A Microphysiological System to Study Leukocyte-Endothelial Cell Interaction during Inflammation
Summary
In this protocol, a biomimetic microfluid assay, which can reproduce a physiologically relevant microvascular environment and reproduce the entire leukocyte adhesion/migration cascade, is employed to study leukocyte-endothelial cell interactions in inflammatory disease.
Abstract
Leukocyte-endothelial cell interactions play an important role in inflammatory diseases such as sepsis. During inflammation, excessive migration of activated leukocytes across the vascular endothelium into key organs can lead to organ failure. A physiologically relevant biomimetic microfluidic assay (bMFA) has been developed and validated using several experimental and computational techniques, which can reproduce the entire leukocyte rolling/adhesion/migration cascade to study leukocyte-endothelial cell interactions. Microvascular networks obtained from in vivo images in rodents were digitized using a Geographic Information System (GIS) approach and microfabricated with polydimethylsiloxane (PDMS) on a microscope slide. To study the effect of shear rate and vascular topology on leukocyte-endothelial cell interactions, a Computational Fluid Dynamics (CFD) model was developed to generate a corresponding map of shear rates and velocities throughout the network. The bMFA enables the quantification of leukocyte-endothelial cells interactions, including rolling velocity, number of adhered leukocytes in response to different shear rates, number of migrated leukocytes, endothelial cell permeability, adhesion molecule expression and other important variables. Furthermore, by using human-related samples, such as human endothelial cells and leukocytes, bMFA provides a tool for rapid screening of potential therapeutics to increase their clinical translatability.
Introduction
Inflammation is the host response to infection and injury, and the endothelium plays an important role in the inflammatory response1,2,3. Inflammatory dysregulation is the underlying cause of a number of disease pathologies such as sepsis, cardiovascular diseases, asthma, inflammatory bowel disease, cancer and COVID-19. Leukocyte-endothelial cell interactions play a central role in these inflammatory diseases. During inflammation, the release of PAMPS (pathogen-associated molecular patterns) from pathogens or DAMPS (damage-associated molecular patterns) from injured tissues activate immune cells to release cytokines/chemokines and other proinflammatory mediators that lead to the activation of endothelium, resulting in alterations in vascular endothelium barrier function and increased permeability3,4. Increased activation of endothelial cells during inflammation results in enhanced leukocyte-endothelial cell interaction leading to excessive migration of activated leukocytes across the vascular endothelium into key organs1,5,6,7.
The recruitment of leukocytes is initiated by chemically diverse chemoattractants composed of bioactive lipids, cytokines, chemokines and complement components8,9. Leukocyte recruitment is a multi-step process that includes five discrete steps: 1) leukocyte margination and capture/attachment, 2) rolling, 3) firm arrest, 4) spreading and crawling and 5) extravasation/migration (Figure 1). Each step of this process requiring crosstalk between leukocytes and endothelial cells to orchestrate this dynamic phenomenon1,9. Ultimately, arrested leukocytes extravasate to inflamed tissues across endothelium via a multi-step process controlled by concurrent chemoattractant-dependent signals, adhesive events and hemodynamic shear forces1,9,10,11,12.
Given the central role of shear stress in regulating endothelial cell function and the significance of the leukocyte-endothelium cell interactions13, several in vitro models have been developed during the last few decades to study various aspects of the leukocyte migration cascade in a more controlled environment14. Traditional fluidic devices to study leukocyte-endothelial cell interactions can be classified into two broad categories14: a) devices for studying leukocyte rolling, adhesion and adhesion molecule expression such as parallel plate flow chambers and b) devices for studying leukocyte migration under static conditions such as transwell chambers. Systems like parallel plate flow chambers have been used to study the roles of adhesion molecules and their ligands in the adhesion cascade under shear forces15. However, a significant drawback is that these simplistic, idealized devices (e.g., straight channel) are not able to reproduce the scale and geometry of the in vivo microvasculature (e.g., successive vascular bifurcations, vascular morphology) and the resulting flow conditions (e.g., converging or diverging flows at bifurcations). As a result, these devices can only model adhesion but not transmigration. Transwell chambers can only study transmigration under static conditions without considering the in vivo geometrical features and flow conditions. Thus, these traditional models do not mimic the microenvironment of living tissues or resolve adhesion and migration cascade in a single assay6.
To address this limitation, we have developed and extensively validated a novel 3D biomimetic microfluidic assay (bMFA) (Figure 2), which realistically reproduces in vivo microvascular networks on a chip16,17,18. The protocol for microfabrication of this device has been published previously17 and is only briefly described here. The microvasculature of mouse cremaster muscle was digitized using a modified Geographic Information System (GIS) approach19. Then, the synthetic microvascular network was generated on polydimethylsiloxane (PDMS) using soft-lithography processes based on the digitized microvascular network14,17,20,21,22. Briefly, the digitized network images were printed on Mylar film, which was then used as a mask to pattern a SU-8 positive photoresist on top of a silicon wafer to create the masters for fabrication. Microfabricated pillars (10 µm diameter, 3 µm tall) were used to create the 3 µm high and 100 µm wide pores, an optimum size for leukocyte migration23,24,25, connecting the vascular channels and tissue compartments. PDMS was prepared according to the manufacturer's instructions and poured over the developed masters. Further, the PDMS was degassed and allowed to cure overnight in an oven (65 °C) to create complementary microchannels in PDMS. Subsequently, the cured PDMS was peeled from the SU-8 master, followed by punching ports for inlets/outlets. Then, the PMDS was plasma bonded to a glass slide. The surface of the microfluidic device comprises native glass and PDMS. In order to promote cell attachment, spreading and proliferation, extracelluar matrix (ECM) coating is required. The bMFA includes a microvascular network and a tissue compartment connected via 3 µm high and 100 µm wide pores (Figure 2). This microfluidic system reproduces the entire leukocyte adhesion/migration cascade in a physiologically relevant 3D environment of a complete microvascular network with interconnecting vessels and bifurcations, including circulation, margination, rolling, adhesion and migration of leukocytes into the extra-vascular tissue compartment in a single system14,16,17,21,26.
It should be noted that even when the flow rate at the inlet of bMFA is fixed, the flow conditions in the network vary at different locations and cannot be calculated by a simple mathematical formula. A computational fluid dynamics (CFD)-based model was developed to calculate different flow parameters (e.g., shear stress, shear rate, velocity) at different locations in the network. This CFD model was used to simulate the dye perfusion patterns and flow parameters in the bMFA. Cross-validation with experimental results suggested that the flow resistances across the network are well predicted by the computational model (Figure 3)17. This CFD model was then used to estimate velocity and shear rate profile in every vessel of bMFA (Figure 4), allowing analysis of the effects of shear flow and geometry on leukocyte rolling, adhesion and migration16. Leukocytes preferentially adhere near bifurcations and in low shear regions in vivo, and these spatial patterns of leukocyte adhesion were successfully demonstrated in bMFA using neutrophils (Figure 5)16. This paper describes the protocol for preparing bMFA to study leukocyte-endothelial cell interaction under inflammatory conditions using human lung microvascular endothelial cells (HLMVEC) and human neutrophils. Microphysiological systems, such as the bMFA, can be used to study endothelial cell interactions with different types of cells such as neutrophils, monocytes, lymphocytes and tumor cells18,27,28,29,30. The bMFA can be seeded with primary endothelial cells from different organs (e.g., lung vs. brain) and different species (e.g., human vs. murine endothelial cells), as well as endothelial cell lines21,27,31,32. The bMFA can be used to study multiple cellular responses, cell-cell interactions, barrier function, drug delivery and drug toxicity.
Protocol
Heparinized human blood is obtained for neutrophil isolation from healthy adult donors (males and females, aged between 21 and 60 years old), following informed consent as approved by the Institutional Review Board of Temple University (Philadelphia, PA, USA).
1. Priming and coating the device with human fibronectin
NOTE: The bMFA has two inlet ports and two outlet ports connected to the vascular compartment. It also has one port connected to the tissue compartment (Figure 2).
- Insert tubing (O.D. of 0.06" and I.D. of 0.02") (Table of Materials) to all the ports except for one inlet port with fine-point forceps. Clamp the two outlet ports and the tissue compartment port with jaw clamps. Ensure that each tubing is ~1 inch in length.
- Dilute human fibronectin to 100 µg/mL with PBS from fibronectin stock solution (1mg/mL). Load a 1 mL syringe with prepared fibronectin solution. Connect the syringe to a 24 G blunt needle and a ~4-inch long tubing.
NOTE: To prevent crosslinking, do not over mix/pipette human fibronectin. Other extracellular matrices (ECM) such as collagen may be used for different cell types. - Insert the tubing into the open inlet port, push the plunger until human fibronectin comes out of the other inlet port, then clamp it. Repeat this process for the rest of the ports until all the channels, tissue compartment and tubing are filled with the human fibronectin solution. Remove the needle but keep the 4-inch long tubing inserted and unclamped.
NOTE: Make sure the tubing is filled with fibronectin before it is clamped. - For degassing, connect the unclamped tubing to a compressed nitrogen tank through the Pneumatic Primer, adjust the pressure to ~5 psi and run for ~15 min.
- After 15 min, check under the microscope to make sure there are no air bubbles trapped in the device. If there are air bubbles trapped in the channel or tissue compartment, reconnect the device to the Pneumatic Primer for degassing again, i.e., repeat step 1.4.
NOTE: Inverted microscope is used in all the steps involving microscopy in this protocol. - Remove the device from the Pneumatic Primer. Incubate the device at 37 °C for 1 h, then flush all the channels and tissue compartment with pre-warmed HLMVEC culture media (Table of Materials), similar to step 1.3. The bMFA is now ready for cell seeding.
NOTE: Before removing/inserting a tubing from port, first place a drop of water with a pipette around the base of the inlet port tubing to prevent air from entering the device.
2. Seeding bMFA with HLMVEC
- Culture HLMVEC to 60%- 80% confluency according to the vendor's protocol in a T-25 flask.
- Aspirate HLMVEC culture media from cell culture flask. Wash twice with PBS.
- Add 2 mL of pre-warmed trypsin-EDTA solution (37 °C) in the T-25 flask. Incubate for 3-5 min in an incubator (37 °C, 5% CO2) until most cells are detached from the flask.
NOTE: Incubation time may vary for different cell types and trypsin-EDTA concentration. - Add 2 mL of Trypsin Neutralization Solution (TNS, 37 °C) to the flask to neutralize trypsin-EDTA.
- Gently tap the flask to help dissociate the cells. Then, transfer the cell suspension solution to a 15 mL conical tube.
- Centrifuge for 5 min at 150 x g to pellet the endothelial cells. Remove the supernatant and resuspend cells in 3 mL of cell culture media. Count the number of cells with a hemocytometer.
- Centrifuge again for 5 min at 150 x g to pellet the cells. Remove the supernatant and resuspend the cells to the desired seeding density of 20 x 106/mL.
NOTE: Depending on the cell types and confluency, about 2 x 106 endothelial cells can be collected from two T-25 flasks, and with the seeding density of 20 x 106/mL, 100 µL of the cell suspension (2 x 106 endothelial cells), is sufficient to seed 5 devices. - Mount a 1 mL syringe in the programmable syringe pump, attach tubing to the blunt needle.
- Draw ~20 µL of the cell suspension into the tubing, making sure the cell suspension is only in the tubing, not in the syringe barrel.
- Take the clamp off an outlet port of the device. Connect the tubing to one inlet of the device. Make sure no air bubbles are introduced into the channel.
- Start the pump with a flow rate of 4-8 µL/min. Observe the device under a microscope.
- When the channels are filled with cells, stop the pump, clamp the outlet, and cut the inlet tubing.
NOTE: Take care not to introduce air bubbles into the device. - Put the device in the incubator for 4 h at 5% CO2 and 37 °C.
- After 4 h incubation, prepare another syringe filled with fresh cell culture media. Mount the syringe on a syringe pump, connect the syringe to the inlet port and remove the clamp from an outlet port.
- Run fresh media through the device for about 5 min at 4-8 µL/min to wash out floating/unattached cells.
3. Cell culture under flow for 48 h
- Prepare a syringe filled with fresh cell culture media. Mount the syringe in a syringe pump, connect the syringe to one inlet port and keep an outlet open. Put the device in the incubator (5% CO2, 37 °C).
- Program the syringe pump for culturing endothelial cells under flow using the following instructions mentioned in Table 1.
- Check bMFA under a microscope after 48 h of culture under flow. HLMVEC should cover vascular channels forming a complete lumen (Figure 6).
NOTE: Other flow regimes may be required for different cell types.
4. Cytokine and/or therapeutic treatment
NOTE: As an example, this section describes using bMFA to study the impact of treating the cells with Tumor Necrosis Factor-alpha (TNF-α) and a novel anti-inflammatory inhibitor (Protein Kinase C delta-TAT peptide inhibitor, PKCδ-i)18,27,32,33,34,35,36.
- Prepare three different bMFA devices using the protocol above (section 1-3).
- Load three 1 mL syringe with cell culture media, (TNF-α) (10 ng/mL), TNF-α (10 ng/mL) + PKCδ-i (5 µM), respectively.
NOTE: Cytokines are reconstituted in cell culture media. - Connect the three syringes to three bMFA devices. Treat HLMVEC with buffer TNF-α or TNF-α + inhibitor for 4 h at 0.1 µL/min.
NOTE: Based on specific study requirements, other cytokines or cytokine cocktails (e.g., TNF-α + Interleukin 1 beta (IL1-β) + Interferon-gamma (IFN-γ)), may be used to treat endothelial cells.
5. Human neutrophil isolation
- Use all reagents at room temperature (RT). Reagents include leukocyte isolation media , 1x HEPES buffer with Ca2+/Mg2+ (Table 2), 6% Dextran in normal saline (0.9% NaCl), 3.6% NaCl solution and de-ionized water (DI) water.
- Pipette 20 mL of leukocyte isolation media into a 50 mL conical tube and carefully layer 25 mL of blood over the leukocyte isolation media. Centrifuge for 40 min at 427 x g at RT.
NOTE: Perform this step slowly and carefully to avoid mixing the blood and leukocyte isolation media. - Aspirate down to ~5 mL above red blood cells (RBC) layer. The white buffy layer on top of the RBC is predominantly neutrophils.
- Add HEPES buffer to double the current volume (existing volume: HEPES buffer = 1:1).
- Add an amount of 6% Dextran, which will be 20% of the final volume. (current volume/4 = volume of added 6% Dextran).
- Invert the tube gently a few times to mix well and let the RBC sediment (~25 min).
- Carefully pipette the upper layer (neutrophil-rich layer) and transfer to a new 50 mL tube, be careful not to contaminate with sedimented RBC.
- Centrifuge for 10 min at 315 x g at RT. Remove the supernatant and resuspend the pellet in 8 mL of HEPES buffer.
- To lyse the residual RBC, add 24 mL of DI water and gently mix for 50 s. Immediately, add 8 mL of 3.6% NaCl solution, mix and centrifuge for 10 min at 315 x g at RT.
- Remove the supernatant, wash the cell pellet with 15 mL of HEPES buffer and centrifuge for 5 min at 315 x g. Resuspend the pellet in HEPES buffer.
6. Neutrophil adhesion and migration experiment with bMFA
- Resuspend the isolated human neutrophils (15 million) in 999 µL of HEPES buffer.
- Add 1 µL CFDA-SE (carboxyfluorescein diacetate succinimidyl ester) dye stock solution (10 mM) to neutrophil suspension to obtain a working concentration of 10 µM CFDA-SE and incubate for 10 min at RT. Wash the cells twice with HEPES buffer by centrifuging for 5 min at 315 x g.
- Count the neutrophils using a hemocytometer and resuspend at 2 million neutrophils/mL with cell culture media, TNF-α (10 ng/mL) and TNF-α (10 ng/mL) +PKCδ-i (5 μM), respectively; incubate for 15 min at RT before the start of the experiment.
- Prepare a syringe filled with 1 µM N-formyl-met-leu-phe (fMLP), the chemoattractant for the study, in cell culture media. Take bMFA and open one inlet port and one outlet port.
NOTE: These experimental conditions are used to mimic the conditions of sepsis that include a systemic inflammatory response with high levels of circulating cytokine/chemokines. Furthermore, fMLP, a Gram-negative bacteria-derived peptide, is used to model the presence of bacteria in the lung (i.e., tissue compartment). - Remove the tissue compartment port tubing and insert the fMLP tubing. Inject ~20 µL of fMLP into the tissue compartment for all bMFA, except the one treated with cell culture media alone. Then cut the tubing and clamp it.
- Fill the syringe with ~200 µL of the neutrophil suspension and mount the syringe on the syringe pump.
- Put the device on the inverted microscope stage (Table of Materials).
- Start the pump at a flow rate is 1 µL/min and wait until a small drop of the neutrophil suspension comes out of the tubing. Insert the tubing into the inlet port. See Figure 7 for the setup.
7. Acquire images
- Use the Scan Large Image function in the microscope image analysis software (Table of Materials) to obtain the adhesion map in bMFA (Figure 8) 10 min after starting the experiment. Most of the neutrophils enter bMFA during the first 10 min.
NOTE: Keep the fluorescence light off when not taking images to reduce photobleaching. - During the next hour, take timelapse images (1 image every 5 min) of the tissue compartment (Figure 8B,C) for migration analysis to obtain the migration map.
8. Digital Image analysis
- For the adhesion map, count the number of adhered neutrophils in each vessel. For each bifurcation, create a circular region of interest (ROI) with a diameter equal to two times the vessel diameter and count the number of adhered neutrophils.
- Use manual count or the automated Object Count function to quantify the number of adhering neutrophils in each vessel and bifurcation region.
- Use the shear rate map of the network generated using CFD simulation17 to determine the shear rate in each vessel. Then plot the number of adhered neutrophils in a given vessel against the shear rate in that vessel to create a shear rate map in bMFA as shown in Figure 9A.
- Neutrophils are counted as migrated if they have crossed through the endothelium into the tissue compartment. Count the number of migrated neutrophils at 10 min, 15 min, 30 min and 60 min. Plot the number of migrated neutrophils versus time as shown in Figure 9B.
Representative Results
After 48 h of culture under shear flow in bMFA, endothelial cells covered the surface of the vascular channels in bMFA and aligned in the direction of flow (Figure 6). Confocal microscopy indicated that all surfaces of the vascular channels were covered by endothelial cells, forming a complete 3D lumen in bMFA18.
Using this protocol, a neutrophil adhesion map can be acquired, showing that there is significant adhesion of neutrophils to endothelial cell in bMFA (Figure 8). Correlating the spatial distribution of neutrophils in this adhesion map with the shear rate map generated from the CFD model shows that neutrophil adhesion is shear dependent, and neutrophils preferentially adhere in low shear rate and bifurcation regions (Figure 9A). The adhesion of neutrophils to TNF-α-activated endothelial cells increased significantly. Analyzing timelapse images indicated that TNF-α activation of endothelial cells increases neutrophil migration in response to the chemoattractant fMLP (Figure 9B). As stated above, bMFA can be used to rapidly test the efficacy of a novel therapeutic for treating inflammatory disease. For example, treatment of endothelial cells and neutrophils with PKCδ-i significantly reduced neutrophil adhesion and migration (Figure 9A,B)27.
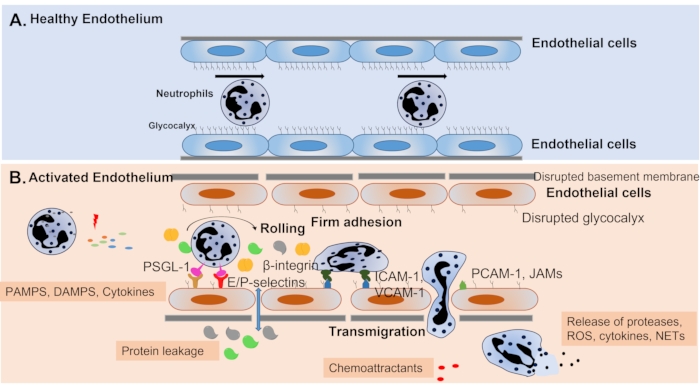
Figure 1: Endothelial cell activation during inflammation. (A) Under normal conditions, the vascular endothelium is covered by the glycocalyx and forms a tight barrier that regulates barrier permeability, leukocyte migration and anti-inflammatory defenses. (B) During inflammation, leukocytes and endothelial cells are activated by PAMPS and DAMPS to produce cytokines and chemoattractants, leading to the elevated expression of surface molecules on both leukocytes and endothelial cells, enhancing leukocyte-endothelial interactions. Interactions of E/P-selectin on endothelial cells and their ligands (e.g., PSGL-1) on leukocytes are involved in rolling, which slows down the neutrophil. Firm adhesion is regulated by endothelium-expressed adhesion molecules, including ICAM-1, ICAM-2 and VCAM-1, and their ligands, β2 integrins. In response to chemoattractant gradients, adhered leukocytes migrate through endothelial junctions regulated by PECAM-1 and JAMs. Activated leukocytes migrate to tissue, where they release cytokines, reactive oxygen/nitrogen species and proteases to fight against infection. However, dysregulated leukocyte migration can damage organ tissue, resulting in organ failure. During inflammation, the glycocalyx is degraded, endothelial cell tight junctions are damaged and there is increased endothelial cell apoptosis, leading to damaged barrier function and increased permeability. This figure has been modified from Yang et al.2. Please click here to view a larger version of this figure.
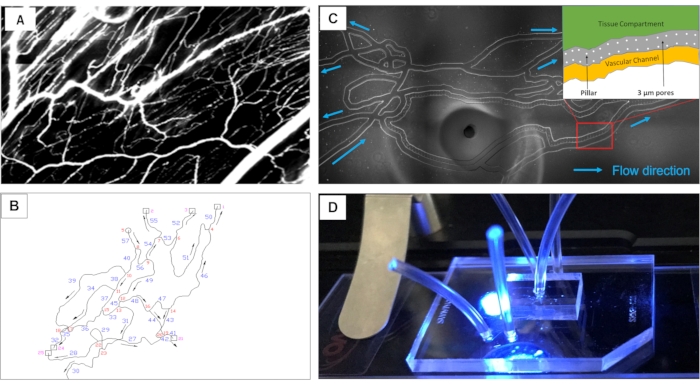
Figure 2: Overview of the bMFA. (A) Images of the mouse cremaster muscle are obtained and (B) digitized using the GIS system to fabricate the network on PDMS. (C) Bright-field image of bMFA network shows that in bMFA, vascular channels are connected to the tissue compartment through a 3 µm barrier (the schematic insert in Panel C). Flow direction of the vascular channels is indicated. (D) A bMFA chip, consisting of a PDMS layer bonded to a microscope slide, is on microscope stage. Tubing is inserted into two inlet ports, two outlet ports and one tissue compartment port. (C: scale bar = 500 µm) Please click here to view a larger version of this figure.
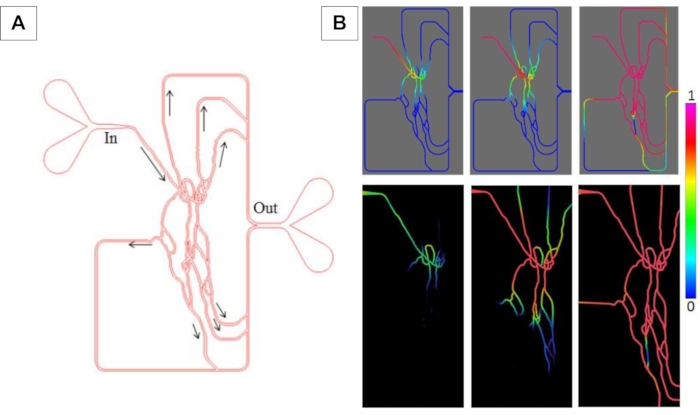
Figure 3: Transient perfusion studies comparing experimental and simulation results in bMFA. (A) The network of bMFA showing the inlet, outlet and flow direction. (B) Top panel: CFD perfusion profiles. Bottom panel: Experimental perfusion profiles. Images are pseudo-colored to show gradients. The scale is normalized with blue (no perfusion; 0) and magenta (complete perfusion; 1). Please click here to view a larger version of this figure.
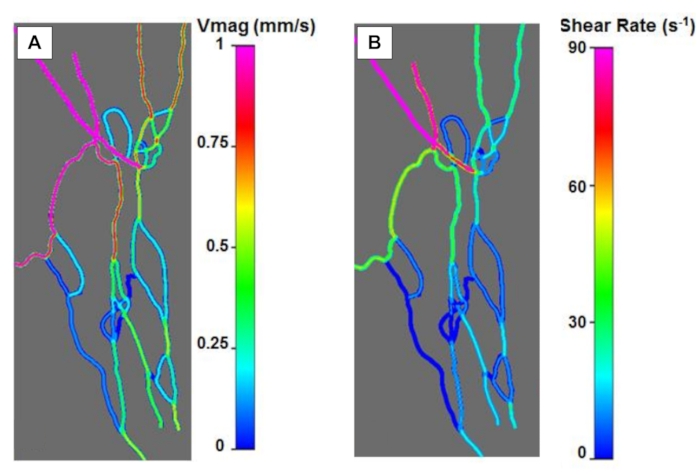
Figure 4: Velocity and shear rates profile in bMFA from CFD simulations. (A) Distribution of velocity magnitudes (Vmag) across the network. (B) Distribution of shear rate indicates heterogeneous shear rates in the network. Please click here to view a larger version of this figure.
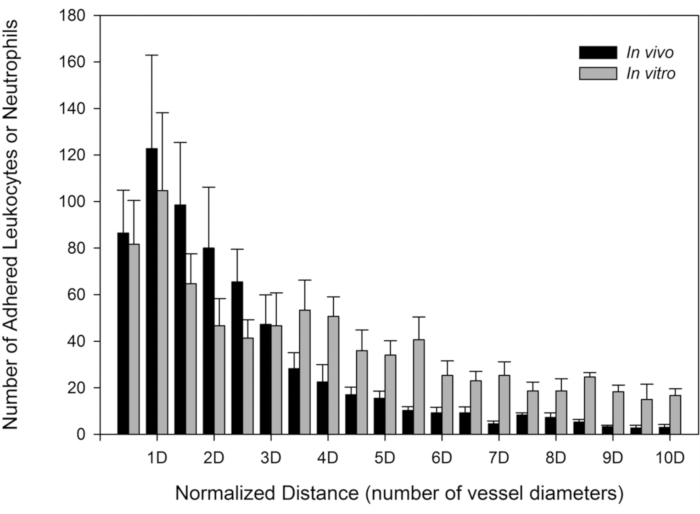
Figure 5: Neutrophil adhesion patterns in bMFA is similar to neutrophil adhesion patterns in vivo. The distributions of the number of adhered neutrophils in vivo and the number of neutrophils in bMFA are both skewed to the left, indicating that neutrophils preferentially adhere near bifurcations with the peak occurring at one vessel or channel diameter from the nearest bifurcation (mean ± SEM; N = 3). The distance of adhered neutrophils to the nearest bifurcation was normalized by the following: normalized distance = distance to the center of the nearest bifurcation/diameter of the channel. This figure has been modified from Lamberti et al.16 (https://pubs.acs.org/doi/10.1021/ac5018716, further permissions related to the material excerpted should be directed to the American Chemical Society). Please click here to view a larger version of this figure.
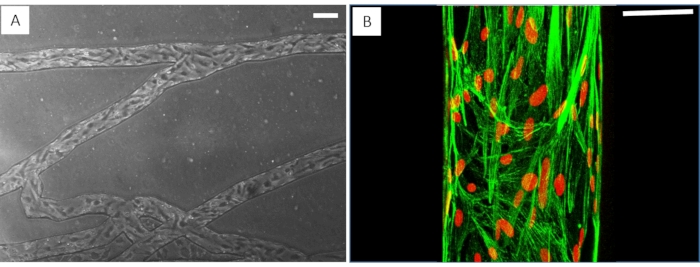
Figure 6: Endothelial cells form a complete 3D lumen in the bMFA. (A) Phase-contrast images show that endothelial cells are lined up in the direction of flow. (B) Fluorescence images show endothelial cells cover the vascular channel; F-actin is labeled green using phalloidin and nuclei is labeled red using Draq5. (A: scale bar = 100 µm, B: scale bar = 50 µm). Please click here to view a larger version of this figure.
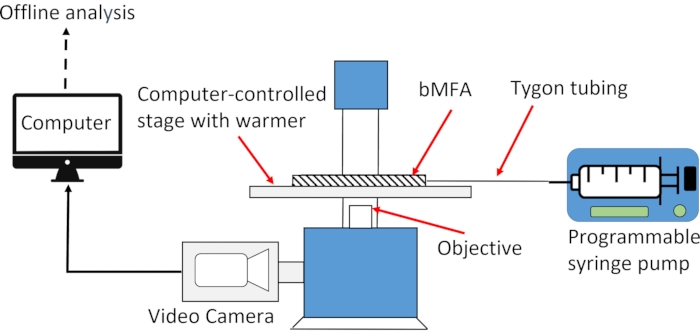
Figure 7: A computer-controlled inverted fluorescence microscope is used to study neutrophil-endothelial interactions in bMFA. A computer-controlled stage with a warmer is used to keep the bMFA at 37 °C. The bMFA is connected to a programmable syringe pump through the tubing. The inverted fluorescence microscope is equipped with a video camera to take images/video for further offline analysis on the computer. Please click here to view a larger version of this figure.
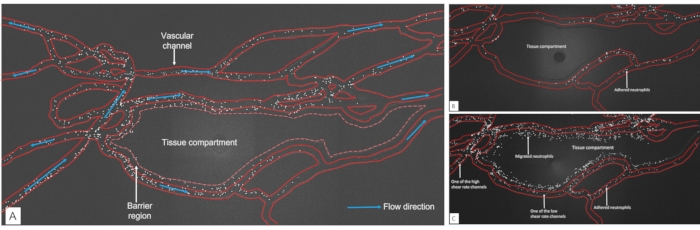
Figure 8: Representative images of neutrophil adhesion and migration map. (A) Neutrophil adhesion map in bMFA. The image was taken 10 min after the start of the experiment using "Scan large map" function of Nikon software. The white dots in the map are fluorescently labeled neutrophils with CFDA-SE. Vascular channel boundary is digitally labeled (scale bar = 100 µm). (B) Neutrophil migration map in bMFA without TNF-α activation. The image was taken 60 min after the start of the experiment. (C) Neutrophil migration map in bMFA with TNF-α activation. The image was taken 60 min after the start of the experiment. Please click here to view a larger version of this figure.
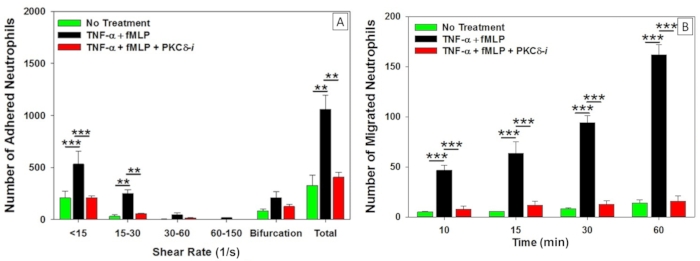
Figure 9: Patterns of neutrophil adhesion and migration in bMFA during inflammation. (A) TNF-α treatment significantly increased human neutrophil adhesion to human pulmonary microvascular endothelial cells, which was inhibited following treatment with a PKCδ inhibitor. Neutrophil adhesion occurred preferentially in vessels with low shear rate and near bifurcations in bMFA. (B) In response to fMLP, human neutrophil migration across TNF-α activated endothelial cells was significantly increased compared to untreated cells. Treatment with the PKCδ inhibitor reduced migration to untreated levels. n = 4, Mean ± SEM, one-way ANOVA, ** p < 0.01, and *** p < 0.001). The figure has been modified from Soroush et al.27. Please click here to view a larger version of this figure.
| Step 1: Constant rate | Comments | |
| Mode | Infuse | |
| Flow rate | 1 µL/min | |
| Time | 10 min | |
| Step 2: Delay (no flow) | ||
| Mode | Delay | |
| Time | 4 h | |
| Step 3: Repeat | Step 1 and Step 2 will be repeated 6 times before going to Step 4. | |
| Mode | Repeat | |
| Repeat from | Step 1 | |
| Repeat | 6 times | |
| Step 4: Constant flow rate | ||
| Mode | Infuse | |
| Flow rate | 0.1 µL/min | |
| Time | 48 h |
Table 1: Syringe pump program
| Formula | Name | MW | Add | Concentration | 10x HEPES buffer (Add DI water to 1000 mL and adjust pH to 7.3. Dilute to 1x before use) |
| NaCl | Sodium Chloride | 58.44 | 87.66 g | 1.5 M | |
| KOH | Potassium Hydroxide | 56.11 | 2.81 g | 50 mM | |
| Hepes | Hepes | 238.3 | 23.83 g | 100 mM | |
| MgCl2 | Magnesium Chloride | 203.31 | 2.44 g | 12 mM | |
| CaCl2 | Calcium Chloride | 147.62 | 1.90 g | 12.9 mM |
Table 2: HEPES buffer composition.
Discussion
The bMFA reproduces the topography and flow conditions of the in vivo microvascular networks and can be used to study leukocyte-endothelial cell interaction and endothelial function in vitro under physiologically realistic conditions. In the microvasculature of either mouse or human, the geometry of the microvascular networks are self-similar and fractal, and the Reynolds number << 1, indicating that vascular geometry does not significantly impact flow patterns. Therefore, the bFMA can be used to study leukocyte-endothelial interactions for different species, including humans, though the microvasculature of mouse cremaster muscle was mapped to manufacture bMFA.
The bMFA allows for the real-time assessment of individual steps of the neutrophil migration cascade in a single assay, including rolling, firm adhesion, spreading and migration of neutrophils into the tissue compartment. Primary human cells and clinically relevant samples can be used in bMFA to increase translatability and to rapidly screen potential therapeutics. Compared to traditional fluidic systems such as parallel plate flow chambers, bMFA has the advantage of using less than 95% of reagents37 due to its small vessel sizes and the fact that it can study the whole neutrophil migration cascade in one single assay. However, this small size also requires users to work with a very small volume of reagents, which can be challenging and requires extensive practice. The most common problem with this protocol is the presence of air bubbles in bMFA, which can block the channel and alter the flow patterns. Therefore, great care should be taken when removing or inserting tubing into the ports, and a drop of water must be placed at the base of the tubing at the ports to prevent air from entering the channel whenever tubing is removed from bMFA.
In addition to the application of bMFA for studying neutrophil-endothelial interactions, the bMFA has also been used to study the integrity of endothelium during inflammation by measuring variables such as permeability and transendothelial endothelial resistance (TEER)27. Furthermore, by functionalizing microparticles with specific antibodies, bMFA can be used to study the expression of adhesion molecules on endothelial cells27. It was demonstrated that the inflammatory response could also be upregulated by other stimuli in the bMFA, such as ionizing radiation, and bMFA has been used to study radiation-induced endothelial cell damage and neutrophil-endothelial interactions29. Another advantage of this device is that endothelial cells, neutrophils and/or other blood cells or drug-carrying particles can be isolated from microvascular channels and/or tissue compartment for further analysis. Furthermore, cells/particles that did not interact with the endothelial cells in the device can be isolated from the effluent exiting the device. Pericytes and the corresponding tissue types can also be cultured in the device to better represent the in vivo conditions. In conclusion, the protocol presented here provides a physiologically relevant environment in bMFA to study leukocyte-endothelial cell interactions during inflammation.
Declarações
The authors have nothing to disclose.
Acknowledgements
This work was supported by the National Institutes of Health, Grant Number: GM114359 and GM134701 (M.F.K. and L.E.K.), 1F31AI164870-01 (J.C.L.), and Defense Threat Reduction Agency, Grant Number: HDTRA11910012 (M.F.K. and L.E.K.).
Materials
| 1 mL syringe | Fisher Scientific | 14-823-30 | |
| Biomimetic microfluidic assay (bMFA) | SynVivo | SMN1-C001 | Exclusive at SynVivo |
| Blunt needle | Jensen Global | JG24-0.5 | |
| Calcium Chloride | Fisher Scientific | C70-500 | |
| CFDA, SE | ThermoFisher | C1157 | |
| Dextran, 250,000, Powder | Spectrum Chemical Mfg. Corp | DE-130 | |
| Ficoll-Paque Premium | GE Health Care | 17-5442-02 | Leukocyte isolation media |
| fMLP | Sigma-Aldrich | F3506 | |
| Hepes | Fisher Scientific | AAJ1692630 | |
| Human fibronectin | Fisher Scientific | 33-016-015 | use vendor recommended ECM for different cell lines |
| Microvascular Endothelial Cell Growth Medium-2 BulletKit | Lonza | cc-3202 | Human lung microvascular endothelial cell culture medium (HLMVEC). |
| Human lung microvascular endothelial cells | Lonza | cc-2527 | use vedor remommended trypsin-EDTA and TNS |
| Magnesium Chloride | Fisher Scientific | BP214-500 | |
| Nikon Eclipse Ti2 | Nikon Instruments Inc. | Microscope | |
| NIS-elements, 5.20.01 | Nikon Instruments Inc. | Imaging software | |
| PBS | Fisher Scientific | MT21040CV | |
| PhD Ultra Syringe Pump | Harvard Apparatus | 70-3007 | Syringe Pump |
| Potassium Hydroxide | Fisher Scientific | 02-003-763 | |
| Recombinant Human TNF-alpha | R&D Systems | 210-TA | |
| Slide clamp | SynVivo | ||
| Sodium Chloride | Fisher Scientific | S640-500 | |
| Synvivo Pneumatic Primer | SynVivo | ||
| Trypsin-EDTA, Trypsin Neutralization Solution(TNS) | Lonza | cc-5034 | |
| Tygon tubing | Fisher Scientific | 50-206-8921 | Tubing |
Referências
- Kolaczkowska, E., Kubes, P. Neutrophil recruitment and function in health and inflammation. Nature Reviews Immunology. 13 (3), 159-175 (2013).
- Yang, Q., Wijerathne, H., Langston, J. C., Kiani, M. F., Kilpatrick, L. E. Emerging approaches to understanding microvascular endothelial heterogeneity: A roadmap for developing anti-inflammatory therapeutics. International Journal of Molecular Sciences. 22 (15), 7770 (2021).
- Joffre, J., Hellman, J., Ince, C., Ait-Oufella, H. Endothelial responses in sepsis. American Journal of Respiratory and Critical Care Medicine. 202, 361-370 (2020).
- Ince, C., et al. The endothelium in sepsis. Shock. 45, 259-270 (2016).
- Ruparelia, N., Chai, J. T., Fisher, E. A., Choudhury, R. P. Inflammatory processes in cardiovascular disease: a route to targeted therapies. Nature Reviews Cardiology. 14 (3), 133-144 (2017).
- Kilpatrick, L. E., Kiani, M. F. Experimental approaches to evaluate leukocyte-endothelial cell interactions in sepsis and inflammation. Shock. 53, 585-595 (2020).
- Langer, H. F., Chavakis, T. Leukocyte-endothelial interactions in inflammation. Journal of Cellular and Molecular Medicine. 13 (7), 1211-1220 (2009).
- Sadik, C. D., Luster, A. D. Lipid-cytokine-chemokine cascades orchestrate leukocyte recruitment in inflammation. Journal of Leukocyte Biology. 91 (2), 207-215 (2012).
- Phillipson, M., Kubes, P. The neutrophil in vascular inflammation. Nature Medicine. 17, 1381-1390 (2011).
- Maniatis, N. A., Orfanos, S. E. The endothelium in acute lung injury/acute respiratory distress syndrome. Current Opinion in Critical Care. 14, 22-30 (2008).
- Ley, K., Laudanna, C., Cybulsky, M. I., Nourshargh, S. Getting to the site of inflammation: the leukocyte adhesion cascade updated. Nature Reviews Immunology. 7, 678-689 (2007).
- Molteni, R., Fabbri, M., Bender, J. R., Pardi, R. Pathophysiology of leukocyte-tissue interactions. Current Opinion in Cell Biology. 18, 491-498 (2006).
- Chistiakov, D. A., Orekhov, A. N., Bobryshev, Y. V. Effects of shear stress on endothelial cells: go with the flow. Acta Physiologica. 219 (2), 382-408 (2017).
- Prabhakarpandian, B., Shen, M. -. C., Pant, K., Kiani, M. F. Microfluidic devices for modeling cell-cell and particle-cell interactions in the microvasculature. Microvascular Research. 82, 210-220 (2011).
- Zou, X., et al. PSGL-1 derived from human neutrophils is a high-efficiency ligand for endothelium-expressed E-selectin under flow. American Journal of Physiology-Cell Physiology. 289, 415-424 (2005).
- Lamberti, G., et al. Bioinspired microfluidic assay for in vitro modeling of leukocyte-endothelium interactions. Analytical Chemistry (Journal). 86, 8344-8351 (2014).
- Prabhakarpandian, B., et al. Synthetic microvascular networks for quantitative analysis of particle adhesion. Biomedical Microdevices. 10 (4), 585-595 (2008).
- Soroush, F., et al. A novel microfluidic assay reveals a key role for protein kinase C delta in regulating human neutrophil-endothelium interaction. Journal of Leukocyte Biology. 100, 1027-1035 (2016).
- Roth, N. M., Kiani, M. F. A "geographic information systems" based technique for the study of microvascular networks. Annals of Biomedical Engineering. 27, 42-47 (1999).
- Prabhakarpandian, B., Wang, Y. I., Rea-Ramsey, A., Sundaram, S., Kiani, M. F., Pant, K. Bifurcations: Focal points of particle adhesion in microvascular networks. Microcirculation. 18, 380-389 (2011).
- Rosano, J., et al. A physiologically realistic in vitro model of microvascular networks. Biomedical Microdevices. 11, 1051-1057 (2009).
- Tousi, N., Wang, B., Pant, K., Kiani, M. F., Prabhakarpandian, B. Preferential adhesion of leukocytes near bifurcations is endothelium independent. Microvascular Research. 80, 384-388 (2010).
- Yona, S., Hayhoe, R., Avraham-Davidi, I. Monocyte and neutrophil isolation and migration assays. Current Protocols in Immunology. 88 (1), 11-14 (2010).
- Entschladen, F., et al. Analysis methods of human cell migration. Experimental Cell Research. 307 (2), 418-426 (2005).
- Chen, H. -. C. Boyden chamber assay. Methods in Molecular Biology. Cell Migration: Developmental Methods and Protocols. , (2005).
- Deosarkar, S. P., Prabhakarpandian, B., Wang, B., Sheffield, J. B., Krynska, B., Kiani, M. F. A novel dynamic neonatal blood-brain barrier on a chip. PloS one. 10, 142725 (2015).
- Soroush, F., et al. Neutrophil-endothelial interactions of murine cells is not a good predictor of their interactions in human cells. The FASEB Journal. 34, 2691-2702 (2020).
- Soroush, F., et al. Protein Kinase C-Delta (PKCδ) tyrosine phosphorylation is a critical regulator of neutrophil-endothelial cell interaction in inflammation. Shock. 51 (5), 538-547 (2019).
- Soroush, F., Tang, Y., Zaidi, H. M., Sheffield, J. B., Kilpatrick, L. E., Kiani, M. F. PKCδ inhibition as a novel medical countermeasure for radiation-induced vascular damage. The FASEB Journal. 32, 6436-6444 (2018).
- Pradhan, S., et al. A microvascularized tumor-mimetic platform for assessing anti-cancer drug efficacy. Scientific Reports. 8 (1), 3171 (2018).
- Tang, Y., et al. A biomimetic microfluidic tumor microenvironment platform mimicking the EPR effect for rapid screening of drug delivery systems. Scientific Reports. 7 (1), 1-14 (2017).
- Tang, Y., et al. Protein kinase C-delta inhibition protects blood-brain barrier from sepsis-induced vascular damage. Journal of Neuroinflammation. 15, 309 (2018).
- Mondrinos, M. J., et al. Pulmonary endothelial protein kinase C-delta (PKCd) regulates neutrophil migration in acute lung inflammation. The American Journal of Pathology. 184, 200-213 (2014).
- Kilpatrick, L. E., et al. Protection against sepsis-induced lung injury by selective inhibition of protein kinase C-d (d-PKC). Journal of Leukocyte Biology. 89, 3-10 (2011).
- Mondrinos, M. J., et al. Biodistribution and efficacy of targeted pulmonary delivery of a protein kinase C-d inhibitory peptide: Impact on Indirect lung Injury. Journal of Pharmacology and Experimental Therapeutics. 355, 86-98 (2015).
- Liverani, E., Mondrinos, M. J., Sun, S., Kunapuli, S. P., Kilpatrick, L. E. Role of protein kinase C-delta in regulating platelet activation and platelet-leukocyte interaction during sepsis. PloS one. 13, 0195379 (2018).
- Smith, A. M., Prabhakarpandian, B., Pant, K. Generation of shear adhesion map using SynVivo synthetic microvascular networks. Journal of Visualized Experiments: JoVE. (87), e51025 (2014).

