Estrogen-Like Effect of Bazi Bushen Capsule in Ovariectomized Rats
Summary
Presented here is a protocol to show that Bazi Bushen capsule (BZBS) can regulate the RANKL/OPG signaling pathway in the ovariectomized rodent model through its estrogen-like effect.
Abstract
This study aims to show the estrogen-like effect of Bazi Bushen capsule (BZBS), a Chinese herbal compound, in ovariectomized mice. Female Sprague-Dawley (SD) rats were randomly divided into six groups: a sham-operated group, a model group (OVX), a progynova group, and BZBS groups (1, 2, and 4 d/kg/d). An ovariectomy was performed on all rats except those in the sham-operated group. Micro-computed tomography (micro-CT) scanning, hematoxylin and eosin (H&E) staining, immunohistochemistry, and enzyme-linked immunosorbent assay (ELISA) detection were performed after 4 months of BZBS treatment. As a result, compared with the OVX group, rats treated with BZBS showed an increased number and area of trabecular bone and bone marrow cells, and a decreased number of adipose cells. The bone volume, trabecular number, and trabecular thickness of the right tibia in the medication groups increased and the trabecular space decreased. The 17β-estradiol and serum calcium levels in the medication groups were elevated, but the levels of serum phosphorus, sclerostin, β-CTX, and TRACP-5b were decreased. In the medication groups, the RANKL and sclerostin levels were decreased, while the osteoprotegerin (OPG) level was increased. In conclusion, this protocol systematically evaluated the therapeutic effects and potential molecular mechanisms of Chinese herbal compounds in ovariectomized rats with a variety of techniques.
Introduction
Postmenopausal osteoporosis (PMOP) is a skeletal system disease caused by inadequate ovarian function, decreased estrogen, and enhanced osteoclast activity1, characterized by low bone mass, microarchitectural degeneration of the bone tissue, and damage of the bone trabecula. PMOP prone to cause fractures and can have serious effects on patients’ quality of life. Osteoporosis affects about 200 million people worldwide2; about 40% of postmenopausal women suffer from osteoporosis3, and fractures occur in 33% of patients with PMOP4. The reduction of estrogen could lead to a relatively enhanced osteoclast activity5 and declined bone mass when bone resorption exceeds bone formation. Thus, osteoclast activation is usually considered a sign of bone loss6. Physiologically, RANKL/OPG serves as an important pathway involving bone remodeling by regulating osteoclast activation to promote bone resorption7. Meanwhile, estrogen can also mediate osteoclast formation and function by regulating RANKL/OPG signaling8.
Over recent years, traditional medicine has been increasingly used to treat different diseases with less adverse reactions and better prognosis9,10. Bazi Bushen capsule (BZBS), a traditional Chinese medicine, could be an alternative approach to prevent and treat PMOP. It is composed of Cuscuta, Fructus lycii, Fructus schisandrae, Fructus cnidii, Fructus rosae laevigatae (Cherokee rose fruit), Raspberry, Semen Allii Tuberosi, Toosendan fructus, Herba epimedii, Morinda officinalis, Herba cistanches, Rehmannia glutinosa, Medicinal cyathula root, Ginseng, Pilose antler, and Hippocampus Kelloggi (Table 1), containing 11 kinds of phytoestrogen with hormone-like effects. A previous study has confirmed that BZBS can delay the formation of atheromatous plaque through an estrogen-like effect11, which is similar to treating osteoporosis with estrogen12. However, the underlying mechanisms of BZBS in the prevention and treatment of osteoporosis caused by the deficiency of estrogen are unclear. Therefore, the present study aimed to verify whether BZBS has a bone-protective effect on ovariectomy-induced rat osteoporosis13,14.
Protocol
The animal experiments were approved by the Committee on Animal Research and Ethics of the Hebei Yiling Medical Research Institute (approval number: N2020150). The 36 Sprague-Dawley (SD) female rats (3 months old, weighing 180-200 g) (see Table of Materials) were fed with normal food and clean water in the new drug evaluation center of Hebei Yiling Medical Research Institute and were exposed to artificial light for 12 h per day in rooms with controlled temperatures (20-26 °C) and relative humidity (40%-70%).
1. Animal experiment
- Divide the rats into six groups randomly, a sham-operated group (SHAM group, 10 mL/kg/d saline), model group (OVX group, 10 mL/kg/d saline), progynova15 group (0.2 mg/kg/d), low-dose BZBS group (BZBSL group, 1 g/kg/d), middle-dose BZBS group (BZBSM group, 2 g/kg/d), and high-dose BZBS group (BZBSH group, 4 g/kg/d).
NOTE: The dose of the BZBSM group was determined from the recommended dosages for humans and calculated as follows: equivalent experimental dose for rats (mg/kg/d) = human dose (mg/kg)/body weight (60 kg) x 6.3. - Employ 3% and 1.5% isoflurane for the induction and maintenance of rat anesthesia throughout the operation, respectively.
NOTE: If the rats do not blink and the pedal reflex is absent, the depth of anesthesia is confirmed and the maintenance of rat anesthesia is conducted, respectively. - Apply local analgesia with a topical injection of 1.5 mg/kg bupivacaine after anesthesia. Remove the fur of the rat's abdomen with a razor and disinfect the skin of the rat's abdomen using ethanol and povidone-iodine 3x with a sterile cotton ball.
- Perform a 2 cm midline incision on the abdomen of the rat with a scalpel in the SHAM group and suture the midline incision using a 4-0 surgical suture.
- For the rats in the other groups, isolate and expose the intact ovary using forceps and ligate at the root of the ovary using a 4-0 surgical suture. Remove the intact ovary using surgical scissors. Suture the midline incision using a 4-0 surgical suture.
NOTE: All postoperative rats were placed in isolation in 37 °C containers until the rats were awake and able to move freely. - After 4 months of intragastric administration of designated drugs (in step 1.1), first anesthetize the rat with 3% isoflurane, and then disinfect the skin of the groin and entire lower limb using ethanol and povidone-iodine 3x with a sterile cotton ball.
NOTE: The drug was administered once a day with a 10 cm stainless steel gavage needle. The gavage dose was adjusted weekly according to the weight of the rats. - Cut and peel the skin of the groin to expose the femoral artery using surgical scissors and tweezers. Collect a blood sample from the femoral artery using a vacuum blood collection tube and then apply pressure to stop bleeding with a cotton ball.
- Continue to cut and peel the skin of the groin and the lower limb to expose the intact tibia using surgical scissors and tweezers. After removing the tibia, wash it three times with saline and continue to remove excess muscle tissue in saline with tweezers and ophthalmic scissors.
- Disinfect the back skin of the lumbar L4 using ethanol and povidone-iodine 3x with a sterile cotton ball. Use surgical scissors and tweezers to cut the back skin and expose the lumbar L4.
- Sever the upper and lower vertebrae of the lumbar L4 with a rongeur and cut off the sternum around the lumbar L4 with surgical scissors. Wash the isolated lumbar L4 with saline three times and continue to remove excess muscle tissue in saline with tweezers and micro scissors.
- Store the tibia and L4 vertebra in 10% formalin tissue fluid (see Table of Materials) at room temperature for 3 days.
- Euthanize the rats with excessive inhalation of carbon dioxide, pack the carcass in a special cadaver bag, and place it in an animal carcass storage cabinet.
2. Micro-CT imaging16
- Press the green power button to turn on the micro-CT scanner (see Table of Materials) and start the software to heat the x-ray source.
NOTE: To avoid scattering of the x-rays, move any metal objects near the imaging area before starting. Micro-CT examination of rat tibial tissue in step 1.11 with insufficient x-ray irradiation can affect the findings. The bone should be kept moist during the micro-CT scanning. The animal bed should be taken out while warming up. Make sure that the key is turned to the ON position on the SAFETY KEY in the lower left corner in front of the CT instrument. - Create a database by clicking on a new one to build a file to save the data to obtain next.
- Set the data acquisition parameters in the software control window as follows: x-ray tube voltage (50 kV); CT x-ray tube current (100 µA); live x-ray tube current (100 µA); field of view (FOV) (18 mm); no gating technique; scanning technique (high resolution 4 min).
- Wash the tibia with saline three times and blot out excess water with filter paper. Wrap the tibia tissue on the bed with plastic film to fix its position. Adjust the tibia tissue to the center of the imaging field by rotating the button in the instrument.
- Close the instrument door and turn on the live mode. Press the capture button to view the tibial tissue.
- Start the scan by clicking the CT scan button. Click Yes to produce the image.
NOTE: Once the x-ray is turned on, the orange light at the top of the instrument will be illuminated and the sliding door will not open for the safety of the operator. When the scan is complete, a new window appears in the 2D Viewer software showing the reconstructed cross-axis, coronal plane, and sagittal plane. Click Paste to the drawing board to save. - Take the tibia out of the animal bed. Open the Analyze 12.0 software and import the objective CT image by clicking File > Load > Process. Click the Image Calculator… and Region Pad options to display the SubRegion dialog box.
- Click the Interactive option and select the bone in the 2 mm thickness area. Click Apply to select Change a Copy of the Loaded Volume. In the Analyze 12.0 screen, click the Apps option to select BMA.
- Click Segment Cortex and Segment Trabeculae, then click Save Final Object Map. Click Measure Bone to display the value of specific parameter indicators, including bone mineral density (BMD, g/cm3), bone volume fraction (bone tissue volume/tissue volume [BV/TV], %), number of bone trabeculae (Tb.N, 1/mm), bone trabecular thickness (TB.Th, µm), and trabecular separation degree (Tb.Sp, µm).
NOTE: Micro-CT was not performed with L4 vertebrae.
3. Hematoxylin and eosin (H&E) staining
- Take out the tibia and L4 vertebra samples of the rat in step 1.11 and store them in 20% formic acid solution for a 4 day decalcification.
NOTE: The 20% formic acid solution is prepared by mixing 2,400 mL of formaldehyde solution and 600 mL of formic acid solution. - Put the processed tibia and L4 vertebra sample of the rat from step 3.1 in the embedding box and wash them with running water for more than 6 h.
- Dehydrate the samples with gradient concentration alcohol solutions (60% ethanol for 1 h, 70% ethanol for 1 h, 90% ethanol for 1 h, 95% ethanol for 2 h, and 100% ethanol for 2 h) using an automated tissue processor.
- Place the tissue specimens in xylene for 2 h to make them translucent. At the end of dehydration, place the permeabilized samples in paraffin wax at 60 °C for 3 h and embed them in an automatic processor.
- Obtain the 4 µm sections by using a rotary slicer. Place the sections in hematoxylin stain for 3-8 min and eosin stain for 1-3 min.
- Transfer the stained sections to pure alcohol and xylene, respectively. Seal and fix the stained sections with neutral gum for pathological examination under an optical microscope.
- Scan the slices with a slide scanner (see Table of Materials) at 40x magnification and use the viewing software to acquire the H&E staining results of the entire tibia and L4 vertebra.
4. Immunohistochemistry
- Perform heat-induced epitope retrieval using the 4 µm tibia sections obtained in step 3.5. Add the appropriate antigen repair buffer (citric acid buffer, pH = 6.0) to a microwaveable container. Place the carrier sheet into the microwaveable container and place the container in the microwave oven. Set the program to hold the antigen repair for 20 min at 98 °C.
- Remove the container and rinse the interior with cold tap water for 10 min. Block endogenous peroxidase activity with hydrogen peroxide (3% H2O2) (see Table of Materials), then incubate the slice for 15 min at room temperature.
- Wash the sections in phosphate-buffered saline (PBS) (see Table of Materials) three times, 5 min each.
- Put the sections into normal goat serum blocking solution (see Table of Materials), then incubate the samples in a moist chamber for 15 min at room temperature.
- Add 200 µL of RANKL antibody (1:200) (see Table of Materials), OPG antibody (1:300) (see Table of Materials), and sclerostin antibody17 (1:200) (see Table of Materials) into the slice samples and incubate for one night at 4 °C in the dark.
NOTE: Sclerostin is a negative regulator of bone growth. It is a secreted glycoprotein with a C-terminal cysteine knot-like domain and has a sequence similarity to the DAN family of bone morphogenetic protein antagonists17. This protein is not a member of the RANKL pathway. - Add biotin-labeled goat anti-rabbit IgG (see Table of Materials) into the sections and incubate at room temperature for 30 min. Rinse the sections with PBS and use diaminobenzidine (DAB) (see Table of Materials) to stain the sections for 3-5 min at room temperature.
- Dye the sections with hematoxylin for 1-2 min. After dehydrating and drying the sections, seal them with neutral gum.
- Scan the sections at 40x magnification using a slide scanner (see Table of Materials) and capture digital images using an automatic microscope imaging system (see Table of Materials).
- Open Imagepro Plus software (see Table of Materials).
- Click File to import the analysis image. Click Measure > Calibration > Intensity Calibration for optical density correction. Click the Measure, IOD, Count/Size, and Select Colors options one by one to display the Segmentation interface.
- Click the Histogram Based option to select HIS and click Load File. Click Count to generate the IOD value and Area value.
5. Enzyme-linked immunosorbent assay (ELISA) protocol
- Ensure that the ELISA kit for detecting 17β-estradiol includes the anti-17β-estradiol IgG-coated microplate (12 x 8 wells), 15 mL of stop solution, 22 mL of 17β-estradiol-HRP conjugate, 15 mL of 3,3',5,5' tetramethylbenzidine (TMB) substrate solution, 50 mL of 10x washing solution, cover foils, a strip holder, the 17β-estradiol control, and a different concentration gradient of 17β-estradiol standard (see Table of Materials).
- Remove the 17β-estradiol kit from the refrigerator and place all the reagents at room temperature (18-25 °C).
- Reagent preparation: Dilute the 10x washing solution with deionized water to prepare a 1x washing solution. Mix 50 mL of 10x washing solution with 450 mL of deionized water to make 500 mL of 1x washing solution.
- Remove the serum sample from the refrigerator and thaw it at room temperature.
- Remove the excess microtiter strips from the plate frame, return them to the foil bag containing the desiccant pack, reseal them, and store at 4 °C.
- Add 25 µL of control, standard, or sample to each of their respective wells. Then, add 200 µL of 17β-estradiol-HRP conjugate to each well.
- Cover all the wells with foil and incubate the plate at 37 °C for 2 h.
- Remove the foil and liquid from each well and wash the well three times with 300 µL of 1x washing solution.
NOTE: Avoid overflowing of the reaction wells. Also, the soak time between each wash cycle should be >5 s. At the end, carefully remove the remaining fluid by tapping tissue paper strips before the next step. - Add 100 µL of TMB substrate solution to each well and incubate in the dark for 30 min at room temperature.
- Add 100 µL of stop solution to each well and gently shake the microtiter plate for 30 s. Measure the absorbance of the sample at 450 nm within 30 min.
- Draw the average absorbance of the standards against concentration. Plot the best-fit curve by the plotted points.
- Obtain the corresponding concentration values expressed in pg/mL by interpolating the values of the samples on the standard curve.
NOTE: The other protocols for detecting serum Ca2+, phosphorus, SOST, TRACP-5b, and β-CTX are the same as the 17β-estradiol measurement, according to the manufacturer's instructions.
6. Statistical analysis
- Data were presented as mean ± standard deviation. Multiple comparisons were performed by one-way ANOVA followed by Tukey's test. p < 0.05 was considered statistically significant.
Representative Results
The bone microstructure of the tibia was evaluated by micro-CT scanning
The treatment of ovariectomized rats with BZBS significantly reduced OVX-induced trabecular structural changes. As shown in the reconstructed micro-CT images of the right tibia (Figure 1A,B), trabecular bone in the OVX group showed a significant decrease in BMD (Figure 1C), BV/TV (Figure 1D), Tb.Th (Figure 1E), Tb.N (Figure 1F), and an increase in Tb.Sp (Figure 1G), compared with the SHAM group. Compared with the OVX group, BV/TV, Tb.N, and Tb.Th. of the right tibia in the BZBS treatment groups were significantly increased, and Tb.Sp. was markedly decreased.
Bone histology of the tibia and L4 vertebra was evaluated by H&E staining
Histomorphological damage in ovariectomized rats was relieved after taking BZBS. As shown in Figure 2A,B, the H&E staining results indicated consistent changes between the tibia and L4 vertebra. Compared with the SHAM group, trabecular bone in the OVX group was sparser and thinner, characterized by a disordered, fractured, and irregular structure with more adipose cells, and a significantly decreased number of bone marrow cells. Compared with the OVX group, BZBS treatment showed an increased number and area of trabecular bone (yellow arrow), decreased number and area of adipose cells (blue arrow), and increased number of bone marrow cells (green arrow). All the above data indicated that BZBS could improve the bone histology of ovariectomized rats.
RANKL, OPG, and SOST protein expressions of the tibia were evaluated by immunohistochemistry
The expression of osteogenic/osteoclast cytokines in ovariectomized rats was improved by BZBS treatment. Compared with the OVX group, the immunohistochemistry results showed a decreased expression of RANKL (Figure 3A), increased expression of OPG (Figure 3B), and reduced expression of SOST (Figure 3C) in the BZBS treatment groups.
Bone metabolic markers in serum were evaluated by ELISA
Biochemical markers of bone metabolism in ovariectomized rats were also improved by BZBS. Compared with the SHAM group, the OVX group had a lower level of 17β-estradiol (Figure 4A) and Ca2+ (Figure 4B), and a higher level of phosphorus (Figure 4C), SOST (Figure 4D), TRACP-5b (Figure 4E), and β-CTX (Figure 4F). Nevertheless, BZBS treatment elevated serum 17β-estradiol and Ca2+ levels, but subdued the levels of phosphorus, SOST, β-CTX, and TRACP-5b.
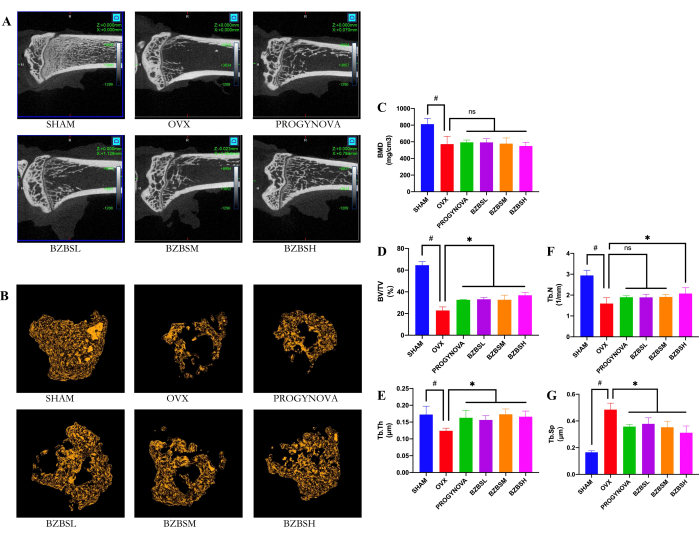
Figure 1: Micro-CT scanning resultson ovariectomized rats with osteoporosis after 4 months of treatment with BZBS. (A) 2D images in the vertical plane of the right tibia. (B) 3D images of the right tibia. The microstructure parameters include BMD (C), BV/TV (D), Tb.Th (E), Tb.N (F), and Tb.Sp (G). n = 3, #p < 0.05, compared with the SHAM group; *p < 0.05, compared with the OVX group. Please click here to view a larger version of this figure.
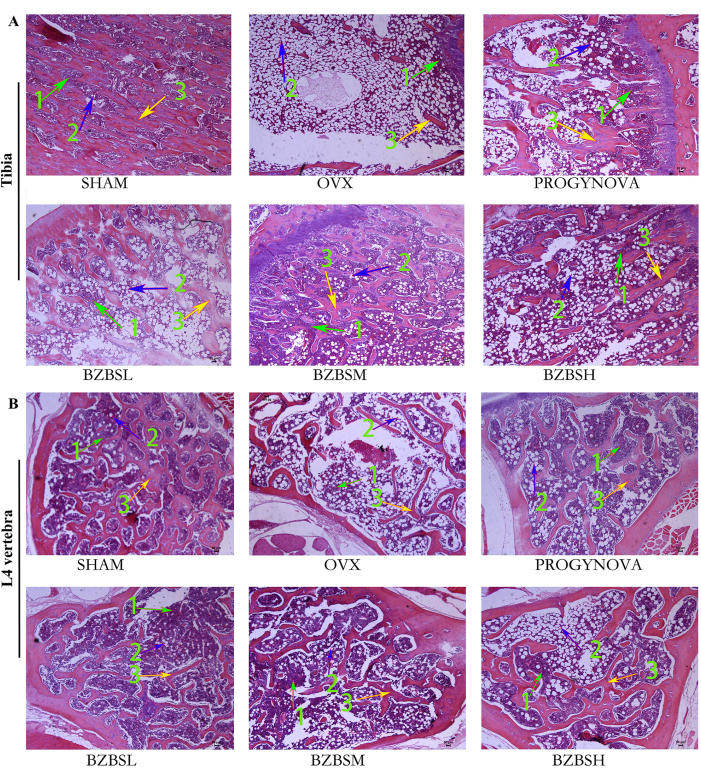
Figure 2: H&E staining images of the tibia (A) and L4 vertebra (B) sections. The number "1" represents bone marrow cells, the number "2" represents adipose cells, and the number "3" represents trabecular bone structure. n = 6. Please click here to view a larger version of this figure.
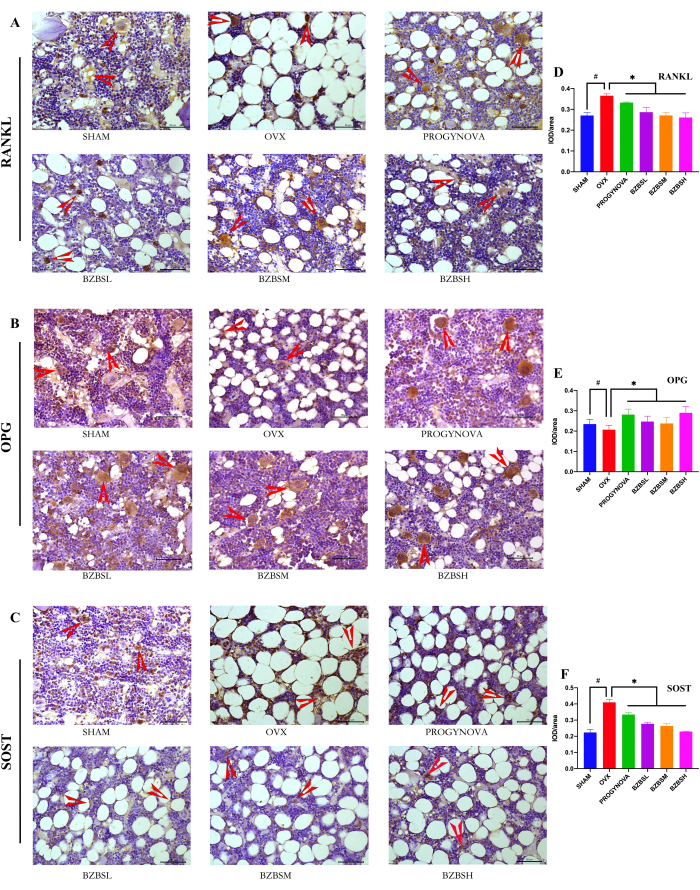
Figure 3: Effect of BZBS on RANKL, OPG, and SOST expression in ovariectomized rats. The expression level of RANKL (A), OPG (B), and SOST (C) were evaluated with immunohistochemistry after 4 months of BZBS treatment. Quantitative analysis of RANKL (D), OPG (E), and SOST (F) expression. Red arrows represent positive expressions. n = 3, #p < 0.05, compared with the SHAM group; *p < 0.05, compared with the OVX group. Please click here to view a larger version of this figure.
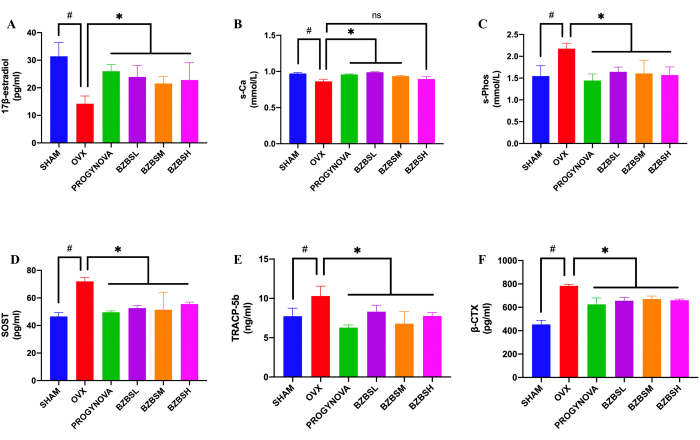
Figure 4: Serum 17β-estradiol, Ca2+, phosphorus, SOST, TRACP-5b, and β-CTX expression in ovariectomized rats in different groups byELISA evaluation. Expression of 17β-estradiol (n = 5) (A), Ca2+ (n = 3) (B), phosphorus (n = 6) (C), SOST (n = 5) (D), TRACP-5b (n = 5) (E), and β-CTX (n = 3) (F). #p < 0.05, compared with the SHAM group; *p < 0.05, compared with the OVX group. Please click here to view a larger version of this figure.
| Latin name | Pinyin | Amount in application (ratio) |
| Cuscuta | Tusizi | 250 |
| Fructus lycii | Gouqizi | 138 |
| Fructus schisandrae | Wuweizi | 46 |
| Fructus cnidii | Shechuangzi | 35 |
| Fructus rosae laevigatae(Cherokee rose fruit) | Jinyingzi | 35 |
| Raspberry | Fupenzi | 35 |
| Semen Allii Tuberosi | Jiucaizi | 35 |
| Toosendan fructus | Chuanlianzi | 23 |
| Herba epimedii | Yinyanghuo | 70 |
| Morinda officinalis | Bajitian | 35 |
| Herba cistanches | Roucongrong | 35 |
| Rehmannia glutinosa | Shengdihuang | 46 |
| Medicinal cyathula root | Chuanniuxi | 35 |
| Ginseng | Renshen | 25 |
| Pilose antler | Lurong | 16 |
| Hippocampus Kelloggi | Haima | 21 |
Table 1: Drug composition and proportion contained in prescription BZBS.
Discussion
After ovariectomy, estrogen levels decreased sharply due to the continuous reduction of cancellous bone and increased bone turnover. Essentially, decreased estrogen secretion after menopause could cause significant bone loss18, leading to Ca2+ loss in ovariectomized rats19,20. It has been reported that taking estrogen can help the metaphyseal fracture recovery of ovariectomized rats, and estrogen replacement therapy can also be used to maintain bone mass and prevent osteoporosis in postmenopausal women. Previous studies have indicated that using estradiol helps improve bone microarchitecture21. This experiment confirmed that BZBS could inhibit bone Ca2+ loss and improve the bone microstructure of ovariectomized rats, which is consistent with the previous study18. For the serum test, BZBS enhanced the decrease of estrogen in ovariectomized rats and regulated the levels of calcium and phosphate. Meanwhile, BZBS could lower the RANKL expression and increase the OPG expression in ovariectomized rats, which would effectively inhibit the rising of bone resorption markers. Furthermore, the micro-CT imaging and H&E staining results showed that BZBS improved the bone microarchitecture of ovariectomized rats, which indicated that BZBS had the effect of preventing and treating osteoporosis, as well as lowering bone resorption markers and inhibiting bone loss. Compared with the model group, BZBS treatment increased serum estradiol levels and lowered bone loss, which suggested that BZBS might protect bone through an estrogen-like effect.
Osteoporosis induced by estrogen deficiency might be related to regulation of the RANKL/OPG signal. The ratio of RANKL to OPG could be a sign of osteoclast activation23, which is responsible for regulating the balance between bone formation and bone resorption. The binding of activated RANKL to RANK due to estrogen deficiency would promote osteoclast differentiation and improve bone resorption, which would lead to the temporary decrease of bone volume24. As a decoy receptor, OPG would competitively bind to RANK and directly block the activation of RANKL/RANK, which would lower osteoclast activity and inhibit bone resorption. In this experiment, increased OPG expression of ovariectomized rats in the BZBS group was found and RANKL was inhibited. It is reported that a raised SOST level could increase RANKL expression and lower OPG expression25. In addition, as the cause of bone loss in ovariectomized rats26, overexpression of sclerostin could be an indicator to accurately evaluate the bone microenvironment. Meanwhile, our results revealed that BZBS could prevent bone loss by inhibiting the overexpression of sclerostin and suppressing osteoclast activity.
In this study, a rat model of osteoporosis induced by ovarian removal was successfully constructed, and estrogen-like regulation of bone homeostasis by the Chinese herbal compound BZBS was further confirmed by integrating micro-CT, H&E staining, and other various molecular biological techniques. This integrated methodological process provides a reference for the development of anti-osteoporosis drugs27. As the key imaging technique for evaluating bone health status, micro-CT can obtain nondestructive 3D imaging of bone trabecular microstructures to quantify micro-changes of bone structure and bone density. However, the following aspects should be paid attention to when performing micro-CT. First of all, in order to prevent the sample from moving, low-density media, such as plastic film, should be used to wrap the sample, and the sample should be placed parallel in the small animal bed. Secondly, it is worth noting that unstable voltage can easily cause malfunction during scanning. Finally, and more notably, the estrogen-deficient osteoporosis model established by ovarian removal cannot fully simulate clinical postmenopausal women with osteoporosis. More gene-edited animal models of hormone-deficient osteoporosis or actual clinical cases should be further tested.
Disclosures
The authors have nothing to disclose.
Acknowledgements
This work was supported by the Strategic Consulting Project of the Chinese Academy of Engineering: Strategic research on anti-aging effect of Traditional Chinese Medicine (Grant No.: 2022-XY-45), the Natural Science Foundation of Hebei Province in China (Grant No.:2022106065), S&T Program of Hebei, China (Grant No.:22372502D) and High-level S&T Innovation and Entrepreneurship Talent Project of Shijiazhuang, Hebei, China (Grant No.: 07202203).
Materials
| 10% neutral buffered formalin | Proteintech Group, Inc. | 23408-1AP-100 | |
| 17-Beta-Estradiol ELISA kits | Proteintech Group, Inc. | 21933-1-AP | |
| 3%H2O2 | ShanDong LIRCON Medical Technology Incorporated Company | 20221027 | |
| Anti-osteoprotegerin antibody | Abcam | ab203061 | |
| Bazibushen capsules | Shijiazhuang Yiling Pharmaceutical Co. Ltd. | XB2103001 | |
| Biotin goat anti-rabbit IgG | Abcam | ab207995 | |
| Calcium assay kits | Nanjing Jiancheng Bioengineering Institute | C004-2-1 | |
| Diaminobenzidine | ZSGB-BIO | ZLI-9018 | |
| Estradiol valerate tablets | Bayer AG | 156A | |
| Gene 1580R centrifuge | GENE Co. Ltd. | GZ422515090077 | |
| Image-Pro Plus | Media Cybernetics | IPP 6.0 | |
| Leica DM6000B Microscope (fully automated upright microscope system) | Leica | 361715 | |
| Normal Goat Serum Blocking Solution | Vector Laboratories | S-1000-20 | |
| Phosphate assay kits | Nanjing Jiancheng Bioengineering Institute | C006-1-1 | |
| Phosphate buffered saline | Servicebio, Wuhan, China | CR10201M | |
| Quantum GX2 microCT Imaging System | PerkinElmer | CLS149276 | |
| RANKL rabbit polyclonal antibody | Elabscience Biotechnology Co. Ltd. | E-EL-R3032-96T | |
| Rat SOST (Sclerostin) ELISA kit | Elabscience Biotechnology Co. Ltd. | E-EL-R1405c-96T | |
| Rat TRACP-5b (Tartrate Resistant Acid Phosphatase 5b) ELISA kit | Abcam | ab108667 | |
| Rat β-CTx (Beta Crosslaps) ELISA kit | Elabscience Biotechnology Co., Ltd. | E-EL-R0939c-96T | |
| Sclerostin rabbit polyclonal antibody | Beijing Vital River Laboratory Animal Technology Co., Ltd. | SCXK(JING)2016-0006 | |
| Slide scanner | Hamamatsu Photonics K.K. | Nano Zoomer-SQ | |
| Sprague Dawley female rats | Beijing Vital River Laboratory Animal Technology Co., Ltd. | ZLI-9381 |
References
- Zhai, X., et al. Muscone ameliorates ovariectomy-induced bone loss and receptor activator of nuclear factor-κb ligand-induced osteoclastogenesis by suppressing TNF receptor-associated factor 6-mediated signaling pathways. Frontiers in Pharmacology. 11, 348 (2020).
- Wang, S., et al. An antioxidant sesquiterpene inhibits osteoclastogenesis via blocking IPMK/TRAF6 and counteracts OVX-induced osteoporosis in mice. Journal of Bone and Mineral Research. 36 (9), 1850-1865 (2021).
- Xia, C., et al. Bushenhuoxue formula promotes osteogenic differentiation of growth plate chondrocytes through β-catenin-dependent manner during osteoporosis. Biomedicine & Pharmacotherapy. 127, 110170 (2020).
- Chen, Y., et al. Shikonin relieves osteoporosis of ovariectomized mice by inhibiting RANKL-induced NF-κB and NFAT pathways. Experimental Cell Research. 394 (1), 112115 (2020).
- Lorenzo, J. From the gut to bone: connecting the gut microbiota with Th17 T lymphocytes and postmenopausal osteoporosis. The Journal of Clinical Investigation. 131 (5), e146619 (2021).
- Meng, X., et al. Estrogen-mediated downregulation of HIF-1α signaling in B lymphocytes influences postmenopausal bone loss. Bone Research. 10 (1), 15 (2022).
- Yue, H., et al. Comparative study of holothurin A and echinoside A on inhibiting the high bone turnover via downregulating PI3K/AKT/β-catenin and OPG/RANKL/NF-κB signaling in ovariectomized mice. Food & Function. 13 (8), 4748-4756 (2022).
- Wang, Z., et al. An emerging role of Prevotella histicola on estrogen deficiency-induced bone loss through the gut microbiota-bone axis in postmenopausal women and in ovariectomized mice. The American Journal of Clinical Nutrition. 114 (4), 1304-1313 (2021).
- Wang, X., et al. Salidroside, a phenyl ethanol glycoside from Rhodiola crenulata, orchestrates hypoxic mitochondrial dynamics homeostasis by stimulating Sirt1/p53/Drp1 signaling. Journal of Ethnopharmacology. 293, 115278 (2022).
- Hou, Y., et al. Salidroside intensifies mitochondrial function of CoCl2-damaged HT22 cells by stimulating PI3K-AKT-MAPK signaling pathway. Phytomedicine. 109, 154568 (2023).
- Huang, D., et al. Bazi Bushen Capsule alleviates post-menopausal atherosclerosis via GPER1-dependent anti-inflammatory and anti-apoptotic effects. Frontiers in Pharmacology. 12, 658998 (2021).
- Genazzani, A. R., Monteleone, P., Giannini, A., Simoncini, T. Hormone therapy in the postmenopausal years: considering benefits and risks in clinical practice. Human Reproduction Update. 27 (6), 1115-1150 (2021).
- Saville, P. D. Changes in skeletal mass and fragility with castration in the rat; a model of osteoporosis. Journal of the American Geriatrics Society. 17 (2), 155-166 (1969).
- Huyut, Z., Alp, H. H., Bakan, N., Yıldırım, S., Şekeroğlu, M. R. Stimulating effects of vardenafil, tadalafil, and udenafil on vascular endothelial growth factor, angiogenesis, vitamin D3, bone morphogenic proteins in ovariectomized rats. Archives of Physiology and Biochemistry. 128 (4), 1121-1127 (2020).
- Tian, Y., Xu, K. H., Qiao, L. Comparative study of effects of hormonal therapies on the healing of fracture in ovariectomized rats and Rats’ endometria. Journal of Sichuan University. Medical Science Edition. 37 (3), 416-420 (2006).
- Clark, D. P., Badea, C. T. Micro-CT of rodents: state-of-the-art and future perspectives. Physica Medica. 30 (6), 619-634 (2014).
- Xu, C., et al. Sclerostin antibody promotes bone formation through the Wnt/β-catenin signaling pathway in femoral trochlear after patellar instability. Connective Tissue Research. 64 (2), 148-160 (2023).
- Shang, Q., et al. Jingui Shenqi pills regulate bone-fat balance in murine ovariectomy-induced osteoporosis with kidney yang deficiency. Evidence-Based Complementary and Alternative Medicine. 2020, 1517596 (2020).
- Ge, G., et al. Theaflavin-3,3′-digallate promotes the formation of osteoblasts under inflammatory environment and increases the bone mass of ovariectomized mice. Frontiers in Pharmacology. 12, 648969 (2021).
- Quintero-García, M., et al. Calcium bioavailability of Opuntia ficus-indica Cladodes in an ovariectomized rat model of postmenopausal bone loss. Nutrients. 12 (5), 1431 (2020).
- Mattix Kramer, ., J, H., Grodstein, F., Stampfer, M. J., Curhan, G. C. Menopause and postmenopausal hormone use and risk of incident kidney stones. Journal of the American Society of Nephrology. 14 (5), 1272-1277 (2003).
- Ohlsson, C., et al. The effects of estradiol are modulated in a tissue-specific manner in mice with inducible inactivation of ERα after sexual maturation. American Journal of Physiology. Endocrinology and Metabolism. 318 (5), E646-E654 (2020).
- Yahiro, Y., et al. BMP-induced Atoh8 attenuates osteoclastogenesis by suppressing Runx2 transcriptional activity and reducing the Rankl/Opg expression ratio in osteoblasts. Bone Research. 8 (1), 32 (2020).
- Zhu, M., et al. Vinpocetine inhibits RANKL-induced osteoclastogenesis and attenuates ovariectomy-induced bone loss. Biomedicine & Pharmacotherapy. 123, 109769 (2020).
- Ardawi, M. S. M., et al. High serum sclerostin predicts the occurrence of osteoporotic fractures in postmenopausal women: the Center of Excellence for Osteoporosis Research Study. Journal of Bone and Mineral Research. 27 (12), 2592-2602 (2012).
- Bai, S. Y., Chen, Y., Dai, H. W., Huang, L. Effect of sclerostin on the functions and related mechanisms of cementoblasts under mechanical stress. West China Journal of Stomatology. 37 (2), 162-167 (2019).
- Wang, T., et al. Therapeutic potential and outlook of alternative medicine for osteoporosis. Current Drug Targets. 18 (9), 1051-1068 (2017).

.
