Utilizing the Precision-Cut Lung Slice to Study the Contractile Regulation of Airway and Intrapulmonary Arterial Smooth Muscle
Summary
The present protocol describes preparing and utilizing mouse precision-cut lung slices to assess the airway and intrapulmonary arterial smooth muscle contractility in a nearly in vivo milieu.
Abstract
Smooth muscle cells (SMC) mediate the contraction of the airway and the intrapulmonary artery to modify airflow resistance and pulmonary circulation, respectively, hence playing a critical role in the homeostasis of the pulmonary system. Deregulation of SMC contractility contributes to several pulmonary diseases, including asthma and pulmonary hypertension. However, due to limited tissue access and a lack of culture systems to maintain in vivo SMC phenotypes, molecular mechanisms underlying the deregulated SMC contractility in these diseases remain fully identified. The precision-cut lung slice (PCLS) offers an ex vivo model that circumvents these technical difficulties. As a live, thin lung tissue section, the PCLS retains SMC in natural surroundings and allows in situ tracking of SMC contraction and intracellular Ca2+ signaling that regulates SMC contractility. Here, a detailed mouse PCLS preparation protocol is provided, which preserves intact airways and intrapulmonary arteries. This protocol involves two essential steps before subjecting the lung lobe to slicing: inflating the airway with low-melting-point agarose through the trachea and infilling pulmonary vessels with gelatin through the right ventricle. The PCLS prepared using this protocol can be used for bioassays to evaluate Ca2+-mediated contractile regulation of SMC in both the airway and the intrapulmonary arterial compartments. When applied to mouse models of respiratory diseases, this protocol enables the functional investigation of SMC, thereby providing insight into the underlying mechanism of SMC contractility deregulation in diseases.
Introduction
Smooth muscle cell (SMC) is a major structural cell type in the lung, primarily residing in the media wall of airways and pulmonary vessels. SMCs contract to alter the luminal caliber, thus regulating air and blood flow1,2. Therefore, contractile regulation of SMCs is essential to maintain the homeostasis of air ventilation and pulmonary circulation. In contrast, aberrant SMC contractility provokes obstructive airway or pulmonary vascular diseases like asthma and pulmonary arterial hypertension. However, the functional assessment of lung SMCs has been challenged by limited access to the lung tissue, especially those small airways and microvessels in the distal part of the lung2,3. Current solutions resort to indirect assays, such as measuring airflow resistance by Flexivent to reflect airway constriction, and checking pulmonary arterial blood pressure by right heart catheterization to assess pulmonary vasocontraction4,5. However, these indirect assays have multiple disadvantages, such as being confounded by structural factors, failing to capture the spatial diversity of airway or vascular responses in the whole lung scale6,7, and unfitting for the mechanistic study of contractile regulation at the cellular level. Therefore, alternative approaches using isolated primary cells, trachea/bronchi muscle strips8,9, or large vascular segments10 have been applied for the SMC study in vitro. Nevertheless, these methods also have limitations. For example, a quick phenotypical adaptation of primary SMCs in the culture condition11,12 makes it problematic to extrapolate findings from cell culture to in vivo settings. In addition, the contractile phenotype of SMCs in the isolated proximal airway or vascular segments may not represent the SMCs in the distal lung6,7. Moreover, the muscle force measurement at the tissue level remains dissociated from molecular and cellular events that are essential for mechanistic insight into contractile regulation.
Precision-cut lung slice (PCLS), a live lung tissue section, provides an ideal ex vivo tool to characterize pulmonary SMCs in a near in vivo microenvironment (i.e., preserved multi-cellular architecture and interaction)13. Since Drs. Placke and Fisher first introduced the preparation of lung slices from agarose-inflated rat and hamster lungs in the 1980s14,15, this technique has been advanced continuously to provide PCLSs with higher quality and greater versatility for biomedical research. One significant improvement is the enhancement of pulmonary arterial preservation by gelatin infusion in addition to lung inflation with agarose via the trachea. As a result, both the airway and pulmonary arteries are kept intact in the PCLS for ex vivo assessement16. Furthermore, the PCLS is viable for a prolonged time in culture. For instance, mouse PCLSs had no significant change in cell viability and metabolism for a minimum of 12 days in culture, as well as, they retained airway contractility for up to 7 days17. In addition, PCLS keeps different-sized airways or vessels for contraction and relaxation assays. Moreover, intracellular Ca2+ signaling of SMCs, the determinant factor of cell contractility, can be assayed with Ca2+ reporter dyes imaged by a confocal or 2-photon microscope13.
Considering the extensive application of the mouse model in lung research, a detailed protocol is described here for preparing mouse PCLS with intact airways and intrapulmonary arteries for ex vivo lung research. Using the prepared PCLSs, we subsequently demonstrated how to evaluate the airway and pulmonary arterial responses to constrictive or relaxant stimuli. In addition, the method of loading the PCLS with Ca2+ reporter dye and then imaging Ca2+ signaling of SMCs associated with contractile or relaxant responses are also described.
Protocol
All animal care was in accordance with the guidelines of the Institutional Animal Care and Use Committee of Massachusetts General Hospital. Wild-type C57/B6 male mice, 8 weeks of age, were used for the present study.
1. Experimental preparation
- Prepare the working solution.
- Prepare 1x Hank's Balanced Salt Solution (HBSS, with Ca2+ and Mg2+, and pH balanced with 20 mM HEPES, see Table of Materials). Use the HBSS solution to prepare and process the PCLS. Keep HBSS solution cold on ice when preparing PCLSs.
- Prepare the culture medium of PCLS by supplementing Dulbecco's Modified Eagle Medium and F-12 (DMEM-F12) with an antibiotic-antimycotic agent (1:100 volume ratio, see Table of Materials).
- Prepare 1.5% agarose and 6% gelatin solution.
- Mix low melting point (LMP) agarose or gelatin powder (see Table of Materials) with HBSS solution separately in 15 mL sterile centrifuge tubes (or 50 mL tubes if solution volume >10 mL) to final concentrations.
NOTE: Total volume of agarose solution = 5 mL/mouse x number of mice; gelatin solution = 2 mL/mouse x number of mice. - Heat the solution tubes in boiling water until the powder completely dissolves. Keep both agarose and gelatin solutions in a 42 °C water bath.
NOTE: A heating lamp at the dissection table can be useful to keep the operating environment warm and prevent agarose solution from solidifying before being injected into the mouse lung.
- Mix low melting point (LMP) agarose or gelatin powder (see Table of Materials) with HBSS solution separately in 15 mL sterile centrifuge tubes (or 50 mL tubes if solution volume >10 mL) to final concentrations.
- Submerge all dissection tools, including a pair of dissecting scissors, two pairs of curved micro-dissecting forceps, and a pair of hemostatic forceps in 70% ethanol solution for at least 20 min for sterilization.
- Keep a vibratome ready (see Table of Materials) to section the lung tissue into thin slices.
- Keep an inverted phase-contrast microscope with a CCD camera to evaluate the airway or vascular contractile responses and a custom-built laser scanning confocal microscope (see Table of Materials) to assess the Ca2+ signaling in pulmonary SMCs18.
2. Inflation of mouse lungs with agarose and gelatin solution
- Euthanize the mouse.
- Place the mouse in a plastic chamber (around 750 cm3) with 5% isoflurane. Keep the mouse in the chamber until it stops breathing for at least 1 min.
NOTE: Other primary euthanasia methods, such as intraperitoneal injection of ketamine (240 mg/Kg) and xylazine (32 mg/Kg)19 or pentobarbital (0.3 mL)20, can be applied as alternatives to inhaled isoflurane. - Place the mouse body on a dissection board in the supine position. Fix the body in position by pinning down the tail, front paws, and head with 25 G syringe needles, and sanitize the body with 70% ethanol spray.
- Cut open the mouse's abdomen with surgical scissors (incision, ~2 cm long x 2 cm wide). Then, cut off the abdominal aorta to deplete the blood volume.
- Place the mouse in a plastic chamber (around 750 cm3) with 5% isoflurane. Keep the mouse in the chamber until it stops breathing for at least 1 min.
- Inflate lung lobes following the steps below.
- Open the chest cavity carefully along the sternum and bilateral inferior rib cage above the diaphragm, and observe the lung lobes collapse when the chest cavity opens.
- Remove part of bilateral ventral rib cages to expose the heart. Avoid lung damage by pointing the sharp scissor tip away from the lung tissue.
- Use forceps to separate the thymus and soft tissue in the mouse neck to expose the trachea.
- Cut a small hole (1.2 mm in diameter) in the upper end of the trachea allowing passage of the tip of a 20 G Y-shaped IV catheter (see Table of Materials).
- Connect one adaptor port of the Y-shaped catheter with a 3 mL syringe prefilled with 0.5 mL of air, and the other port with a 3 mL syringe prefilled with 2 mL of warm 1.5% agarose solution (42 °C).
- Inject agarose solution to fill the catheter and then push the catheter through the pre-cut opening into the trachea for 5-8 mm in length.
- Slowly inject the agarose solution at around 1 mL/5 s. The lung will expand along the proximal-to-distal axis. Stop injection when the edge of each lung lobe is inflated.
NOTE: Do not overinflate the lung, as this could rupture it. The volume of agarose solution to inflate the whole lung of a young adult mouse is about 1.3 ± 0.1 mL. - Inject 0.2-0.3 mL of air from the other syringe to push the residual agarose in the catheter and conductive airways to the distal alveoli space.
- Clip close the trachea with a pair of curved hemostatic forceps (see Table of Materials).
- Infill the pulmonary vasculature.
- Fill a 1 mL syringe with warm 6% gelatin. Connect to a needle scalp vein catheter, infill the catheter with gelatin solution, and then puncture the right ventricle close to the inferior wall with the needle.
- Push the needle into the right ventricle for 2-3 mm in length and point the needle tip to the main pulmonary artery.
- Inject 0.2 mL of gelatin solution slowly into the right ventricle and pulmonary arterial vessels.
NOTE: The lung lobes appear slightly paler with appropriate gelatin inflation. - Keep the needle in place for 5 min after injection, cool the lung lobes by pouring ice-cold HBSS solution on the heart and lung, then place the body with the dissection board in a 4 °C refrigerator or on ice for a total of 10 min.
- Remove the mouse lung and heart from surrounding connective tissues with scissors. Then, separate each lung lobe and keep them in HBSS solution on ice.
3. Sectioning of lung lobes to thin slices
- Trim and attach the lung lobe on the top of the specimen column with superglue, and orient the lobe (Figure 1A,B) to allow the cutting direction to be perpendicular to most airways from the hila to the lung surface.
- Use a vibratome with a fresh thin razor blade to cut the lung lobe into 150 µm slices. Collect the slices in 100 mm sterile Petri dishes prefilled with cold HBSS solution.
NOTE: Set appropriate blade moving speed and oscillation frequency before starting slicing. A typical setting for a Compresstom is Speed level 4 and Oscillation level 4. - Transfer slices to Petri dishes filled with DMEM/F-12 culture medium (20 slices/15 mL medium/dish) and maintain them in a 37 °C incubator overnight before experiments.
NOTE: Mouse PCLSs can be maintained in culture for about 7 days without losing airway contractile responses17. The longevity of pulmonary arteries has not been thoroughly evaluated. In our observation, they retain intact structure and vasoreaction for at least 3 days in DMEM/F12 medium. For long-term storage, mouse PCLSs can be placed in cryovials filled with DMEM/F12 medium containing 10% DMSO (5 slices in 1 mL medium per vial), frozen in an isopropyl alcohol-filled container at -80 °C overnight, and cryopreserved in liquid nitrogen for weeks to months21.
4. Analyzing contractile responses of intrapulmonary airways and arteries
- Place PCLSs in an HBSS-filled 24-well culture plate, one in each well. Locate the PCLS in the middle of the well, then remove the HBSS solution with a pipette.
- Find the target airway and vessel in the slice under the microscope, then cover the slice using a nylon mesh with a pre-cut central hole (2-3 mm in diameter) to expose the target area.
- Lay down a hollow metal washer on top of the mesh to hold the slices in place (Figure 2A).
- Add 600 μL of HBSS solution to submerge the PCLS. Rest the slice for 10 min, then record the baseline images of airways or vessels.
- Induce the airway or vascular contraction by cautiously suctioning out the blank HBSS solution with a pipette and adding 600 μL of HBSS with an agonist, such as 1 µM of methacholine (MCh) or 10 nM of endothelin (see Table of Materials).
NOTE: KCl stimulation as a tool-set standard is not required prior to methacholine or endothelin exposure. 100 mM of KCl stimulation in mouse airways only induces a small and irregular airway contraction (10%-15%)20. Likewise, a priming methacholine stimulation is not obligated as we have confirmed that the same agonist, for instance, methacholine or serotonin, triggers similar airway contraction on the first and repetitive exposures20,22. - Observe the reaction under the microscope until the luminal area change reaches an equilibratory status, and then record the airway or vascular images for analysis.
- Remove MCh or endothelin solution with a pipette and add a new 600 µL HBSS solution with the same agonist concentration and a relaxant; observe and record the airway or vascular relaxation.
- Quantify the airway or vascular reaction following the steps below.
- Load the images to NIH-sponsored free software FIJI.
- Select the Polygon Selection in the toolbar to outline the airway or vascular lumen on each frame.
- Choose Analyze > Set Measurements > Area.
- Choose Analyze > Measure to quantify the area of interest. Results are shown in a separate window for statistical analysis.
5. Analyzing Ca2+ signaling of airway or vascular SMCs
- Prepare the Ca2+ dye loading buffer.
- Dissolve Ca2+ dye, Oregon Green 488 BAPTA-1-AM (see Table of Materials), 50 µg (one vial) with 10 µL of DMSO.
- Dissolve 0.2 g of Pluronic F-12 powder (see Table of Materials) in 1 mL of DMSO to generate a 20% Pluronic solution.
- Mix 10 µL of 20% Pluronic solution with 10 µL of Ca2+ dye-DMSO solution.
- Make Ca2+ dye loading buffer by adding 20 µL of the mixture to 2 mL of HBSS solution with 200 µM of sulfobromophthalein (see Table of Materials).
- Place 15 mouse PCLSs in 2 mL of Ca2+ loading buffer and incubate at 30 °C for 1 h. Then move the slices to 2 mL of HBSS solution with 100 µM of sulfobromophthalein for an additional 30 min for fluorescent dye de-esterification at room temperature.
- Detect Ca2+ signaling of SMCs.
- Place Ca2+ dye-loaded slices on a large cover glass.
- Fill a 3 mL syringe with high vacuum silicone grease. Squeeze the grease through an 18 G blunted needle to draw two parallel lines across the cover glass above and below the slice.
- Cover the slice using a nylon mesh between two grease lines. Place the second cover glass on top of the mesh to generate a chamber sealed by grease on the sides (Figure 3A).
NOTE: The nylon mesh is essential to keep the slice attached to the bottom coverslip and thus stay within the working distance of a high magnitude inverted objective, such as 40x oil. - Add HBSS or agonist solution into the chamber from one end by pipetting manually or via a perfusion system. Remove the fluid from the chamber by suctioning from the other end with tissue paper or vacuum in the case of continuous fluid perfusion.
- Put the PCLS chamber on the microscope stage. Detect the Ca2+ signaling of SMC23 with a custom-built resonant scanning confocal microscope with a laser scan rate of 15 or 30 images/s.
NOTE: Alternatively, a high-speed commercial laser scanning confocal microscope, widely available at present, can be applied in the Ca2+ signaling assay of pulmonary SMCs in PCLS.
- Quantify the Ca2+ fluorescence in SMCs.
- Load the recorded image sequence to FIJI and choose the Image > Type > 8-bit grayscale.
- Select the Rectangle Selection in the toolbar and define a 5 pixel x 5 pixel region of interest (ROI) in a smooth muscle cell.
- Choose Analyze > Set Measurements > Mean gray value, representing the Ca2+ fluorescence intensity.
- If the position of an ROI changes with SMC contraction, choose Analyze > Measure the Ca2+ fluorescent intensity frame-by-frame.
- If the ROI of SMC stays in a similar position in a stack of images, choose Image > Stacks > Measure stack of the Ca2+ fluorescent intensity.
NOTE: A custom-written macro can be plugged in to track the ROI within the SMC when it moves with contraction in an image stack and automatically measures the Ca2+ intensity (gray value) of ROI frame-by-frame.
Representative Results
Mouse PCLS preparation preserving intact intrapulmonary airways and arteries
A 150 µm thick PCLS was observed under the inverted phase-contrast microscope. In mouse lungs, conductive airways are accompanied by intrapulmonary arteries, running from the hilus to the peripheral lung. A representative pulmonary airway-artery bundle in a mouse PCLS is shown in Figure 2B. The airway can be easily identified by cuboidal epithelial cells with active cilial beating lining the inner surface of the lumen. In contrast, the nearby pulmonary artery is featured by flat endothelium. When reaching the peripheral lung field, the conductive airways branch into respiratory ducts and sacs, surrounding the small intra-acinar arterioles (Figure 2C).
Utilizing mouse PCLS to assess the pulmonary airway and arterial contraction
Methacholine (MCh, 1 µM) induced airway contraction is demonstrated in Figure 2B. The airway contractile responses are quantified by the percentage of luminal area reduction following MCh exposure (Figure 2D). In contrast, the pulmonary artery presents no response to MCh stimuli (Figure 2B). Airways maintain similar dose-dependent contractile responses to MCh in the PCLS following 1-day or 5-day culture (Figure 2D). When the PCLS is exposed to endothelin (10 nM), both airways and pulmonary arteries constrict (Figure 2C,E), followed by NOC-5 (100 µM) induced relaxation (Figure 2E).
Utilizing mouse PCLS to assess the Ca2+ signaling of the airway and arterial SMC
The Ca2+ dye-loaded PCLS is observed under a confocal fluorescent microscope. The Ca2+ fluorescence in the airway (Figure 3B) and vascular (Figure 3C) SMCs is low at resting status, with no focal spark of intracellular Ca2+ signaling notable. Upon exposure to agonists, the Ca2+ fluorescence intensity elevates in the SMC (Figure 3B,C), usually from one spot and then propagating to the entire cell. The Ca2+ fluorescent waves repeatedly appear in the same cell as oscillatory signals (Figure 3D,E). In general, the frequency of Ca2+ oscillation increases as the agonist concentration rises until reaching a plateau level24. Airway SMC relaxation is associated with decreasing or cessation of Ca2+ oscillations25.
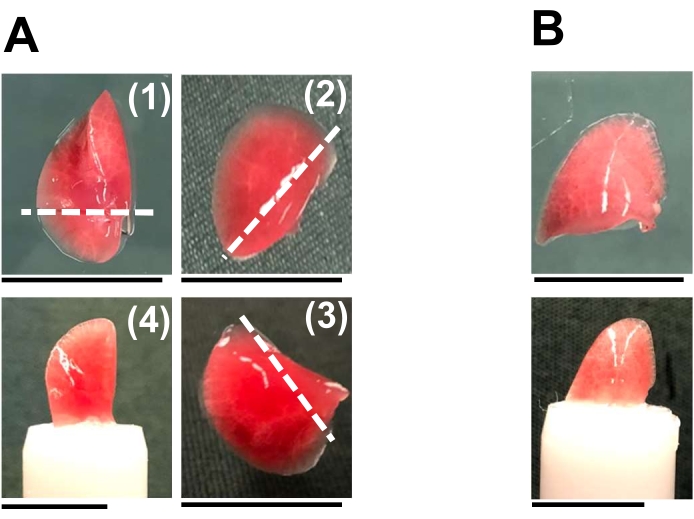
Figure 1: Orientation of mouse lung lobes for vibratome slicing. The mouse lung lobes are separated into individual ones for sections. (A) The left (1), right cranial (2), and caudal lobes (3) are trimmed near the hilum along the white dotted lines before sticking the flat cutting surface on the sample column. The placement of the left lobe is shown in (4). (B) The right middle lobe can be directly glued to the sample column. The right accessory lobe was not commonly used due to its small size. Appropriate orientation of different lobes ensures that most airways and pulmonary vessels present transverse sections in the PCLS. Scale bar = 1 cm. Please click here to view a larger version of this figure.
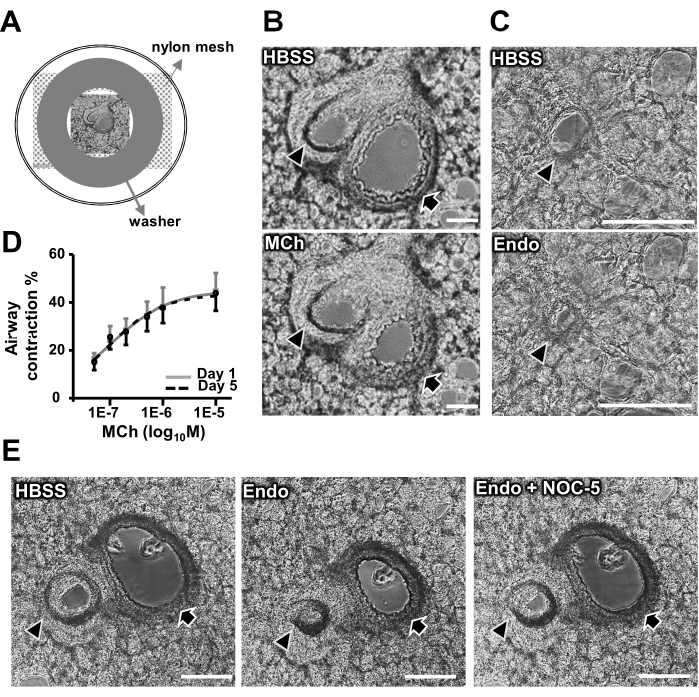
Figure 2: Contractile and relaxant responses of airways and pulmonary arteries in mouse PCLS. (A) Schematic showing the placement of a PCLS in a culture plate well for contraction assay. (B) Representative images showing an airway (black arrows) with a nearby pulmonary artery (black arrowheads) in HBSS at rest and following exposure to 1 µM methacholine (MCh). (C) Representative images showing an intra-acinar arteriole in HBSS at rest and upon exposure to 10 nM endothelin (Endo). (D) Dose-dependent airway contractile responses to MCh in PCLSs following 1-day (grey line) and 5-day (black dotted line) culture. Each point represents the average ± SEM of nine airways from two mice. (E) Representative images showing 10 nM endothelin-induced airway and pulmonary arterial contraction, followed by 100 µM NOC-5, a nitric oxide donor, induced relaxation. Scale bar = 100 μm. Please click here to view a larger version of this figure.
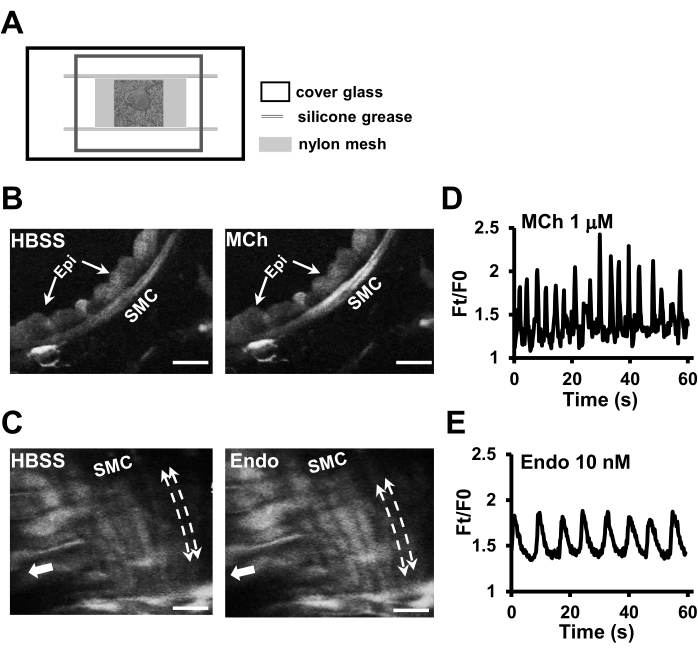
Figure 3: Ca2+ signaling of airway and pulmonary arterial SMCs in PCLS. (A) Schematic showing the setup of a chamber with a top and a bottom cover glass, grease seal, and a nylon mesh to hold a PCLS in the focal plane for Ca2+ imaging of SMCs. (B) Representative fluorescent images showing the Ca2+ signaling of airway SMCs at rest and following exposure to 1 µM MCh. Epi, epithelial cell. (C) Ca2+ fluorescent images of pulmonary arterial SMCs at rest and following exposure to 10 nM endothelin (Endo). The bold white arrows indicate the longitudinal axis of the pulmonary artery, and the dotted lines with end arrows indicate the helical distribution of vascular SMCs around the arterial wall. Scale bar = 20 µm. Oscillatory elevation of Ca2+ fluorescence intensity (Ft), in ratio to the fluorescence intensity at rest condition (F0), in an airway SMC to 1 µM MCh (D) and in a pulmonary arterial SMC in response to 10 nM endothelin stimulation (E). Please click here to view a larger version of this figure.
Discussion
The preparation of PCLS involves several critical steps. First, it is essential to inflate the lung lobe homogeneously to avoid the variation of tissue stiffness from uneven agarose distribution. As the liquid agarose rapidly gels in thin catheters or airways at a temperature below 37 °C, the resultant filling defect in the distal lung field could increase the disparity of lung tissue stiffness and cause tissue tearing during the vibratome section. Therefore, keeping the low-melting agarose solution at 42 °C in a water bath and using a heating lamp at the dissection table can be practiced to avoid quick agarose gelling. A quick injection could push more agarose to the lung parenchyma with higher compliance, hence needs to be avoided. The manual agarose injection usually takes around 5-7 s. Second, a necessary step at the end of agarose infilling is to push a small amount of air (~0.2 mL) to flush the agarose from the conductive airway to the distal alveoli space. Otherwise, the agarose would gel and stay in the lumen to resist the airway contraction. It is also worth noting that the agarose gelling inside the alveoli stays in place and never melts again at 37 °C in the incubator. The agarose gel plays an essential role in holding the 3D structure of lung tissue as in vivo maintained by the negative intrathoracic pressure. Third, perfusing pulmonary arteries with gelatin solution is essential to keep the arterial lumen open in the PCLS. The gelatin solution gels at room temperature as a mechanical blocker to resist vasoconstriction upon chemical and physical stimuli during the tissue section. Without gelatin inflation, the pulmonary arteries in lung slices usually collapse and detach from the surrounding interstitial tissue, even in the presence of high doses of mixed vasodilatory agents, including phentolamine, epinephrine, and nifedipine13,16. In contrast to an agarose gel, gelatin gel melts at 37 °C, flowing out of the vascular lumen after overnight incubation, leaving the arterial lumen free of obstruction prior to vascular reaction assay.
As the PCLS preserves pulmonary SMCs in situ and retains their contractile function in a nearly in vivo condition, it has been applied as a powerful platform to investigate the regulation of SMC contraction, especially the regulation via Ca2+ dependent mechanisms26. In particular, with a low-magnification multi-channel confocal or two-photon microscope, agonist-induced Ca2+ signaling in airway SMCs and associated luminal constriction can be captured simultaneously for mechanistic study13,20. Airway or vascular responsiveness measured using the PCLS method is expected to reflect cellular properties free of influences from the lung environment, such as the inflammatory milieu, and neural innervation of the response in the lung27,28. As such, the PLCS provides an experimental system to help distinguish intrinsic vs. secondary SMCs modification. In addition to investigating the contractile regulation of SMCs in health and disease models, PCLSs have been collected from different age groups to explore the functional adaptation of airway SMC during postnatal lung development and in response to environmental insults27. Moreover, PCLSs contain different-sized airways and blood vessels from the peripheral to the proximal lung field, enabling the investigation of a region-specific mechanism to regulate the pulmonary SMCs contractility in homeostasis and under pathogenic stimuli. Furthermore, as mouse PCLSs retain airway contractility in the culture medium for 7 days17, they have been used as an ex vivo model to examine or validate risk factors for SMC deregulation, such as cytokine or virus exposure29. Lastly, PCLS provides an ideal platform to screen vasodilatory or bronchodilatory medications. In particular, the bioassays using PCLS preparation are highly cost-effective, as one adult mouse can generate hundreds of lung slices. Using neighbor PCLSs in the control and treatment groups also significantly reduces the experimental bias from intergroup sample variation.
Considering the difference between rodent and human lung anatomy, the human PCLS is a more powerful tool for translational research. However, the limited availability of human lung tissue, especially diseased lung samples, remains a challenge. In contrast, mouse lung tissue, mouse models of human diseases, and transgenic mouse models are widely applied in biomedical and pharmacological research, making mouse PCLS an accessible and disease-relevant system13,30. In addition, the preservation of intrapulmonary arteries has been successful only in mouse PCLS preparation, which makes it a unique tool to explore the vascular deregulation in pulmonary vascular diseases such as pulmonary hypertension. Therefore, despite caveats associated with any disease models, a protocol for mouse PCLS preparation is invaluable to establish an ex vivo platform to investigate airway and pulmonary arteries in health and disease. We and others have reported lung research with human PCLSs31,32,33,34. In our experience, the protocol of human PLCS preparation is similar to the mouse one, except applying a higher agarose concentration of 2%, more agarose solution (3 L for one lung), and much larger-sized catheters to cannulate main,lobar, or segmental bronchi for agarose injection. Experience in mouse PLCS preparation helps tremendously with human lung slice preparation.
Despite numerous advantages of using PCLS preparation in pulmonary SMC research, it is crucial to be aware of the limitations of this technique. First, the PCLS remains a static system, lacking physiological breathing cycles that periodically stretch the lung parenchymal and airways. Neither does it contain blood circulation, which generates pulsive pressure on the vascular SMCs and shearing forces on endothelial cells. These mechanical variations in the PCLS could modify the contractile regulation of SMCs. Even though a full establishment of the air ventilation and blood circulation in PCLSs is unachievable, at least for the airway SMC study, vagarious efforts have been placed in the past decade to generate a "breathing" PCLS by stretching the tissue section with a variety of devices35,36,37. Second, pulmonary SMCs maintain their contractility only for a limited period. This time limit prevents the PLCS from modeling the subacute changes of SMCs, for instance, a process taking more than 6-7 days to modify airway SMCs. Since previous research reveals SMCs lose contractility due to a diminution of contractile proteins, optimizing the culture medium with an additional low dose of insulin has been shown to sustain the airway SMC contraction for up to 12 days17. Lastly, the genetic manipulation of pulmonary SMCs by plasmid or siRNA transfection of the PCLS remains unsuccessful. The technical barrier to transfect SMCs in the PCLS certainly warrants further investigation, as this method provides an alternative approach to the transgenic animal model for the mechanistic study of pulmonary SMCs. More importantly, this technique is the exclusive measure to achieve genetic modulation of human pulmonary SMCs in translational research.
This article provides a comprehensive description of preparing mouse PCLSs with well-preserved airways and pulmonary arteries and applying them in the contraction and Ca2+ signaling assays. PCLS preparation supports the smooth muscle functioning in a live lung environment while allowing mechanistic study access to cells in situ. This unique feature has made the PCLS preparation a versatile tool for studying the pulmonary airway and vascular SMCs in health and diseases.
Declarações
The authors have nothing to disclose.
Acknowledgements
This work is supported by NIH grants, K08135443 (Y.B), 1R01HL132991 (X.A).
Materials
| 1 mL syringe | BD | 309626 | |
| 15 mL sterile centrifuge tubes | Celltreat | 229411 | |
| 3 mL syringe | BD | 309585 | |
| 50 mL sterile centrifuge tubes | Celltreat | 229422 | |
| Acetyl-beta-methacholine | Millipore Sigma | 62-51-1 | |
| Antibiotic-anitmycotic | Thermo Fisher | 15240-062 | |
| CCD-camera | Nikon | Nikon Ds-Ri2 camera | |
| Cover glassess | Fisher Scientific | 12-548-5CP; 12-548-5PP | |
| Cryogenic vials | Fisher Scientific | 430488 | |
| Custom-built laser scanning confocal microscope | Details in Reference 18 | ||
| DMEM/F12 | Fisher Scientific | MT-10-092-CM | |
| Endothelin 1 | Millipore Sigma | E7764 | |
| Fine dissecting scissor | Fisher Scientific | NC9702861 | |
| Freezing container | Sigma-Aldrich | C1562 | |
| Gelatin from porcine skin | Sigma-Aldrich | 9000-70-8 | |
| Hanks' Balanced Salt Solution (HBSS) | Thermo Fisher | 14025092 | |
| Hemostatic forcep | Fisher Scientific | 16-100-117 | |
| HEPES | Thermo Fisher | 15630080 | |
| High vaccum silicone grease | Fisher Scientific | 146355d | |
| Isopropyl alcohol | Sigma-Aldrich | W292907-1KG-K | |
| Metal washers | Home Depot Product Authority | 800442 | Everbilt Flat Washers #10 |
| Micro-dissecting forcep | Sigma-Aldrich | F4142 | |
| Needle scalp vein set (25 G) | EXELINT | 26708 | |
| NOC-5 | Cayman Chemical | 16534 | |
| Nylon mesh | Component Supply | U-CMN-300 | |
| Oregon green 488 BAPTA-1 AM | Life Technologies | o-6807 | |
| Phase-contrast microscope | Nikon | Nikon Eclipse TS 100 | |
| Pluronic F-127 | Thermo Fisher | P-6867 | |
| Razor blades | Personna | Personna Double Edge Razor Blades in White Wrapper 100 count | |
| Sulfobromophthalein | Sigma-Aldrich | S0252 | |
| Superglue | Krazy Glue | Krazy Glue, All purpose | |
| Ultrapure low melting point agarose | Thermo Fisher | 16520050 | |
| Vibratome | Precisionary | VF 310-0Z | |
| Vibratome chilling block | Precisionary | SKU-VM-CB12.5-NC | |
| Vibratome specimen tube | Precisionary | SKU VF-SPS-VM-12.5-NC | |
| Y shaped IV catheter | BD | 383336 | BD Saf-T-Intima closed IV catheter |
Referências
- Prakash, Y. S. Emerging concepts in smooth muscle contributions to airway structure and function: implications for health and disease. American Journal of Physiology Lung Cellular and Molecular Physiology. 311 (6), 1113-1140 (2016).
- Lechartier, B., et al. Phenotypic diversity of vascular smooth muscle cells in pulmonary arterial hypertension: implications for therapy. Chest. 161 (1), 219-231 (2022).
- Doeing, D. C., Solway, J. Airway smooth muscle in the pathophysiology and treatment of asthma. Journal of Applied Physiology. 114 (7), 834-843 (2013).
- McGovern, T. K., et al. Evaluation of respiratory system mechanics in mice using the forced oscillation technique. Journal of Visualized Experiments: JoVE. (75), e50172 (2013).
- Bikou, O., et al. Induction and characterization of pulmonary hypertension in mice using the hypoxia/SU5416 model. Journal of Visualized Experiments: JoVE. (160), e59252 (2020).
- Stenmark, K. R., et al. Dynamic and diverse changes in the functional properties of vascular smooth muscle cells in pulmonary hypertension. Cardiovascular Research. 114 (4), 551-564 (2018).
- Bai, Y., Zhang, M., Sanderson, M. J. Contractility and Ca2+ signaling of smooth muscle cells in different generations of mouse airways. American Journal of Respiratory Cell and Molecular Biology. 36 (1), 122-130 (2007).
- Chin, L. Y., et al. Human airway smooth muscle is structurally and mechanically similar to that of other species. The European Respiratory Journal. 36 (1), 170-177 (2010).
- Wang, P., et al. Inflammatory mediators mediate airway smooth muscle contraction through a G protein-coupled receptor-transmembrane protein 16A-voltage-dependent Ca(2+) channel axis and contribute to bronchial hyperresponsiveness in asthma. The Journal of Allergy and Clinical Immunology. 141 (4), 1259-1268 (2018).
- Currigan, D. A., et al. Vasoconstrictor responses to vasopressor agents in human pulmonary and radial arteries: an in vitro study. Anesthesiology. 121 (5), 930-936 (2014).
- Halayko, A. J., et al. Divergent differentiation paths in airway smooth muscle culture: induction of functionally contractile myocytes. The American Journal of Physiology. 276 (1), 197-206 (1999).
- Worth, N. F., et al. Vascular smooth muscle cell phenotypic modulation in culture is associated with reorganisation of contractile and cytoskeletal proteins. Cell Motility and the Cytoskeleton. 49 (3), 130-145 (2001).
- Sanderson, M. J. Exploring lung physiology in health and disease with lung slices. Pulmonary Pharmacology and Therapeutics. 24 (5), 452-465 (2011).
- Placke, M. E., Fisher, G. L. Adult peripheral lung organ culture-a model for respiratory tract toxicology. Toxicology and Applied Pharmacology. 90 (2), 284-298 (1987).
- Fisher, G. L., Placke, M. E. In vitro models of lung toxicity. Toxicology. 47 (1-2), 71-93 (1987).
- Perez, J. F., Sanderson, M. J. The contraction of smooth muscle cells of intrapulmonary arterioles is determined by the frequency of Ca2+ oscillations induced by 5-HT and KCl. The Journal of General Physiology. 125 (6), 555-567 (2005).
- Li, G., et al. Preserving airway smooth muscle contraction in precision-cut lung slices. Scientific Reports. 10 (1), 6480 (2020).
- Sanderson, M. J., Parker, I. Video-rate confocal microscopy. Methods in Enzymology. 360, 447-481 (2003).
- Kolbe, U., et al. Early cytokine induction upon pseudomonas aeruginosa infection in murine precision cut lung slices depends on sensing of bacterial viability. Frontiers in Immunology. 11, 598636 (2020).
- Perez, J. F., Sanderson, M. J. The frequency of calcium oscillations induced by 5-HT, ACH, and KCl determine the contraction of smooth muscle cells of intrapulmonary bronchioles. The Journal of General Physiology. 125 (6), 535-553 (2005).
- Rosner, S. R., et al. Airway contractility in the precision-cut lung slice after cryopreservation. American Journal of Respiratory Cell and Molecular Biology. 50 (5), 876-881 (2014).
- Bai, Y., Sanderson, M. J. Modulation of the Ca2+ sensitivity of airway smooth muscle cells in murine lung slices. American Journal of Physiology-Lung Cellular and Molecular Physiology. 291 (2), 208-221 (2006).
- Sanderson, M. J., et al. Fluorescence microscopy. Cold Spring Harbor Protocols. 10, 071795 (2014).
- Sanderson, M. J., Bai, Y., Perez-Zoghbi, J. Ca(2+) oscillations regulate contraction of intrapulmonary smooth muscle cells. Advances in Experimental Medicine and Biology. 661, 77-96 (2010).
- Perez-Zoghbi, J. F., Bai, Y., Sanderson, M. J. Nitric oxide induces airway smooth muscle cell relaxation by decreasing the frequency of agonist-induced Ca2+ oscillations. The Journal of General Physiology. 135 (3), 247-259 (2010).
- Lam, M., Lamanna, E., Bourke, J. E. Regulation of airway smooth muscle contraction in health and disease. Advances in Experimental Medicine and Biology. 1124, 381-422 (2019).
- Patel, K. R., et al. Targeting acetylcholine receptor M3 prevents the progression of airway hyperreactivity in a mouse model of childhood asthma. FASEB Journal: Official Publication of the Federation of American Societies for Experimental Biology. 31 (10), 4335-4346 (2017).
- Aven, L., et al. An NT4/TrkB-dependent increase in innervation links early-life allergen exposure to persistent airway hyperreactivity. FASEB Journal: Official Publication of the Federation of American Societies for Experimental Biology. 28 (2), 897-907 (2014).
- Liu, G., et al. Use of precision cut lung slices as a translational model for the study of lung biology. Respiratory Research. 20 (1), 162 (2019).
- Wu, X., et al. Mouse lung tissue slice culture. Methods in Molecular Biology. 1940, 297-311 (2019).
- Bai, Y., et al. CD38 plays an age-related role in cholinergic deregulation of airway smooth muscle contractility. The Journal of Allergy and Clinical Immunology. 6749 (21), 01760-01767 (2021).
- Khan, M. M., et al. An integrated multiomic and quantitative label-free microscopy-based approach to study pro-fibrotic signalling in ex vivo human precision-cut lung slices. The European Respiratory Journal. 58 (1), (2021).
- Kennedy, J. L., et al. Effects of rhinovirus 39 infection on airway hyperresponsiveness to carbachol in human airways precision cut lung slices. The Journal of Allergy and Clinical Immunology. 141 (5), 1887-1890 (2018).
- Bai, Y., et al. Cryopreserved Human precision-cut lung slices as a bioassay for live tissue banking. a viability study of bronchodilation with bitter-taste receptor agonists. American Journal of Respiratory Cell and Molecular Biology. 54 (5), 656-663 (2016).
- Mondoñedo, J. R., et al. A high-throughput system for cyclic stretching of precision-cut lung slices during acute cigarette smoke extract exposure. Frontiers in Physiology. 11, 566 (2020).
- Davidovich, N., Huang, J., Margulies, S. S. Reproducible uniform equibiaxial stretch of precision-cut lung slices. American Journal of Physiology-Lung Cellular and Molecular Physiology. 304 (4), 210-220 (2013).
- Ram-Mohan, S., et al. Tissue traction microscopy to quantify muscle contraction within precision-cut lung slices. American Journal of Physiology-Lung Cellular and Molecular Physiology. 318 (2), 323-330 (2020).

