Detection of SARS-CoV-2 Receptor-Binding Domain Antibody using a HiBiT-Based Bioreporter
Summary
The outlined protocol describes the procedure for producing the HiBiT-receptor-binding domain protein complex and its application for fast and sensitive detection of SARS-CoV-2 antibodies.
Abstract
The emergence of the COVID-19 pandemic has increased the need for better serological detection methods to determine the epidemiologic impact of severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2). The increasing number of SARS-CoV-2 infections raises the need for better antibody detection assays. Current antibody detection methods compromise sensitivity for speed or are sensitive but time-consuming. A large proportion of SARS-CoV-2-neutralizing antibodies target the receptor-binding domain (RBD), one of the primary immunogenic compartments of SARS-CoV-2. We have recently designed and developed a highly sensitive, bioluminescent-tagged RBD (NanoLuc HiBiT-RBD) to detect SARS-CoV-2 antibodies. The following text describes the procedure to produce the HiBiT-RBD complex and a fast assay to evaluate the presence of RBD-targeting antibodies using this tool. Due to the durability of the HiBiT-RBD protein product over a wide range of temperatures and the shorter experimental procedure that can be completed within 1 h, the protocol can be considered as a more efficient alternative to detect SARS-CoV-2 antibodies in patient serum samples.
Introduction
The recent emergence of a new coronavirus, SARS-CoV21, has caused more than 2,800,000 fatalities and 128 million infections as of March 30th, 20212. Due to the lack of a reliable and well-established treatment procedure for SARS-CoV-2 clinical therapies, many endeavors have been made to restrict further viral transmission and more importantly, to develop an effective and robust treatment or a vaccine3. To date, there are more than 50 COVID-19 vaccine candidates in trials reported by the World Health Organization4. Detection of antibodies against SARS-CoV-2 is of paramount importance to determine the long-term stability of humoral response upon administration of the vaccine as well as in recovered patients of COVID-195. Some studies have demonstrated that there is a possibility that recovered SARS-CoV-2 patients lose most of the RBD-binding antibodies after 1 year5,6,7,8,9. Further investigation is required to better understand lasting immunity, and more sensitive antibody detection platforms can help further such work. Reports of sustained immunity of mild SARS-CoV-2 infections, which suggest long-term antibody responses, is also an interesting and worthwhile area of study. A fast and accurate method of detection is essential for monitoring antibodies in individuals' sera to provide more information about immunity in the population.
Like other coronaviruses, SARS-CoV-2 uses protruding spike glycoprotein to bind to angiotensin-converting enzyme-2 (ACE2) to initiate a cascade of events that lead to the fusion of the viral and cell membranes6,7. Several studies have recently proved the RBD of the Spike protein to have a crucial role in eliciting powerful and specific antibody response against SARS-CoV28,9,10,11. In particular, correlations observed by Premkumar et al. between the titer of RBD-binding antibody and SARS-CoV-2 neutralization potency of patients' plasma are consistent with RBD being an immunogenic compartment of the virus structure9. With that in mind, many diagnostic tests available for SARS-CoV-2 antibody detection are time and cost-intensive, require a lengthy procedure of incubation and washing (enzyme-linked immunosorbent assay [ELISA]), or lack sensitivity and accuracy (lateral flow immunoassay [LFIA])12. Therefore, a quantitative and rapid complementary serological method of COVID-19-derived antibody detection with high sensitivity, fast response, and relatively low cost would serve the need for a reliable serologic test for SARS-CoV-2 epidemiologic surveillance.
Collectively, the limitations of current serological assays prompted the investigation of the bioluminescent reporting system as a potential diagnostic agent in future serosurveys. Bioluminescence is a naturally occurring enzyme/substrate reaction, with light emission. Nanoluc luciferase is the smallest (19 kDa), yet the brightest system compared to Renilla and firefly luciferase (36 kDa and 61 kDa, respectively)13,14. Further, Nanoluc has the highest signal to noise ratio and stability among the previously mentioned systems. The high signal intensity of Nanoluc supports the detection of even very low amounts of reporter fusions15. Nanoluc Binary Technology (NanoBiT) is a split version of the Nanoluc system, which is comprised of two segments: small BiT (11 amino acids; SmBiT) and large BiT (LgBiT) with relatively low-affinity interactions (KD = 190 µM ) to form a luminescent complex16. NanoBiT is extensively used in various studies involving the identification of protein-protein interactions15,17,18,19 and cellular signaling pathways11,20,21.
Recently, another small peptide with a distinctly higher affinity to LgBiT (KD = 0.7 nM ) was introduced, namely the HiBiT Nano-Glo system, in place of SmBiT. The high affinity and strong signal of the Nano-Glo "add-mix-read" assay makes HiBiT a suitable, quantitative, luminescent peptide tag. In this approach, the HiBiT tag is appended to the target protein by developing a construct imposing minimal structural interference. HiBiT-protein fusion would actively bind to the LgBiT counterpart, producing a highly active luciferase enzyme to generate detectable bioluminescence in the presence of detection reagents (Figure 1). Similarly, we developed a HiBiT Nano-Glo-based system to readily measure the neutralizing antibody titer in the sera of SARS-CoV-2 recovered individuals and recently developed a HiBiT-tagged SARS-CoV-2 RBD. This paper describes the protocol for producing the HiBiT-RBD bioreporter using standard laboratory procedures and equipment, and shows how this bioreporter can be used in a fast and efficient assay to detect SARS-CoV-2 RBD-targeting antibodies.
Protocol
NOTE: The protocol described below adheres to all ethics guidelines according to protocol code 20200371-01H.
1. Production and evaluation of the HiBiT-RBD bioreporter
- Producing a sufficient quantity of HiBiT-RBD bioreporter
- Prepare for cell culture
- Prepare complete Dulbecco's modified Eagle medium (DMEM) containing 10% fetal bovine serum and 1% penicillin/streptomycin. Then, warm the media in a 37 °C water bath.
- Turn the biological safety cabinet (BSC) on, and use 70% v/v ethanol for sterilizing the cabinet surface.
- Culture the cell line.
- Take out the cell line from -80 °C or liquid nitrogen, and thaw it in a 37 °C water bath.
NOTE: An appropriate cell line is easy to maintain in culture, has high transfection efficiency, and is suitable for exogenous protein production. Human embryonic kidney, HEK293 cells were used for this protocol. - Mix the thawed cells with at least 10 mL of complete medium, pipette the cell suspension to a 10 cm Petri dish, and swirl the plate to distribute cells in the dish uniformly. Place the dish in a cell culture incubator at 37 °C, 5% CO2, and 85-95% humidity.
- Observe the cells under the microscope until the confluency level reaches 80-90%. At high confluency, remove the medium, wash the cells with warm phosphate-buffered saline (PBS), and add 1 mL of 0.25% trypsin-ethylenediamine tetraacetic acid to detach the cells from the surface.
NOTE: The HEK293 cells are fairly easily detached. Hence, the washing step should be done very gently to prevent accidental detachment and loss of cells. - After approximately 5 min, look at the cells under a microscope. If all cells are floating, add at least 4 mL of medium, and transfer the cell suspension into a new sterile tube. Count the cells using a hemocytometer, and add 1 × 106 cells into each well of a 6-well plate for transfection.
NOTE: After 24 h, cells should be at 80% or more confluent.
- Take out the cell line from -80 °C or liquid nitrogen, and thaw it in a 37 °C water bath.
- Transfection of the HiBiT-RBD plasmid
- Use 1 µg of the HiBiT-RBD expression plasmid with a suitable transfection reagent. Incubate for 10-15 min at room temperature, and then add the total volume to each well of the plate, drop-wise.
NOTE: Follow the manufacturer's transfection protocol. In this case, a mixture (a specified amount) of the transfection reagent with DMEM was added to the diluted plasmid (1 µg) in DMEM (see the Table of Materials). Use a marker containing (e.g., green fluorescent protein [GFP]) plasmid as a control to monitor the transfection efficiency. - On the next day, replace the medium containing the transfection mixture with complete medium. Observe the transfection control well 48 h after transfection.
NOTE: If the transfection was positive and efficient (more than 80% GFP-positive), the cells should be ready for harvesting the HiBiT-RBD bioreporter. The construct also contains His-tag, which can be used to obtain purified protein in place of the total supernatant. - Collect the supernatant in 1.5 mL microtubes. Add 500 µL of 1x passive lysis buffer (PLB) to the cells; incubate and shake the plate for 15 min at room temperature for cell lysis.
NOTE: The supernatant and the lysate solution can be preserved at -20 °C with minor loss of integrity for at least 6 months. According to Azad et al.22, the reporter is stable at a wide range of pH (4-12) and temperature (4-42 °C).
- Use 1 µg of the HiBiT-RBD expression plasmid with a suitable transfection reagent. Incubate for 10-15 min at room temperature, and then add the total volume to each well of the plate, drop-wise.
- Prepare for cell culture
- Evaluation of the luminescent signal from the bioreporter by luciferase assay
- Preparing the reaction components
- Use the supernatant as the source of the bioreporter.
NOTE: Both supernatant and lysate contain the HiBiT-RBD bioreporter and can be used for the assay. However, the supernatant is recommended as the source due to reasons explained in the following notes. - Dilute the LgBiT and substrate to 1x before use (stock concentration is 100x).
NOTE: See the Table of Materials for details about the LgBiT.
- Use the supernatant as the source of the bioreporter.
- Luciferase assay
- Transfer 50 µL of the supernatant from each well or tube to a 96-well plate, and add 50 µL of 1x LgBiT to each well. Incubate for 5 min at room temperature.
- Open the luminometer software, add 50 µL of 1x substrate (furimazine) to each well, place the plate in the luminometer, and run the software.
NOTE: Add the substrate immediately before reading the plate to prevent consumption of the substrate by the active enzymes before signal measurement.
- Preparing the reaction components
2. Detecting anti-RBD antibody with a fast and sensitive assay
- HiBiT-RBD antibody detection assay
- Prepare the HiBiT-RBD bioreporter as described in section 1.1 of the protocol.
NOTE : It is recommended to use the supernatant for the following assay as it is simpler to collect and contains the mature glycosylated version of the protein. Moreover, the lysate has several other proteins that could interfere with the HiBiT-RBD-antibody interaction. - Combine 50 µL of the HiBiT-RBD-containing supernatant with 1 µg of the commercial SARS-CoV-2-RBD antibody in a 1.5 mL microtube. Add 20 µL of immunoglobulin-binding protein (protein G) to the solution. Bring up the total volume to 300 µL by adding PBS.
NOTE: The total volume of the mixture can be decreased to 150 µL. Lower total volumes are not recommended as it could result in inadequate mixing of antibodies with the bioreporter. - Incubate the tube(s) on a tube shaker or rotator for 30 min. Centrifuge at 12,000 × g for 30 s, discard the supernatant, and wash with PBS. Repeat the process three times to remove free HiBiT-RBD. Resuspend in 50 µL of PBS and transfer to a 96-well plate.
- Add 50 µL of 1x LgBiT and wait for 5 min. Then, add 50 µL of 1x NanoLuc substrate. Immediately read the luminescent signal with a luminometer.
- Prepare the HiBiT-RBD bioreporter as described in section 1.1 of the protocol.
3. High-throughput detection of the SARS-CoV-2-specific antibodies from patient serum samples
- Prepare larger quantities of the HiBiT-RBD bioreporter for a high-throughput assay by following section 1.1 of the protocol.
- Combine 20 µL of magnetic protein G with 50 µL of the HiBiT-RBD supernatant in a well of 96-well plate for each sample. Add 10 µL of the serum sample to each well and bring up the total volume to 150 µL by adding PBS.
NOTE: Use both nonspecific IgG (negative control) and neutralizing SARS-CoV-2 antibody (positive control) at 1 µg/mL concentration as described in step 2.1.2. Moreover, serum samples from vaccinated mice can also be assessed for antibodies. In this test, serum samples were obtained from Ottawa Hospital General Campus under an approved procedure and with informed consent from individuals. - Incubate for 30 min on a shaker at room temperature. Place the plate on a magnetic washer to precipitate the protein G-antibody complex.
NOTE: The specific structure of the magnetic washer will precipitate the complex on the side walls of each well. - Discard the solution in the middle section of each well, and add PBS for washing. Repeat the washing step at least three times to remove excess HiBiT-RBD. Add 50 µL of 1x PBS and 50 µL of 1x LgBiT. Incubate for at least 5 min at room temperature.
- Prepare the luminometer software, and then add 50 µL of 1x substrate. Place the plate in the machine, run the software, and record the signals. Compare the signals from serum samples, control samples, and background (empty wells).
Representative Results
The signals from both the HiBit-RBD-containing cell lysate and supernatant of the transfected cells were recorded (Figure 2) to evaluate the appropriate protein source. HiBiT-RBD and LgBit were separately used as controls, and the data showed low background compared to a strong signal when both parts were combined. Hence, HiBiT-RBD interaction with LgBiT is necessary to generate active enzyme for substrate digestion and bioluminescence activity (Figure 1).
The addition of protein G will help antibody precipitation (Figure 3). The assay was used to compare the signal from a commercial SARS-CoV-2-neutralizing antibody with a control IgG. The specific antibody signal was robust, while the control antibody had close to the luminescent background level (Figure 4A). Recombinant attenuated oncolytic vesicular stomatitis virus (VSV) with a mutation at position 51 of the M protein (Δ51) expressing exogenous RBD was used to vaccinate mice. The serum collected from vaccinated mice produced a robust signal compared to no signal in mice injected with control VSV (Figure 4B).
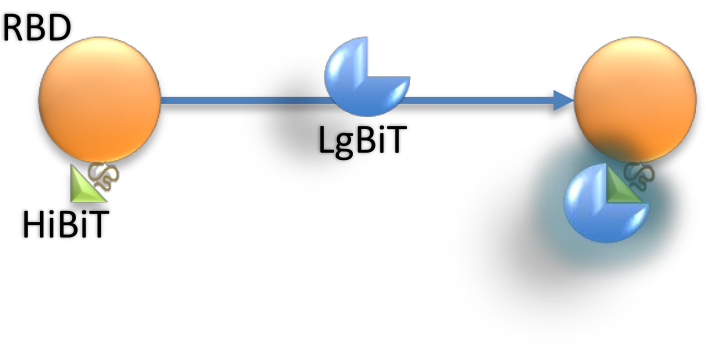
Figure 1: LgBiT interaction with HiBiT connected to RBD. Upon interaction of the small portion of the nanoluciferase, HiBiT, with the large subunit of the enzyme, LgBiT, the active enzyme complex can produce a luminescent signal after substrate consumption. The RBD does not interfere with this process. Abbreviations: RBD = receptor-binding domain. Please click here to view a larger version of this figure.
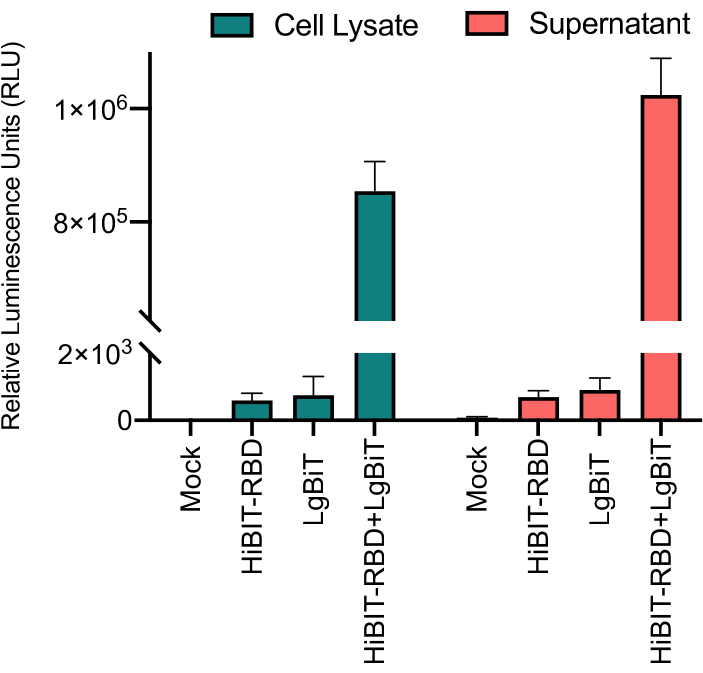
Figure 2: Robust reporter activity from both lysate and supernatant of transfected cells. The protein is present in both supernatant and lysate and produces a strong signal. Control groups' luminescent signals were close to the background. Error bars represent Standard Deviation (SD). Please click here to view a larger version of this figure.
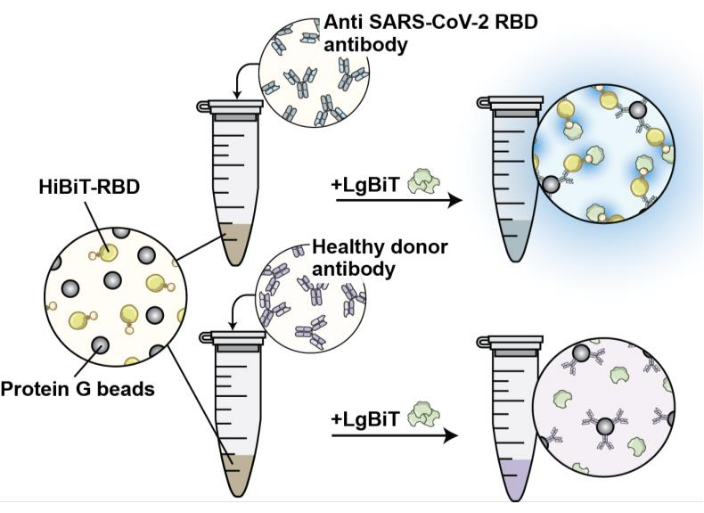
Figure 3: Schematic of the HiBiT-RBD interaction with antibodies bound to protein G. The schematic depicts the antibody precipitation by protein G and interaction with HiBiT-RBD. The addition of the LgBit to the mixture will produce a robust signal when the antibody is specific for RBD. Please click here to view a larger version of this figure.
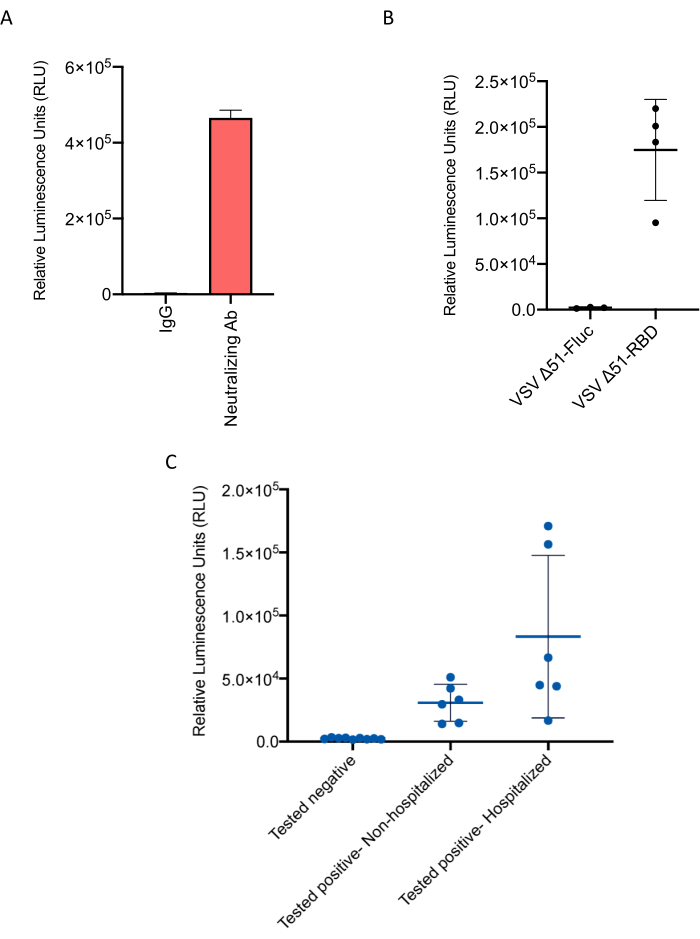
Figure 4: HiBiT-RBD bioreporter generates strong bioluminescence with purified neutralizing antibodies, vaccinated mouse serum, and patient serum samples. (A) HiBiT-RBD interacts with RBD-specific neutralizing antibody and generates significantly high signal compared to the negligible signal for nonspecific IgG. (B) The bioreporter can detect SARS-CoV-2 antibodies in vaccinated mouse serum. Abbreviations: RBD = receptor-binding domain; IgG = immunoglobulin G; Ab = antibody; VSV = vesicular stomatitis virus; Fluc = firefly luciferase. (C) Detection of the SARS-CoV-2 antibodies in patient serum samples, reproduced from Azad et al.22. Please click here to view a larger version of this figure.
Discussion
The increasing number of people infected with the SARS-CoV-2 and the ongoing effort for global vaccination necessitates sensitive and fast serologic tests that can be used in large-scale serosurveys. Recent research shows that split nanoluciferase-based bioreporters can be used to develop such assays. We recently developed the HiBiT-RBD bioreporter to design a test that can be used to detect SARS-CoV-2-specific antibodies in patient serum in a fast and reliable fashion (Figure 4C).
There are a few critical steps in this assay. Because the system’s efficiency depends on RBD protein expression, the protein levels should be validated by western blotting. Moreover, it is necessary to use a positive control, such as a commercial antibody against RBD, and a negative control antibody. Addition of a Nanoluciferase protein is recommended to be used as a positive control for bioluminescence detection. The protein product also contains a His-tag, which can be used for purification for a large number of serum samples.
There are several advantages to using this bioreporter compared to other competing methods. First, an experienced user can perform the complete assay procedure in less than one hour, which is considerably faster than existing tests such as ELISA. Second, the minimum preparation and testing requirements make this test highly valuable for large-scale production at a low cost. Moreover, the detection limit of the assay is as low as 1 ng of the SARS-CoV-2-neutralizing antibody as described by Azad et al.22. A drawback of this approach is the inability to differentiate between different antibody isotypes. Moving forward, the sensitivity of the test should be compared to other routinely used serologic tests.
Azad et al.22 had used serum samples from patients to evaluate the applicability of the assay. It is also essential that the system is tested for antibody detection in blood samples from patients. This tool could also be very impactful in the assessment of the correlation between the severity of COVID-19 and the presence of SARS-CoV-2-specific antibodies. Overall, such serologic tests could have a substantial impact in estimating the epidemiological impact of the SARS-CoV-2 and can be a convenient substitute for time-consuming and less sensitive detection methods.
Declarações
The authors have nothing to disclose.
Acknowledgements
We appreciate and thank the technical assistance of Xiaohong He, Ricardo Marius, Julia Petryk, Bradley Austin, and Christiano Tanese De Souza. We also thank Mina Ghahremani for Graphic Design. We would also like to thank all the individuals who participated and donated their blood samples for this study. DWC is supported in part by uOttawa Faculty and Department of Medicine.
Materials
| 5x Passive Lysis Buffer | Promega | E194A | 30 mL |
| Bio-Plex Handheld Magnetic Washer | Bio-Rad | 171020100 | |
| DMEM | Sigma | D6429-500ml | |
| Dual-Glo luciferase Assay System | Promega | E2940 | 100 mL kit |
| Fetal Bovine Serum (FBS) | Sigma | F1051 | |
| HiBiT-RBD Plasmid | gacggatcgggagatctcccgatcccctatggt gcactctcagtacaatctgctctgatgccgcata gttaagccagtatctgctccctgcttgtgtgttgg aggtcgctgagtagtgcgcgagcaaaattta agctacaacaaggcaaggcttgaccgacaa ttgcatgaagaatctgcttagggttaggcgttttg cgctgcttcgcgatgtacgggccagatatacgc gttgacattgattattgactagttattaatagt aatcaattacggggtcattagttcatagcccat atatggagttccgcgttacataacttacggtaa atggcccgcctggctgaccgcccaacgaccc ccgcccattgacgtcaataatgacgtatgttccc atagtaacgccaatagggactttccattgacgtc aatgggtggagtatttacggtaaactgcccact tggcagtacatcaagtgtatcatatgccaagta cgccccctattgacgtcaatgacggtaaatgg cccgcctggcattatgcccagtacatgaccttat gggactttcctacttggcagtacatctacgtat tagtcatcgctattaccatggtgatgcggtttt ggcagtacatcaatgggcgtggatagcggtttg actcacggggatttccaagtctccaccccattg acgtcaatgggagtttgttttggcaccaaaatc aacgggactttccaaaatgtcgtaacaactccg ccccattgacgcaaatgggcggtaggcgtgta cggtgggaggtctatataagcagagctctctgg ctaactagagaacccactgcttactggcttatcg aaattaatacgactcactatagggagacccaa gctggctagcgtttaaacttaagcttggtaccga gctcggatccgccaccATGGAGACAGA CACACTCCTGCTATGGGTACTGC TGCTCTGGGTTCCAGGTTCCAC TGGTGACtctggctctagcggctctggctct agcggcggcATGGTGAGCGGCTG GCGGCTGTTCAAGAAGATTAGC tctagcggcGACTACAAGGACC ACGACGGTGACTACAAGGACCA CGACATCGACTACAAGGACGAC GACGACAAGggcagcggctccggca gcagcggaggaggaggctctggaggagga ggctctagcggcggcaacatcacaaatctgtg cccattcggcgaggtgtttaacgccaccagat ttgccagcgtgtatgcctggaaccggaagaga atctctaattgcgtggccgactatagcgtgct gtacaatagcgcctccttctctacctttaagt gctatggcgtgtcccccacaaagctgaacgac ctgtgcttcaccaacgtgtacgccgactcttttgt gatcaggggcgatgaggtgcgccagatcgc acctggacagacaggcaagatcgccgactac aactataagctgccagacgatttcaccggct gcgtgatcgcctggaatagcaacaatctggatt ccaaagtgggcggcaactacaattatctgtac cggctgttcagaaagagcaacctgaagccctt tgagcgggatatcagcacagagatctaccag gcaggctccaccccttgcaacggagtggagg gcttcaattgttattttcccctgcagagctacggc ttccagcctacaaatggcgtgggctatcagcca tacagggtggtggtgctgtcctttgagctgctg cacgcacctgcaaccgtgtcctctggacacatc gagggccgccacatgctggagatgggccatc atcaccatcatcaccaccaccaccactgatag cggccgctcgagtctagagggcccgtttaaac ccgctgatcagcctcgactgtgccttctagtt gccagccatctgttgtttgcccctcccccgtg ccttccttgaccctggaaggtgccactcccac tgtcctttcctaataaaatgaggaaattgcat cgcattgtctgagtaggtgtcattctattctgggg ggtggggtggggcaggacagcaaggggga ggattgggaagacaatagcaggcatgctggg gatgcggtgggctctatggcttctgaggcggaa agaaccagctggggctctagggggtatcccca cgcgccctgtagcggcgcattaagcgcggcg ggtgtggtggttacgcgcagcgtgaccgctac acttgccagcgccctagcgcccgctcctttcg ctttcttcccttcctttctcgccacgttcgccggctt tccccgtcaagctctaaatcgggggctcccttta gggttccgatttagtgctttacggcacctcgacc ccaaaaaacttgattagggtgatggttcacgta gtgggccatcgccctgatagacggtttttcgcc ctttgacgttggagtccacgttctttaatagtg gactcttgttccaaactggaacaacactcaacc ctatctcggtctattcttttgatttataagggatttt gccgatttcggcctattggttaaaaaatgagctg atttaacaaaaatttaacgcgaattaattctgt ggaatgtgtgtcagttagggtgtggaaagtccc caggctccccagcaggcagaagtatgcaaag catgcatctcaattagtcagcaaccaggtgtgg aaagtccccaggctccccagcaggcagaagt atgcaaagcatgcatctcaattagtcagcaac catagtcccgcccctaactccgcccatcccgc ccctaactccgcccagttccgcccattctccgcc ccatggctgactaattttttttatttatgcagaggc cgaggccgcctctgcctctgagctattccagaa gtagtgaggaggcttttttggaggcctaggcttttg caaaaagctcccgggagcttgtatatccattttc ggatctgatcaagagacaggatgaggatcgttt cgcatgattgaacaagatggattgcacgcagg ttctccggccgcttgggtggagaggctattcggc tatgactgggcacaacagacaatcggctgctct gatgccgccgtgttccggctgtcagcgcagggg cgcccggttctttttgtcaagaccgacctgtccgg tgccctgaatgaactgcaggacgaggcagcg cggctatcgtggctggccacgacgggcgttcct tgcgcagctgtgctcgacgttgtcactgaagcg ggaagggactggctgctattgggcgaagtgcc ggggcaggatctcctgtcatctcaccttgctcctg ccgagaaagtatccatcatggctgatgcaatg cggcggctgcatacgcttgatccggctacctgc ccattcgaccaccaagcgaaacatcgcatcg agcgagcacgtactcggatggaagccggtct tgtcgatcaggatgatctggacgaagagcat caggggctcgcgccagccgaactgttcgcca ggctcaaggcgcgcatgcccgacggcgagg atctcgtcgtgacccatggcgatgcctgcttg ccgaatatcatggtggaaaatggccgctttt ctggattcatcgactgtggccggctgggtgt ggcggaccgctatcaggacatagcgttggct acccgtgatattgctgaagagcttggcggcg aatgggctgaccgcttcctcgtgctttacgg tatcgccgctcccgattcgcagcgcatcgcc ttctatcgccttcttgacgagttcttctgagcg ggactctggggttcgaaatgaccgaccaag cgacgcccaacctgccatcacgagatttcgat tccaccgccgccttctatgaaaggttgggctt cggaatcgttttccgggacgccggctggatga tcctccagcgcggggatctcatgctggagt tcttcgcccaccccaacttgtttattgcagctta taatggttacaaataaagcaatagcatcacaa atttcacaaataaagcatttttttcactgcatt ctagttgtggtttgtccaaactcatcaatgtat cttatcatgtctgtataccgtcgacctctagct agagcttggcgtaatcatggtcatagctgtttc ctgtgtgaaattgttatccgctcacaattccacac aacatacgagccggaagcataaagtgtaaag cctggggtgcctaatgagtgagctaactcacat taattgcgttgcgctcactgcccgctttccagtc gggaaacctgtcgtgccagctgcattaatgaa tcggccaacgcgcggggagaggcggtttgcg tattgggcgctcttccgcttcctcgctcactgactc gctgcgctcggtcgttcggctgcggcgagcggt atcagctcactcaaaggcggtaatacggttatc cacagaatcaggggataacgcaggaaagaa catgtgagcaaaaggccagcaaaaggccag gaaccgtaaaaaggccgcgttgctggcgtttt tccataggctccgcccccctgacgagcatcac aaaaatcgacgctcaagtcagaggtggcgaa acccgacaggactataaagataccaggcgtt tccccctggaagctccctcgtgcgctctcctgtt ccgaccctgccgcttaccggatacctgtccgcc tttctcccttcgggaagcgtggcgctttctcat agctcacgctgtaggtatctcagttcggtgtag gtcgttcgctccaagctgggctgtgtgcacgaa ccccccgttcagcccgaccgctgcgccttatcc ggtaactatcgtcttgagtccaacccggtaag acacgacttatcgccactggcagcagccactg gtaacaggattagcagagcgaggtatgtaggc ggtgctacagagttcttgaagtggtggcctaact acggctacactagaagaacagtatttggtatc tgcgctctgctgaagccagttaccttcggaaa aagagttggtagctcttgatccggcaaacaaa ccaccgctggtagcggtggtttttttgtttgca agcagcagattacgcgcagaaaaaaaggat ctcaagaagatcctttgatcttttctacggggt ctgacgctcagtggaacgaaaactcacgttaa gggattttggtcatgagattatcaaaaaggatct tcacctagatccttttaaattaaaaatgaagtt ttaaatcaatctaaagtatatatgagtaaactt ggtctgacagttaccaatgcttaatcagtgagg cacctatctcagcgatctgtctatttcgttcatcca tagttgcctgactccccgtcgtgtagataactac gatacgggagggcttaccatctggccccagtg ctgcaatgataccgcgagacccacgctcacc ggctccagatttatcagcaataaaccagccag ccggaagggccgagcgcagaagtggtcctg caactttatccgcctccatccagtctattaattgtt gccgggaagctagagtaagtagttcgccagtt aatagtttgcgcaacgttgttgccattgctacag gcatcgtggtgtcacgctcgtcgtttggtatgg cttcattcagctccggttcccaacgatcaaggc gagttacatgatcccccatgttgtgcaaaaaag cggttagctccttcggtcctccgatcgttgtca gaagtaagttggccgcagtgttatcactcatggt tatggcagcactgcataattctcttactgtcatg ccatccgtaagatgcttttctgtgactggtgagta ctcaaccaagtcattctgagaatagtgtatgcg gcgaccgagttgctcttgcccggcgtcaatacg ggataataccgcgccacatagcagaactttaa aagtgctcatcattggaaaacgttcttcggggc gaaaactctcaaggatcttaccgctgttgagat ccagttcgatgtaacccactcgtgcacccaact gatcttcagcatcttttactttcaccagcgtttc tgggtgagcaaaaacaggaaggcaaaatgc cgcaaaaaagggaataagggcgacacgga aatgttgaatactcatactcttcctttttcaat attattgaagcatttatcagggttattgtc tcatgagcggatacatatttgaatgtattt agaaaaataaacaaataggggttccgcgca catttccccgaaaagtgccacctgacgtc | ||
| LgBiT | Promega | N3030 | |
| penicillin Streptomycin | Thermo Fisher Scientific | 15140122 | |
| Pierce Protein G Magnetic Beads | Thermo Fisher Scientific | 88848 | |
| PolyJet In Vitro DNA Transfection Reagent | Signagen | SL100688.5 | |
| SARS-CoV-2 (2019-nCoV) Spike Neutralizing Antibody, Mouse Mab | SinoBiological | 40592-MM57 | |
| Synergy Mx Microplate Reader | BioTek | 96-well plate reader luminometer | |
| Trypsin-EDTA | Thermo Fisher Scientific | 2520056 | 0.25% |
Referências
- Ullah, H., Ullah, A., Gul, A., Mousavi, T., Khan, M. W. Novel coronavirus 2019 (COVID-19) pandemic outbreak: A comprehensive review of the current literature. Vacunas. , (2020).
- Coronavirus update (Live). Worldometer Available from: https://www.worldometers.info/coronavirus/ (2021)
- Cacciapaglia, G., Cot, C., Sannino, F. Second wave COVID-19 pandemics in Europe: a temporal playbook. Scientific Reports. 10 (1), 15514 (2020).
- COVID-19 vaccines. World Health Organization Available from: https://www.who.int/emergencies/diseases/novel-coronavirus-2019/covid-19-vaccines (2021)
- Hueston, L., et al. The antibody response to SARS-CoV-2 infection. Open Forum Infectious Diseases. 7 (9), (2020).
- Lan, J., et al. Structure of the SARS-CoV-2 spike receptor-binding domain bound to the ACE2 receptor. Nature. 581 (7807), 215-220 (2020).
- Azad, T., et al. Implications for SARS-CoV-2 vaccine design: fusion of Spike glycoprotein transmembrane domain to receptor-binding domain induces trimerization. Membranes. 10 (9), 215 (2020).
- Piccoli, L., et al. Mapping neutralizing and immunodominant sites on the SARS-CoV-2 Spike receptor-binding domain by structure-guided high-resolution serology. Cell. 183 (4), 1024-1042 (2020).
- Premkumar, L., et al. The receptor-binding domain of the viral spike protein is an immunodominant and highly specific target of antibodies in SARS-CoV-2 patients. Science Immunology. 5 (48), (2020).
- Walls, A. C., et al. Elicitation of potent neutralizing antibody responses by designed protein nanoparticle vaccines for SARS-CoV-2. Cell. 183 (5), 1367-1382 (2020).
- Azad, T. Nanoluciferase complementation-based biosensor reveals the importance of N- linked glycosylation of SARS-CoV-2 Spike for viral entry. Mol Ther. , 0074-0075 (2021).
- Bastos, M. L., et al. Diagnostic accuracy of serological tests for covid-19: systematic review and meta-analysis. BMJ. 370, 2516 (2020).
- Bioluminescent Reporters | Reporter Gene Applications | An Introduction to Reporter Genes. Promega Available from: https://www.promega.ca/resources/guides/cell-biology/bioluminescent-reporters/#references-6d127eb8-eeae-40b7-86e9-fe300545e8fa (2021)
- Fleiss, A., Sarkisyan, K. S. A brief review of bioluminescent systems. Current Genetics. 65 (4), 877-882 (2019).
- Nouri, K., et al. A kinome-wide screen using a NanoLuc LATS luminescent biosensor identifies ALK as a novel regulator of the Hippo pathway in tumorigenesis and immune evasion. The FASEB Journal. 33 (11), 12487-12499 (2019).
- Boute, N., et al. NanoLuc Luciferase – a multifunctional tool for high throughput antibody screening. Frontiers in Pharmacology. 7, 27 (2016).
- Nouri, K., et al. Identification of celastrol as a novel YAP-TEAD inhibitor for cancer therapy by high throughput screening with ultrasensitive YAP/TAZ-TEAD biosensors. Cancers. 11 (10), 1596 (2019).
- Azad, T., et al. SARS-CoV-2 S1 NanoBiT: A nanoluciferase complementation-based biosensor to rapidly probe SARS-CoV-2 receptor recognition. Biosensors and Bioelectronics. 180, 113122 (2021).
- Brown, E. E. F., et al. Characterization of critical determinants of ACE2-SARS CoV-2 RBD interaction. International Journal of Molecular Sciences. 22 (5), 2268 (2021).
- Azad, T., et al. A gain-of-functional screen identifies the Hippo pathway as a central mediator of receptor tyrosine kinases during tumorigenesis. Oncogene. 39 (2), 334-355 (2020).
- Schwinn, M. K., et al. CRISPR-Mediated tagging of endogenous proteins with a luminescent peptide. ACS Chemical Biology. 13 (2), 467-474 (2018).
- Azad, T., et al. A high-throughput NanoBiT-based serological assay detects SARS-CoV-2 seroconversion. Nanomaterials. 11 (3), 807 (2021).

