Analysis of Raw and Processed Cyperi Rhizoma Samples Using Liquid Chromatography-Tandem Mass Spectrometry in Rats with Primary Dysmenorrhea
Summary
Here, a comparative analysis of raw and processed Cyperi rhizoma (CR) samples is presented using ultra-high performance liquid chromatography-high-resolution tandem mass spectrometry (UPLC-MS/MS) in rats with primary dysmenorrhea. The changes in blood levels of the metabolites and the sample constituents were examined between rats treated with CR and CR processed with vinegar (CRV).
Abstract
Cyperi rhizoma (CR) is widely used in gynecology and is a general medicine for treating women’s diseases in China. Since the analgesic effect of CR is enhanced after processing with vinegar, CR processed with vinegar (CRV) is generally used clinically. However, the mechanism by which the analgesic effect is enhanced by vinegar processing is unclear. In this study, the ultra-high pressure liquid chromatographytandem mass spectrometry (UPLC-MS/MS) technique was used to examine changes in the blood levels of the exogenous constituents and metabolites between CR-treated and CRV-treated rats with dysmenorrhea. The results revealed differing levels of 15 constituents and two metabolites in the blood of these rats. Among them, the levels of (-)-myrtenol and [(1R,2S,3R,4R)-3-hydroxy-1,4,7,7-tetramethylbicyclo[2.2.1]hept-2-yl]acetic acid in the CRV group were considerably higher than in the CR group. CRV reduced the level of 2-series prostanoids and 4-series leukotrienes with proinflammatory, platelet aggregation, and vasoconstriction activities and provided analgesic effects by modulating arachidonic acid and linoleic acid metabolism and the biosynthesis of unsaturated fatty acids. This study revealed that vinegar processing enhances the analgesic effect of CR and contributes to our understanding of the mechanism of action of CRV.
Introduction
Primary dysmenorrhea (PD) is the most prevalent condition in clinical gynecology. It is characterized by backache, swelling, abdominal pain, or discomfort before or during menstruation without pelvic pathology in the reproductive system1. A report on its prevalence showed that 85.7% of students suffer from PD2. Low-dose oral contraceptives are the standard therapy, but their adverse side effects, such as deep vein thrombosis, have drawn increasing attention3. The prevalence of deep vein thrombosis among oral contraceptive users is >1 per 1,000 women, and the risk is highest during the first 6-12 months and in users older than 40 years4.
Long used in traditional Chinese medicine (TCM), Cyperi rhizoma (CR) is derived from the dried rhizome of the Cyperus rotundus L. of the Cyperaceae family. CR regulates menstrual disorders and relieves depression and pain5. CR is widely used in gynecology and is considered a general medicine to treat women's diseases6. CR processed with vinegar (CRV) is typically used clinically. Compared with CR, CRV shows enhanced regulation of menstruation and pain relief. Modern studies have shown that CR inhibits cyclooxygenase-2 (COX-2) and the subsequent synthesis of prostaglandins (PGs), thus achieving an anti-inflammatory effect. Meanwhile, CR exhibits an analgesic effect without side effects7, making CR a good choice for dysmenorrhea patients. However, the mechanism underlying the regulation of menstruation and provision of pain relief by CRV is unclear. CR research has mainly focused on changes in its active chemical components and pharmacologic activities, such as its anti-inflammatory, antidepressant, and analgesic effects8,9,10,11,12.
Although the ingredients of TCM are complex, they are absorbed into the blood and must reach a specific blood concentration to be effective13. The scope of screening the active ingredients of TCM can be narrowed by utilizing the strategy of constituent determination in the blood. Blindness can be avoided in studying the chemical components in vitro, and one-sidedness can be avoided in studying the individual constituents14. By comparing the compositions of CR and CRV in the blood, changes in the active ingredients of the processed CR can be detected effectively and quickly. Drug efficacy is the process by which a drug influences the body. Changes in the drug components due to the body's metabolic response, which may be related to the action mechanism of the drug, can be determined with metabonomics. Metabonomics aims to measure the overall and dynamic metabolic responses, which is consistent with determining the overall efficacy of traditional Chinese medicine15. Furthermore, metabolites are the final product of gene expression, which is most closely related to phenotypes16. Thus, metabonomics may be suitable for exploring the differences in the metabolic pathways between CR and CRV in the treatment of PD. Liquid chromatography-high-resolution tandem mass spectrometry (LC-MS/MS)-based untargeted metabolomics is characterized by high throughput, high sensitivity, and high resolution and can be used to measure many different small molecular components17,18. This method can simultaneously determine the endogenous metabolites and exogenous constituents absorbed into the blood. Metabonomics has been widely used in studies on TCM19, drug toxicology20, health management21, sports22, food23, and other fields.
In this study, the differences in the exogenous constituents absorbed into the blood and the endogenous metabolites were measured between CR-treated and CRV-treated dysmenorrhea model rats using LC-MS/MS-based untargeted metabolomics to reveal the mechanisms of the analgesic effects of CRV.
Protocol
All animal-related experiments were conducted with approval from the Experiment Ethics Committee of Chongqing Institute of TCM. Twenty-four female Sprague Dawley rats (SD) that were 8-10 weeks old and weighed 200 g ± 20 g were used in this experiment.
1. Preparation of the extraction
- Calculation
- Plan to administer the CR or CRV extract to a treatment group of six Sprague-Dawley rats (10 g/[kg∙day]) for 3 days. Use a CR or CRV extract concentration of 1 g/mL (1 mL of the extract is obtained from 1 g of herbs).
NOTE: The dosage of CR is 6-10 g. In this study, the maximum dose of 10 g was used as the dosage. As the average weight of an adult person is 60 kg, the adult dose is 0.1667 g/kg. According to the weight conversion algorithm24, as the dose conversion coefficient between humans and rats is 6.3, the dosage for rats is 1.05 g/kg. The drug dosage was increased by 10 times to 10.5 g/kg for rats. The dosage was set as 10 g/kg for the convenience of calculation and the actual experiment. For example, by the calculation, if a total of 36 g of CR or CRV is needed, the Chinese herbal medicine should be prepared at least twice. Thus, 200 g of CR was required-100 g of CR was used as the CR, and 100 g of CR was processed into CRV. - Calculate the volume of CR or CRV to be applied per rat using equation (1):
V = 10 g/(kg∙day) × 200 g/(1 g/mL) = 2 mL (1)
- Plan to administer the CR or CRV extract to a treatment group of six Sprague-Dawley rats (10 g/[kg∙day]) for 3 days. Use a CR or CRV extract concentration of 1 g/mL (1 mL of the extract is obtained from 1 g of herbs).
- Processing of the CRV
- Mix 100 g of CR and 20 g of vinegar (>5.5 g acetic acid/100 mL) thoroughly and incubate for 12 h.
NOTE: To ensure that the interior of the CR was moistened by vinegar after 12 h, the CR and vinegar were mixed, stirred well, and then stirred again until the inner portion was moist. - Stir-fry the mixture in an iron pan for 10 min at 110-120 °C. Then, take out the mixture, and let it cool at room temperature.
NOTE: To prevent the CR from scorching, it is necessary to stir continuously while heating. If the processed CRV is too wet, it can be dried at 60 °C. When the surface of the CR is sepia, the stir-frying can be stopped.
- Mix 100 g of CR and 20 g of vinegar (>5.5 g acetic acid/100 mL) thoroughly and incubate for 12 h.
- Extraction
- CR extract
- Add 10 times (the amount of CR) pure water to the CR, and soak for 2 h. Make sure that the medicinal materials are below the liquid level when soaking.
NOTE: The CR only needs to be cut in half before extraction. The purpose of soaking is to extract the active constituents more effectively. The process of soaking is essential. - Bring the mixture of water and medicine to the boil over a high heat, and keep it boiling over a low heat for 20 min. Filter with a filter cloth (100 mesh), and collect the filtrate.
NOTE: When decocting, a high heat was used before boiling, and a low heat was used to maintain the boiling. - Repeat step 1.3.1.2 once, and combine the filtrates.
- Concentrate the extract with a rotary evaporator to 1 g/mL (based on the original medicine, the concentration temperature must be below 60 °C).
NOTE: The active components in CR are volatile, so the concentration temperature should not be higher than 60 °C.
- Add 10 times (the amount of CR) pure water to the CR, and soak for 2 h. Make sure that the medicinal materials are below the liquid level when soaking.
- CRV extract
- Perform the same steps (1.3.1.1-1.3.1.3) as for the CR extraction method.
- CR extract for testing
- Pipette 500 µL of the CR extract and 500 µL of methanol into a 1.5 mL microcentrifuge tube, and vortex for 30 s to mix.
NOTE: A mixture of methanol and water extracts the active components better. The extract should not be directly filtered for testing. - Centrifuge each sample for 15 min at 1,6502 × g at 4 °C. Filter the supernatant, and then transfer it to the sample vial for testing.
NOTE: After the high-speed centrifugation of the mixture of methanol and extract, the supernatant can be directly transferred to the sample bottle for determination without filtration. Due to the heat generated by the centrifugation process, it is preferable to use a cryogenic centrifuge.
- Pipette 500 µL of the CR extract and 500 µL of methanol into a 1.5 mL microcentrifuge tube, and vortex for 30 s to mix.
- CRV extract for testing
- Perform steps 1.3.3.1-1.3.3.2 to prepare the CRV extract for testing.
- CR extract
2. Animals
- Calculation
- Take 50 mg of estradiol benzoate, and add it to 50 mL of olive oil to prepare a 1 mg/mL solution. Take 50 mg of oxytocin, and add it to 50 mL of normal saline to prepare a 1 mg/mL solution.
NOTE: The dose of the intraperitoneal injection of estradiol benzoate and oxytocin is 10 mg/(kg∙day). Estradiol benzoate is dissolved in olive oil, and oxytocin is dissolved in normal saline. Estradiol benzoate is not easy to dissolve in olive oil and can be treated with ultrasound to accelerate dissolution. Both the estradiol benzoate and oxytocin solutions must be prepared daily. - Calculate the volume of estradiol benzoate solution to be applied per rat (i.e., V = 10 mg/[kg∙day] × 200 g/[1 g/mL] = 2 mL). Calculate the volume of oxytocin solution to be applied per rat (i.e., V = 10 mg/[kg∙day] × 200 g/[1 g/mL] = 2 mL).
- Take 50 mg of estradiol benzoate, and add it to 50 mL of olive oil to prepare a 1 mg/mL solution. Take 50 mg of oxytocin, and add it to 50 mL of normal saline to prepare a 1 mg/mL solution.
- Animal grouping and administration
NOTE: Ten days were assigned for administration25,26. During treatment, the rats had unrestricted access to standard chow and water. Within 30 min of oxytocin administration, each rat's writhing activity was tracked. The PD rat model was developed successfully, as evidenced by the model rats' twisting responses, which included uterine contraction, one limb rotation, hind limb extension, a hollow trunk, and abdominal contraction26.- Assign 24 female Sprague-Dawley rats (SD rats, 8-10 weeks of age, weighing 200 g ± 20 g) into four groups at random-control, model, CR, and CRV-and feed them for 7 days.
- Animal administration
- Intraperitoneally inject rats in the model, CR, and CRV groups with 2 mL of estradiol benzoate solution every day. Intraperitoneally inject the rats in the control group with 2 mL of normal saline.
- From day 8, complete step 2.2.2.1. Then, administer intragastrically 2 mL of CRV extract to the rats in the CRV group, 2 mL of CR extract to the rats in the CR group, and 2 mL of normal saline to the rats in the control and model groups.
- On day 10, complete step 2.2.2.2. Then, intraperitoneally inject the rats in the model, CR, and CRV groups with 2 mL of oxytocin solution and the rats in the control group with 2 mL of normal saline.
- Record the writhing times of the rats within 30 min of the oxytocin injection.
- Sample collection
- Collect abdominal aorta blood samples, excise the uterus rapidly and completely, and carefully separate the connective tissue and fat adhering to the uterine wall.
NOTE: Blood was collected as close to within 1 h after the final dose. - Use microcentrifuge tubes to preserve and transfer the uterine tissue to liquid nitrogen. Store the tissue samples at −80 °C.
- Centrifuge the blood samples for 10 min at 4,125 × g. Remove the serum-containing supernatant, and centrifuge at 16,502 × g for 10 min at 4 °C. Centrifuge the serum, and then keep it in the tube at −80 °C for storage.
NOTE: The blood samples must be left at room temperature for 1 h for reprocessing.
- Collect abdominal aorta blood samples, excise the uterus rapidly and completely, and carefully separate the connective tissue and fat adhering to the uterine wall.
- Sample processing
- Serum samples
- Put 400 µL of methanol and 200 µL of serum into a microcentrifuge tube, and vortex for 30 s to mix. Centrifuge each sample for 15 min at 16,502 × g at 4 °C. Fill sample bottles with the supernatant after collection and filtration. Mix all the supernatants from each sample with the same volume to prepare quality control samples for testing.
- Uterine tissue samples
- Take 100 mg of uterine tissue from the ipsilateral segment and grind it with a ninefold volume of normal saline. Centrifuge the homogenate for 10 min at 4,125 × g, and collect the supernatant. Place the supernatant in a refrigerator at 4 °C for testing or at −80 °C if not to be tested on the same day.
- Place normal saline, tissue, and steel balls in a 2 mL microcentrifuge tube, put the microcentrifuge tube into liquid nitrogen for 3-5 s, and then put the tissue into a tissue grinder for grinding.
- Serum samples
- Sample testing
- Use an enzyme-linked immunosorbent assay (ELISA) to measure the PGF2α and PGE2 content in the uterine tissues of the rat samples.
NOTE: The rat PGE2 ELISA kit was used to measure the PGE2 content, and the rat PGF2α ELISA kit was used to measure the PGF2α level. The detailed steps can be found in the manufacturer's instructions. The details of the kit are given in the Table of Materials. - Assess the serum sample, the CR extract, and the CRV extract using UPLC-MS/MS.
- For UPLC, use a C18 column (2.6 µm, 2.1 mm x 100 mm) and a binary gradient method with mobile phase A containing 0.1% formic acid and mobile phase B containing acetonitrile. Set the elution gradient as follows: from 0 min to 1 min, 15% B; from 1 min to 8.5 min, 15% to 85% B; from 8.5 min to 11.5 min, 85% B; from 11.5 min to 11.6 min, 99% to 1% B; and from 11.6 min to 15 min, 15% B. Set the flow rate to 0.35 mL/min and the injection volume to 2 µL.
- For MS, set the temperature to 600 °C, the curtain gas (CUR) flow rate to 0.17 MPa, and both the sheath and auxiliary gas flow rates to 0.38 MPa. Set the ion spray voltage of the positive ion mode and negative ion mode to 5.5 kV and −4.5 kV, respectively, the declustering potential (DP) voltage to 80 V or −80 V, the collision energy (CE) to 40 eV or −25 eV, and the collision energy superposition (CES) to 35 eV ± 15 eV.
- Perform the test following the manufacturer's protocol using UPLC-MS/MS. Perform MS for a mass range of 50-1,000 m/z.
- Obtain the UPLC-MS/MS results using the matching workstation program in conjunction with the detection mode's information-dependent acquisition, multiple mass defect filter, and dynamic background deduction. Use the quality control samples of pooled serum to test the repeatability and stability of the UPLC-MS/MS equipment to verify the conventional approach. Before the research samples, inject the quality control samples for four successive runs, and then inject them after every five serum samples.
- Use an enzyme-linked immunosorbent assay (ELISA) to measure the PGF2α and PGE2 content in the uterine tissues of the rat samples.
3. Data processing
- Data preparation
- Use conversion software to convert the raw data to the mzXML format. Normalize the total ion current (TIC) data of each sample.
NOTE: An internal R-based application (www.lims2.com) built on XCMS was used to analyze the information for the integration, alignment, extraction, and peak detection27. - Perform metabolite annotation using a proprietary MS2 database (www.lims2.com). Set the annotation threshold to 0.328.
NOTE: The endogenous metabolites were identified in each group.
- Use conversion software to convert the raw data to the mzXML format. Normalize the total ion current (TIC) data of each sample.
- Principal component analysis and orthogonal partial least square discriminant analysis
- Use analysis software to perform the principal component analysis (PCA) and the modeling. Import the standardized data of the metabolites to the analysis software. Then, use autofit to build the analysis model. Finally, use score to obtain the score scatter plot of the PCA.
NOTE: The clustering of each group was obtained with the score scatter plot of the PCA. PCA is an unsupervised analysis mode that mainly groups the samples through dimension reduction without intervention. - Use the analysis software to perform the orthogonal partial least square discriminant analysis (OPLS-DA).
- Import the standardized data on the metabolites in the CR and CRV groups into the analysis software.
- Import the data from the CR group into the created CR group, and import the data from the CRV group into the created CRV group.
NOTE: OPLS-DA is a supervised analysis mode, and grouping of the samples is necessary. - Then, use autofit to build the analysis model, and use score to obtain the score scatter plot of the OPLS-DA. Finally, use VIP to obtain the variable significance in the projection (VIP) value in the OPLS-DA.
NOTE: The variable significance in the projection (VIP) values of the metabolites in the CR and CRV groups were obtained through the OPLS-DA.
- Use analysis software to perform the principal component analysis (PCA) and the modeling. Import the standardized data of the metabolites to the analysis software. Then, use autofit to build the analysis model. Finally, use score to obtain the score scatter plot of the PCA.
- Identification of the potential differential metabolites
- Screen out the metabolites with VIP values greater than 1 in step 3.2.2.3.
- Use statistical software to calculate the P value of the metabolites that were screened out in step 3.3.1 by the Student's t-test.
NOTE: Significant differences in the potential differential metabolites in the CR and CRV groups were determined by a Student's t-test. The potential differential metabolites were those with a Student's t-test p-value < 0.05 and a variable significance in the projection (VIP) greater than 1. The representation was accomplished using a volcanic plot.
- Identification of the differential metabolites
- Screen out the potential differential metabolites in step 3.3. Use the results of step 3.1.2 to identify these differential metabolites.
NOTE: A small number of potential differential metabolites were identified, and they became the candidate differential metabolites. - Screen out the differential metabolites to be matched in the KEGG database. Show the changes in the differential metabolites in the CR and CRV groups by drawing a heatmap.
NOTE: A small number of candidate differential metabolites were matched, and they became the differential metabolites.
- Screen out the potential differential metabolites in step 3.3. Use the results of step 3.1.2 to identify these differential metabolites.
- Examination of the potential metabolic pathways
- Upload the different metabolites to the Metaboanalyst (www.metaboanalyst.ca) database.
- Use Pathways Analysis to obtain the potential metabolic pathways.
NOTE: The potential metabolic pathways were obtained in the CR and CRV groups. The P value and impact value were two very important indicators in the selection of the critical pathways. The P value was more important than the impact value. Significance was defined by a P value < 0.05; bigger impact values were associated with better correlations. - Analyze the potential metabolic pathways by uploading the different metabolites to the KEGG (http://www.kegg.jp/kegg/pathway.html) database.
NOTE: The relationship between the function of the metabolic pathway and PD should also be considered. The critical metabolic pathways were obtained.
- Identification of the constituents absorbed into the blood
- Identify the chemical constituents in the CR and CRV extracts using the in-house MS2 database (www.lims2.com), the Human Metabolome Database (HMDB), and the Massbank and Chemspider databases.
- Determine the constituents absorbed into the blood in the CR and CRV groups, and compare the constituents between the CR and CRV groups.
NOTE: The constituents absorbed into the blood must be detected in the CR or CRV groups but cannot be detected in the control group.
- Statistical analysis
- Analyze the data using univariate analysis (UVA) and multivariate statistical methods, including the analysis of variance (ANOVA) and the Student's t-test.
NOTE: The experimental information was presented using statistical software as mean ± standard deviation (±SD). P < 0.01 was considered a highly significant difference, and P < 0.05 represented a significant difference28.
- Analyze the data using univariate analysis (UVA) and multivariate statistical methods, including the analysis of variance (ANOVA) and the Student's t-test.
Representative Results
Analysis of the dysmenorrhea model experiment
There was no writhing response within 30 min in the control group because these rats were not intraperitoneally injected with oxytocin and estradiol benzoate to cause pain. The rats in the model, CR, and CRV groups displayed substantial writhing reactions following the oxytocin injection. These outcomes demonstrate the efficacy of the estradiol benzoate and oxytocin combination for inducing dysmenorrhea. The differences in the PGF2α, PGE2, and PGF2α/PGE2 levels between the model and the control groups were significant (P < 0.001, P < 0.05), demonstrating the model's efficacy (Figure 1).
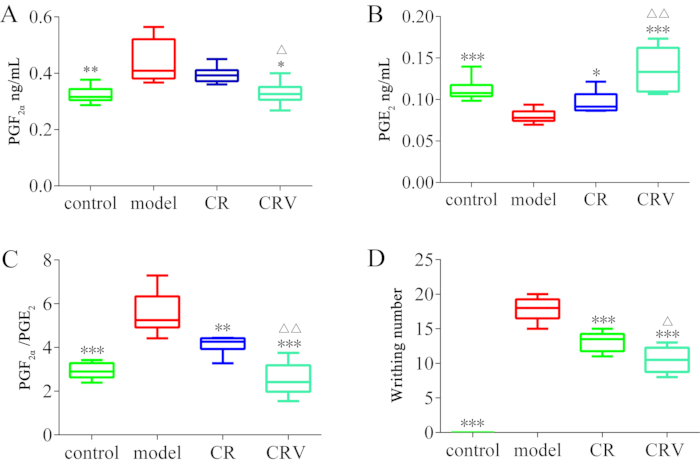
Figure 1: The PGF2α and PGE2 content in the uterine tissue and the writhing number in each group. (A) The PGF2α content in the uterine tissue in each group. (B) The PGE2 content in the uterine tissue in each group. (C) The PGF2α/PGE2 content in the uterine tissue in each group. (D) The writhing number in each group. The columns represent the mean ± SEM from four groups (six mice per group). * P < 0.05 or ** P < 0.01 represent a significant difference compared to the model group, and ΔP < 0.05 or ΔΔP < 0.01 represent a significant difference compared to the CR group. The rats in the model, CR, and CRV groups displayed a substantial writhing reaction following the oxytocin injection. Abbreviations: PG = prostaglandin; CR = Cyperi rhizome; CRV = CR processed with vinegar. Please click here to view a larger version of this figure.
The concentrations of PGF2α and PGF2α/PGE2 were reduced in the CRV and CR groups, and this reduction was more marked in the CRV group. There were also significant (P < 0.001 and P < 0.05) differences in the concentrations of PGF2α and PGF2α/PGE2 between the CRV and the CR groups. Compared to the model group, the PGE2 concentration was significantly greater in the CRV and CR groups (P < 0.001), with the level increasing the most in the CRV group.
Quality control
A good correspondence was observed between the retention times of the BPC chromatographic peaks and signal intensities in the quality control samples, demonstrating a high level of instrument stability and remarkably consistent data quality (Supplementary File 1-Supplementary Figure 1 and Supplementary Figure 2). Furthermore, all the quality control samples were within ±2 standard deviations (Figure 2A,B), and the correlation between the quality control samples was greater than 0.7 (Figure 2C,D). These results suggest that the procedure was reliable and that the results were credible.
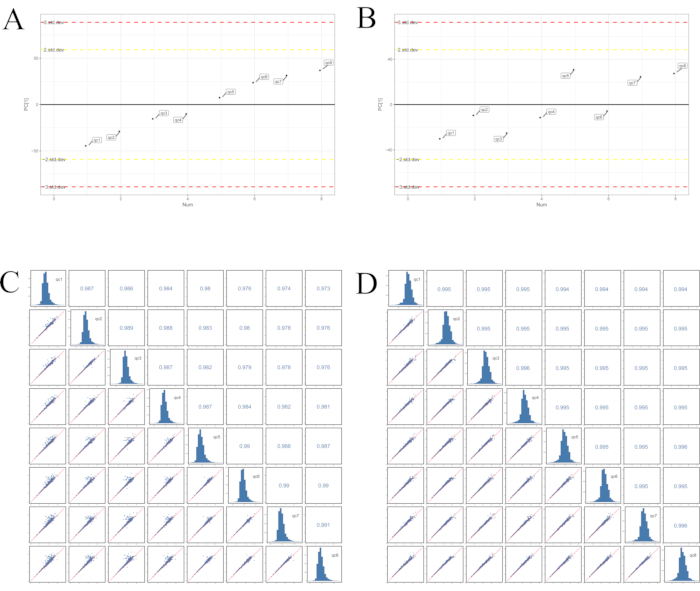
Figure 2: PCA-X one-dimensional distribution of the quality control samples. (A) Positive mode, (B) negative mode; correlation analysis of the quality control samples in (C) positive mode and (D) negative mode. All the quality control samples were within ±2 standard deviations, and the correlations between the quality control samples were greater than 0.7. These results suggested that the procedure was reliable and the information was credible. Abbreviations: PC = principal component; PCA = principal component analysis; quality control; CR = Cyperi rhizome; CRV = CR processed with vinegar. Please click here to view a larger version of this figure.
Principal component analysis
As shown in Figure 3, the abscissa PC (1) indicated the scores of the first principal components, while the ordinate PC (2) indicated the scores of the second principal components. In the figure, the green circle indicates the control group, the red circle represents the model group, the blue circle indicates the CR group, the white circle denotes the CRV group, and the pink circle indicates the quality control group.
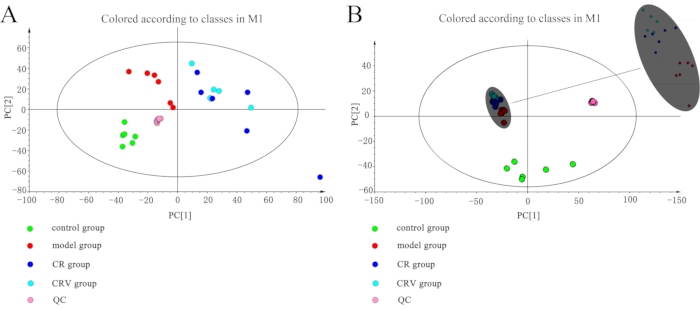
Figure 3: Score scatter plot of the PCA. (A) Positive mode and (B) negative mode. The quality control samples overlap, which indicates that the instrument was very stable. Each group is distributed in its own area, with only the CR group and the CRV group occasionally crossing paths. Abbreviations: PC = principal component; PCA = principal component analysis; QC = quality control; CR = Cyperi rhizome; CRV = CR processed with vinegar. Please click here to view a larger version of this figure.
The PCA analysis results demonstrated that the clusters of the CR and the model groups were significantly separated in both the positive and negative ion modes. The clusters were significantly separated between the CRV and model groups in the positive and negative ion modes. The quality control, model, and control groups were separated from the other treatment groups (CR, CRV). The CR and CRV groups occasionally overlapped in the positive ion mode. In the negative ion mode, each group was separated significantly.
Orthogonal partial least squares method discriminant analysis
OPLS-DA was used to screen for metabolic differences. The OPLS-DA scatter plot showed that all the samples were within the 95% confidence interval (Hotelling's T-square ellipse). Figure4A,C demonstrate that the CR and CRV groups were separated. Model overfitting was tested using the permutation test (n = 200), and the model's statistical significance was assessed. In Figure 4B,D, the ordinate depicts the value of R2Y or the Q2 value, and the abscissa indicates the replacement retention. R2Y is represented by the green dot, Q2 is represented by the blue square dot, and the two dashed lines represent their corresponding regression lines. In the positive and negative modes, the R2Y values were 0.8 and 0.84, and the Q2 values were −0.59 and −0.57, respectively. This demonstrates the model's high reliability and the absence of overfitting.
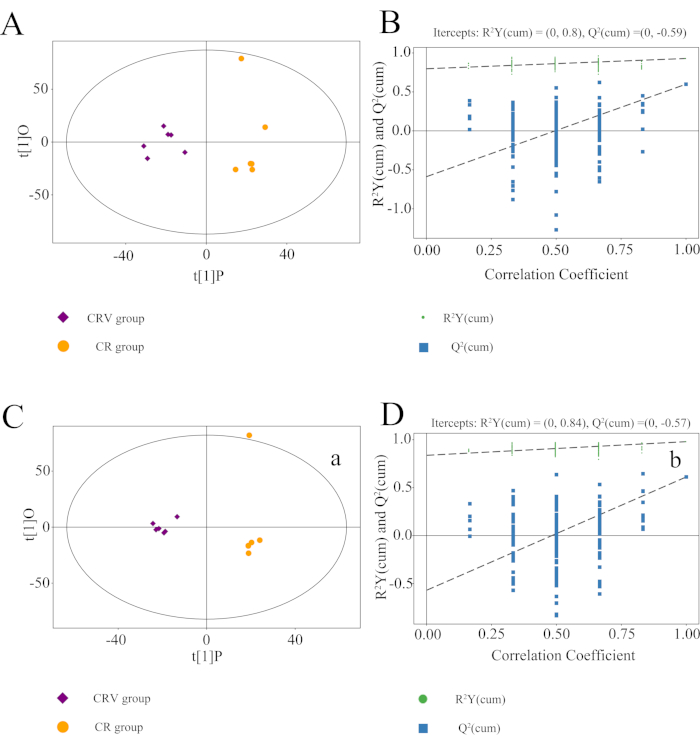
Figure 4: OPLS-DA models for the CR group versus the CRV group. Score scatter plot in (A) positive mode and (C) negative mode. Permutation test of the OPLS-DA model for the CR group versus the CRV group in (B) positive mode and (D) negative mode. R2Y was 0.8 and 0.84, and Q2 was −0.59 and −0.57 in the positive and negative modes, respectively. This demonstrates the model's high reliability and the absence of overfitting behavior. Abbreviations: OPLS-DA = orthogonal partial least squares discriminant analysis; CR = Cyperi rhizome; CRV = CR processed with vinegar. Please click here to view a larger version of this figure.
Univariate statistical analysis
Univariate statistical analyses were performed to identify metabolic variations. Under the standard screening of VIP > 1 and P < 0.05, 339 and 394 potential differential metabolites were detected in the positive and negative ion modes, respectively. A volcano plot is shown in Figure 5, wherein each point corresponds to a different metabolite. The ordinate shows the P value of the Student's t-test, and the abscissa reflects multiple changes in the level of each molecule in the group. The scatter size represents the VIP value of the OPLS-DA model; the VIP value increases with the size of the scatter. The red dots indicate an increase, the blue dots indicate a decrease, and the gray indicates no significant difference.
A qualitative analysis of the candidate differential metabolites was performed using secondary mass spectrometry. Significant changes were observed in 63 metabolites in positive mode (Supplementary File 1-Supplementary Table 1) and in 30 metabolites in negative mode (Supplementary File 1-Supplementary Table 2). The differential metabolites were determined using the KEGG and HDMB databases. The accurately matched compounds were identified as differential metabolites, and these are listed in Table 3 and Table 4.
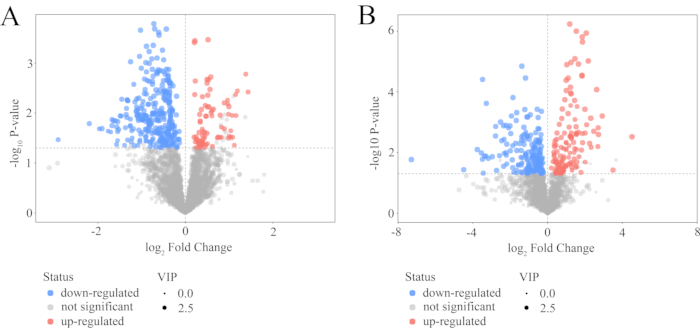
Figure 5: Volcano plot for the CR group versus the CRV group. (A) Positive mode and (B) negative mode. In the volcano graph represented, each point corresponds to a different metabolite. The ordinate shows the P value of the Student's t-test, and the abscissa reflects the multiple changes in each substance in the group. The scatter size represents the VIP value of the OPLS-DA model. The VIP value increases with the size of the scatter. Red dots indicate increases, blue dots indicate decreases, and gray indicates no significant differences. Abbreviations: OPLS-DA = orthogonal partial least squares discriminant analysis; VIP = variable significance in the projection; CR = Cyperi rhizome; CRV = CR processed with vinegar. Please click here to view a larger version of this figure.
Metabolite comparisons
The Euclidean distance matrix of the quantitative values of the differential metabolites between the CR and CRV groups was calculated, and the differential metabolites were clustered using a comprehensive correlation approach.
The abscissa represents different experimental groups, while the ordinate represents the relative level in Figure 6. The placement of the color patches indicates how each metabolite is expressed relative to the others. In the positive ion mode, compared with the CR group, the levels of four differential metabolites in the CRV group increased, while the levels of 11 differential metabolites decreased. In the negative ion mode, compared with the CR group, the levels of four differential metabolites in the CRV group increased, and the levels of 7 differential metabolites decreased.
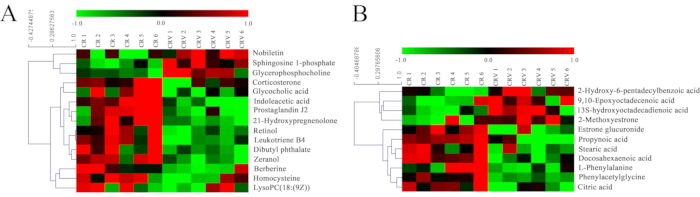
Figure 6: Heatmap analysis for the CRV group versus the CR group. (A) Positive mode and (B) negative mode. The abscissa represents the different experimental groups, and the ordinate represents the relative expression levels. The placement of the color patches indicates how each metabolite is expressed relative to the others. Abbreviations: CR = Cyperi rhizome; CRV = CR processed with vinegar. Please click here to view a larger version of this figure.
Compared with the CR group, the metabolites that increased in the CRV group were sphingosine 1-phosphate, nobiletin, glycerophosphocholine, lysopc(18:1(9z)), 2-hydroxy-6-pentadecylbenzoic acid, 9,10-epoxyoctadecenoic acid, 13s-hydroxyoctadecadienoic acid, and 2-methoxyestrone, while those that decreased were corticosterone, indoleacetic acid, glycocholic acid, berberine, dibutyl phthalate, retinol, leukotriene B4, prostaglandin J2, 21-hydroxypregnenolone, zeranol, homocysteine, propynoic acid, stearic acid, docosahexaenoic acid, phenylacetylglycine, L-phenylalanine, estrone glucuronide, and citric acid (Figure 7).
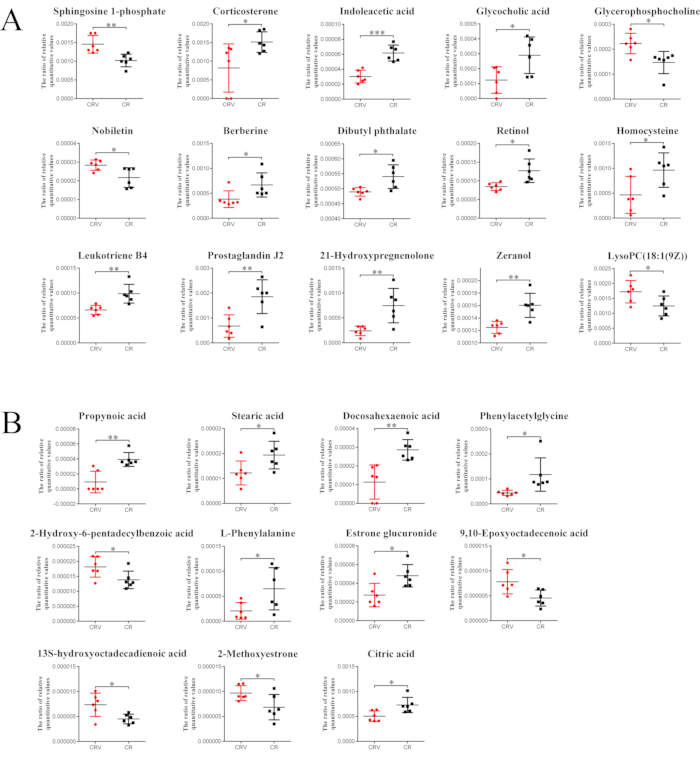
Figure 7: The relative level trends of the potential metabolites in the CRV and CR groups. (A) Positive modeand (B) negative mode. In the positive mode, compared with the CR group, the levels of four differential metabolites increased in the CRV group, and the levels of 11 decreased. In the negative mode, the levels of four differential metabolites increased in the CRV group, and the levels of seven differential metabolites decreased. The columns represent the mean ± SEM from four groups (six mice per group). * P < 0.05, ** P < 0.01, or *** P < 0.01 represent a significant difference between the CRV and CR groups. Abbreviations: CR = Cyperi rhizome; CRV = CR processed with vinegar. Please click here to view a larger version of this figure.
KEGG pathway analysis
The differential metabolites were annotated, and the KEGG pathways were analyzed. The results showed that the differential metabolites were associated with nine pathways in the positive and negative modes, respectively (Table 1 and Table 2). In Figure 8, the metabolic pathways are each represented by a bubble in the bubble chart, with a more significant scale indicating a greater effect factor. The size of the path influencing the factors in the topology analysis is represented by the abscissa of the bubble chart and the bubble size. The ordinate of the bubble chart and the bubble color indicates the P value of the enrichment analysis (taking the negative natural logarithm, i.e., −ln (p)). The degree of enrichment is more significant, the P value is smaller, the ordinate value is more prominent, and the color is darker. The P value and impact value are two very important indicators in the selection of critical pathways. Generally, the P value is more important than the impact value.
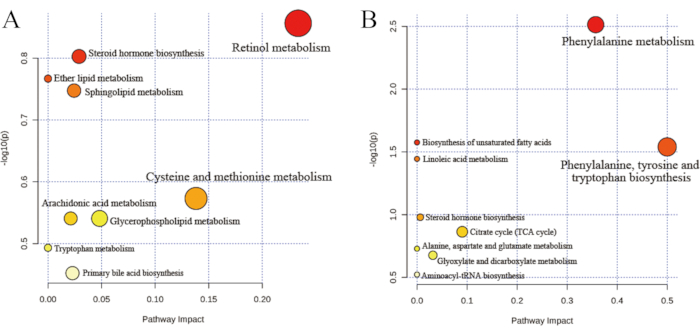
Figure 8: Pathway analysis for the CRV group versus the CR group. (A) Positive mode and (B) negative mode. The metabolic pathways are each represented by a bubble in the bubble chart. The abscissa of the bubble chart and the bubble size indicate the size of the path-influencing factors in the topology analysis. The ordinate of the bubble chart and the bubble color indicate the P value of the enrichment analysis. The key metabolic pathways include the biosynthesis of unsaturated fatty acids and linoleic acid metabolism. Abbreviations: CR = Cyperi rhizome; CRV = CR processed with vinegar. Please click here to view a larger version of this figure.
Table 3 and Table 4 show metabolic pathways with significant differences-phenylalanine metabolism and linoleic acid metabolism, as well as the biosynthesis of unsaturated fatty acids, phenylalanine, tyrosine, and tryptophan. Although the metabolic pathways included steroid hormone biosynthesis, sphingolipid and arachidonic acid metabolism, there were no significant differences, which were related to dysmenorrhea. The results showed that linoleic acid metabolism and the biosynthesis of unsaturated fatty acids were the critical metabolic pathways associated with the enhanced efficacy of CRV.
Constituents absorbed into the blood
Fifteen constituents and two products of metabolism from the CRV and CR extracts were detected in the rat serum in the CR and CRV groups (Table 5). All 17 components were found in positive ion mode.
The relative constituent levels in the blood samples of the two groups were compared using the Student's t-test. The abscissa in Figure 9 displays various experimental groups; the ordinate represents the response value of the mass spectrum. There were two components with significant differences. The analysis showed that the levels of these two components were elevated in the CRV group compared to the CR group, while the levels of 15 elements showed no significant changes.
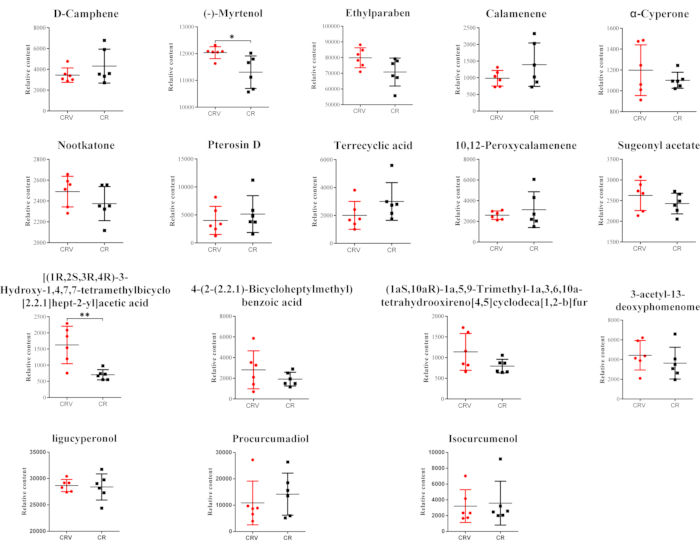
Figure 9: Constituents absorbed into the blood for the CRV group versus the CR group. There were 15 constituents and two products of metabolism from the CRV and CR extracts in the rat serum in the CR and CRV groups. The analysis showed that blood levels of two components were increased in the CRV group compared to the CR group, while the blood levels of 15 components showed no significant changes. Among them, the level of (-)-myrtenol and [(1R,2S,3R,4R)-3-hydroxy-1,4,7,7-tetramethylbicyclo[2.2.1]hept-2-yl]acetic acid in the CRV group was considerably higher than in the CR group. * P < 0.05 or ** P < 0.01 represent a significant difference between the CRV group and the CR group. Abbreviations: CR = Cyperi rhizome; CRV = CR processed with vinegar. Please click here to view a larger version of this figure.
Table 1: Metabolic differences in positive mode. Abbreviations: KEGG = Kyoto Encyclopedia of Genes and Genomes; VIP = variable significance in the projection; RT = retention time. Please click here to download this Table.
Table 2: Metabolic differences in negative mode. Abbreviations: KEGG = Kyoto Encyclopedia of Genes and Genomes; VIP = variable significance in the projection; RT = retention time. Please click here to download this Table.
Table 3: Metabolic pathway analysis in positive mode. Abbreviation: KEGG = Kyoto Encyclopedia of Genes and Genomes. Please click here to download this Table.
Table 4 Metabolic pathway analysis in negative mode. Abbreviation: KEGG = Kyoto Encyclopedia of Genes and Genomes. Please click here to download this Table.
Table 5: Identification of the prototype ingredients and metabolites in rat serum. Note: M1 and M2 are metabolites; other constituents are prototype ingredients. Please click here to download this Table.
Supplementary File 1: BPC diagrams and MS2 chromatogram of the constituents absorbed into the blood of nude mice. BPC diagrams of all the quality control samples in positive and negative modes; MS2 chromatogram of the 17 constituents absorbed into the blood of the rats: D-camphene, (-)-myrtenol, ethylparaben, calamenene, α-cyperone, (+)-nootkatone, (1R,2S,3R,4R)-3-hydroxy-1,4,7,7-tetramethylbicyclo[2.2.1]hept-2-yl]acetic acid, 4-(2-(2.2.1)-bicycloheptylmethyl)benzoic acid, 10,12-peroxycalamenene, (1aS,10aR)-1a,5,9-trimethyl-1a,3,6,10a-tetrahydrooxireno[4,5]cyclodeca[1,2-b]furan-10(2H)-one, pterosin D, terrecyclic acid, sugeonyl acetate, 3-acetyl-13-deoxyphomenome, isocurcumenol, ligucyperonol, procurcumadiol. The differential metabolites identified by secondary mass spectrometry, as well as the HDMB and KEGG databases, in the positive and negative ion modes are also included. Please click here to download this File.
Discussion
Due to the wide variety and different nature of TCMs, these herbs sometimes do not work in clinical practice, and this may be due to the inappropriate processing and decocting of TCMs. The mechanisms of TCM are becoming more apparent with the use of contemporary science and technology29,30. This study shows that both CR and CRV have therapeutic effects in PD model rats and that the therapeutic effect of CRV is more substantial. The mechanism of action of CRV could be related to the fact that vinegar processing may influence the constituents of CR that are absorbed into the blood and could be associated with linoleic acid metabolism and the biosynthesis of unsaturated fatty acids. Figure 10 depicts the potential pathways of CRV's action in pain relief.
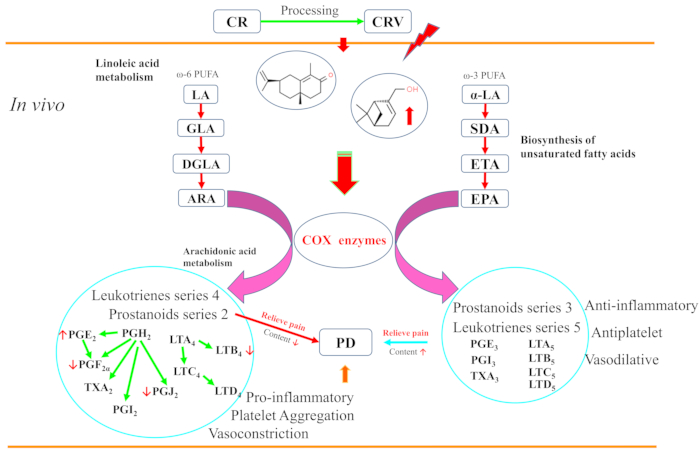
Figure 10: Mechanisms of the enhanced analgesic effect of CRV. The results showed that 15 constituents and two metabolites were found in the blood. Among them, the levels of (-)-myrtenol and [(1R,2S,3R,4R)-3-hydroxy-1,4,7,7-tetramethylbicyclo[2.2.1]hept-2-yl]acetic acid in CRV group were considerably higher than in the CR group. CRV can reduce the level of 2-series prostanoids and 4-series leukotrienes made from ARA and achieve analgesic effects via the modulation of arachidonic acid metabolism, the biosynthesis of unsaturated fatty acids, and linoleic acid metabolism. Abbreviations: ARA = arachidonic acid; COX = cyclooxygenase; LA = linoleic acid; PUFA = polyunsaturated fatty acids; GLA =γ–linolenic acid; DGLA = dihomo-γ-linolenic; PD = primary dysmenorrhea; PG = prostaglandin; LT = leukotrienes; TX = thromboxane; SDA = stearidonic acid; ETA = eicosatetraenoic acid; EPA = eicosapentaenoic acid; CR = Cyperi rhizome; CRV = CR processed with vinegar. Please click here to view a larger version of this figure.
Precautions during the experiment
Since the oil of the effective component of CR is volatile, the time for extracting the CR should not exceed 20 min, and when it boils, it should be on a low heat with a temperature not exceeding 60°C during concentration. To ensure the successful extraction of the effective ingredients, the herbs must be soaked in water for at least 2 h before decoction so that the herbs are wet when soaking is complete. During the CR processing, the vinegar must be well mixed with the herbs so that the vinegar can fully penetrate into them. If the vinegar volume is too low to wet the herbs thoroughly, a small amount of water can be added to dilute the vinegar, and then the vinegar can be fully mixed with the herbs. When mixing is complete, the herbs will absorb all the vinegar. Since vinegar contains acetic acid, the mixture should not come into contact with iron to avoid a chemical reaction.
In the rat experiment, the first intraperitoneal injection of estradiol benzoate is given on day 10, then the intragastric administration of the extract of CR or CRV is given, and, finally, the intraperitoneal injection of oxytocin is administered. After the intraperitoneal injection of oxytocin, the animal is observed for 30 min, and blood is immediately taken. Usually, the blood concentration reaches a peak within 1 h after intragastric administration13, which is the best time to take blood.
When determining the extract and serum samples by LC-MS/MS, they should be determined in the same batch to ensure that the retention time of the same component in different samples is consistent. In this experiment, the identification of the components was a difficult point. Although a relatively mature database can be used for endogenous metabolites, there was no matching database for identifying the constituents absorbed into the blood, so more care should be taken in identification.
Differences in the constituents absorbed into the blood between the CR and CRV groups
To determine the active ingredient of CR, this experiment investigated the constituents absorbed into the blood in dysmenorrhea model rats. In this experiment, it was found that 15 constituents and two metabolites in the blood differed between the CR and CRV groups. Among them, the levels of (-)-myrtenol and [(1R,2S,3R,4R)-3-hydroxy-1,4,7,7-tetramethylbicyclo[2.2.1]hept-2-yl]acetic acid in the CRV group were considerably higher than in the CR group, but the levels of other components were not significantly different. (-)-Myrtenol and [(1R,2S,3R,4R)-3-hydroxy-1,4,7,7-tetramethylbicyclo[2.2.1]hept-2-yl]acetic acid are terpenoids and are considered the effective components of CRV.
Ligucyperonol and procurcumadiol are produced by the oxidation of α-cyperone and isocurcumenol, respectively, and α-cyperone has a strong analgesic effect. The proposed mechanism of action may reduce LPS-induced COX-2 expression and PGE2 synthesis through the negative regulation of NF-kB signaling31. (+)-Nootkatone also inhibits COX-2 activity32,33. Isocurcumenol is the core component of Curcumae rhizoma in treating dysmenorrhea3, and its metabolite procurcumadiol may have an analgesic effect. Compared to the CR group, the CRV group showed considerably higher levels of (-)-myrtenol, which has an analgesic effect34. The possible mechanism of action may be that changes in the expression of COX-235 increase the levels of anti-inflammatory cytokine (IL-10, IFN-γ) and reduce the levels of pro-inflammatory cytokines (TNF-α, IL-1β) levels36. Processing with vinegar improves the levels of the active ingredients in the blood, which may be why products produced with vinegar are more effective.
Differences in the metabolic pathways between the CR and CRV groups
Pathway analysis showed significantly different metabolic pathways between the CR and CRV groups, including in terms of phenylalanine metabolism, the biosynthesis of unsaturated fatty acids, phenylalanine, tyrosine, and tryptophan biosynthesis, and linoleic acid metabolism. However, the metabolic pathways of phenylalanine metabolism and phenylalanine, tyrosine, and tryptophan biosynthesis are not related to PD. These results show that the metabolic pathways associated with the enhanced efficacy of CRV are linoleic acid metabolism and the biosynthesis of unsaturated fatty acids.
Unsaturated fatty acids include two types: omega-3 and omega-637. Three precursors of mediators, including eicosapentaenoic acid (20:5ω3; EPA), docosahexaenoic acid (22:5ω3; DHA), and arachidonic acid (20:5ω6; ARA), are involved in the biosynthesis of unsaturated fatty acids pathway37,38. EPA and ARA metabolism both produce prostaglandins and leukotrienes under the action of COX. ARA produces 2-series prostanoids, including PGF2α, PGE2, PGI2, and thromboxane (TXA2, TXB2), and 4-series leukotrienes, including leukotriene A4 (LTA4) leukotriene B4 (LTB4), leukotriene C4 (LTC4), and leukotriene D4 (LTD4). The 3-series prostanoids include prostaglandin E3 (PGE3), prostacyclin I3 (PGI3), and thromboxane A2 (TXA3), and the 5-series leukotrienes include leukotriene A5 (LTA5) leukotriene B5 (LTB5), leukotriene C5 (LTC5), and leukotriene D5 (LTD5), which are largely produced by EPA.
The transformation process of EPA is the same as that of ARA and is mediated by similar enzymes39. The 2-series prostanoids and 4-series leukotrienes mainly have pro-inflammatory, platelet aggregation, and vasoconstriction effects. In contrast, 3-series prostanoids and 5-series leukotrienes show anti-inflammatory, antiplatelet, and vasodilatory effects38. TXA2 and TXB2 are produced from ARA, causing the blood vessels to narrow. EPA-derived PGI3, PGE3, and TXA3 act only as vasodilators38,40. EPA and ARA compete for conversion into PGs by the COX enzyme. When the membrane EPA/AA ratio is increased, the eicosanoids PGI2 and TXA2, which promote aggregation, can be transformed into TXA3 and PGI3, which promote anti-aggregation, resulting in anti-inflammatory and anti-aggregatory effects40. Additionally, the combined use of EPA, DHA, and linoleic acid (C18:2ω6; LIN) can reduce PGF2α and PGE2 release in bovine endometrium and trophoblasts41,42.
PD is likely caused by the production of PGs and leukotrienes43,44,45, particularly PGs46. Meanwhile, an imbalance in vasopressin, β-endorphins, estrogen, progesterone, neurotransmitters, IL, ET-1, and NO may also be related to dysmenorrhea47. According to the ELISA results, the PGF2α and PGF2α/PGE2 levels in the CRV group decreased, while the PGE2 level increased relative to the CR group. Additionally, leukotriene B4 (C02165) and prostaglandin J2 (C05957) were lower in the CRV group. This indicates that, in the CRV group, there were lower levels of 2-series prostanoids made from ARA, including PGF2α, which is the most important factor for dysmenorrhea. Therapy with PGE2 at high concentrations dilates the blood vessels, and PGE2 causes vasoconstriction at low concentrations48. Therefore, the uterus is relaxed with vasodilation, and dysmenorrhea is relieved.
As the human body cannot synthesize a sufficient amount of C18 unsaturated fatty acids, linoleic acid and α-linolenic acid, which are the only source of C18 unsaturated fatty acids, are very important38. Linoleic acid and α-linolenic acid are the source of omega-6 and omega-3 unsaturated fatty acid metabolism, respectively. In particular, the precursor of prostaglandin synthesis is ARA, and ARA is synthesized from linoleic acid49. Linoleic acid is a precursor, and a series of metabolites are synthesized from it, including ARA, prostaglandins (PGF2α, PGE2), prostacyclin (PGI2), and thromboxane (TXA2)50. Linoleic acid is closely related to the enhanced analgesic effect of CRV. Furthermore, the metabolic pathway of steroid hormone biosynthesis can produce progesterone51,52,53 and sphingosine-1-phosphate (C06124), which are produced by the metabolic pathway of sphingolipid metabolism, induce COX-2, and produce PGE2 through TNF54,55,56,57. These may also be related to PD.
There are some limitations to the protocol. Only the relative levels of the constituents were compared, and the standard sample was not used to quantify the constituents in the herb and those absorbed into the blood. The feces and urine of rats were not collected in the animal experiments, resulting in the discovery of fewer metabolites of CR in vivo. Validation experiments should further confirm the mechanism discovered by the metabonomics analysis.
The strategy and protocol used in this study avoided blindness in the study of chemical components in vitro and the one-sided study of individual components in vivo. Therefore, it was very suitable for mechanism exploration. The strategy can save a lot of time and work and can accurately identify the critical constituents and mechanism for therapeutic efficacy.
In this study, it was found that there were 15 constituents and two metabolites in the blood. Among them, the levels of (-)-myrtenol and [(1R,2S,3R,4R)-3-hydroxy-1,4,7,7-tetramethylbicyclo[2.2.1]hept-2-yl]acetic acid were significantly increased in the CRV group compared to the CR group, showing that processing with vinegar can increase the levels of the active ingredients in the blood. After CR was processed with vinegar, the levels of 2-series prostanoids and 4-series leukotrienes with pro-inflammatory, platelet aggregation, and vasoconstriction properties decreased, including PGF2α, leukotriene B4, and prostaglandin J2. This may be the mechanism by which CRV demonstrates an enhanced analgesic effect.
Declarações
The authors have nothing to disclose.
Acknowledgements
This work was supported by the Chongqing Municipal Health and Family Planning Commission Chinese Medicine Science and Technology Project (Project Number: ZY201802297), General project of Chongqing Natural Science Foundation (Project Number: cstc2019jcyj-msxmX065), Chongqing Modern Mountain Area Characteristic High-efficiency Agricultural Technology System Innovation Team Building Plan 2022 [10], and Chongqing Municipal Health Commission Key Discipline Construction Project of Chinese Materia Medica Processing.
Materials
| Acetonitrile | Fisher Scientific, Pittsburg, PA, USA | 197164 | |
| BECKMAN COULTER Microfuge 20 | Beckman Coulter, Inc. | MRZ15K047 | |
| Estradiol benzoate | Shanghai Macklin Biochemical Co., Ltd | C10042616 | |
| formic acid | Fisher Scientific, Pittsburg, PA, USA | 177799 | |
| LC 30A system | Shimadzu, Kyoto, Japan | 228-45162-46 | |
| Olive oil | Shanghai Yuanye Biotechnology Co., Ltd | H25A11P111909 | |
| Oxytocin synthetic | Zhejiang peptide biology Co., Ltd | 2019092001 | |
| Rat PGF2α ELISA kit | Shanghai lmai Bioengineering Co., Ltd | 202101 | |
| Rat PGFE2 ELISA kit | Shanghai lmai Bioengineering Co., Ltd | EDL202006217 | |
| SPF Sprague-Dawley rats | Hunan SJA Laboratory Animal Co., Ltd | Certificate number SCXK (Hunan) 2019-0004 | |
| Tecan Infinite 200 PRO | Tecan Austria GmbH, Austria | 1510002987 | |
| Triple TOF 4600 system | SCIEX, Framingham, MA, USA | BK20641402 | |
| water | Fisher Scientific, Pittsburg, PA, USA | 152720 |
Referências
- Yu, W. Y., et al. Acupuncture for primary dysmenorrhea: A potential mechanism from an anti-inflammatory perspective. Evidence-Based Complementary and Alternative. 2021, 1907009 (2021).
- Rafique, N., Al-Sheikh, M. H. Prevalence of primary dysmenorrhea and its relationship with body mass index. Journal of Obstetrics and Gynaecology Research. 44 (9), 1773-1778 (2018).
- Tong, H., et al. Bioactive constituents and the molecular mechanism of Curcumae Rhizoma in the treatment of primary dysmenorrhea based on network pharmacology and molecular docking. Phytomedicine. 86, 153558 (2021).
- Ferries-Rowe, E., Corey, E., Archer, J. S. Primary dysmenorrhea: Diagnosis and therapy. Obstetrics & Gynecology. 136 (5), 1047-1058 (2020).
- Lu, J., et al. The association study of chemical compositions and their pharmacological effects of Cyperi Rhizoma (Xiangfu), a potential traditional Chinese medicine for treating depression. Journal of Ethnopharmacology. 287, 114962 (2021).
- Lu, J., et al. Quality status analysis and intrinsic connection research of growing place, morphological characteristics, and quality of Chinese medicine: Cyperi Rhizoma (Xiangfu) as a case study. Evidence-Based Complementary and Alternative. 2022, 8309832 (2022).
- Taheri, Y., et al. Cyperus spp.: A review on phytochemical composition, biological activity, and health-promoting effects. Oxidative Medicine and Cellular Longevity. 2021, 4014867 (2021).
- El-Wakil, E. A., Morsi, E. A., Abel-Hady, H. Phytochemical screening, antimicrobial evaluation and GC-MS analysis of Cyperus rotundus. World Journal Of Pharmacy And Pharmaceutical Sciences. 8 (9), 129-139 (2019).
- Rocha, F. G., et al. Preclinical study of the topical anti-inflammatory activity of Cyperus rotundus L. extract (Cyperaceae) in models of skin inflammation. Journal of Ethnopharmacology. 254, 112709 (2020).
- Hao, G., Tang, M., Wei, Y., Che, F., Qian, L. Determination of antidepressant activity of Cyperus rotundus L extract in rats. Tropical Journal of Pharmaceutical Research. 16 (4), 867-871 (2017).
- Kakarla, L., et al. Free radical scavenging, α-glucosidase inhibitory and anti-inflammatory constituents from Indian sedges, Cyperus scariosus R.Br and Cyperus rotundus L. Pharmacognosy Magazine. 12 (47), 488-496 (2016).
- Shakerin, Z., et al. Effects of Cyperus rotundus extract on spatial memory impairment and neuronal differentiation in rat model of Alzheimer’s disease. Advanced Biomedical Research. 9 (1), 17-24 (2020).
- Li, J., et al. Pharmacokinetics of caffeic acid, ferulic acid, formononetin, cryptotanshinone, and tanshinone IIA after oral Administration of naoxintong capsule in rat by HPLC-MS/MS. Evidence-Based Complementary and Alternative. 2017, 9057238 (2017).
- Zhang, A., et al. Metabolomics: Towards understanding traditional Chinese medicine. Planta Medica. 76 (17), 2026-2035 (2010).
- Li, L., Ma, S., Wang, D., Chen, L., Wang, X. Plasma metabolomics analysis of endogenous and exogenous metabolites in the rat after administration of Lonicerae Japonicae Flos. Biomedical Chromatography. 34 (3), 4773 (2020).
- Guijas, C., Montenegro-Burke, J. R., Warth, B., Spilker, M. E., Siuzdak, G. Metabolomics activity screening for identifying metabolites that modulate phenotype. Nature Biotechnology. 36 (4), 316-320 (2018).
- Hu, L., et al. Functional metabolomics decipher biochemical functions and associated mechanisms underlie small-molecule metabolism. Mass Spectrometry Reviews. 39 (5-6), 417-433 (2020).
- Cui, L., Lu, H., Lee, Y. Challenges and emergent solutions for LC-MS/MS based untargeted metabolomics in diseases. Mass Spectrometry Reviews. 37 (6), 772-792 (2018).
- Liu, F., et al. Metabonomics study on the hepatoprotective effect of Panax notoginseng leaf saponins using UPLC/Q-TOF-MS analysis. The American Journal of Chinese Medicine. 47 (3), 559-575 (2019).
- Zhao, L., Hartung, T. Metabonomics and toxicology. Methods in Molecular Biology. 1277, 209-231 (2015).
- Martin, F. J., Montoliu, I., Kussmann, M. Metabonomics of ageing – Towards understanding metabolism of a long and healthy life. Mechanisms of Ageing and Development. 165, 171-179 (2017).
- Heaney, L. M., Deighton, K., Suzuki, T. Non-targeted metabolomics in sport and exercise science. Journal of Sports Sciences. 37 (9), 959-967 (2019).
- Yang, Y., et al. Metabonomics profiling of marinated meat in soy sauce during processing. Journal of the Science of Food and Agriculture. 98 (4), 1325-1331 (2018).
- Xu, S. Y. . Methodology of Pharmacological Experiment. , (2002).
- Ma, B., et al. An integrated study of metabolomics and transcriptomics to reveal the anti-primary dysmenorrhea mechanism of Akebiae Fructus. Journal of Ethnopharmacology. 270, 113763 (2021).
- Li, X., et al. Regulation of mild moxibustion on uterine vascular and prostaglandin contents in primary dysmenorrhea rat model. Evidence-Based Complementary and Alternative. 2021, 9949642 (2021).
- Smith, C. A., Want, E. J., O’Maille, G., Abagyan, R., Siuzdak, G. XCMS: Processing mass spectrometry data for metabolite profiling using nonlinear peak alignment, matching, and identification. Analytical Chemistry. 73 (3), 779-787 (2006).
- Wang, D., et al. UPLC-MS/MS-based rat serum metabolomics reveals the detoxification mechanism of Psoraleae Fructus during salt processing. Evidence-Based Complementary and Alternative Medicine. 2021, 5597233 (2021).
- Wang, X., et al. Rhodiola crenulata attenuates apoptosis and mitochondrial energy metabolism disorder in rats with hypobaric hypoxia-induced brain injury by regulating the HIF-1α/microRNA 210/ISCU1/2(COX10) signaling pathway. Journal of Ethnopharmacology. 241, 111801 (2019).
- Xie, H., et al. Raw and vinegar processed Curcuma wenyujin regulates hepatic fibrosis via bloking TGF-β/Smad signaling pathways and up-regulation of MMP-2/TIMP-1 ratio. Journal of Ethnopharmacology. 246, 111768 (2020).
- Jung, S. H., et al. α-Cyperone, isolated from the rhizomes of Cyperus rotundus, inhibits LPS-induced COX-2 expression and PGE2 production through the negative regulation of NFkappaB signalling in RAW 264.7 cells. Journal of Ethnopharmacology. 147 (1), 208-214 (2013).
- Dantas, L. B. R., et al. Nootkatone inhibits acute and chronic inflammatory responses in mice. Molecules. 25 (9), 2181 (2020).
- Xu, Y., et al. Nootkatone protects cartilage against degeneration in mice by inhibiting NF- κB signaling pathway. International Immunopharmacology. 100, 108119 (2021).
- Heimfarth, L., et al. Characterization of β-cyclodextrin/myrtenol complex and its protective effect against nociceptive behavior and cognitive impairment in a chronic musculoskeletal pain model. Carbohydrate Polymers. 244, 116448 (2020).
- Viana, A., et al. (-)-Myrtenol accelerates healing of acetic acid-induced gastric ulcers in rats and in human gastric adenocarcinoma cells. European Journal of Pharmacology. 854, 139-148 (2019).
- Bejeshk, M. A., et al. Anti-inflammatory and anti-remodeling effects of myrtenol in the lungs of asthmatic rats: Histopathological and biochemical findings. Allergologia et Immunopathologica. 47 (2), 185-193 (2019).
- Christie, W. W., Harwood, J. L. Oxidation of polyunsaturated fatty acids to produce lipid mediators. Essays in Biochemistry. 64 (3), 401-421 (2020).
- Wiktorowska-Owczarek, A., Berezinska, M., Nowak, J. Z. PUFAs: Structures, metabolism and functions. Advances in Clinical and Experimental. 24 (6), 931-941 (2015).
- Araujo, P., et al. The effect of omega-3 and omega-6 polyunsaturated fatty acids on the production of cyclooxygenase and lipoxygenase metabolites by human umbilical vein endothelial cells. Nutrients. 11 (5), 966 (2019).
- Shahidi, F., Ambigaipalan, P. Omega-3 polyunsaturated fatty acids and their health benefits. Annual Review of Food Science and Technology. 9, 345-381 (2018).
- Meier, S., Ledgard, A. M., Sato, T. A., Peterson, A. J., Mitchell , M. D. Polyunsaturated fatty acids differentially alter PGF(2α) and PGE2 release from bovine trophoblast and endometrial tissues during short-term culture. Animal Reproduction Science. 111 (2), 353-360 (2009).
- Cheng, Z., et al. Altering n-3 to n-6 polyunsaturated fatty acid ratios affects prostaglandin production by ovine uterine endometrium. Animal Reproduction Science. 143 (1-4), 38-47 (2013).
- Sultan, C., Gaspari, L., Paris, F. Adolescent dysmenorrhea. Endocrine Development. 22, 171-180 (2012).
- Zeev, H. M. D., Craig, L. M. D., Suzanne, R. M. D., Rosalind, V. M. D., Jeffrey, D. M. D. Urinary leukotriene (LT) E4 in adolescents with dysmenorrhea: A pilot study. Journal of Adolescent Health. 27 (3), 151-154 (2000).
- Fajrin, I., Alam, G., Usman, A. N. Prostaglandin level of primary dysmenorrhea pain sufferers. Enfermería Clínica. 30, 5-9 (2020).
- Iacovides, S., Avidon, I., Baker, F. C. What we know about primary dysmenorrhea today: a critical review. Human Reproduction Update. 21 (6), 762-778 (2015).
- Barcikowska, Z., Rajkowska-Labon, E., Grzybowska, M. E., Hansdorfer-Korzon, R., Zorena , K. Inflammatory markers in dysmenorrhea and therapeutic options. International Journal of Environmental Research and Public Health. 17 (4), 1191 (2020).
- Wang, T., et al. Arachidonic acid metabolism and kidney inflammation. International Journal of Molecular Science. 20 (15), 3683 (2019).
- Szczuko, M., et al. The role of arachidonic and linoleic acid derivatives in pathological pregnancies and the human reproduction process. International Journal of Molecular Sciences. 21 (24), 9628 (2020).
- Serrano-Mollar, A., Closa, D. Arachidonic acid signaling in pathogenesis of allergy: Therapeutic implications. Current Drug Targets-Inflammation and Allergy. 4 (2), 151-155 (2005).
- Toit, R. L., Storbeck, K. H., Cartwright, M., Cabral, A., Africander, D. Progestins used in endocrine therapy and the implications for the biosynthesis and metabolism of endogenous steroid hormones. Molecular and Cellular Endocrinology. 441, 31-45 (2017).
- Ghayee, H. K., Auchus, R. J. Basic concepts and recent developments in human steroid hormone biosynthesis. Reviews in Endocrine and Metabolic Disorders. 8 (4), 289-300 (2007).
- Liang, J. J., Rasmusson, A. M. Overview of the molecular steps in steroidogenesis of the GABAergic neurosteroids allopregnanolone and pregnanolone. Chronic Stress. 2, 2470547018818555 (2018).
- Pettus, B. J., et al. The sphingosine kinase 1/sphingosine-1-phosphate pathway mediates COX-2 induction and PGE2 production in response to TNF-α. The FASEB Journal. 17 (11), 1411-1421 (2003).
- Zeidan, Y. H., et al. Acid ceramidase but not acid sphingomyelinase is required for tumor necrosis factor-α-induced PGE2 production. Journal of Biological Chemistry. 281 (34), 24695-24703 (2006).
- Kawamori, T., et al. Role for sphingosine kinase 1 in colon carcinogenesis. The FASEB Journal. 23 (2), 405-414 (2009).
- Hannun, Y. A., Obeid, L. M. Sphingolipids and their metabolism in physiology and disease. Nature Reviews Molecular Cell Biology. 19 (3), 175-191 (2018).

