Production of Modified Autologous Conditioned Serum and Ex Vivo Assessment of Its Healing Potential in Murine Corneal Epithelium
Summary
This article describes a protocol to simplify the process and render the preparation of autologous conditioned serum (ACS) less expensive. No special syringes or surface-coated glass beads are needed. Moreover, the modified ACS (mACS) has competitive advantages over conventional autologous serum in the corneal wound healing of murine eyes ex vivo.
Abstract
Human blood-derived topical therapies have been a boon to clinicians in recent decades. Autologous serum (AS) and platelet-rich plasma (PRP) are enriched in epitheliotropic growth factors that are essential in corneal wound healing. Unlike AS, PRP is based on a differential centrifugation system, yielding more platelet-derived growth factors. Autologous conditioned serum (ACS) not only preserves the preparation of AS and PRP, but also focuses on immune-modulating properties, which are important in inflammatory diseases.
The lack of standardized protocols and high preparation costs are limitations for the clinical application of ACS. This video experiment demonstrates a standard operating procedure for preparing modified autologous conditioned serum (mACS) eye drops. First, glycerol was added into heparin syringes as the blood cell stabilizer during hypoxic incubation. To activate the blood cells, a 4 h incubation at 37 °C was initiated. Then, the blood samples were centrifuged at 3,500 × g for 10 min at room temperature. After filtration of the supernatant through a 0.22 µm filter, the mACS eye drops were fully prepared.
A tentative try-out of the therapeutic effect of mACS showed that it may have competitive advantages over conventional AS in the corneal wound healing in ex vivo mouse eyes. The AS used in this study was prepared according to published studies and the clinical practice in our hospital. Therefore, the efficacy of mACS on ocular surface diseases could be evaluated in future research through in vivo animal studies and clinical trials.
Introduction
The therapeutic effects of autologous serum (AS) in dry eye diseases were first reported in the 1980s by Fox et al.1. It is believed that both the lubricating property and the essential epitheliotropic biochemical components in AS, mimicking natural tears, benefit the proliferation of corneal epithelial cells. Over the past decades, several studies have been performed on this basis. Trophic components include epidermal growth factor (EGF), vitamin A, transforming growth factor β (TGF- β), and other cytokines. Interestingly, the serum is rich in TGF- β and vitamin A, which are believed to play a pivotal role in epidermal proliferation2,3,4,5. In addition, when treating patients with ocular surface diseases, several studies have showed some advantages of AS eye drops in patient-reported outcomes, other objective dry eye parameters6,7, and microscopic findings such as cell density8. Meta-analysis studies revealed that there might be some benefits in improving patient's syndromes with AS eye drops treatment, but long-term results and observations are still lacking9,10.
Unlike AS, platelet-rich plasma (PRP) is derived from adding an anticoagulant during preparation, with further differential centrifugation and chemical activation of the platelets. Compared with AS, numerous chemicals and growth factors, such as TGF- β, vascular endothelial growth factor (VEGF), and EGF, are present in PRP. It has also been applied to ocular surface diseases with clinical benefits in symptom relief11.
The cross-link between epithelial defects and inflammation is complex. Notably, immunopathophysiology is another important issue in ocular surface diseases. Pro-inflammatory cytokines, such as IL-1β and IFN-γ, are believed to be pivotal mediators in inflammatory cascades12. New avenues of treatment are thus opened based on understanding the immune mechanism. Strategies to stop this inflammatory process, including the production of interleukin-1 receptor antagonist (IL-1Ra) and other anti-inflammatory cytokines, may also play an important role in ocular surface diseases13,14,15.
Since 1998, Orthokine, a commercialized autologous conditioned serum (ACS), has been used clinically in orthopedic patients suffering from osteoarthritis (OA), rheumatoid arthritis (RA), and spinal disorders13. Compared with AS and PRP, treatment with chemically coated glass beads and hypoxic incubation to activate monocytes are the specific features of ACS16. Theoretically, more anti-inflammatory factors can be secreted by adding survival stress to the cells, resulting in a higher concentration of essential immune-modulating components, including IL-1Ra. The improved therapeutic benefits of ACS in OA, compared with AS, have also been reported17. Ocular surface diseases share similar immune backgrounds with orthopedic inflammatory diseases in some respects. Therefore, based on the successful results of human blood-derived therapy in the orthopedic field, ACS might have advantages over conventional treatments in clinical practice by epitheliotropic and immune-modulating properties. Although ACS has been widely used in orthopedic inflammatory diseases, its clinical applications in ophthalmology still need to be explored, which may be hindered by its high cost, lack of literature support, and lack of standardization of the preparation process, resulting in diverse performance.
In this video article, a novel, cost-effective, and convenient method was demonstrated to generate the modified ACS (mACS), or plasma rich in growth factors (PRGF), producing an eye drop solution with a comparable practical value to commercialized ACSs. The key ideas of adding anticoagulants and triggering the blood cells to secrete anti-inflammatory cytokines by stressed incubation were retained, but unlike the chemically-induced methods, such as those based on CrSO4-coated glass beads and commercial kits, the critical stress status is physically induced by hypoxic incubation in this method. Moreover, glycerol was added to provide extra benefits, including an increase in the stability of the membrane of blood cells, maintenance of a proper osmotic extracellular fluid pressure18, and an appropriate source of nutrients in hypoxic conditions that avoid overstressing the cells.
Protocol
The research was performed in compliance with institutional guidelines at the beginning of the protocol section. All protocols and procedures were carried out according to the Declaration of Helsinki and were reviewed and approved by the Chang Gung Medical Foundation Institutional Review Board. All volunteers were informed of the nature of this study and signed an informed consent form prior to their inclusion. The consumables required for the entire experimental procedure are presented in Figure 1 and Figure 2, as well as in the Table of Materials.
1. Preparation of the materials needed to produce mACS eye drops
- Prepare 250 mL of 10% glycerol solution and keep a 21 G butterfly-winged infusion set, a 3 mL syringe without the needle, and six 10 mL vacutainer tubes containing heparin 158 USP units ready (Figure 1).
- Connect the 21 G blood collection needle to the 3 mL syringe and withdraw 3 mL of 10% glycerol solution into the prepared syringe.
NOTE: All materials must be sterilized before the needle is inserted. - Distribute the 10% glycerol solution into the vacutainer tubes in sequence, with approximately 0.5 mL of 10% glycerol solution in each (Figure 3A).
NOTE: Because of the negative pressure in the test tube, the needle must come out immediately after it goes in to evenly distribute 3 mL of glycerol solution to the six test tubes. - Sterilize the patient's skin with 75% alcohol sterile cotton swabs. Puncture the superficial vein of the patient's upper limbs with the 18 G blood collection needle. Draw 60-70 mL of venous blood from the superficial vein in total.
2. Preparation for mACS eye drops
- Sequentially inject 10 mL of the drawn venous blood into each of the six vacutainer tubes (Figure 3B).
NOTE: This step relies on the negative pressure of the vacuum to fill the tubes. To avoid blood cell disruption and hemolysis, do not apply any positive pressure. - Place the six vacutainer tubes in the incubator with a constant temperature of 37 °C for 4 h (Figure 3C).
NOTE: The hypoxic status is maintained and stabilized by the remaining negative pressure in a sealed tube that received glycerol. - Remove the tubes from the incubator after 4 h and centrifuge them at 3,500 × g for 10 min at room temperature.
- At this point, prepare the materials for mACS extraction, including the sterilized eye drop bottles, a 3 mL syringe with the needle, a 0.22 µm filter, an 18 G needle, and a pair of sterile gloves (Figure 2).
NOTE: The operating table should be wiped with 75% alcohol to ensure an aseptic environment. The supernatant, after centrifugation, is already a semi-finished product of mACS. - Place the six tubes on the tube rack and open the caps after complete centrifugation (Figure 3D).
NOTE: Sterility is not required in this step. - Put on sterile gloves and draw out the mACS one by one using a 3 mL syringe with an 18 G needle.
NOTE: Be careful not to draw the lower blood cell layer during this step (Figure 3E). - Pull out the needle, connect it to the 0.22 µm filter, and connect the 23 G, 1.5 in blood collection needle with the original 3 mL syringe to the outlet below (Figure 3F).
- Push the needle gently through the 0.22 µm filter into the prepared sterile eye drop bottles (Figure 3G).
- Repeat the above steps until all the mACS has been filtered and stored in eye drop bottles (Figure 3H).
- Store the mACS eye drops at 4 °C for immediate use; store at -20 °C for long-term preservation.
NOTE: Do not keep them for more than 2 weeks at 4 °C or for longer than 3 months at -20 °C9,19.
3. Ex vivo wound healing model of murine corneal epithelium
NOTE: The following ex vivo animal model was based on prior experience from Hung et al. on mechanical injuries of the corneal epithelium20. The following steps should be performed under the microscope to create a well-circumscribed and consistent corneal epithelial wound.
- Anesthetize the C57BL/6 mice with 3%-4% isoflurane. Indent the skin biopsy punch over the murine central cornea, leaving a shallow circle on the epithelium as a uniform wound margin.
NOTE: Be gentle to avoid eyeball rupture. - Debride the corneal epithelium within the confirmed area down to the Bowman's layer, with a corneal rust ring remover equipped with a 0.5 mm burr.
- To establish the ex vivo animal model of the mechanical corneal wound, harvest the eyeball and prepare the culture well.
- Euthanize the mice first; then, gently press the superior and inferior orbital rims of the mice, introduce the tip of the forceps over the retrobulbar space, and out the eyeball and hold it by the forceps.
- Cut the optic nerve and periorbital soft tissue with corneal scissors to isolate the eyeball perfectly.
- Prepare a 96-well plate with melted wax inside it. Quickly create a round hole using the tips of forceps and then wait for solidification.
- Prepare the media to be tested: 0.5% mACS, 0.5% AS for comparison, normal saline as a negative control, and Dulbecco's modified Eagle's medium (DMEM).
NOTE: mACS is obtained using the protocol mentioned above. - For the ex vivo culture, place the harvested eyeball onto the prepared 96-well plate. Add 200 µL of each medium into the 96-well plate.
- Place the 96-well culture plate into the incubator at 37 °C with 5% CO2. Ensure that the culture media is changed every 24 h.
- To confirm the sequential wound healing effect, monitor the epithelial wound area under the microscope every 8 h by fluorescein staining.
- Dissolve the fluorescein on the fluorescein paper with normal saline.
- Drop the fluorescein dye onto the murine central cornea, then observe and document it under a microscope. A typical result is shown in Figure 4.
NOTE: One drop (approximately 0.05 mL) of fluorescein dye is enough for the observation.
Representative Results
Figure 1 and Figure 2 show the materials needed for the experiment, and Figure 3 displays the sequential steps and the successful mid-products during the preparation of mACS. First, 0.5 mL of 10% glycerol solution was added into each 10 mL sterile test tube (Figure 3A). Then, 60-70 mL of venous blood was obtained from the patient, and 10 mL of blood was injected into each tube (Figure 3B). The patient's blood must be subjected to thorough, regular laboratory examinations before the preparation to ensure that the quality of the blood is up to standard. The most common low-quality blood product is due to dyslipidemia, which results in a cloudy upper layer of serum after centrifugation that is difficult to remove; further preparation of eye drops cannot be continued (Figure 5A).
Then, the sealed test tube is placed in an incubator at 37 °C for 4 h (Figure 3C). After incubation and centrifugation, the sample could be separated into an upper layer containing the preliminary product of mACS and a lower blood cell layer (Figure 3D). The upper layer fluid was then collected. Aseptic techniques are essential hereafter. Because the serum is a very nutritious breeding environment for microorganisms, any surface that comes into contact with the needle must be disinfected with 75% alcohol first, and sterile gloves must be used during the process. Importantly, disturbance of the blood cells from the lower layer should be avoided when removing the supernatant (Figure 3E).
At this step, it is expected that the supernatant is clear or pale yellow. However, if a slight red color is seen, hemolysis may have occurred, and the preparation may not be suitable for the subsequent steps (Figure 5B). The collected serum could be centrifuged again to ensure the supernatant is free of red blood cells. The serum was filtered through a 0.22 µm filter into a sterile eye drop bottle (Figures 3F,G). The final product, mACS eye drops, was then refrigerated or frozen as soon as possible, depending on the purpose (Figure 3H). The eye drops could not be stored for too long because of their richness in labile nutrients and the lack of preservatives.
In the ex vivo surface healing model, the mACS eye drop showed a superior outcome in corneal wound healing compared with the AS eye drop. Harvesting the eyeballs of C57BL/6 mice was done after creating a concentric corneal wound by physical abrasion under the microscope. Then, the sacrificed mice eyes were cultured in four different media, including pure normal saline, DMEM, 0.5% AS, and 0.5% mACS.
The AS medium used here was prepared based on the literature21 and the clinical practice in Chang Gung Memorial Hospital Linkou. Venous blood, 40 mL in total, was drawn into vacutainer tubes from volunteers after skin sterilization. No anticoagulant was added to the tubes. The blood was then stored for 30 min at room temperature to ensure complete clotting followed by centrifugation at 3,500 × g for 10 min. After centrifugation, the pure AS occupied the upper layer of the tubes, which was diluted by BSS (sterile irrigating solution) to 0.5% in this ex vivo animal model.
Corneal wounds were sequentially observed at six different time points, namely 0, 8, 16, 24, 32, and 48 h. The preliminary results showed that, at 16 h, the corneal wounds healed faster under the 0.5% mACS eye drop than the other groups. At 24 h, the 0.5% AS and DMEM groups had comparable therapeutic effects with the 0.5% mACS eye drop. Healing of the corneal wounds, close to full recovery, was observed at 32 h, except for the normal saline group (Figure 4).
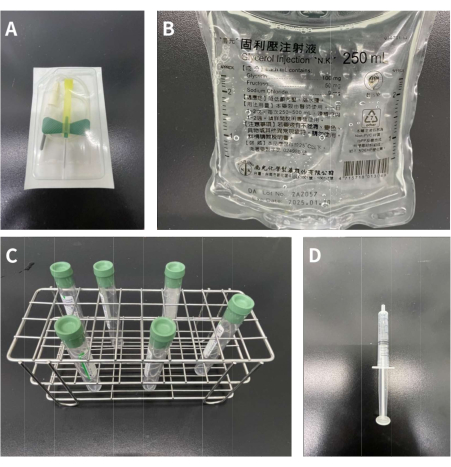
Figure 1: Materials needed for adding 10% glycerol solution into the tubes. (A) 21 G butterfly-winged infusion set. (B) 250 mL of 10% glycerol solution. (C) Six 10 mL vacutainer tubes containing heparin 158 USP units. (D) 3.0 mL syringe. Please click here to view a larger version of this figure.
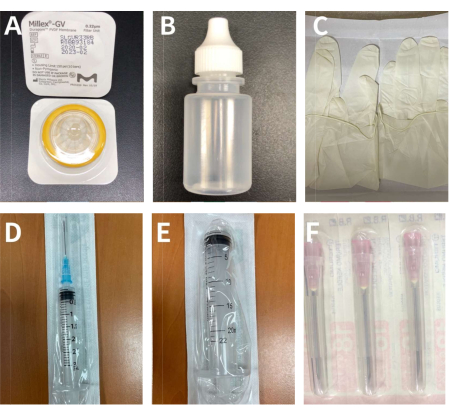
Figure 2: The materials for extraction and filtration of the supernatant of the blood sample. (A) A 0.22 µm filter. (B) A 5 mL sterilized eye drop bottle. (C) A pair of sterile gloves. (D) A 3 mL syringe with the 23 G needle. (E) A 20 mL syringe. (F) 18 G needles. Please click here to view a larger version of this figure.
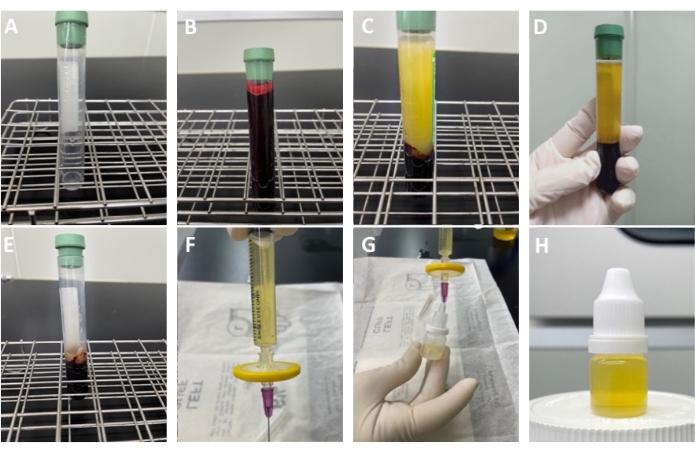
Figure 3: Procedures for blood centrifugation and preparation for the production of modified autologous conditioned serum eye drops. (A) 0.5 mL of 10% glycerol solution was added into each test tube. (B) The blood sample collected from the patient was divided equally into six test tubes (only one shown in the image). (C) Appearance of the test tube after being placed in a 37 °C incubator for 4 h. (D) The serum and blood cell layers after centrifugation. (E) The residue after aspirating the supernatant. One should be careful not to aspirate the lower blood cell layer. (F) The supernatant is collected in the 3 mL syringe and a 0.22 µm filter and 23 G needle are inserted below it. (G) The needle is gently pushed into the sterile eye drop bottle. (H) Final product of the modified ACS eye drops. Please click here to view a larger version of this figure.

Figure 4: Sequential changes of corneal wounds (green areas in the figures after fluorescein staining) in murine eyes in four different culture media over time. No significant difference was observed in the saline group, while wound healing (arrows) over time was observed in the DMEM, 0.5% AS, and 0.5% mACS groups. In the 0.5% mACS group, the wound healing (arrows) was significantly more complete at 16 h than those treated with AS and DMEM. At 24 h, the DMEM, 0.5% mACS, and 0.5% AS groups showed further healing effects (arrows), especially in the mACS group. Near total recovery was observed at 32 h for all groups, except for the saline group. Please click here to view a larger version of this figure.
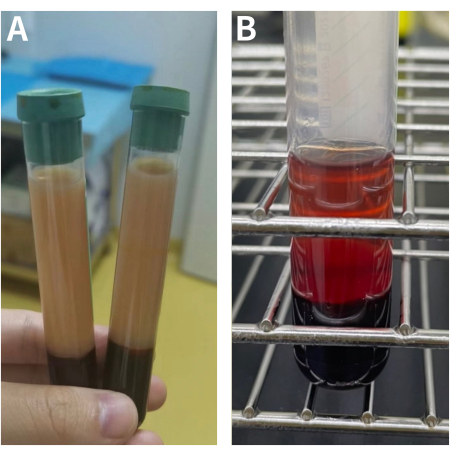
Figure 5: Examples of poor blood products after centrifugation. (A) Dyslipidemia. The most common defective products are due to obesity or diabetes mellitus, as the upper serum contains high fat and cholesterol. (B) Hemolysis. This is also a common sub-optimal product. The main reason can be attributed to using small bore needles for blood drawing or using inappropriate external force in injecting blood products. Please click here to view a larger version of this figure.
Discussion
In this study, a protocol for the preparation of mACS is described and the benefit of mACS eye drops in the wound healing of animal models is further shown. The crucial modification of this mACS protocol is the addition of approximately 0.5 mL of 10% glycerol solution in each test tube, which creates suitable hypoxic conditions during the 4 h incubation at 37 °C. This setting provides the AS with proper stress and prompt cells to secrete the necessary growth factors that help wound healing. The 0.22 µm filter can help eliminate the macromolecular proteins, blood cells, and impurities, thus making the final product pure and less adhesive.
The blood is incubated for 4 h at 37 °C, and further centrifugation and filtration are performed. Strict attention to sterility during the preparation and storage of the final products at a proper temperature is also crucial. The high nutrient content in the final serum products is an excellent culture medium for the bacteria. Thus, improper storage or contamination during preparation may spoil the eye drops. Contaminated mACS can cause diseases such as conjunctivitis or infectious keratitis.
The quality of the patient's blood is also important; therefore, laboratory tests are needed before the preparation of mACS to ensure the safety of blood products. During drawing and when operating the patient's blood samples, operators should be aware of hemolysis. Any pressure during preparation can easily cause hemolysis; therefore, the syringe should be pulled and pushed slowly during blood collection. Hypoxia should also be controlled to obtain high concentrations of growth factors and anti-inflammatory cytokines. Based on experience and previously published studies22,23, the application of a 4 h hypoxic incubation is the ultimate setting. Glycerol could provide more suitable hypoxic stress to the cell, due to the stabilization effect and the proper osmotic extracellular fluid pressure it can provide18. However, the optimal concentration of glycerol and how it correlates with the cytokine concentration still need to be checked. In the future, in vivo animal studies of wound healing and a larger-scale randomized control trial will be warranted to confirm its efficacy.
So far, research has mainly focused on applying serum products to ocular surface diseases using traditional AS and the emerging PRP eye drops24,25,26,27. Traditional AS without any anticoagulant and using no filter during preparation is amber in its pure form, but often makes patients uncomfortable due to its adhesiveness. As a result, diluted AS (20%-50%) is usually used, which lowers the concentrations of the nutrients. PRP, however, is less adhesive and usually used in its pure form (100%). Recent literature reports mostly point out that PRP provides better anti-inflammatory and wound healing effects than AS, mainly due to its rich content in anti-inflammatory cytokines, growth factors, and its ability to attenuate IL-1β-induced inflammation16,26. A randomized controlled trial conducted by García-Conca et al. has already found that the application of PRP in severe or moderate hyposecretory dry eye diseases shows satisfactory results28. Although ACS, as an improvement over AS, has rarely been reported in inflammatory and degenerative ocular surface diseases, it has been widely used in the treatment of degenerative arthritis and wound healing in orthopedics29,30,31. The most common ACS has been manufactured and patented as a commercial kit13,32. mACS was designed after adjusting the process of ACS preparation to combine the strengths of both ACS and PRP. mACS can be produced at a more economical cost because no special reagents are needed, such as CrSO4-coated glass beads or any patented kits, which are needed in the preparation of both ACS and PRP.
Aside from the economical concern, the mACS eye drops also provided satisfactory preliminary results in an ex vivo experiment, and may be a revolutionary alternative to the current, emerging PRP eye drops. Of note, ACS has demonstrated better outcomes than most established treatments in inflammatory joint diseases both qualitatively and quantitatively because of its anti-inflammatory ability and improved endogenous repair mechanisms17. Although there is no literature evidence of therapeutic effects of ACS in ophthalmology to date, it is reasonable to assume the potential in its application to patients with corneal epithelial defects due to its anti-inflammatory mechanism and the success in orthopedics33. Only AS was compared with the mACS in our ex vivo study; however, PRP and ACS produced via commercial kits can also be compared. Future research in analyzing the serum levels of anti-inflammatory cytokines and growth factors in mACS would be useful in confirming its similarity to ACS or PRP, and potential superiority to AS.
Declarações
The authors have nothing to disclose.
Acknowledgements
The authors thank Ya-Lan Chien and Chia-Ying Lee for excellent technical assistance, and OnLine English company for the linguistic edition. This study was funded in part by Chang Gung Medical Research Project (Grant No. CMRPG3L1491).
Materials
| 96-well culture plate | Merck KGaA, Germany | CLS3997 | |
| Barraquer lid speculum | katena | K1-5355 | 15 mm |
| Barraquer needle holder | Katena | K6-3310 | without lock |
| Barron Vacuum Punch 8.0 mm | katena | K20-2108 | for cutting filter paper |
| BD 10.0 mL vacutainer tubes containing heparin 158 USP units | Becton,Dickinson and Company, US | 367880 | At least 6 tubes, necessary to collect blood for subsequent experiments and to avoid blood agglutination |
| BD 21 G butterfly-winged infusion set | Becton,Dickinson and Company, US | 367281 | For even distribution of glycerol solution |
| C57BL/6 mice | National Laboratory Animal Center | RMRC11005 | for mouse model |
| Castroviejo forceps 0.12 mm | katena | K5-2500 | |
| Centrifuge | Eppendorf, Germany | 5811000428 | 3,500 x g for 10 min |
| Cheng Yi 10.0 mL sterilized eye dropper bottle | Cheng Yi Chemical, Taiwan | CP405141 | Must be sterile and as the storage container for the final product |
| Corneal rust ring remover with 0.5 mm burr | Algerbrush IITM; Alger Equipment Co., Inc. Lago Vista, TX | CHI-675 | for debridement of the corneal epithelium |
| Dulbecco's modified minimal essential medium | Merck KGaA, Germany | D6429 | |
| Filter paper | Toyo Roshi Kaisha,Ltd. | 1.11 | |
| Fluorescein sodium ophthalmic strips U.S.P | OPTITECH | OPTFL100 | staining for corneal epithelial defect |
| Incubator | Firstek, Taiwan | S300S | 37 °C for 4 h |
| Kanam sterile gloves | Kanam Latex Industries, India | EN455 | For aseptic operation |
| Merck 0.22 µm filter | Merck KGaA, Germany | PR05359 | At least 2 filters for mACS filtration |
| Nang Kuang 250 mL 10% glycerol solution | Nang Kuang Pharmaceutical, Taiwan | 19496 | To offer suitable membrane stabilization effect and extracellular osmotic pressure for blood cells |
| Normal saline | TAIWAN BIOTECH CO., LTD. | 100-120-1101 | |
| Skin biopsy punch 2mm | STIEFEL | 22650 | |
| Stereomicroscope | Carl Zeiss Meditec, Dublin, CA | SV11 | microscope for surgery |
| Terumo 18 G needle | Terumo, Taiwan | SMACF0120-18BX | 3.0 mL syringe with 18 G needle to extract the supernatant after centrifugation |
| Terumo 20.0 mL syringe | Terumo, Taiwan | MDSS20ES | Could be used to collect serum after initial centrifugation and use it for secondary centrifugation. |
| Terumo 3.0 mL syringe with the 23 G needle | Terumo, Taiwan | MDSS03S2325 | 3.0 mL syringe is used to extract the supernatant after centrifugation. Then connect the filter and the 23 G needle for injection into the eye drop bottles. |
| Westcott Tenotomy Scissors Medium | katena | K4-3004 |
Referências
- Fox, R. I., Chan, R., Michelson, J. B., Belmont, J. B., Michelson, P. E. Beneficial effect of artificial tears made with autologous serum in patients with keratoconjunctivitis sicca. Arthritis and Rheumatology. 27 (4), 459-461 (1984).
- Noble, B. A., et al. Comparison of autologous serum eye drops with conventional therapy in a randomised controlled crossover trial for ocular surface disease. The British Journal of Ophthalmology. 88 (5), 647-652 (2004).
- Bradley, J. C., Bradley, R. H., McCartney, D. L., Mannis, M. J. Serum growth factor analysis in dry eye syndrome. Clinical & Experimental Ophthalmology. 36 (8), 717-720 (2008).
- Alshammari, T. M., Al-Hassan, A. A., Hadda, T. B., Aljofan, M. Comparison of different serum sample extraction methods and their suitability for mass spectrometry analysis. Saudi Pharmaceutical Journal. 23 (6), 689-697 (2015).
- Tsubota, K., et al. Treatment of dry eye by autologous serum application in Sjögren’s syndrome. The British Journal of Ophthalmology. 83 (4), 390-395 (1999).
- Urzua, C. A., Vasquez, D. H., Huidobro, A., Hernandez, H., Alfaro, J. Randomized double-blind clinical trial of autologous serum versus artificial tears in dry eye syndrome. Current Eye Research. 37 (8), 684-688 (2012).
- Cui, D., Li, G., Akpek, E. K. Autologous serum eye drops for ocular surface disorders. Current Opinion in Allergy and Clinical Immunology. 21 (5), 493-499 (2021).
- Jirsova, K., et al. The application of autologous serum eye drops in severe dry eye patients; subjective and objective parameters before and after treatment. Current Eye Research. 39 (1), 21-30 (2014).
- Pan, Q., Angelina, A., Marrone, M., Stark, W. J., Akpek, E. K. Autologous serum eye drops for dry eye. Cochrane Database of Systematic Reviews. 2 (2), (2017).
- Wang, L., et al. Autologous serum eye drops versus artificial tear drops for dry eye disease: a systematic review and meta-analysis of randomized controlled trials. Ophthalmic Research. 63 (5), 443-451 (2020).
- Soni, N. G., Jeng, B. H. Blood-derived topical therapy for ocular surface diseases. The British Journal of Ophthalmology. 100 (1), 22-27 (2016).
- Solomon, A., et al. anti-inflammatory forms of interleukin-1 in the tear fluid and conjunctiva of patients with dry-eye disease. Investigative Ophthalmology and Visual Science. 42 (10), 2283-2292 (2001).
- Meijer, H., Reinecke, J., Becker, C., Tholen, G., Wehling, P. The production of anti-inflammatory cytokines in whole blood by physico-chemical induction. Inflammation Research. 52 (10), 404-407 (2003).
- Bielory, B. P., Shah, S. P., O’Brien, T. P., Perez, V. L., Bielory, L. Emerging therapeutics for ocular surface disease. Current Opinion in Allergy and Clinical Immunology. 16 (5), 477-486 (2016).
- Stevenson, W., Chauhan, S. K., Dana, R. Dry eye disease: an immune-mediated ocular surface disorder. Archives of Ophthalmology. 130 (1), 90-100 (2012).
- Yang, J., Guo, A., Li, Q., Wu, J. Platelet-rich plasma attenuates interleukin-1β-induced apoptosis and inflammation in chondrocytes through targeting hypoxia-inducible factor-2α. Tissue and Cell. 73, 101646 (2021).
- Shakouri, S. K., Dolati, S., Santhakumar, J., Thakor, A. S., Yarani, R. Autologous conditioned serum for degenerative diseases and prospects. Growth Factors. 39 (1-6), 59-70 (2021).
- Gull, M., Pasek, M. A. The role of glycerol and its derivatives in the biochemistry of living organisms, and their prebiotic origin and significance in the evolution of life. Catalysts. 11 (1), 86 (2021).
- Drew, V. J., Tseng, C. L., Seghatchian, J., Burnouf, T. Reflections on dry eye syndrome treatment: therapeutic role of blood products. Frontiers in Medicine. 5, 33 (2018).
- Hung, K. H., Yeh, L. K. Ex vivo and in vivo animal models for mechanical and chemical injuries of corneal epithelium. Journal of Visualized Experiments. (182), e63217 (2022).
- Geerling, G., Maclennan, S., Hartwig, D. Autologous serum eye drops for ocular surface disorders. The British Journal of Ophthalmology. 88 (11), 1467-1474 (2004).
- Rutgers, M., Saris, D. B., Dhert, W. J., Creemers, L. B. Cytokine profile of autologous conditioned serum for treatment of osteoarthritis, in vitro effects on cartilage metabolism and intra-articular levels after injection. Arthritis Research & Therapy. 12 (3), R114 (2010).
- Antebi, B., et al. Short-term physiological hypoxia potentiates the therapeutic function of mesenchymal stem cells. Stem Cell Research & Therapy. 9 (1), 265 (2018).
- Chen, Y. M., Wang, W. Y., Lin, Y. C., Tsai, S. H., Lou, Y. T. Use of autologous serum eye drops with contact lenses in the treatment of chemical burn-induced bilateral corneal persistent epithelial defects. BioMed Research International. 2022, 6600788 (2022).
- Diaz-Valle, D., et al. Comparison of the efficacy of topical insulin with autologous serum eye drops in persistent epithelial defects of the cornea. Acta Ophthalmologica. 100 (4), e912-e919 (2022).
- Metheetrairut, C., et al. Comparison of epitheliotrophic factors in platelet-rich plasma versus autologous serum and their treatment efficacy in dry eye disease. Scientific Reports. 12 (1), 8906 (2022).
- NaPier, E., Camacho, M., McDevitt, T. F., Sweeney, A. R. Neurotrophic keratopathy: current challenges and future prospects. Annals of Medicine. 54 (1), 666-673 (2022).
- Garcia-Conca, V., et al. Efficacy and safety of treatment of hyposecretory dry eye with platelet-rich plasma. Acta Ophthalmologica. 97 (2), e170-e178 (2019).
- Gholian, S., et al. Use of autologous conditioned serum dressings in hard-to-heal wounds: a randomised prospective clinical trial. Journal of Wound Care. 31 (1), 68-77 (2022).
- Raeissadat, S. A., Rayegani, S. M., Jafarian, N., Heidari, M. Autologous conditioned serum applications in the treatment of musculoskeletal diseases: a narrative review. Future Science OA. 8 (2), 776 (2022).
- Tokawa, P. K. A., Brossi, P. M., Baccarin, R. Y. A. Autologous conditioned serum in equine and human orthopedic therapy: A systematic review. Research in Veterinary Science. 146, 34-52 (2022).
- Evans, C. H., Chevalier, X., Wehling, P. Autologous conditioned serum. Physical Medicine and Rehabilitation. Clinics of North America. 27 (4), 893-908 (2016).
- Coskun, H. S., Yurtbay, A., Say, F. Platelet rich plasma versus autologous conditioned serum in osteoarthritis of the knee: clinical results of a five-year retrospective study. Cureus. 14 (4), e24500 (2022).

