absolute differences in elasticity between ECM and PCM was observed (Figure 4A). Furthermore, the results do not show any significant association concerning the ECM/PCM ratio or associated cellular spatial changes (r = -0.099, p = 0.281).
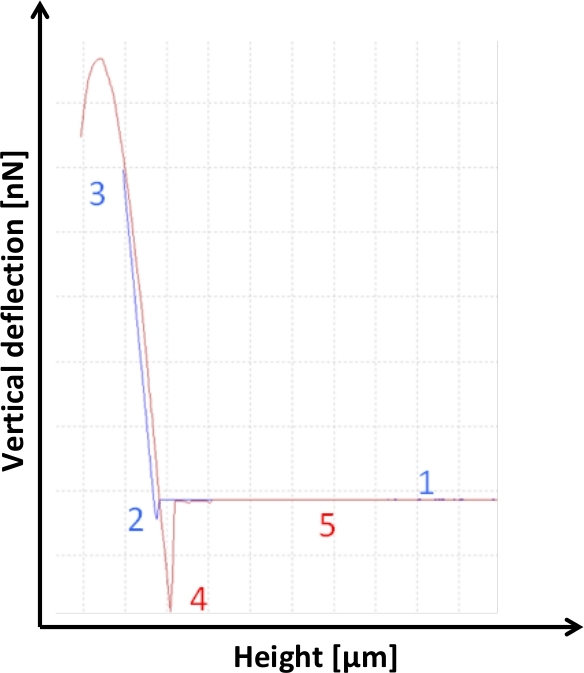
Figure 1: Schematic representation of a force-distance curve in the AFM contact mode. As the probe approaches the surface, the forces are too small to give a measurable deflection of the tip at first, thus leaving the tip in its undisturbed position (1). Then, when the cantilever is very close to the sample, due to adhesive forces active between the tip and the probe, the cantilever actually quickly snaps towards the sample (2). With the probe further approaching the sample, the repulsive deflection then faces against the movement of direction, with an almost linear function of height and deflection until the vertical deflection reaches the relative set point value (3). When retracting (4), in addition to the lowering deflecting forces, adhesion forces are also present while the cantilever is retracted in the Z-axis from the sample. As the AFM probe is pulled off the contact with the sample, it first gets “stuck” before it is able to loosen from the adhesion at the interface, even leading to a short negative deflection of the cantilever, before again reaching its unbent neutral position without contact with the probe (5). The extent of deflection is expressed in the force working on the cantilever expressed in nanonewtons. Please click here to view a larger version of this figure.
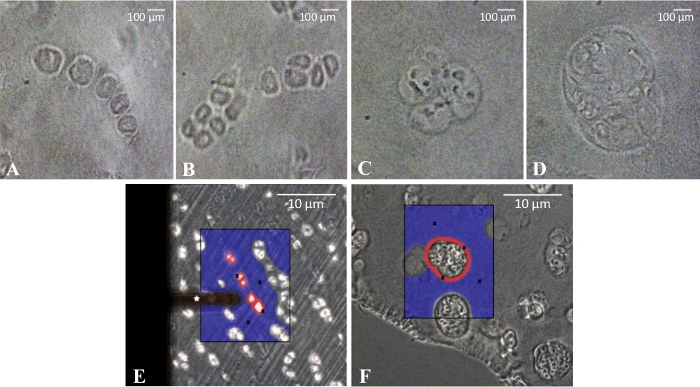
Figure 2: Representative spatial characterization of chondrocytes and AFM measurements of the extracellular matrix (ECM) and pericellular matrix (PCM). (A-D) Characterization of the cellular patterns: strings (A), double strings (B), small clusters (C), and big clusters (D). The elastic moduli of the PCM (red circles) and ECM (blue area) (E/F) were assessed for the different cellular patterns in osteoarthritic cartilage. Measurement sites for the ECM and PCM were selected by the experimenter and are graphically indicated by black crosses. The cantilever tip used for the measurements is marked by a white star. Scale bars represent 10 µm (A-D), and 100 µm (E/F). The figure is adapted and modified from Danalache et al.9. Please click here to view a larger version of this figure.
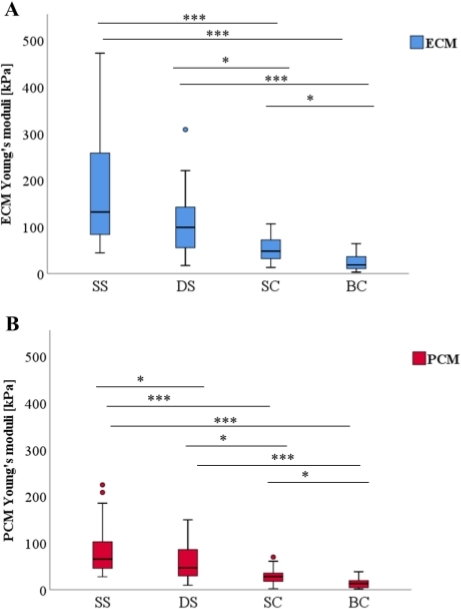
Figure 3: Comparison of the quantified Young’s moduli of the extracellular matrix (ECM) and the pericellular matrix (PCM) as a function of spatial chondrocyte organization. With progressive pathological spatial chondrocyte organization, a gradual decrease of elasticity was noted in the boxplots for both the ECM (A) and the PCM (B) (*p < 0.05, ***p < 0.001). Abbreviations: SS: single strings, DS: double strings, SC: small clusters, BC: big clusters. The figures are taken from Danalache et al.9. Please click here to view a larger version of this figure.
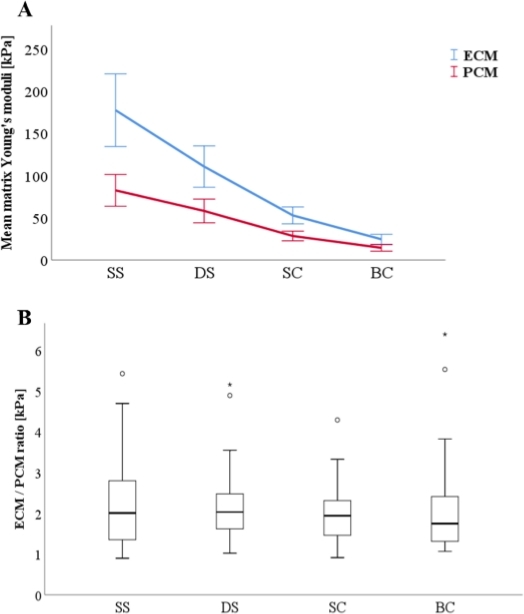
Figure 4: Relationship of the Young’s moduli of the extracellular matrix (ECM) and the pericellular matrix (PCM) as a function of cellular spatial organization. An increasingly pathological spatial chondrocyte organization was associated with a decrease of the Young’s moduli for both ECM and PCM (A). While these spatial changes took place, the ratio of ECM and PCM elasticity remained constant, showing no significant changes (B). The data are presented as a line diagram with mean ± standard error (A) and boxplots (B). Abbreviations: SS: single strings, DS: double strings, SC: small clusters, BC: big clusters. The figures are taken from Danalache et al.9. Please click here to view a larger version of this figure.
Supplemental Figure 1: Representative force curve obtained by indentation of the pericellular matrix (PCM) of a single string pattern showing the fit results (orange arrow) as well as the residual root mean square (residual RMS; black arrow). The fit results include the contact point between sample and tip, the Young’s Modulus, and the baseline. The residual RMS displayed below describes the difference between the fit and the force data, thereby representing the quality of a force curve fit. Please click here to download this figure.
| Table 1. Parameters for gluing a microsphere on the AFM-probe | |
| Parameters | Value |
| Setpoint | 5.0 V |
| Adjust baseline | 1 |
| Pulling length | 90.0 µm |
| Z movement | Constant speed |
| Extend speed | 5.0 µm/s |
| Extend time | 18.0 s |
| Contact time | 90.0 s |
| Delay mode | Constant force |
| Sample rate | 2000 Hz |
Table 1. Parameters for gluing a microsphere on the AFM-probe.
| Table 2. Approach parameters | |
| Approach Parameters | Value |
| Approach IGain | 5.0 Hz |
| Approach PGain | 0.0002 |
| Approach target height | 10.0 µm |
| Approach setpoint | 5.00 V |
| Approach baseline | 0.00 V |
Table 2. Approach parameters.
| Table 3. Run parameters | |
| Parameters | Value |
| Setpoint | 1.0 V |
| Adjust baseline | 1 |
| Pulling length | 90.0 µm |
| Z movement | Constant speed |
| Extend speed | 5.0 µm/s |
| Extend time | 18.0 s |
| Contact time | 0.0 s |
| Delay mode | Constant force |
| Sample rate | 2000 Hz |
Table 3. Run parameters.
Discussion
Using AFM as a novel and powerful technique to measure the biomechanical properties of biological materials at a nanoscale level, we measured the elastic properties of the ECM and PCM in human osteoarthritic articular cartilage. Cartilage samples were selected according to their predominant spatial pattern of chondrocyte organization as an image-based biomarker for local tissue degeneration. As expected, a strong decline in the values of elasticity of both ECM and PCM was observed along spatial chondrocyte reorganization. These observations clearly highlight that the deviations in spatial arrangement of chondrocytes were not only associated with changes in the elastic properties of the cellular microenvironment (PCM), but also throughout the entire cartilage (ECM). Furthermore, the ECM/PCM ratio did not show any significant changes in spite of the robust changes in the elastic moduli of PCM and ECM during OA. These findings indicate that the changes in the mechanical properties of the ECM and the PCM occurred unidirectionally and at the same time, which might mean that the nature of progressive destruction is similar for both the PCM and ECM. OA initiation and progression thus triggers significant PCM and ECM degradation and ultimately destruction. Both losses were associated with a significant loss of biomechanical properties of the articular cartilage such as its elasticity, as shown in the present study. This emphasizes the functional relevance of the spatial organization of chondrocytes as a marker for biomechanical properties. Conversely, it allows us to use local elasticity measurements to draw conclusions about the locally predominant spatial patterns and thus the local tissue degeneration of the cartilage.
Atomic force microscopy (AFM) has emerged as a high-resolution tool to study tissues in a nondestructive way. It operates by physically probing samples with a delicate and pliable cantilever that reflects a laser onto a photodiode. Any changes in this reflection are registered and converted into an electrical signal. While AFM is a powerful tool to conduct nanoscale measurements, it does not come without its limitations and pitfalls. Especially critical is the cantilever preparation by gluing the microspheres. In the context of this method, microspheres are used to modify the indentation depth and local pressure during measurements. Using small microspheres attached to the tip of the probe allows the measurement of the biomechanical properties of a fiber network rather than the elasticity of single fiber when using the tip alone. It also prevents tissue damage during the measurement process. Due to the delicate nature of the cantilever sensors, an attentive and careful mode of preparation needs to be established in order to obtain consistent and accurate measurements. In order to prevent the microspheres from detaching from the sensor’s tip, we recommend freshly mixed glue not older than one week. Furthermore, it is vital for the sensor’s functionality to place the microsphere at the tip’s center as lateral deviation in microsphere attachment easily results in inconsistent measurements.
Placing the cantilever on the glass block and fixing it with a fitting spring is a delicate and error-prone process that needs meticulous attention and steady hands. Because cantilevers are very likely to be destroyed by untrained operators, we recommend conducting several test runs and practice to comfortably handle the AFM’s easily breakable components.
A clean glass block is imperative to properly calibrate the device and obtain reliable measurements. Dirt or dust on the block’s optical surface may prevent proper laser alignment on the photodetector. Therefore, if problems during the laser alignment are encountered, washing off the glass block with ethanol a second time might be necessary.
Data analysis or determination of the elastic modulus can be performed using the Hertz model as described previously11,12. In short, the data generated by indentation are plots of force over movement of the cantilever tip. During the measurements, the cantilever is moved in the direction of the sample. This leads to the cantilever making contact with the sample and subsequently to its bending in the direction opposite to the one in which it originally moved. Simultaneously, an indentation of the sample by a certain amount occurs. In order to use the Hertz-fit model, the sample’s indentation has to be calculated and adjusted to isolate the cantilever bending parameter. The parameter describing the sample is Poisson’s ratio, which depends on the material investigated. For soft biological samples, Poisson’s ratio is often set to 0.515. As mentioned above, the shape of the used indenter is relevant to the calculation of Young’s modulus, as it dictates the extensions that have to be made to the original Hertz equation. In case of the described experiment, a spherical indenter shape is assumed due to the usage of microspheres.
While AFM may offer new and interesting possibilities of gathering data, the consistency and reliability of the yielded data strongly depend on the experience of the respective operator. Several of the steps outlined above are prone to human error and require patience and meticulousness to execute them properly.
Due to the many sensitive variables that can affect measurement results, the absolute force values reported in this study cannot be generalized but are rather specific for our experimental setup. When using this technique to evaluate the tissue degeneration of cartilage, some normalizing measurements on different spatial patterns first need to be performed to scale the results to the specific experimental measurement settings present. The relationship of the different elasticity moduli and the spatial patterns will, however, not be affected.
Declarações
The authors have nothing to disclose.
Acknowledgements
We thank our co-authors from the original publication for their help and support.
Materials
| Amphotericin B | Merck | A2942 | |
| Atomic Force Microscope (AFM) | CellHesion 200, JPK Instruments, Berlin, Germany | JPK00518 | |
| AFM head | (CellHesion 200) JPK | JPK00518 | |
| Biocompatible sample glue | JPK Instruments AG, Berlin, Germany | H000033 | |
| Cantilever | tip C, k ¼ 7.4 N/m, All-In-One-AleTl, Budget Sensors, Sofia, Bulgaria | AIO-TL-10 | |
| Dulbecco's modified Eagle's medium (DMEM) | Gibco, Life Technologies, Darmstadt, Germany | 41966052 | |
| Inverted phase contrast microscope (Integrated with AFM) | AxioObserver D1, Carl Zeiss Microscopy, Jena, Germany | L201306_03 | |
| Leibovitz's L-15 medium without L-glutamine | (Merck KGaA, Darmstadt, Germany) | F1315 | |
| Microspheres | Polysciences | 07313-5 | |
| Penicillin-Streptomycin | Sigma | P4333 | |
| Petri dish heater associated with AFM | JPK Instruments AG, Berlin, Germany | T-05-0117 | |
| Scalpel | Feather | 2023-01 | |
| Tissue culture dishes | TPP Techno Plastic Products AG, Trasadingen, Switzerland | TPP93040 | |
| Tissue-tek O.C.T. Compound | Sakura Finetek, Alphen aan den Rijn, Netherlands | SA6255012 |
Referências
- Guilak, F., et al. The pericellular matrix as a transducer of biomechanical and biochemical signals in articular cartilage. Annals of the New York Academy of Sciences. 1068, 498-512 (2006).
- Peters, H. C., et al. The protective role of the pericellular matrix in chondrocyte apoptosis. Tissue Engineering Part A. 17 (15-16), 2017-2024 (2011).
- Larson, C. M., Kelley, S. S., Blackwood, A. D., Banes, A. J., Lee, G. M. Retention of the native chondrocyte pericellular matrix results in significantly improved matrix production. Matrix Biology. 21 (4), 349-359 (2002).
- Rolauffs, B., Williams, J. M., Grodzinsky, A. J., Kuettner, K. E., Cole, A. A. Distinct horizontal patterns in the spatial organization of superficial zone chondrocytes of human joints. Journal of Structural Biology. 162 (2), 335-344 (2008).
- Schumacher, B. L., Su, J. L., Lindley, K. M., Kuettner, K. E., Cole, A. A. Horizontally oriented clusters of multiple chondrons in the superficial zone of ankle, but not knee articular cartilage. The Anatomical Record. 266 (4), 241-248 (2002).
- Rolauffs, B., et al. Onset of preclinical osteoarthritis: the angular spatial organization permits early diagnosis. Arthritis Rheumatology. 63 (6), 1637-1647 (2011).
- Maver, U., Velnar, T., Gaberšček, M., Planinšek, O., Finšgar, M. Recent progressive use of atomic force microscopy in biomedical applications. TrAC Trends in Analytical Chemistry. 80, 96-111 (2016).
- Polini, A., Yang, F., Ramalingam, M., Ramakrishna, S. Physicochemical characterization of nanofiber composites. Nanofiber Composites for Biomedical Applications. , 97-115 (2017).
- Danalache, M., Jacobi, L. F., Schwitalle, M., Hofmann, U. K. Assessment of biomechanical properties of the extracellular and pericellular matrix and their interconnection throughout the course of osteoarthritis. Journal of Biomechanics. 97, 109409 (2019).
- Danalache, M., et al. Changes in stiffness and biochemical composition of the pericellular matrix as a function of spatial chondrocyte organisation in osteoarthritic cartilage. Osteoarthritis Cartilage. 27 (5), 823-832 (2019).
- Lin, D. C., Dimitriadis, E. K., Horkay, F. Robust strategies for automated AFM force curve analysis–I. Non-adhesive indentation of soft, inhomogeneous materials. Journal of Biomechanical Engineering. 129 (3), 430-440 (2007).
- Darling, E. M., Topel, M., Zauscher, S., Vail, T. P., Guilak, F. Viscoelastic properties of human mesenchymally-derived stem cells and primary osteoblasts, chondrocytes, and adipocytes. Journal of Biomechanics. 41 (2), 454-464 (2008).
- Thambyah, A., Nather, A., Goh, J. Mechanical properties of articular cartilage covered by the meniscus. Osteoarthritis Cartilage. 14 (6), 580-588 (2006).
- Choi, A. P., Zheng, Y. P. Estimation of Young’s modulus and Poisson’s ratio of soft tissue from indentation using two different-sized indentors: finite element analysis of the finite deformation effect. Medical Biological Engineering Computing. 43 (2), 258-264 (2005).
- Jin, H., Lewis, J. L. Determination of Poisson’s ratio of articular cartilage by indentation using different-sized indenters. Journal of Biomechanical Engineering. 126 (2), 138-145 (2004).

