Studying the Activity of Neuropeptides and Other Regulators of the Excretory System in the Adult Mosquito
Summary
This protocol outlines methodologies behind the Ramsay assay, ion-selective microelectrodes, Scanning Ion-selective Electrode Technique (SIET) and in vitro contraction assays, applied to study the adult mosquito excretory system, comprised of the Malpighian tubules and hindgut, to collectively measure ion and fluid secretion rates, contractile activity, and transepithelial ion transport.
Abstract
Studies of insect physiology, particularly in those species that are vectors of pathogens causing disease in humans and other vertebrates, provide the foundation to develop novel strategies for pest control. Here, a series of methods are described that are routinely utilized to determine the functional roles of neuropeptides and other neuronal factors (i.e., biogenic amines) on the excretory system of the mosquito, Aedes aegypti. The Malpighian tubules (MTs), responsible for primary urine formation, can continue functioning for hours when removed from the mosquito, allowing for fluid secretion measurements following hormone treatments. As such, the Ramsay assay is a useful technique to measure secretion rates from isolated MTs. Ion-selective microelectrodes (ISME) can sequentially be used to measure ion concentrations (i.e., Na+ and K+) in the secreted fluid. This assay allows for the measurement of several MTs at a given time, determining the effects of various hormones and drugs. The Scanning Ion-selective Electrode Technique uses ISME to measure voltage representative of ionic activity in the unstirred layer adjacent to the surface of ion transporting organs to determine transepithelial transport of ions in near real time. This method can be used to understand the role of hormones and other regulators on ion absorption or secretion across epithelia. Hindgut contraction assays are also a useful tool to characterize myoactive neuropeptides, that may enhance or reduce the ability of this organ to remove excess fluid and waste. Collectively, these methods provide insight into how the excretory system is regulated in adult mosquitoes. This is important because functional coordination of the excretory organs is crucial in overcoming challenges such as desiccation stress after eclosion and before finding a suitable vertebrate host to obtain a bloodmeal.
Introduction
Maintenance of salt and water levels in insects allows them to succeed in many ecological and environmental niches, utilizing a variety of feeding strategies1. Most insects have evolved mechanisms to regulate the composition of their haemolymph within narrow limits in order to withstand the different challenges associated with their particular environment2. Terrestrial insects are often faced with the challenge of conserving water and the excretory system undergoes anti-diuresis to prevent loss of water and some essential salts, therefore, avoiding desiccation. In contrast, diuresis occurs when the insect feeds and is challenged with excess water and potentially salts3,4. Through their specialized and highly active excretory system, insects have evolved regulatory mechanisms acting to counter their osmoregulatory challenges. In adult Aedes aegypti mosquitoes, the excretory system is comprised of the Malpighian tubules (MTs) and hindgut, the latter of which is made up of the anterior ileum and posterior rectum5. MTs are responsible for generating primary urine, usually rich in NaCl and/or KCl. The primary urine is then modified through secretory and reabsorptive processes as it travels downstream of the tubule and enters the hindgut5. The final excreta can be hyper- or hypoosmotic to the haemolymph, depending on feeding/environmental conditions, and is enriched in toxic and nitrogenous wastes2.
MTs are ideal for studying many features of epithelial fluid and solute transport as they carry out a great variety of transport and excretory functions2,6. Through hormonal regulation2, MTs function by secreting ions and other solutes from the blood into the tubule lumen7, providing an osmotic gradient allowing water to be transported by aquaporins8,9, which collectively creates the primary urine, before traveling toward the reabsorptive hindgut2. Thus, by collecting the secreted fluid from isolated MTs, one can continuously monitor transepithelial transport of fluid and ions. Measuring secretion rate and urine composition provides insight on mechanisms responsible for transepithelial ion and fluid transport. A popular method for studying fluid secretion rates is the Ramsay assay, which was first introduced by Ramsay in 195310. In this method, the distal (closed) end of the tubule is treated with a hormone (or other test compound/drug), while the proximal (open) end is wrapped around a pin in water-saturated paraffin oil, which secretes the primary urine, accumulating as a droplet on the tip of the pin. Isolated MTs are able to survive and function for long periods (up to 24 h) under optimized in vitro conditions, which make them suitable and efficient models for fluid secretion measurement. Insects have open circulatory systems, thus the MTs are easily dissected and removed as they are usually freely floating in the haemolymph6. Additionally, with the exception of aphids—which lack MTs11—the number of MTs in a given insect species can vary considerably from four to hundreds (five in Aedes mosquitoes) allowing for multiple measurements from one insect.
The MTs in Aedes mosquitoes, in common with other endopterygote insects, are composed of two cell types forming a simple epithelium2,12; large principal cells, which facilitate active transport of cations (i.e., Na+ and K+) into the lumen, and thin stellate cells, which aid in transepithelial Cl– secretion13. The MTs are not innervated2, and instead are regulated by several hormones including both diuretic and anti-diuretic factors, allowing for the control of ion transport (mainly Na+, K+, and Cl–) and osmotically-obliged water2. Numerous studies have examined the hormonal regulation of Aedes MTs to understand the role of endocrine factors on transepithelial transport14,15,16,17,18. As shown in the representative results, the protocols herein demonstrate the effects of different hormonal factors on isolated MTs from adult female A. aegypti mosquitoes, including both diuretic and anti-diuretic control (Figure 1). The Ramsay assay is used to demonstrate how an anti-diuretic hormone, AedaeCAPA-1, inhibits fluid secretion of MTs stimulated by diuretic hormone 31 (DH31) (Figure 1).
The smaller size of insects has required the development of micro methods for measuring ionic activity and concentrations in fluid samples, or near the surface of isolated tissues such as the MTs and gut. Varying methods have been implemented, including the use of radioisotopes of ions19, which requires collection of the secreted fluid drops for measurement of ion concentrations20. Stimulated Aedes tubules in vitro typically secrete ~0.5 nL/min21, thus handling of such small volumes can pose a challenge and potentially introduce error upon transfer. As a result, ion-selective microelectrodes (ISMEs) have been extensively used to measure ion concentrations in secreted droplets of MTs in vitro. In this method, a reference electrode and ISME, filled with the appropriate backfill solution and ionophore, are positioned into the secreted urine droplet to determine ion concentrations22. Adapted from Donini and colleagues23, this current protocol uses a Na+-selective ionophore to measure ion activity in secreted droplets from stimulated MTs in adult Aedes mosquitoes. Since ion-selective microelectrodes measure ion activity, this data can be expressed as ion concentrations following the assumption that the calibration solutions and experimental samples share the same ion activity coefficient21 (Figure 1B,C).
The Scanning Ion-selective Electrode Technique (SIET) also makes use of ISMEs to measure ion concentration gradients in the unstirred layer adjacent to organs, tissues, or cells that are transporting ions. The ISMEs measure voltage gradients which can then be used to calculate the ion concentration gradients and direction and magnitude of ion flux across the organ, tissue, or cell20. In this technique, the ISME is mounted to a three axes manipulator controlled by computerized micro-stepper motors so that its 3D position is controlled to the micrometer level20. Voltages are measured at two points within the unstirred layer using a sampling protocol programmed into and controlled by computer software. The two points are typically separated by a distance of 20–100 µm with one point within 5–10 µm of the surface of the organ, tissue, or cell and the second point a further 20–100 µm away. The difference in magnitude of voltages between the two points is calculated to obtain a voltage gradient24,25,26, which is then used to calculate the concentration gradient and subsequently the net flux using Fick’s Law24,27. This method is useful for assessing the transport of specific ions across different regions of the insect gut and MTs, or at specific timepoints following a bloodmeal or treatment exposure. For instance, the SIET can be used to understand how absorptive and secretory processes in the mosquito excretory system are regulated by hormones28 as well as different feeding behaviors and rearing conditions25. Previous work utilizing the SIET revealed sites involved in ion transport along the anal papillae and rectum of larval and adult mosquitoes24,28. The current protocol, described previously by Paluzzi and colleagues26, measures Na+ flux across the rectal pad epithelia of the adult female rectum (Figure 2).
The final segment of the mosquito excretory system requires coordinated muscular movement to help mix food and secrete waste26. Non-absorbable products of digestion from the midgut, along with primary urine secreted by the MTs, are passed through the pyloric valve and delivered to the hindgut2. Spontaneous hindgut contractions begin at the pyloric valve and occur in peristaltic waves, which are relayed over the ileum through the coordinated contraction of circular and longitudinal muscles surrounding the basal surface of epithelial cells26. Finally, the muscles within the rectum help to propel and eliminate waste through the anal canal. Although insect hindgut motility is myogenic, requiring extracellular Ca2+ to produce spontaneous contractions, these processes can also be regulated neuronally26,29,30. This exogenous regulation by the nervous system is important after feeding, as the animal must expel wastes from the gut and restore haemolymph balance31. As a result, performing in vitro bioassays to identify myostimulatory or myoinhibitory neuropeptides is useful in assessing how neurochemicals influence hindgut motility. The current protocol, performed by Lajevardi and Paluzzi28, uses video recordings to examine ileal motility in response to neuropeptides (Figure 3). Similarly, a force transducer or impedance converter may also be used to observe traces of contractions through a data acquisition software32,33. However, using video technology allows us to visually assess the organ and further analyze using a subset of parameters to identify the role of hormones on hindgut motility.
Using these techniques can help characterize factors that regulate and coordinate fluid and ion transport along the excretory system along with hindgut motility. Importantly, a functional link between the diuretic response by the MTs and hindgut motility is supported, as diuretic hormones, such as DH31 and 5HT, characterized by their ability to stimulate fluid secretion by the MTs, have also been found to exhibit myotropic actions along the mosquito hindgut21,34,35. These findings highlight the importance of stringent coordination between the MTs and hindgut during events such as post-prandial diuresis in insects requiring rapid waste elimination.
Herein, the detailed approach behind the Ramsay assay technique to measure fluid secretion rate in the mosquito, A. aegypti, and the use of ion-selective microelectrodes to determine Na+ concentrations within the secreted fluid of the MTs are described, which when combined allows for transepithelial ion transport rates to be determined. Additionally, the Scanning Ion-selective Electrode Technique and hindgut contraction assays are described to measure ion flux and motility, respectively, which helps to elucidate hormonal regulation of the hindgut (Figure 4).
Protocol
1. Making silicone-lined dishes
NOTE: This step should be done prior to the experiments. These dishes will be made to prepare the assay dish for dissections, and for contraction assay experiments.
- Prepare the silicone using a silicone elastomer kit following the manufacturer’s recommendations. Mix the two-part liquid components at a ratio of 10 to 1. Mix gently and thoroughly by inverting but be sure to minimize bubble formation.
- Pour the silicone elastomer into standard 60 mm disposable polystyrene culture dishes to a depth of normally between 7–9 mm. Prepare as many dishes as required. Remove any air bubbles using a fine pin under a microscope shortly after pouring silicone into dishes. Place the dishes in a dust-free chamber or cabinet at room temperature (RT) for at least 48 h or in oven at 50 °C for about 4 h to cure (harden) (Figure 5A).
2. Making the Ramsay assay dish
NOTE: The dish can be re-used from experiment to experiment, thus, repeat this step only if the dish is damaged or breaks. A separate dish is used for dissections.
- Using a scalpel (with standard sharps precautions), create wells in one of the silicone-coated Petri dishes to prepare the assay dish, by removing enough of the silicone, about ~5 mm, from the top of the dish (this should fit ~20 µL bathing droplet). Prior to this, use a permanent marker to mark the positions under the dish to note where to make the wells. The wells should be ~1 cm apart, with a diameter of about ~4 mm to allow for the distal end of the tubule to fit. Each well will contain one fluid-secreting tubule, thus a dish containing 20 wells will allow for up to 20 MTs to be analyzed per experiment.
- To prepare the Minutien pins, place the 0.1 mm stainless steel pins in a row on a piece of labeling tape, perpendicular to the tape’s axis. Using scissors, cut along the length of the tape, creating equal halves of the pins. Use half a pin for each well.
- Using a blunt pair of forceps, insert each half-pin into the silicone-coated assay dish, beside each well. Depending on the length of the insect tubule, insert the half-pin at a distance that will allow the distal end of the tubule to immerse in the bathing saline and proximal end to wrap around the pin. This step can be done under a microscope to visualize wells easily. The pins can be re-used, however, replace the half-pin if damaged or lost. (Figure 5B)
3. Making the poly-L-lysine-coated SIET dish
NOTE: This step is important for the organ to adhere to the bottom of the dish during SIET measurements ensuring the site of measurement remains the same for each sample. Preparation of these dishes should be done at least 2 days prior to the experiments. Each dish should only be used once when applying a specific treatment. Dispose following every sample or if the poly-L-lysine coat is scratched off or damaged.
- For every sample, place a 35 mm Petri dish on a level surface. Remove the lids and pipette 70 µL of poly-L-lysine (0.1 mg/mL) in the center of each dish. Place the lids back on and let the poly-L-lysine dry (~48 h) undisturbed.
- Once dry, mark the dish bottom with a circle outlining the dried poly-L-lysine coat to better visualize where the excised organ should be placed following dissections. If necessary to minimize volumes of saline and treatment solutions in experiments, use a hot glue gun to encircle the poly-L-lysine coating, leaving enough room for the electrode to move and take background measurements (~20–25 mm diameter of the glued circle is enough). These coated dishes can be stored indefinitely at RT (Figure 5C).
4. Preparing the Ramsay and contraction assay dishes for experiments
NOTE: The dish can be re-used (provided appropriate washing to remove previously used saline/treatments), thus only repeat this step if the plate is damaged or breaks. A separate dish is used for dissections. This step is performed on the day of the experiment.
- For the contraction assay dish, use a scalpel (with standard sharps precautions), create wells in one of the silicone-coated Petri dishes to prepare the assay dish. Angle the scalpel in a way that the wells become triangle-shaped to facilitate pinning the dissected tissue on the edges. Wells should be big enough to hold up to 250 µL and fit the entire alimentary canal (rather than digging too deep, make the well wider to about 0.5–1 cm) (Figure 5D).
- For Ramsay plate, use a pipette and fill up wells up to 20 µL of solution (18 µL of 1X Aedes saline:Schneider’s medium prepared in step 5.2, and 2 µL of hormone/drug). For unstimulated controls, fill up wells with 20 µL of 1X Aedes saline:Schneider’s medium. Once all the wells are filled up, pour hydrated mineral oil into the assay dish until the wells and Minutien pins are submerged. Use a 20 µL pipette tip to pop or remove air bubbles that could interfere with the secreted droplets.
5. Preparing solutions
- Prepare 2X Aedes aegypti saline and 10x glucose, as described in Table 1, adapted from Petzel and colleagues36. Stock solutions should be stored at 4 °C. Make working stocks of 1X Aedes aegypti saline (Table 1), and store at 4 °C. On experiment day, allow the working stocks to come to RT before use. Prepare fresh if there is any evidence of fungal or bacterial growth.
- To prepare the bathing droplet, mix Aedes saline (from step 5.1) with Schneider’s medium, at a 1:1 ratio. Prepare in small aliquots, depending on how much is needed. Keep Schneider’s medium in the fridge, discard if there is any evidence of fungal or bacterial growth. (This step should be done on the day of the experiment).
- For Na+ flux measurements using the SIET, prepare a modified Ca2+-free Aedes saline (Table 2), as previously described26.
6. Mosquito MTs and hindgut dissections
- For adult mosquito dissections, collect pupa in a small beaker and place in a jar containing a sucrose solution for feeding. To determine the age of mosquito, isolate hatched mosquitoes and place in a different jar noting the age and sex of mosquitoes for future dissections.
- Briefly place mosquitoes to be dissected on a CO2 pad and wait until unresponsive. Using fine forceps, pick up the mosquito from its leg or wing and place on a silicone elastomer-coated dissecting dish prepared in step 1. Place the mosquito on one side (lateral side up), and with a Minutien pin, impale into the thorax to secure the mosquito in place. Optionally, remove wings and legs off mosquitoes for easier dissection.
- Using a Pasteur pipette, add a drop of RT Aedes saline to fully immerse the mosquito in saline. Make sure the dissection is all under saline (see Table 1 for making Aedes saline).
- Place the dissecting dish under a stereoscopic microscope. Pinch the last abdominal segment with fine forceps and slowly pull away from the mosquito. This step is facilitated by simultaneously looking under the microscope. The midgut, attached with the MTs and hindgut, should be exposed (Figure 6).
- For the Ramsay assay, carefully remove each tubule from the midgut-hindgut junction, making sure to not damage the organs. Use sharp fine forceps to remove the tubules individually – do not use damaged tubules for the assay. (See step 7 for setup.)
- For the SIET, dissect the hindgut in Ca2+-free Aedes saline (step 5.3). Ensure that the poly-L-lysine dish also has a fixed volume of Ca2+-free Aedes saline. Carefully remove the cuticle from the rectum and place the hindgut (can remove any other attached organs, depending on what is being measured) into the poly-L-lysine dish. If measuring ion transport along the rectal pad epithelia, position the haemolymph-facing side of at least one rectal pad to be accessible to the microelectrode (Figure 7).
- Using forceps to grab the most posterior end, bring the hindgut down to the bottom of the dish (without damaging any structures that will be measured). As soon as the organ touches the poly-L-lysine coat, it will adhere. This dissected sample is ready for measurements. (See steps 14–16 for SIET setup and measurements.)
- For the ileum contraction assay, ensure that the hindgut remains attached to the midgut during the dissection (Figure 4D). Carefully remove the MTs to have an unimpeded view of the hindgut. This organ will be transferred to a new dish prepared as described in step 4.1. (See step 17 for obtaining assay videos.)
7. Setting up the Ramsay assay
- Using the tip of the forceps, carefully lift the proximal (open) end of the tubule by draping it over the forceps (do not pinch the tubule), and transfer into a well of the assay dish. This step can also be done using fine glass probes.
- Once the tubule is immersed in the well, pick up the proximal end of the tubule with the forceps, remove it from the bathing droplet, and wrap the end around the pin. Wrap the tubule around the pin twice – and keep the length of tubule remaining in bathing droplet consistent with the other tubules. At this point, the distal end of the tubule should be in the bathing droplet, while the proximal end wrapped around the pin away from the bathing droplet allowing the secreted fluid to accumulate on the tip of the pin from the open proximal end.
- Immediately after the tubule is wrapped around the pin, note down the wells (e.g., A, B, C), the time (which will be the start time of when the fluid will begin to be secreted from the tubule), and any other identifying information (e.g., hormone, genotype, etc.).
- Each mosquito has five tubules, thus repeat steps 7.1–7.3 with the rest of the tubules. For control and experimental treatments, split the five tubules within the different treatments. Continue with the next dissection, until the entire assay dish is filled. With practice, this technique should take typically 2–3 min to dissect the tubules, transfer them to the bathing saline, and wrap around the pin, thus creating a 2–3 min difference in the start time between each tubule.
NOTE: Before starting the experiment, calibrate the ocular micrometer of the microscope with a stage micrometer under the selected objective. Secreted droplets are routinely measured under a 40x–50x total magnification. - After the allotted incubation time, the secreted droplet can be measured. Using the ocular micrometer of the microscope, measure and record the diameter of the secreted droplet. Calculate the diameter (d) of the secreted droplet by multiplying the ocular unit diameter measured by the calibration conversion (Table 3). Calculate the volume of the secreted droplet using the equation, V = πd3/6.
- To calculate the secretion rate (nL min-1), use the equation, fluid secretion rate = V/secretion time, where V is the volume of the secreted droplet calculated in step 7.5, and the secretion time refers to the incubation period of the tubule.
8. ISME setup
- Place micromanipulators on either side of the stereomicroscope to setup the ISME station. Chloriding the silver wires can be achieved by immersing in a solution of ferric chloride and thread each silver wire into each of the microelectrode holders (or solder the wires onto the coaxial cables, if necessary). Repeat this step whenever the silver wires become un-chlorided.
- Connect the silver wires to an amplifier, which will read to a data acquisition system. Set up and calibrate the system according to the manufacturer’s instructions. In case of increased electrical interference, use a properly grounded Faraday cage.
9. Preparing the microelectrodes for ISME and SIET
- Using a P-97 Flaming Brown pipette puller, pull ~5–6 unfilamented borosilicate glass capillaries (outer diameter 1.5 mm, inner diameter 1.12 mm, length 100 mm) with a tip of 1–5 µm. These will be used as the ion-selective microelectrodes. For SIET microelectrodes, the resulting electrode should have a tip opening of ~5 µm, characterized by a short shank.
- In a hood, place the microelectrodes onto a hotplate (setting down carefully as to not break the tips of the electrodes). Add dichlorodimethylsilane inside a 15 cm glass Petri dish and invert over the electrodes on the hotplate. Use dichlorodimethylsilane to silanize the ion-selective electrodes by adding a hydrophobic coat to all surfaces of the electrode, allowing it to retain the hydrophobic ionophore37. Unused silanized electrodes can be left for a few weeks; therefore, this step is performed every few weeks, or for newly pulled electrodes.
NOTE: Use caution when handling dichlorodimethylsilane – flammable, corrosive, and toxic, see MSDS for safe handling.- Turn the hotplate to 350 °C and leave in place for 75 min. Use a 1:2 ratio of number of microelectrodes to volume (µL) of dichlorodimethylsilane to add onto the glass Petri dish. Thus, for 10 microelectrodes, add 20 µL of dichlorodimethylsilane.
- After 75 min, turn off the hot plate. After the electrodes and hot plate have cooled down, remove the glass Petri dish and transfer the electrodes (with forceps) into a storage box with molding clay. Ensure the tip of the electrode is pointing up to prevent any damage.
- To make the reference electrodes for ISME, pull ~4–5 filamented borosilicate glass capillary tubes (outer diameter 1 mm, inner diameter 0.58 mm, length 100 mm). These electrodes do not have to be silanized. Label and store in a similar box, avoiding any damage to the tip.
- SIET reference electrodes are made from standard glass capillaries (outer diameter 2 mm, inner diameter 1.12 mm, length 102 mm). The capillaries are filled with molten 1 M KCl + 3% agar and should be stored in a 50 mL centrifuge tube containing 1 M KCl (fully submerged) until use. They can be used repeatedly until bubbles begin to form or if the agar breaks. If there is any air space, dispose of the glass and use a new one to ensure that the circuit is complete.
10. Preparing the backfill syringes
NOTE: This step is done to create fine-tipped syringes to backfill electrodes for ISME.
- Use a 1 mL slip-tip syringe with a disposable needle. Discard the needle and pull back the syringe until the 1 mL mark. Under the fumehood, turn on the Bunsen burner, and heat up the plastic tip of the syringe until it starts to melt. Using an old pair of forceps, carefully pinch the melted syringe tip and pull down to stretch the tip.
- Using scissors, cut the melted tip to the desired length, leaving the diameter small enough to fit inside the electrodes (See diagram). After the syringe has cooled down, test the pressure of the syringe with water to ensure there is no blockage. Create one for each backfill solution required and label accordingly (Figure 8).
11. Filling the Ion-Selective and Reference Microelectrode for ISME and SIET
NOTE: The microelectrode may be re-used as long as it is still working (calibrate before each experiment). For ISME, this step can be done while the MTs are incubated in the Ramsay assay.
- To make a Na+-selective microelectrode, use the 1 mL syringe created in step 10 to backfill electrode with 100 mM NaCl. Ensure the backfill solution fills until the tip of the electrode. If air bubbles appear, gently flick the microelectrode or remove the solution and re-fill. This should be done under a stereoscopic microscope to visualize better.
- Under a stereoscopic microscope, dip a 10 µL pipette tip into the Na+-selective ionophore solution. Lining up the electrode perpendicular to the pipette, place a gloved finger over the bottom of the tip to create pressure and expel a small drop of ionophore. Viewing under high objective of a microscope, carefully touch the drop of ionophore to the microelectrode tip, avoiding breaking the tip. Normally, microelectrodes are forward filled with an ionophore cocktail column length of between 150–300 µm, until the ionophore/backfill solution border is flat.
CAUTION: This ionophore is toxic, see MSDS for safe handling. - In a small beaker, fill up halfway with 100 mM NaCl, and place some modeling clay onto the inside at the top of the beaker. After the Na+ ionophore has been taken up, place the electrode tip-down onto the wall of the beaker, letting the tip sit within the 100 mM NaCl, and keep ISME in the beaker until it is ready to use.
- To make the ISME reference electrode, backfill an electrode with 500 mM KCl ensuring that the solution is filled to the tip (follow the same protocol as above). Store the KCl in a beaker.
12. Calibrating electrodes for ISME
NOTE: This step is performed right before taking measurements of the secreted fluid (~10–15 min before). Calibrations should be done every ~5–6 measurements to ensure that the slope is consistent.
- Coat the electrode tips using a solution of ~3.5% (w/v) polyvinyl chloride dissolved in tetrahydrofuran, to avoid displacement of the ionophore when submerged in paraffin oil.
CAUTION: This is flammable, see MSDS for safe handling. - To calibrate the Na+ electrode, place 10 µL droplets of the following NaCl standard concentrations onto the edge of the Ramsay dish with the incubated MTs: 200 mM NaCl and 20 mM + 180 mM LiCl. Place the standard droplets ~ 2 cm apart, with the higher concentration on top.
- Insert both reference electrode and ion-selective electrode over the chlorided silver wires and fasten them securely using electrode holders that are attached to micromanipulators. Navigate both the electrodes toward the 200 mM NaCl droplet using the micromanipulators, ensuring that the electrode tips do not touch the bottom of the dish. Turn on the electrometer to start recording and allow the reading to stabilize.
- Record the reading and continue to the next standard (20 mM NaCl + 180 mM LiCl). Calculate the slope, and if electrode reading is stable and slope is within range, proceed to use the electrode for the secreted droplet measurements. If the reading is unstable, not giving a proper recording or slope, or taking a while to equilibrate, prepare either a new ion-selective electrode or break the very tip of the reference electrode by running a tissue across the tip. The slope for Na+ should be ~58–60 mV38, although an acceptable range is 50–60 mV.
13. ISME recordings and calculations
- Following measurements of the fluid secretion rate (see step 7.7), carefully move both the reference and ion-selective electrode into the secreted droplet using the micromanipulators. Turn on the recording and allow the reading to stabilize and record value. Repeat this step for the other droplets (repeat calibration measurements every ~5–6 measurements).
- Cation concentrations are calculated using the equation described previously22,38, [ion] = [C] x 10ΔV/m, where [C] is the concentration in mmol L-1 of the calibration solution used to calibrate the ISME, ΔV is the change in voltage between the voltage recorded from the secreted fluid droplet and the voltage of the same calibration solution, and m is the voltage difference between the two standard calibrations, which is also the slope.
14. SIET setup
NOTE: The SIET system has been described previously27,39. To reduce background noise, a Faraday cage is installed around the light microscope and headstage. Experiments presented in this paper use the following settings on the Automated Scanning Electrode Technique (ASET) software 2.0: a 4 second wait period to allow ion gradients to fully re-establish following microelectrode movements, with voltage being recorded for 0.5 s following the wait period, an excursion distance of 100 µm and three repetitions for every recording. Certain settings can be user-modified in ASET, as needed.
- Turn on the IPA-2 Ion/Polarographic Amplifier, the light microscope (with an attached video camera), and the computer(s) connected to each of these running ASET and cellSens.
- Prepare microelectrodes and reference electrodes described in steps 9.1–9.3, 9.5, 10, and 11.1–11.3. The ion-selective microelectrode will contain Na+ ionophore with 100 mM NaCl backfill and the reference electrode (with the agar bridges) will always be filled with 3 M KCl.
- Place Na+-selective microelectrode on holder consisting of a silver chloride (AgCl) wire and attach it into female connector jack. Using a syringe, fill the reference electrode holder with fresh 3 M KCl (can be made prior to experiment day and stored at RT).
NOTE: Following experiments, wash out the reference electrode holder with ddH2O. - Remove one reference electrode from the beaker containing all reference electrodes submerged in 3 M KCl, placing one finger at one end and tilting the glass capillary towards this finger to prevent the agar from falling out. Carefully place one end into the holder, ensuring that no bubbles form. If there is a bubble, remove the reference electrode, re-fill the holder with more 3 M KCl and repeat. Place the electrode holder into the female connector jack.
15. Calibrating electrodes for SIET
- For Na+ measurements, use a 150 mM NaCl and 15 mM NaCl + 135 mM LiCl solutions24. There should be a 10-fold difference in Na+ concentrations between the two solutions, encompassing the range of Na+ (20 mM) levels in the saline (Table 2).
- Store calibration solutions at RT in a 50 mL centrifuge tube and aliquot in a Petri dish when conducting the experiment. This aliquot is disposed of at the end of the day. If there is any evidence of contamination in the stock solutions, dispose and make fresh solutions.
- To calibrate, aliquot the two calibration solutions (step 15.1) into individual 35 mm Petri dishes, and place these on the microscope stage. Using the manual adjustment knobs that control the stepper motors with “disable” selected on the computer motion control unit, ensure that the microelectrode tip is placed inside the calibration solution. Do not submerge the tip too far, as it may break. If the tip breaks, prepare a new ion-selective microelectrode and re-calibrate.
- On the computer connected to the amplifier, open the ASET software. Open Calibration settings. Select Nernst Slope for the calibration type, set the sample for 3 seconds, and enter the concentrations of the calibration solutions. For instance, for Na+ measurements, enter 150 mM in solution 1, place the Na+-selective microelectrode tip and reference electrode inside this calibration solution. The voltage reading on the amplifier should be stable. Click on Solution 1, wait for voltage (in mV) to be recorded. Next, place the microelectrode and reference electrode inside the 15 mM NaCl + 135 mM LiCl solution, enter 15 mM in solution 2 and click on Solution 2 to obtain the voltage reading. The slope will be the difference between these two voltages, calculated in these settings. Following calibration, click on OK.
- Slopes for Na+-selective microelectrodes for a tenfold change in ion concentration should be around 59.0 ± 1.624. If the Nernst slope greatly deviates from this range, prepare a new microelectrode, or aliquot new standards (as they may be contaminated). Calibration is required before every 2–3 samples, depending on voltage stability. If the voltage becomes unstable and begins to fluctuate, prepare a new ion-selective microelectrode.
16. SIET measurements
NOTE: The motor switch on the Computer Motion Control unit should always be switched to Disable except when manipulating the electrode using computer keys through ASET, or during measurement recordings (at which point the key should be switched to Enable).
- Following calibration, dissect the organ (see step 6.6). Place the poly-L-lysine dish with the dissected sample on the microscope stage and insert the tip of the reference electrode inside the saline. Submerge the tip of the ion selective microelectrode in the saline taking care not to break the tip.
- Use the manual adjustment knobs to adjust the microelectrode position while looking under the light microscope. Adjust the vertical position of the microelectrode such that its tip is on the same plane as the organ or tissue. Once at the desired position, turn the motor switch to Enable. At this point, the stepper motors can only be operated using the computer software.
- Using the computer arrow keys, move the microelectrode horizontally to a position 3 mm away from the tissue to measure background recordings. Insert notes by pressing F2. When ready, begin to record (by pressing F5). Obtain five measurements of background activity. The average background voltage activity for every sample will be subtracted from the voltage gradients measured along the tissue for the same sample.
- Move the microelectrode tip back close to the tissue, being careful not to pierce the organ. Reduce the keyhit sensitivity to place the microelectrode tip 2 µm directly to the right, perpendicular to the tissue. Obtain three recordings at each site along the rectal pad to identify the site of greatest ion activity. Obtain baseline saline measurements at the site displaying greatest activity (the “hotspot” site).
- Switch the motor to Disable and add the appropriate treatment into the dish to obtain the final desired dose. Following application, create a new note (i.e., treatment name and dose), switch motor back to Enable and take voltage recordings using F5. If the goal is to observe changes over time, continue to take measurements at specific time intervals. Multiple protocols can be created for specific applications using the SIET, depending on the aim of the researcher.
- Watch the ASET software record the voltage at the site along the tissue and subtract this from the voltage at an excursion distance (Figure 7) of 100 µm away from the tissue. Since the first recording is taken at the tissue, a positive voltage difference indicates that the voltage is higher at the epithelium (tissue) (Figure 7A) than away from the epithelium (Figure 7B), and thus the cation is being absorbed. A negative voltage gradient is indicative of cation secretion.
- Obtain background recordings again following the treatment (3 mm away) and use these when calculating ion flux following the treatment. Make one set of background recordings in “baseline” and one set following the treatment. Switch the motor to Disable anytime the microelectrode is not being adjusted with the computer keys or when the voltage is not being recorded. Export the data into a text file and open using Excel to conduct all calculations.
- Calculate ion flux using equations described previously24,26. The net ion flux for each treatment can be expressed as an absolute change from baseline ion flux measurements. To verify significance of results, a vehicle control (i.e., adding saline alone into the dish instead of a treatment) can be used. Changes in ion transport following the hormonal treatment should be assessed relative to any changes in response to this control.
17. Hindgut contraction assays
- Fill one of the wells in the dish described in step 4.1 with a known volume of Aedes saline (step 5.1). Following dissection (step 6.7), carefully transfer the dissected hindgut attached to the midgut to the well in the other dish, making sure not to pinch the organ being examined (ileum). Submerge the gut into the saline inside the well, and place Minutien pins into the midgut and rectum. By doing so, the ileum should not be under tension, and spontaneous contractions, originating at the pyloric valve at the anterior ileum, should be observed.
- Connect a video camera to a stereoscopic microscope, and record videos using a video capture software (e.g., Luminera’s INFINITY CAPTURE). Place the dish containing the dissected organ under the microscope and record for 2 min. This will be the “Baseline” condition.
- Add a known volume of 1X Aedes saline (step 5.1) to the well to account for any changes in ileal motility upon increasing volume within the well. Wait for 1 min following application, then record again for 2 min. This is the “saline” condition.
- Add a known volume of treatment (from a 10x stock concentration to obtain the desired dose in the well). Wait for 1 min following application, then record again for 2 min. This will be the “treatment” condition.
- Save all the videos and convert them to MP4. To analyze, count the number of contractions in the 2 min intervals (defined as a peristaltic wave initiating at the pyloric valve and extending throughout the ileum). Divide this number by 2 min to obtain the contraction rate. Other parameters can also be assessed, such as contraction or relaxation duration. Following collection of raw data, express each parameter relative to the data for “baseline” conditions for every sample. Plot the fold-change for the “saline” and “treatment” all relative to the “baseline”.
Representative Results
Application of DH31 against unstimulated MTs results in a significant increase in fluid secretion rate, confirming its role as a diuretic hormone in Aedes mosquitoes (Figure 1A). When tubules are treated with AedaeCAPA-1, a reduction in secretion rate is observed in DH31-stimulated MTs. Figure 1B demonstrates the use of ion-selective electrodes to measure Na+ concentrations in the secreted droplets. Treatment of DH31 on the MTs had no effect on the Na+ concentration in the secreted droplet; however, with the application of AedaeCAPA-1, the Na+ concentration in the secreted fluid was significantly increased. Additionally, compared with unstimulated controls, DH31 led to a significantly greater Na+ transport rate, whereas AedaeCAPA-1 abolished this increase in DH31-stimulated tubules (Figure 1C). Together, these sample results demonstrate how fluid secretion rates following application of diuretic and anti-diuretic hormones can be measured as well as the ion concentration within the secreted droplets. Three to six-day old females were dissected and between 23–35 MTs were analyzed for the experiment. Differences were denoted as statistically significant if p < 0.05 using a one-way ANOVA and unpaired t-tests.
The SIET was used to assess changes in Na+ transport along the rectal pad epithelia of adult female mosquitoes. Most unstimulated recta examined exhibited haemolymph-directed Na+ transport (absorption), and so only these preparations were examined. Due to variability in baseline activity, the difference in ion flux, following either saline or peptide application, relative to initial transport activity in saline was calculated. These values yield a negative change in ion flux (ion flux following treatment – ion flux in baseline conditions), although all organs remained absorptive following treatments. A leucokinin analog (Drosokinin) was used to examine changes in Na+ absorption, which resulted in a four-fold decrease in Na+ absorption compared to saline control (Figure 2).
To assess the role of a neuropeptide, pyrokinin-2 (PK2), on ileal motility, a Rhodnius prolixus analog was used, which was previously shown to activate the A. aegypti PK2 receptor, enriched in the mosquito ileum28. Relative to baseline levels, PK2 significantly inhibits ileal contractions (Figure 3).
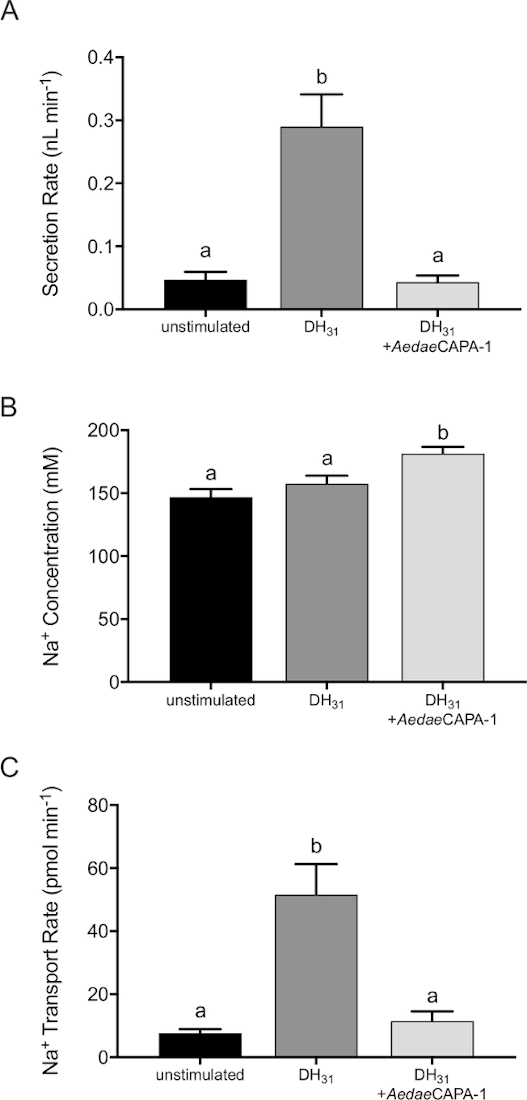
Figure 1: Effect of diuretic and anti-diuretic neuropeptides on in vitro fluid secretion rate, Na+ concentration and transport rate by adult female A. aegypti MTs. AedaeCAPA-1 (0.1 fM) was applied to MTs stimulated with DromeDH31 (25 nM). (A) MTs were incubated for 60 min with neuropeptides and 120 min for unstimulated controls. (B) Na+ concentrations in the secreted droplet were measured using ISME and values were used to calculate transport rate (C). Columns that are significantly different from controls are denoted with the same letter, as determined by a one-way ANOVA and Bonferroni post-test (n = 23–35). Please click here to view a larger version of this figure.
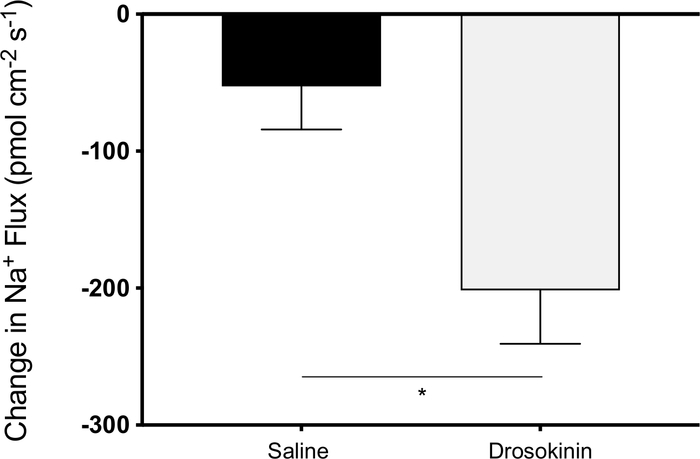
Figure 2: Changes in Na+ transport across female rectal pad epithelia measured using the SIET. Na+ absorption was recorded in unstimulated baseline saline conditions, followed by treatment with either additional saline (vehicle control) or 1 µM Drosokinin (n = 10–12). Changes in haemolymph-directed Na+ flux were measured for each treatment. There was a four-fold decrease in Na+ absorption in response to Drosokinin, as determined by an unpaired two-tailed t-test. Please click here to view a larger version of this figure.
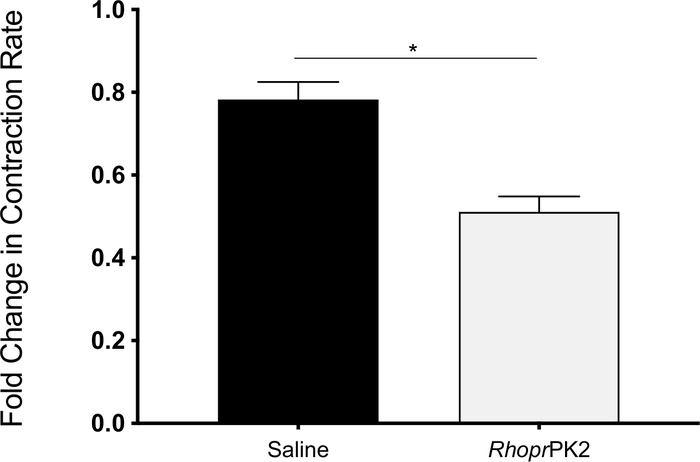
Figure 3: Change in ileal motility of female mosquitoes following saline (vehicle control) and RhoprPK2 application. The change in contraction frequency following both treatments was recorded for every sample relative to baseline conditions (n = 32). RhoprPK2 decreased ileal motility, as determined by a paired two-tailed t-test. Please click here to view a larger version of this figure.
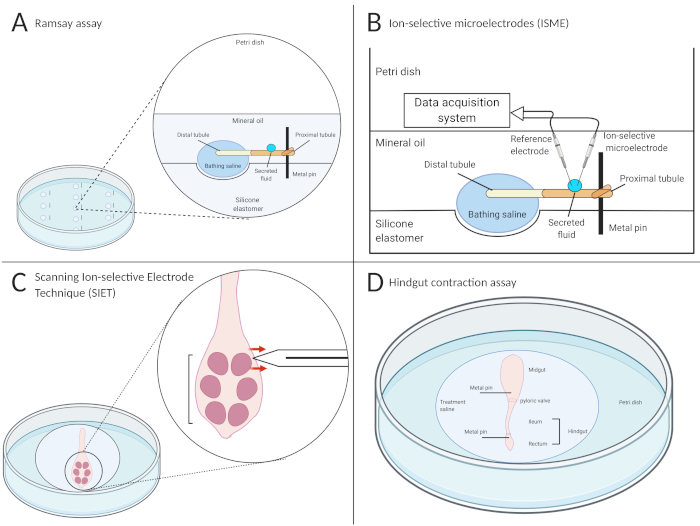
Figure 4: Schematic of the Ramsay assay, use of ion-selective microelectrodes (ISME), the scanning ion-selective electrode technique (SIET), and hindgut contraction assays. Illustrations of the four methods utilized to determine the functional roles of endocrine factors on the excretory system of the mosquito, Aedes aegypti. (A) The Ramsay assay is used to measure fluid secretion rates in isolated Malpighian tubules. The proximal (open) end of the tubule is wrapped around a pin, surrounded by mineral oil, while the distal (closed) end is bathed in the treatment saline. After a set time period, the proximal end will secrete the primary urine, accumulating as droplets on the pin. (B) A reference electrode and ISME are placed in the secreted droplet, connected to a data acquisition system, to measure ion concentration. (C) The SIET measures ion transport using an ion-selective microelectrode perpendicularly placed ~2 µm away from the organ adhered to the bottom of a poly-L-lysine dish. The direction of the red arrows in this schematic indicate that Na+ is being absorbed into the saline, indicated by a greater voltage recording along the epithelium relative to the region away from organ surface. (D) Hindgut contraction assays are conducted using video recordings to assess changes in contraction frequency, length, and relaxation duration. In this schematic, the organs are placed inside a well, and a metal pin is inserted directly into the midgut and rectum to allow the ileum to freely float and contract. Treatments are added into the well to assess changes in motility both qualitatively and quantitatively. Created using BioRender. Please click here to view a larger version of this figure.
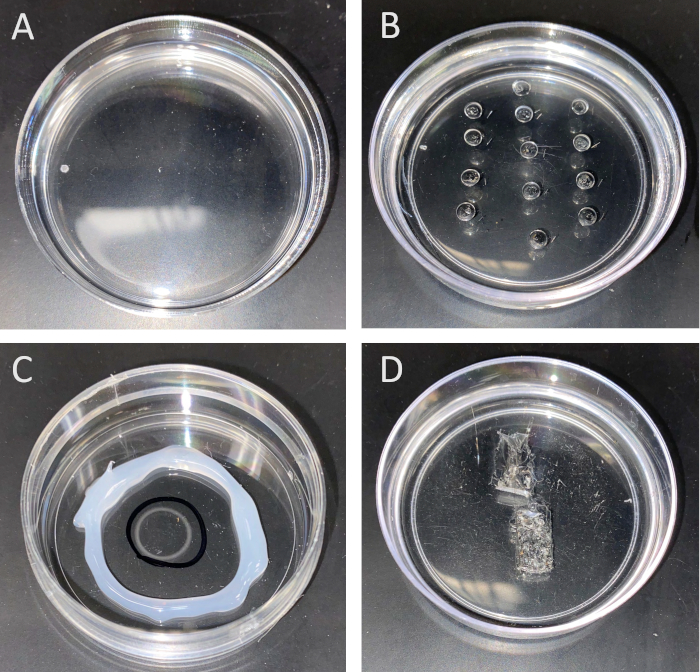
Figure 5: Dishes used for (A) dissecting mosquitoes, (B) Ramsay assay, (C) SIET and (D) contraction assay measurements. A, B and D are completely coated with silicone, whereas C is coated with poly-L-lysine in the center. To prepare the dish used for the Ramsay assay (B), a small silicone-coated Petri dish is utilized, and small ~4–5 mm wells are carved 1 cm apart (around ~20 wells can be carved/dish). Minutien metal pins are placed beside each well to hold onto the isolated tubule to measure fluid secretion rate. The poly-L-lysine dish (C) allows the organ to adhere to the bottom during SIET measurements. The wells in the contraction assay dish (D) are filled with saline and any following treatments. The contracting organ placed inside the well is observed using a video camera. Please click here to view a larger version of this figure.
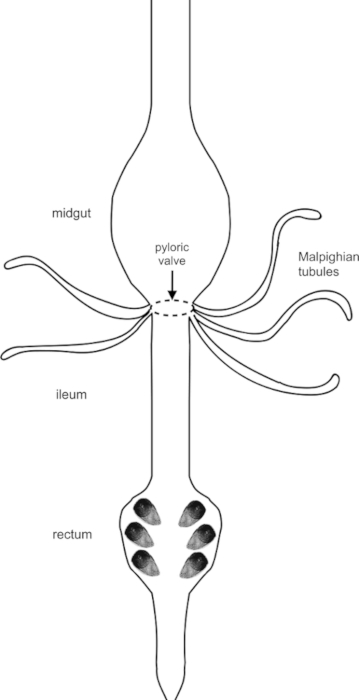
Figure 6: The excretory system of mosquito, Aedes aegypti. After a bloodmeal, solutes, water, and nutrients are passed along the alimentary canal, which includes (but is not limited to) the midgut, pyloric valve, MTs, and hindgut. The midgut is the main site for food digestion and nutrient absorption. The five MTs transport water, solutes, and waste from the surrounding haemolymph and secrete this primary urine into the hindgut, made up of an anterior ileum and posterior rectum, the latter of which includes four (male) or six (female) rectal pads. The hindgut acts as the final reabsorption site before waste is excreted. Please click here to view a larger version of this figure.
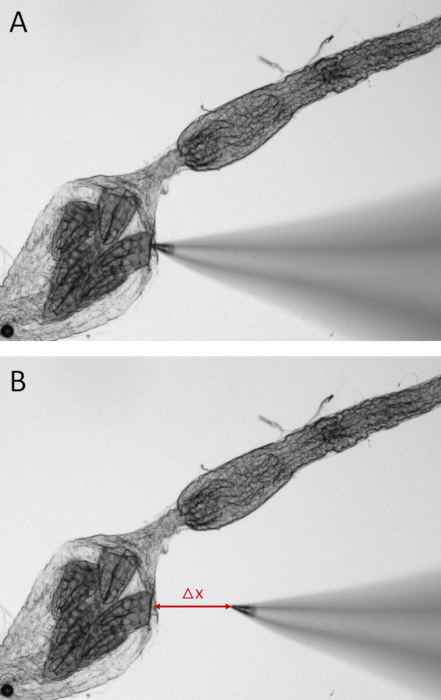
Figure 7: Still images of a dissected mosquito hindgut during SIET measurements. To measure ion activity along the epithelia of one of the six rectal pads, the Na+-selective microelectrode is placed 2 µm away from the tissue (A). Voltage is recorded at this site before the microelectrode tip is moved at an excursion distance (Δx) of 100 µm away from this point (B). The voltage gradient between these two points is used to calculate the concentration gradient and ion flux. Please click here to view a larger version of this figure.
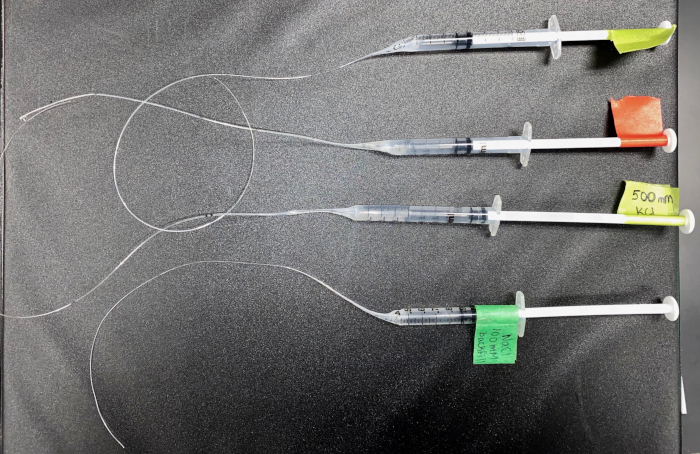
Figure 8: The backfill syringes for ISME and SIET. A picture of the syringes used to backfill NaCl and KCl solutions into the microelectrodes. This shows four 1 mL slip-tip syringes with disposable needles, with a modified stretched tip to allow for insertion into an electrode. Please click here to view a larger version of this figure.
| 2X Aedes aegypti saline | ||
| For 500 mL: | ||
| In a beaker, add 400 mL of ddH2O, then add the following solutes: | ||
| Component | Weight (g) | Final concentration (mM) |
| NaCl | 8.766 | 150 |
| HEPES | 5.957 | 25 |
| KCl | 0.253 | 3.4 |
| NaOH | 0.3 | 7.5 |
| NaHCO3 | 0.151 | 1.8 |
| MgSO4 | 0.12 | 1 |
| CaCl2-2H20 | 0.249 | 1.7 |
| Stir, and adjust pH to 7.1 | ||
| Add ddH2O to final volume of 500 mL to make 2x Aedes saline | ||
| 10X glucose | ||
| For 500 mL: | ||
| In a beaker, add 450 mL of ddH2O, then add: | ||
| Component | Weight (g) | Final concentration (mM) |
| Glucose | 4.5 | 5 |
| Stir and add ddH2O to final volume of 500 mL | ||
| To make 1X Aedes aegypti saline: | ||
| For 100 mL: | ||
| In a beaker, add 40 mL of ddH2O, then add: | ||
| Component | Volume (mL) | |
| 2x Aedes saline | 10 | |
| Stir, adjust pH to 7.1 | ||
| Filter sterilize | ||
Table 1: Making Aedes aegypti saline. To make Aedes saline, separately prepare 2X Aedes saline and 10x glucose, store in 4 °C fridge. Use these two solutions to prepare working stocks (1x Aedes saline), used for dissecting tissues, Ramsay assays, and contraction assays.
| Calcium-free Aedes aegypti Saline + NMDG | ||
| For 500 mL: | ||
| In a beaker, add 400 mL of ddH2O, then add the following solutes: | ||
| Component | Weight (g) | Final concentration (mM) |
| NaCl | 1.17 | 20 |
| NMDG | 25.38 | 130 |
| HEPES | 5.957 | 25 |
| KCl | 0.253 | 3.4 |
| NaHCO3 | 0.151 | 1.8 |
| MgSO4 | 0.12 | 1 |
| Glucose | 0.9 | 5 |
| Stir, and adjust pH to 7.1 | ||
| Add ddH2O to final volume of 500 mL | ||
Table 2: Modified Aedes aegypti saline used for SIET measurements. This Ca2+-free saline is used to prevent spontaneous hindgut contractions during SIET measurements. This saline is specific for measuring Na+ transport, as it consists of reduced Na+ (20 mM) made up by equimolar substitution with N-methyl-D-glucamine (NMDG) to reduce background noise.
| DH31-stimulated MTs | Droplet diameter (units) | Droplet diameter (um) | Droplet volume (um3) | Secretion Rate (nl/min) |
| n | Treatment #1 (60 min) | Treatment #1 (60 min) | Treatment #1 (60 min) | Treatment #1 (60 min) |
| 1 | 16 | 313.73 | 16167692.56 | 0.27 |
| 2 | 14 | 274.51 | 10831090.91 | 0.18 |
| 3 | 17 | 333.33 | 19392547.25 | 0.32 |
| 4 | 13 | 254.90 | 8671977.67 | 0.14 |
| 5 | 14 | 274.51 | 10831090.91 | 0.18 |
| 6 | 22 | 431.37 | 42029685.15 | 0.70 |
| 7 | 15 | 294.12 | 13321768.16 | 0.22 |
| 8 | 16 | 313.73 | 16167692.56 | 0.27 |
| 9 | 20 | 392.16 | 31577524.53 | 0.53 |
| Mean | 16.33333333 | 320.26 | 18776785.52 | 0.31 |
| STD ERR | 0.06 |
Table 3: Sample Ramsay assay data. Table showing sample data collected from DH31-stimulated MTs from adult female mosquitoes. When measuring the diameter of the secreted droplet, first measure with the stereomicroscope ocular micrometer and note down the value. Next, multiply the ocular unit diameter with the calibration conversion conducted before the experiment to get the diameter of the droplet in µm. Calculate the volume of the equation next, using the equation noted in step 7.7, followed by the secretion rate, using the equation noted in step 7.8.
Discussion
When ingesting a blood meal, haematophagus insects face the challenge of excess solutes and water in their haemolymph2. To cope with this, they have a specialized excretory system, which is tightly controlled by hormonal factors, allowing the insects to rapidly initiate post-prandial diuresis. The Ramsay assay and use of ion-selective microelectrodes allows for measurement of fluid secretion rates along with ion concentrations and transport rates in isolated insect MTs. Critical steps within these approaches include ensuring proper dissection of the insect MTs. Individually removing each tubule from the insect must be done carefully, as any damage to the tubule will cause the fluid to be secreted from the tear or lead to a non-functional preparation. Aedes MTs do not secrete readily in unstimulated controls, thus hormones and drugs may be applied to stimulate secretion. However, even with diuretic application or careful dissections, some MTs may fail to secrete if damaged (even if not at all obvious with visual inspection), thus multiple rounds of the assay may have to be conducted. For proper use of ion-selective electrodes during both ISME and SIET measurements, ensure that the slope of the standards is within the normal range reported for the ionophore, which will give the most accurate readings. However, if the readings are unstable, examine the electrode under the microscope to confirm the ionophore has been taken up and is not leaking. A new electrode should be prepared if there are air bubbles present, the tip is broken, or the ionophore has not been taken up properly. If the ionophore/backfill border is convex, the electrodes are under-silanized and more dichlorodimethylsilane should be used. If the border is concave, the electrodes are over-silanized, and less dichlorodimethylsilane should be used. Additionally, some electrodes may be less stable than others, such as the Na+-selective electrode, whereby the slope deteriorates quicker compared to K+ or H+.
These approaches provide unlimited opportunities to studying the mechanisms behind fluid secretion by the MTs. Due to recent advances in genomic editing, reverse genetic strategies such as CRISPR-Cas9 gene-editing and RNA interference may be used to investigate the regulatory and/or functional role of specific genes expressed in excretory organs. Studies have used such tools to understand the hormonal regulation in Aedes40 and Drosophila41 MTs. Additionally, the bathing droplet may be modified to look at the specific roles of hormones14,15,18,21,42, second-messenger systems, and drugs on secretion rate and ion concentrations. These methodologies have been applied in various other insects such as Rhodnius prolixus38, larval and adult mosquito species, Drosophila41, and mayfly species (Hexagenia rigida)43. Similarly, different ions may be examined, such as Cl–, K+, H+ and NH4+.
During SIET measurements, there may be variability between every sample during baseline conditions. Differences could still arise when controlling the age, sex, and feeding state of the mosquito, particularly when examining different regions of the gut (i.e. certain distances anterior or posterior from the ileum-rectum junction). These differences can be accounted for by expressing ion flux following treatment as a change relative to baseline conditions. Over time, ion activity may also fluctuate and therefore it is important to assess changes in comparison to a vehicle control (saline), as shown in the results presented here. If voltage readings begin to fluctuate drastically, prepare a new ion-selective microelectrode and re-calibrate. Using a third calibration solution (diluted 10-fold from the lowest calibration solution concentration) can further help establish a more accurate Nernst slope. Since Na+ activity is typically highly susceptible to background noise, a modified Aedes saline consisting of less Na+ is used (Table 2). It is also important to keep a distance from the Faraday cage during measurements to avoid interference and reduce any background. Additionally, if the organ still produces spontaneous contractions when adhered to the poly-L-lysine at the bottom of the dish, adding a calcium chelator to the saline will further help to prevent this. If examining ion transport along the rectal pad epithelia, as shown in the results presented here, it is important to identify the “hotspot” site exhibiting greatest ion activity by moving the microelectrode tip in small increments along the haemolymph-facing side of the rectal pad. If unable to detect any large changes in ion activity, the organ may have been damaged or the microelectrode tip is not in the same three dimensional plane as the rectal pad opening, and thus another mosquito should be dissected.
The SIET can be used for various experiments, such as examining ion activity in mosquitoes reared in different conditions25 or fed different diets (i.e., sucrose-fed vs. blood-fed vs. unfed), which can be useful to identify ion transport mechanisms critical following a bloodmeal. It is therefore important to ensure that measurements are obtained from the same site between samples. For instance, measurements can be taken from the ileum-rectum junction or at specific distances located anteriorly from this site (along the ileum) or posteriorly (along the rectal epithelium). These measurements can also be obtained along the rectal pad epithelium that requires different positioning of the organ in the dish. Additionally, it is important to note that the SIET is not restricted to measuring ion activity solely along the hindgut; other organs, including the midgut and MTs can also be examined. Similarly, K+, H+, NH4+, and Cl– flux can be measured using the SIET25,44,45 and measurements can be taken at set time intervals following a treatment44. This technique has been used on both larval and adult mosquitoes26,44, as well as various other insects, including Chironomus riparius46, Trichoplusia ni45,47 and Hexagenia rigida43. These experiments can be used together with the Ramsay assay and ISME to gain a better understanding of the role of different treatments and environmental factors on fluid secretion, ion concentration, and ion transport.
Lastly, contraction assays are useful to identify neuroendocrine factors that influence gut motility. It is crucial that the organ under study is not damaged since this assay relies on movements generated by musculature. Therefore, forceps should only be used to grab surrounding structures when transferring the organs to the assay dish. Gripping these adjacent organs with a pin inside the well also prevents them from moving and influencing movement of the isolated organ being examined. If no spontaneous contractions are observed, the tissue may have been damaged, and a new dissection is required. Other methods commonly used to examine insect gut contractile activity include the use of a force transducer32 or impedance monitor33. These methods can also be used to measure contraction rate in the mosquito hindgut; however, if the goal is to visually examine these changes through video recordings, the methods described in this protocol will be beneficial and can be used for further analysis48.
The four techniques outlined in this protocol have helped deduce the role of a number of neuropeptides in the excretory system of various insects. Using these methods in conjunction with additional techniques utilized by insect physiologists, such as quantitative PCR and immunohistochemistry, can provide a more comprehensive understanding of underlying pathways and signaling systems, which can serve as novel potential targets for vector control.
Declarações
The authors have nothing to disclose.
Acknowledgements
This research was funded by Natural Sciences and Engineering Research Council of Canada (NSERC) Discovery Grants to AD and J-PP. AL and FS received NSERC CGS-M awards in support of their graduate research.
Materials
| 1 mL syringes | Fisher Scientific | 14955456 | |
| 35 mm Petri dishes | Corning Falcon (Fisher Scientific) | C351008 | |
| Borosillicate glass capillary filamented tubes (OD 1 mm, ID 0.58 mm, length 100 mm) |
World Precision Instruments | 1B100F-4 | used for ISME reference electrodes |
| Borosillicate glass capillary filamented tubes (OD 2 mm, ID 1.12 mm, length 102 mm) |
World Precision Instruments | 1B200F-4 | used for SIET reference electrodes |
| Borosillicate glass capillary unfilamented tubes (OD 1.5 mm, ID 1.12 mm, length 100 mm) |
World Precision Instruments | TW150-4 | used for ISME and SIET electrodes |
| CO2 pad | Diamed | GEN59-114 | |
| Dimethyltrimethylsilylamine solution | Sigma-Aldrich | 41716 | |
| Faraday cage | Custom | Can be fabricated by local machine shop | |
| Ferric chloride | Sigma-Aldrich | 157740 | |
| Forceps (Dumont #5) | Fine Science Tools | 91150-20 | |
| Glass Petri dish | Fisher Scientific | 08-748A | |
| Hydrated mineral oil | Fisher Scientific | 8042-47-5 | Specific brand is not important |
| INFINITY1-2CB video camera | Luminera | INFINITY1-2CB | |
| Micromanipulators (left and right handed) | World Precision Instruments | MMJL and MMJR | Specific brand is not important so long as high quality manipulator |
| Mineral Oil, Light | Fisher Scientific | 0121-4 | |
| Minutien pins (0.1 mm stainless steel) | Fine Science Tools | 26002-10 | |
| Non-hardening modeling clay | Sargent Art | Specific brand is not important | |
| Olympus light microscope (FOR SIET) | Olympus | customized system | |
| Plastic Pasteur (transfer) pipette | Fisher Scientific | 13-711-7M | |
| Poly-L-lysine solution (0.1 mg/mL) | Sigma-Aldrich | A-005-M | 84 kDa |
| Polyvinyl chloride (PVC) | Sigma-Aldrich | 81395 | |
| Scalpel Blade | Fine Science Tools | 10050-00 | |
| Scalpel Handle | Fine Science Tools | 10053-09 | |
| Schneider's Drosophila medium | Sigma-Aldrich | S0146 | |
| SIET system | Applicable Electronics | customized system | Details available at: http://www.applicableelectronics.com/overview |
| Silver wire | World Precision Instruments | AGW1010 | |
| Sodium ionophore II cocktail A | Fluka | 99357 | |
| Standard polystyrene Petri (culture) dishes | Fisherbrand | FB012921 | Any size would work, but 60 mm dishes are good for both dissections and assay |
| Stereomicroscope with ocular micrometer | Nikon | SMZ800 | |
| Sutter P-97 Flaming Brown Pipette puller | Sutter Instruments | FGPN7 | |
| Sylgard 184 Silicone Elastomer Kit | Dow Chemical Company | NC9285739 | |
| Tetrahydrofuran | Sigma-Aldrich | 401757 | |
| VWR advanced hotplate stirrer – aluminum | VWR | 9578 | Specific brand is not important |
Referências
- Paluzzi, J. P. V. Anti-diuretic factors in insects: the role of CAPA peptides. General and Comparative Endocrinology. 176, 300-308 (2012).
- Beyenbach, K. W. Transport mechanisms of diuresis in Malpighian tubules of insects. Journal of Experimental Biology. 206, 3845-3856 (2003).
- Bradley, T. J. Physiology of osmoregulation in mosquitoes. Annual Review of Entomology. 32, 439-462 (1987).
- Patrick, M. L., Aimanova, K., Sanders, H. R., Gill, S. S. P-type Na+/K+-ATPase and V-type H+-ATPase expression patterns in the osmoregulatory organs of larval and adult mosquito Aedes aegypti. Journal of Experimental Biology. 209, 4638-4651 (2006).
- Coast, G. The endocrine control of salt balance in insects. General and Comparative Endocrinology. 152, 332-338 (2007).
- Maddrell, S. H. P., Overton, J. A. Methods for the study of fluid and solute transport and their control in insect Malpighian tubules. Methods in Enzymology. , 617-632 (1990).
- Beyenbach, K. W., Oviedo, A., Aneshansley, D. J. Malpighian tubules of Aedes aegypti: Five tubules, one function. Journal of Insect Physiology. 39, 639-648 (1993).
- Misyura, L., Yerushalmi, G. Y., Donini, A. A mosquito entomoglyceroporin, Aedes aegypti AQP5, participates in water transport across the Malpighian tubules of larvae. Journal of Experimental Biology. 220, 3536-3544 (2017).
- Drake, L. L., Rodriguez, S. D., Hansen, I. A. Functional characterization of aquaporins and aquaglyceroporins of the yellow fever mosquito, Aedes aegypti. Scientific Reports. 5, 7795 (2015).
- Ramsay, J. A. Active transport of water by the Malpighian tubules of the stick insect, Dixippus morosus (Orthoptera, Phasmidae). Journal of Experimental Biology. 31, 104-113 (1954).
- Jing, X., White, T. A., Yang, X., Douglas, A. E. The molecular correlates of organ loss: the case of insect Malpighian tubules. Biology Letters. 11, 20150154 (2015).
- Halberg, K. A., Terhzaz, S., Cabrero, P., Davies, S. A., Dow, J. A. T. Tracing the evolutionary origins of insect renal function. Nature Communications. 6, 6800 (2015).
- O’Connor, K. R., Beyenbach, K. W. Chloride channels in apical membrane patches of stellate cells of Malpighian tubules of Aedes aegypti. Journal of Experimental Biology. 204, 367-378 (2001).
- Ionescu, A., Donini, A. AedesCAPA-PVK-1 displays diuretic and dose dependent antidiuretic potential in the larval mosquito Aedes aegypti (Liverpool). Journal of Insect Physiology. 58, 1299-1306 (2012).
- Coast, G. M., Garside, C., Webster, S. G., Schegg, K. M., Schooley, D. A. Mosquito natriuretic peptide identified as a calcitonin-like diuretic hormone in Anopheles gambiae (Giles). Journal of Experimental Biology. 208, 3281-3291 (2005).
- Clark, T. M., Bradley, T. J. Additive effects of 5-HT and diuretic peptide on Aedes Malpighian tubule fluid secretion. Comparative Biochemistry and Physiology. 119, 599-605 (1998).
- Veenstra, J. A. Effects of 5-hydroxytryptamine on the Malpighian tubules of Aedes aegypti. Journal of Insect Physiology. 34, 299-304 (1988).
- Pollock, V. P., et al. Conservation of capa peptide-induced nitric oxide signalling in Diptera. Journal of Experimental Biology. 207, 4135-4145 (2004).
- Maddrell, S. H. P., Gardiner, B. O. C. The passive permeability of insect Malpighian tubules to organic solutes. Journal of Experimental Biology. 60, 641-652 (1974).
- O’Donnell, M. J. Too much of a good thing: How insects cope with excess ions or toxins in the diet. Journal of Experimental Biology. 212, 363-372 (2009).
- Sajadi, F., Curcuruto, C., Al Dhaheri, A., Paluzzi, J. P. V. Anti-diuretic action of a CAPA neuropeptide against a subset of diuretic hormones in the disease vector Aedes aegypti. Journal of Experimental Biology. 221, (2018).
- Donini, A., O’Donnell, M. J., Orchard, I. Differential actions of diuretic factors on the Malpighian tubules of Rhodnius prolixus. Journal of Experimental Biology. 211, 42-48 (2008).
- Donini, A., et al. Secretion of water and ions by Malpighian tubules of larval mosquitoes: Effects of diuretic factors, second messengers, and salinity. Physiological and Biochemical Zoology. 79, 645-655 (2006).
- Donini, A., O’Donnell, M. J. Analysis of Na+, Cl-, K+, H+ and NH4+ concentration gradients adjacent to the surface of anal papillae of the mosquito Aedes aegypti: Application of self-referencing ion-selective microelectrodes. Journal of Experimental Biology. 208, 603-610 (2005).
- Durant, A. C., Donini, A. Development of Aedes aegypti (Diptera: Culicidae) mosquito larvae in high ammonia sewage in septic tanks causes alterations in ammonia excretion, ammonia transporter expression, and osmoregulation. Scientific Reports. 9, (2019).
- Paluzzi, J. P. V., Vanderveken, M., O’Donnell, M. J. The heterodimeric glycoprotein hormone, GPA2/GPB5, regulates ion transport across the hindgut of the adult mosquito, Aedes aegypti. PLoS One. 9, 86386 (2014).
- Nguyen, H., Donini, A. Larvae of the midge Chironomus riparius possess two distinct mechanisms for ionoregulation in response to ion-poor conditions. American Journal of Physiology: Regulatory, Integrative and Comparative Physiology. 299, 762-773 (2010).
- Lajevardi, A., Paluzzi, J. -. P. V. Receptor characterization and functional activity of pyrokinins on the hindgut in the adult mosquito, Aedes aegypti. Frontiers in Physiology. 11, 490 (2020).
- Cook, B. J., Holman, G. M. The role of proctolin and glutamate in the excitation-contraction coupling of insect visceral muscle. Comparative Biochemistry and Physiology – Part C: Toxicology and Pharmacology. 80, 65-73 (1985).
- Robertson, L., Rodriguez, E. P., Lange, A. B. The neural and peptidergic control of gut contraction in Locusta migratoria: The effect of an FGLa/AST. Journal of Experimental Biology. 215, 3394-3402 (2012).
- Te Brugge, V. A., Schooley, D. A., Orchard, I. Amino acid sequence and biological activity of a calcitonin-like diuretic hormone (DH31) from Rhodnius prolixus. Journal of Experimental Biology. 211, 382-390 (2008).
- Lange, A. B., et al. The distribution and physiological effects of the myoinhibiting peptides in the kissing bug, Rhodnius prolixus. Frontiers in Neuroscience. 6, 98 (2012).
- Robertson, L., Chasiotis, H., Galperin, V., Donini, A. Allatostatin A-like immunoreactivity in the nervous system and gut of the larval midge Chironomus riparius: Modulation of hindgut motility, rectal K+ transport and implications for exposure to salinity. Journal of Experimental Biology. 217, 3815-3822 (2014).
- Kwon, H., Pietrantonio, P. V. Calcitonin receptor 1 (AedaeGPCRCAL1) hindgut expression and direct role in myotropic action in females of the mosquito Aedes aegypti (L.). Insect Biochemistry and Molecular Biology. 43, 588-593 (2013).
- Messer, A. C., Brown, M. R. Non-linear dynamics of neurochemical modulation of mosquito oviduct and hindgut contractions. Journal of Experimental Biology. 198, 2325-2336 (1995).
- Petzel, D. H., Berg, M. M., Beyenbach, K. W. Hormone-controlled cAMP-mediated fluid secretion in yellow-fever mosquito. The American Journal of Physiology: Regulatory, Integrative and Comparative Physiology. 253, 701-711 (1987).
- Schellinger, J. N., Rodan, A. R. Use of the Ramsay assay to measure fluid secretion and ion flux rates in the Drosophila melanogaster Malpighian tubule. Journal of Visualized Experiments: JoVE. (105), e53144 (2015).
- Paluzzi, J. -. P. V., Naikkhwah, W., O’Donnell, M. J. Natriuresis and diuretic hormone synergism in R. prolixus upper Malpighian tubules is inhibited by the anti-diuretic hormone, RhoprCAPA-α2. Journal of Insect Physiology. 58, 534-542 (2012).
- Rheault, M. R., O’Donnell, M. J., Morris, C. E. Organic cation transport by Malpighian tubules of Drosophila melanogaster: application of two novel electrophysiological methods. Journal of Experimental Biology. 207, 2173-2184 (2004).
- Sajadi, F., et al. CAPA neuropeptides and their receptor form an anti-diuretic hormone signaling system in the human disease vector, Aedes aegypti. Scientific Reports. 10, 1755 (2020).
- Rodan, A. R., Baum, M., Huang, C. L. The Drosophila NKCC Ncc69 is required for normal renal tubule function. American Journal of Physiology. 303, 883-894 (2012).
- Yu, M. J., Beyenbach, K. W. Effects of leucokinin-VIII on Aedes Malpighian tubule segments lacking stellate cells. Journal of Experimental Biology. 207, 519-526 (2004).
- Nowghani, F., et al. Impact of salt-contaminated freshwater on osmoregulation and tracheal gill function in nymphs of the mayfly Hexagenia rigida. Aquatic Toxicology. 211, 92-104 (2019).
- D’Silva, N. M., O’Donnell, M. J. Mechanisms of transport of H+, Na+ and K+, across the distal gastric caecum of larval Aedes aegypti. Journal of Insect Physiology. 121, 103997 (2020).
- Kolosov, D., O’Donnell, M. J. Malpighian tubules of caterpillars: blending RNAseq and physiology to reveal regional functional diversity and novel epithelial ion transport control mechanisms. Journal of Experimental Biology. 222, (2019).
- Jonusaite, S., Kelly, S. P., Donini, A. Tissue-specific ionomotive enzyme activity and K+ reabsorption reveal the rectum as an important ionoregulatory organ in larval Chironomus riparius exposed to varying salinity. Journal of Experimental Biology. 216, 3637-3648 (2013).
- O’Donnell, M. J., Ruiz-Sanchez, E. The rectal complex and Malpighian tubules of the cabbage looper (Trichoplusia ni): regional variations in Na+ and K+ transport and cation reabsorption by secondary cells. Journal of Experimental Biology. 218, 3206-3214 (2015).
- Simo, L., Park, Y. Neuropeptidergic control of the hindgut in the black-legged tick Ixodes scapularis. International Journal for Parasitology. 44, 819-826 (2014).

