Author Spotlight: Exploring Venous Waveforms for Non-Invasive Respiratory Monitoring in Pigs
Summary
Infusing oleic acid continuously into the pulmonary artery of an anesthetized adult pig induces acute respiratory failure, enabling controlled experimentation during acute respiratory decompensation.
Abstract
This protocol outlines an acute respiratory distress model utilizing centrally administered oleic acid infusion in Yorkshire pigs. Prior to experimentation, each pig underwent general anesthesia, endotracheal intubation, and mechanical ventilation, and was equipped with bilateral jugular vein central vascular access catheters. Oleic acid was administered through a dedicated pulmonary artery catheter at a rate of 0.2 mL/kg/h. The infusion lasted for 60-120 min, inducing respiratory distress. Throughout the experiment, various parameters including heart rate, respiratory rate, arterial blood pressure, central venous pressure, pulmonary artery pressure, pulmonary capillary wedge pressure, end-tidal carbon dioxide, peak airway pressures, and plateau pressures were monitored. Around the 60 min mark, decreases in partial arterial oxygen pressure (PaO2) and fraction of oxygen-saturated hemoglobin (SpO2) were observed. Periodic hemodynamic instability, accompanied by acute increases in pulmonary artery pressures, occurred during the infusion. Post-infusion, histological analysis of the lung parenchyma revealed changes indicative of parenchymal damage and acute disease processes, confirming the effectiveness of the model in simulating acute respiratory decompensation.
Introduction
The utilization of porcine models in translational research holds significant importance in advancing our understanding of human medicine1. Porcine models, due to their physiological and anatomical similarities to humans, provide a valuable platform for studying complex diseases and therapeutic interventions1. In the context of acute respiratory failure, porcine models offer a unique opportunity to investigate the pathophysiological mechanisms, evaluate treatment strategies, and assess potential interventions1,2,3. The ability to replicate key aspects of human respiratory physiology and responses to various stimuli in porcine models allows for a comprehensive evaluation of therapeutic modalities before progressing to human trials1,2,3. This research paradigm enables researchers to bridge the gap between preclinical investigations and clinical application, facilitating the development of novel therapies and improving patient outcomes1. Therefore, the establishment of an efficient, effective, and reproducible acute respiratory failure porcine model serves as a crucial tool in advancing the knowledge of respiratory diseases and guiding the development of effective interventions in human medicine1.
Respiratory distress, a critical medical condition, has witnessed limited advancements in its diagnosis and management over the past three decades4. The currently employed evaluation and triage metrics, which include subjective symptoms, physical examination findings, SpO2, and respiratory rate, often exhibit limitations in detecting acute pulmonary conditions at an early stage5,6,7. This inadequacy not only hampers efficient triage and resource allocation but also fails to provide effective, quantitative monitoring of disease progression and treatment response in patients with chronic pulmonary diseases. The emerging landscape of chronic respiratory conditions, such as long COVID, along with the burden of acute respiratory insufficiencies on hospital resources, underscores the urgent need to expand translational research and foster innovation in respiratory disease management.
The direct infusion of oleic acid into a pig's bloodstream has been recognized as a robust method to induce acute respiratory distress8. Oleic acid, a monounsaturated fatty acid, has demonstrated the ability to trigger significant pulmonary injury and compromise respiratory function when introduced into the pulmonary circulation8. Upon infusion, oleic acid provokes vasoconstriction, resulting in increased pulmonary arterial pressure and resistance, leading to impaired gas exchange and oxygenation9. Furthermore, oleic acid promotes the activation of inflammatory pathways, including the release of pro-inflammatory mediators and recruitment of immune cells, which contribute to the development of lung injury and respiratory distress10. All of this results in severe hypoxemia, increases in pulmonary arterial pressures, and the accumulation of extravascular lung water11. Histological evaluation of the lung parenchyma has demonstrated injury that is indistinguishable from human acute respiratory distress9.
This article details a method involving the direct administration of oleic acid into the pulmonary artery to induce acute respiratory distress, avoiding untreatable, severe hemodynamic compromise. The described method is anticipated to be a valuable tool for future researchers exploring the underlying pathophysiological mechanisms of acute respiratory failure and assessing potential therapeutic interventions and innovations.
Protocol
The protocol received approval from the Vanderbilt University Institutional Animal Care and Use Committee (protocol M1800176-00) and strictly adhered to the National Institute of Health Guidelines for the Care and Use of Laboratory Animals. Male and female Yorkshire pigs, weighing approximately 40-45 kg, were utilized in this experiment. The animals were obtained from a commercial source (see Table of Materials). The current practice does not involve screening for any pre-existing medical conditions in the acquired swine. While it is acknowledged that this practice could potentially interfere with or mask intended results, it is considered unlikely according to the vendor, and this limitation is accepted.
1. Animal preparation
- Anesthesia and ventilation
- Anesthetize the pig with intramuscular injection of ketamine (2.2 mg/kg) / xylazine (2.2 mg/kg) / telazol (4.4 mg/kg) and position the animal in a supine (lying down) posture on the operating table.
- Maintain general anesthesia by initiating inhalational anesthetic, 1% isoflurane.
- Expose the vocal cords through the mouth using a laryngoscope and intubate12 with a 6.5 mm endotracheal tube (see Table of Materials). Inflate the tube cuff with 3-5 mL of air using a syringe without a needle attached.
NOTE: Immediately perform carbon dioxide (CO2) capnography post-tracheal cannulation to ensure proper tube placement, and measure CO2 with ventilation indicating appropriate ventilation. - Use an 18 G to 24 G intravenous (IV) catheter placed in the central or marginal ear vein on the posterior side of the auricle to administer intraoperative (as needed) and euthanasia drugs to the pig. Secure the IV catheter with 1-inch adhesive tape.
- Set mechanical ventilation to volume-controlled ventilation settings with a tidal volume of 8 mL/kg.
NOTE: Anesthesia monitoring is conducted throughout the experiment. Vital signs, response to stimulus, presence/absence of movement, jaw tone laxity, changes in heart rate, end-tidal CO2, and respiratory rate variation are monitored by an independent animal lab technician. Adjustments to the inhaled anesthetic dose are made based on these assessments. Analgesic administration of buprenorphine via bolus is performed. Adjust the respiratory rate on the mechanical ventilator to maintain an end-tidal CO2 of 35-40 mmHg throughout the experiment.
- Cannulation and monitoring
- Disinfect the entire anterior neck with a 2% chlorhexidine scrub solution, followed by a spray of 5% providone-iodine solution.
- Surgically expose both the right and left internal and external jugular (IJ and EJ) veins and carotid arteries (CA) with a vertical incision, approximately 7-8 cm, immediately lateral to the trachea on either side using a No. 23 blade to the sternum13.
NOTE: A surgical cut-down approach is chosen for vascular access due to the challenges of a percutaneous, ultrasound-guided Seldinger technique14 in swine. The tough skin and vascular size make a cut-down approach more feasible. Cervical vessels are preferred for bilateral pulmonary artery catheters (PAC), though femoral access is an option. The choice which jugular vein (IJ or EJ) is at the discretion of procedurelist. Which ever has the larger diameter can be cannulated and used for heart catheterization. - Dissect the strap muscles and tract as needed using Kelly tissue scissors and Lahey retractors13 (see Table of Materials).
- After exposure, place two 8.5 Fr cannulas and two PACs using the Seldinger technique14.
NOTE: Right jugular vein catheter and PAC are dedicated for volume administration and hemodynamic monitoring. Left jugular vein catheter and corresponding PAC are used for oleic acid administration. Dual-PAC placement is confirmed using fluoroscopy. - Using the Seldinger technique, place an arterial line in the right CA for invasive blood pressure monitoring throughout the experiment.
- Attach all desired monitoring equipments. Monitor heart rate (HR) with telemetry leads. Monitor systolic blood pressure (SBP), diastolic blood pressure (DBP), and mean arterial pressure (MAP) by connecting a pressure transducer to the CA catheter. Monitor mean pulmonary artery pressure (MPAP) and central venous pressure (CVP) using an independent pressure transducer/amplifier system set-up.
NOTE: Calculate pulse pressure as the difference between SBP and DBP, and pulse pressure variability (PPV) by calculating the difference between peak pulse pressure at inspiration and expiration during the respiratory cycle. Perform thermodilution cardiac output (CO) using device-specific volume temperature/volume calibration. To obtain a pulmonary capillary wedge pressure (PCWP), inflate the PAC balloon with 1.5 mL of air, advance the catheter until visualization of both "v" and "a" waves, representing restricted right-to-left blood flow, and record the PCWP as the pressure value of the "a" wave at end expiration16. - To monitor urine output, place a Foley catheter (see Table of Materials) in the pig's urethra. For male pigs, surgical suprapubic catheterization is required17.
- Administer crystalloids (PlasmaLyte, see Table of Materials) at a rate of 100 mL over 10 min to achieve a PCWP of 8-12 mmHg (euvolemia) before initiating oleic acid. Check PCWP every 100 mL until 10 mmHg is achieved.
2. Oleic acid infusion
- Oleic acid preparation
- Prepare the oleic acid solution (16%) by combining 16 mL of oleic acid with 84 mL of normal saline.
NOTE: Handle oleic acid with care, ensuring personnel wear protective gloves, safety goggles, and masks to prevent direct skin contact, inhalation, or eye exposure. Frequent agitation is necessary to prevent separation, and dimethyl sulfoxide may be added if separation occurs. - Prime the oleic acid solution into an IV fluid line and connect it to the distal port of the left jugular pulmonary artery catheter (see Table of Materials).
- Prepare the oleic acid solution (16%) by combining 16 mL of oleic acid with 84 mL of normal saline.
- Oleic acid initiation
- Start the oleic acid infusion at a rate of 0.2 mL/kg/h, marking the official start time18,19,20(Figure 1).
NOTE: Confirm hemodynamic monitoring and catheter placement before starting the oleic acid infusion. Techniques such as fluoroscopy or transthoracic echocardiography can ensure proper placement21,22. - Immediately after starting oleic acid, set ventilator settings to mimic room air conditions (FiO2 = 21%, PEEP = 0 cm H2O).
- Start the oleic acid infusion at a rate of 0.2 mL/kg/h, marking the official start time18,19,20(Figure 1).
- Hemodynamic and respiratory monitoring during oleic acid infusion
- Continuously monitor heart rate (HR), fraction of oxygen-saturated hemoglobin (SpO2), respiratory rate (RR), end-tidal carbon dioxide (ETCO2), central venous pressure (CVP), systolic blood pressure (SBP), diastolic blood pressure (DBP), mean arterial pressure (MAP), pulse pressure variability (PPV), and mean pulmonary artery pressure (MPAP).
- Measure partial arterial oxygen pressure (PaO2), pH, lactate, base excess, cardiac output (CO), and pulmonary capillary wedge pressure (PCWP) every 30 min over the first 60-90 min and then every 15 min thereafter until sacrifice (Figure 1). Record peak airway pressure and plateau pressure at the same time PaO2 is measured.
NOTE: Typical starting values for continuously monitored variables are 95%-100%, 15-20 breaths per minute, 25-35 mmHg, 70-80 mmHg, 40-50 mmHg, 55-65 mmHg, 1%-4%, and 10-20 mmHg, respectively. Starting values for intermittent variables are 10 mmHg, 7.4, 0-2 mg/dL, -2 +2 mEq/L, >5 L/min, and 8-10 mmHg, respectively.
- Measure partial arterial oxygen pressure (PaO2), pH, lactate, base excess, cardiac output (CO), and pulmonary capillary wedge pressure (PCWP) every 30 min over the first 60-90 min and then every 15 min thereafter until sacrifice (Figure 1). Record peak airway pressure and plateau pressure at the same time PaO2 is measured.
- Consider the experiment complete once arterial PaO2/fraction of inspired O2 (P/F) is less than <10023.
NOTE: At this point, animal can be euthanized (see below), and pulmonary pathology samples can be obtained, if needed (Figure 2).
- Continuously monitor heart rate (HR), fraction of oxygen-saturated hemoglobin (SpO2), respiratory rate (RR), end-tidal carbon dioxide (ETCO2), central venous pressure (CVP), systolic blood pressure (SBP), diastolic blood pressure (DBP), mean arterial pressure (MAP), pulse pressure variability (PPV), and mean pulmonary artery pressure (MPAP).
3. Venous waveform analysis and ventilator management procedure
- Respiratory non-Invasive Venous waveform Analysis
- Respiratory Index (RIVA-RI)
NOTE: Our research team employs this model to investigate changes in venous waveforms during respiratory distress. Peripheral venous waveforms are noninvasively captured at the upper arm of a pig using a piezoelectric sensor (see Table of Materials). Signal processing and amplification are required for analysis of these low-amplitude waveforms. Fourier transformation is then applied to present the data in the frequency domain, revealing a low-amplitude waveform at approximately 0.2 Hz (termed "fR0") corresponding to respiration. This hypothesis suggests that this wave results from retrograde propagation of negative intrathoracic pressure during inspiration from the right atrium/vena cava throughout the venous system. The weighted contributions of the amplitudes of the respiratory signal (fR0) can be ratiometrically normalized to compare data on a common scale and improve performance, and to the amplitude of the frequency of the pulse rate (f0) to produce a RIVA-RI7.- Place the piezoelectrode on the anterior upper extremity venous plexus immediately proximal to the elbow.
NOTE: Ensure recording and uploading abilities with the piezoelectrode device. Examples of previous recording prototypes can be found in referenced literature7,24,25,26,27. - Start recording venous waveforms with LabChart software (see Table of Materials) whenever venous waveforms are desired during experimentation.
- Place the piezoelectrode on the anterior upper extremity venous plexus immediately proximal to the elbow.
- Euthanasia
- Confirm the maintenance of isoflurane at 1%.
- Induce cardiac arrest by IV injection of sodium pentobarbital (125 mg/kg) (following institutionally approved protocols).
- Confirm the lack of vitals post-injection to verify demise.
Representative Results
Early single pig, pilot data demonstrates an increase in RIVA-RI prior to changes in other respiratory monitoring measures (RR and SpO2), in line with changes in PaO2 (Figure 3). The drop in PaO2 is the "positive" result this model intends to achieve. Preliminary data also shows that RIVA-RI increases and the PaO2 decreases with disease progression starting at the 30-min mark (Figure 3; red arrow). PaO2 is the hemodynamic parameter of most interest in the setting of monitoring the respiratory condition secondary to the fact it reflects the balance between oxygen delivery and consumption no matter the clinical state of a patient28,29. It also drives the diagnosis of acute respiratory distress and the implementation of treatments30. Both values changed well before decreases in SpO2 in the porcine model, which was delayed to the 60-min mark31 (Figure 3). The preliminary data also supports the previously reported limitation in the singular use of SpO2 as the clinical indicator of pulmonary disease severity29,32,33,34. These data support that this described model is successful in achieving an acute respiratory failure using oleic acid delivery directly through a PAC.
In addition to successfully achieving acute respiratory failure using gas exchange definitions, histological evaluation of porcine lung tissue5,6 (formalin-fixed, paraffin-embedded and stained with hematoxylin and eosin) after successful achievement of respiratory distress demonstrated changes consistent with human acute respiratory distress (Figure 2). This supports previous literature of oleic acid use for acute respiratory distress9.
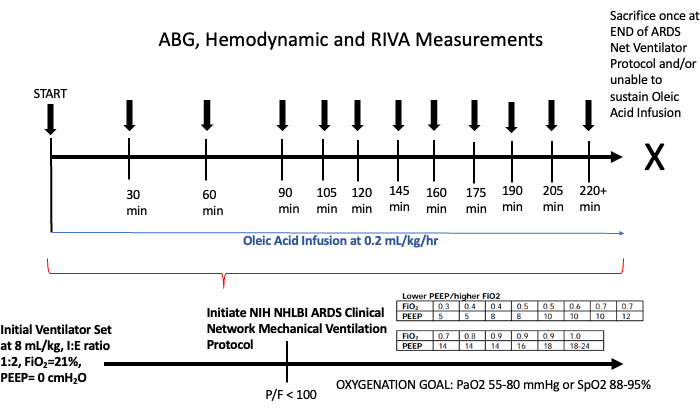
Figure 1: Timeline of a porcine acute respiratory distress study in Yorkshire pigs. RIVA-RI values, hemodynamic data (heart rate, respiratory rate, blood pressure, SpO2, pulmonary artery pressures, pulmonary capillary wedge pressure, central venous pressure, pleural pressure, plateau pressure, and peak pressure), and arterial blood gas analysis (PaO2, PaCO2, Lactate, Base Excess, and pH) were obtained every 30 min for the first 1.5 h and then every 15 min thereafter until sacrifice. The NHLBI ARDS Clinical Network Mechanical Ventilation Protocol will be initiated when the P/F ratio is <100. Ventilator adjustments will follow this protocol with goal oxygenation of PaO2 55-80 mmHg or SpO2 88%-95%. A minimum of 2 min for venous waveform capture will occur prior to any escalation of FiO2 and/or PEEP. Abbreviations: ABG = arterial blood gas; RIVA = Respiratory non-Invasive Venous waveform Analysis; min = minute; PaO2 = partial pressure of oxygen; PaCO2 = partial pressure of carbon dioxide; mL = millimeter; kg = kilogram; h = hour; FiO2 = fraction of inspired oxygen; PEEP = positive end-expiratory pressure. Please click here to view a larger version of this figure.
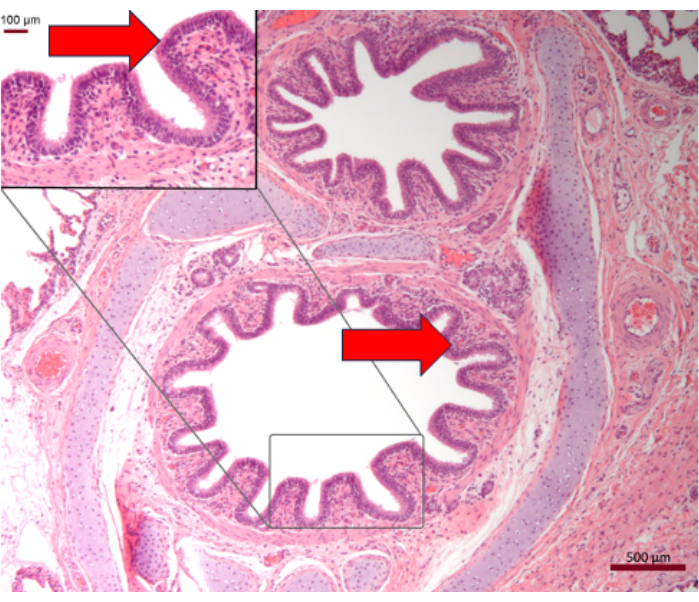
Figure 2: Histology of porcine lung post oleic acid infusion with PaO2/FiO2 <100. Histological changes of porcine lung tissue validates this oleic acid model with demonstration of alveolar expanded alveolar septa by myxoid-appearing fibrous tissue, pneumocyte hyperplasia, and lymphocytic infiltration (red arrows)-classical findings of acute respiratory distress. Scale bar = 500 µm; magnified part = 100 µm. Please click here to view a larger version of this figure.
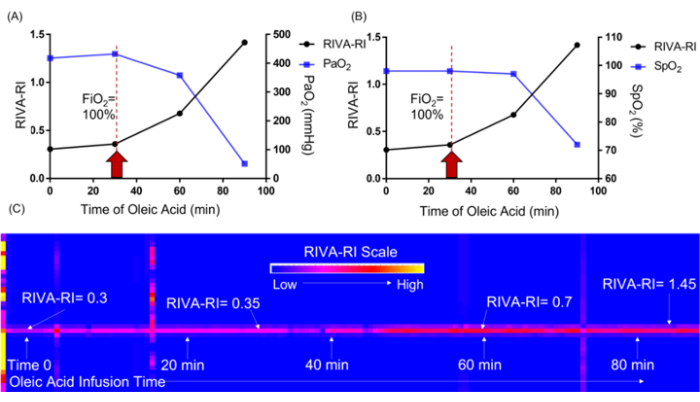
Figure 3: Graphical relationship between RIVA-RI and PaO2 and SpO2 over time of oleic acid infusion in a porcine model of one pig. As acute respiratory distress is induced through the direct infusion of oleic acid into the pulmonary artery, RIVA-RI increases as PaO2 (A) and SpO2 (B) decrease. When RIVA-RI was visualized in the color spectrum (C) over the entire course of oleic acid infusion, an increase in the RIVA-RI value was observed after 40 min of infusion and increased throughout the development of respiratory distress. Red arrows indicate the point at 30 min where RIVA-RI and PaO2 started to change, but SpO2 remained the same. Abbreviations = RIVA-RI = Respiratory non-Invasive Venous waveform Analysis Respiratory Index, PaO2 = partial pressure of oxygen, SpO2 = oxygen hemoglobin saturation, min= minutes. Please click here to view a larger version of this figure.
Discussion
The key element in this protocol is to closely monitor the hemodynamic condition of the pig during the administration of oleic acid to induce respiratory distress15. It is of the utmost importance for researchers to take the necessary time to appropriately position the hemodynamic monitoring devices. One specific drawback of this model is the potential hemodynamic instability that may arise as a result of inflammation and injury to the respiratory vasculature8,10,18. Although the direct administration of oleic acid into the pulmonary artery leads to less instability than its administration at the jugular/atrial junction, the possibility of such instability should still be acknowledged. Typically, around 45-60 min after the initiation of the procedure, a significant increase in MPAP occurs. Therefore, it may be necessary to provide hemodynamic support in order to achieve respiratory failure in pigs. Normally, administering 50-70 mcg of norepinephrine and/or 10-30 mcg of epinephrine for SBP below 60 mmHg and/or HR below 50 bpm will rectify hemodynamic deterioration and allow for the continuation of the experiment. Development of hemodynamic instability has been identified as the primary limitation of this model.
Another limitation of this model is the duration required for the experiment. Based on previous experience, it typically takes an average of 60 min for the early signs of respiratory distress to manifest. In most cases, the PaO2 continues to decline; however, there have been isolated instances in which some form of presumed adaptation occurs, leading to a temporary rebound in PaO2. To date, this rebound has not persisted for longer than 30 min during ongoing oleic acid infusion. It is important to appreciate that the use of oleic acid is directly toxic to the endothelial cells, and this injury is followed by an increased pulmonary microvascular permeability and intrapulmonary shunt11. For this reason, it is believed a more direct administration leads to a more stable and isolated pulmonary injury and acute respiratory failure – the goal of this protocol.
This method was conceived based on the fusion of multiple reports using oleic acid to induce porcine respiratory distress. Boker et. al. used oleic acid-induced lung injury to investigate changes in PaO2, lung compliance, and proinflammatory cytokines for comparing mechanical ventilation using ARDSnet low tidal volumes20. They used a continuous infusion of oleic acid but defined respiratory failure as a PaO2 less than or equal to 65 mmHg for two consecutive measurements, 5 min apart20. They supported their hemodynamics with a continuous dopamine infusion (5-12 μg/kg/min), titrated to control the MAP greater than 50 mmHg20. Mutch et. al.19 also investigated mechanical ventilation strategies after acute respiratory distress using an oleic acid model. They placed a 5 Fr catheter into the femoral vein but their catheter ended within the right atria and oleic acid was administered at a rate of 0.2 mL/kg/h19. Dopamine was also used for hemodynamic support with MAP goals of 60 mmHg19. Respiratory failure was defined as a PaO2 equal to or less than 60 mmHg for two consecutive measurements, 5 min apart19. Multiple differences exist between the model described herein and these two prior examples of oleic acid use. First, there was an obvious difference in the maintenance of hemodynamic control. Prophylactic vasoactive infusion was not desired, and a more reactive administration at the first sign of hemodynamic distress was chosen for this protocol to spare the pig of vasoactive medications if not required. A second difference was observed in oleic acid delivery. They chose to administer them more systemically through a 5 Fr single-lumen catheter that ended approximately 1-2 cm above the diaphragm through the femoral vein and more hemodynamic instability with systemic administration has been observed. In contrast, it is significantly less when administered directly into the pulmonary artery through a dedicated pulmonary artery catheter.
Another method of inducing acute lung injury in swine was described by Rissel et. al.11, where they performed both bronchoalveolar lavage and an injection of oleic acid. Their double-hit method clearly causes severe lung injury by mimicking the two central elements of the pathomechanism of Acute Respiratory Distress Syndrome (ARDS)11 and showed similar PaO2/FiO2 at 1 h mark11. However, their described goal is to cause severe lung injury in pigs that is suitable to study different treatment options in ARDS11. The goal of the described protocol is to create isolated acute respiratory failure; therefore, the addition of another step to ensure surfactant depletion as well as alveolar collapse has not been found to be necessary and has not been performed to date in this described model.
In light of the necessity for innovation and research in the field of respiratory diseases, the establishment of an efficient, reliable, and reproducible animal model is of paramount significance. The use of oleic acid to induce acute respiratory failure has been extensively studied8,10,11,18,19,20. The present report provides a detailed account of cannulation, hemodynamic monitoring, and oleic acid administration strategies employed to induce acute respiratory failure in adult pigs. Together with existing literature, this comprehensive model description aims to facilitate the investigation of pivotal hypotheses within the field by future scientists.
Disclosures
The authors have nothing to disclose.
Acknowledgements
The authors would like to thank Dr. José A. Diaz, Jamie Adcock, Mary Susan Fultz and the S.R. Light Laboratory at Vanderbilt University Medical Center for their assistance and support. This work was supported by a grant from the National Heart, Lung, and Blood Institute of the National Institutes of Health (BA; R01HL148244). The content is the sole responsibility of the authors and does not necessarily represent the official views of the National Institutes of Health.
Materials
| 1% Isoflurane | Primal, Boston, MA, USA | 26675-46-7 | https://www.sigmaaldrich.com/US/en/product/aldrich/792632?gclid=Cj0KCQjw9fqnBhDSARIsAHl cQYS_W-q6tS2s6LQw2Qn7Roa3T GIpTLPf52351vrhgp44foEcRozPqt YaAtvfEALw_wcB |
| Arterial Catheter | Merit Medical, South Jordan, UT, USA | MAK401 | MAK Mini Access Kit 4F |
| Blood Pressure Amp | AD Instruments, Colorado Springs, CO, USA | FE117 | https://www.adinstruments.com/products/bp-blood-pressure-amp |
| Central Venous Catheter | Arrow International, Cleveland, OH, USA | AK-09800 | 8.5 Fr. x 4" (10 cm) Arrow-Flex |
| Disposable Pressure Transducers | AD Instruments, Colorado Springs, CO, USA | MLT0670 | https://www.adinstruments.com/products/disposable-bp-transducers |
| Edwards Lifesciences Triple Stage Venous Cannulas | Edwards Life Sciences, Irvine, CA | TF293702 | https://www.graylinemedical.com/products/edwards-lifesciences-triple-stage-venous-cannulas-venous-dual-stage-cannula-tf293702?variant=31851942576185&gad=1& gclid=Cj0KCQiAr8eqBhD3ARIsAIe -buNdmkzavUBaIx-1be7boWn2kW hbUR6QCjaobB08uuK9qJW66JvY TM4aAufGEALw_wcB |
| Kelly Scissors | MPM Medical Supply, Freehold, NJ 07728 | 104-5516 | https://www.mpmmedicalsupply.com/products/kelly-scissors |
| Kendall 930 FoamElectrodes | Covidien, Mansfield, MA, USA | 22935 | https://www.cardinalhealth.com/en/product-solutions/medical/patient-monitoring/electrocardiography/monitoring-ecg-electrodes/radiolucent-electrodes/kendall-930-series-radiolucent-foam-electrodes.html |
| Ketamine Hydrochloride 100 mg/mL, Injectable Solution, 10 mL | Patterson Veterinary, Loveland, CO 80538 | 07-894-8462 | https://www.pattersonvet.com/ProductItem/078948462?omni=ketamine |
| LabChart 8 software | AD Instruments, Colorado Springs, CO, USA | N/A | https://www.adinstruments.com/products/labchart |
| Lahey Retractor | BOSS Instruments LTD, Gordonsville, VA 22942 | 18-1210 | https://bossinstruments.com/product/7-3-4-lahey-thyroid-retractor-6mmx28mm/ |
| Oleic Acid | Sigma-Aldrich, Merck, Darmstadt, Germany | O1008 | https://www.sigmaaldrich.com/US/en/product/sial/o1008?gclid=CjwKCAjwzJmlBhBBEiwAEJy Lu2047wRpXqF_Z2BegUyhgZJ _WygsWfErhgrGCIyMp8PxwNH sTZ8qARoCl1QQAvD_BwE&gcl src=aw.ds |
| Peripheral IV Catheter Angiocath 18-24 G 1.16 inch | McKesson, Irving, TX, USA | 329830 | https://mms.mckesson.com/product/329830/Becton-Dickinson-381144 |
| Piezoelectrode | MuRata Manuractoring Co, Ltd., Nagaokakyo, Kyoto, Japan | 7BB-12-9 | https://www.murata.com/en-us/products/productdetail?partno=7BB-12-9 |
| PlasmaLyte | Baxter International, Deerfield, IL, USA | 2B2544X | https://www.ciamedical.com/baxter-2b2544x-each-solution-plasma-lyte-a-inj-ph-7-4-1000ml |
| Pulmonary Artery Catheter | Edwards Life Sciences, Irvine, CA | 131F7 | Swan Ganz 7F x 110cm |
| Standard Endotracheal Tubes | Teleflex, Morrisville, NC 27560 | 5-10313 | https://www.teleflex.com/usa/en/product-areas/anesthesia/airway-management/endotracheal-tubes/standard-tubes/index.html |
| SurgiVet Clearview Foley Catheter, 8 Fr, 55 cm Silicone | Penn Veterinary Supply, Inc, West Rendering, PN 13971 | SVCFC1030 | https://www.pennvet.com/customer/portal/catalog/home?urile=wcm:path%3APennVet+Catalog/Super+Sku+Catalog/SS0672/Surgivet+Clearview+Silicone+Foley+Catheters |
| Telazol (Tiletamine HCl and Zolazepam HCl), Injectable Solution, 5 mL | Patterson Veterinary, Loveland, CO 80538 | 07-801-4969 | https://www.pattersonvet.com/ProductItem/078014969?omni=telazol |
| Welch Allyn E-MacIntosh Standard Laryngoscope Blade | MFIMedical, San Diego, CA 92131 | WLA-69242 | https://mfimedical.com/products/welch-allyn-e-macintosh-standard-laryngoscope-blade?variant=12965771870285¤cy =USD&utm_medium=product_sync &utm_source=google&utm_content =sag_organic&utm_campaign=sag _organic&gclid=Cj0KCQiAr8eqBhD 3ARIsAIe-buMhpgM96qRXkCUKA 6Mhmdat0p93JbecCGTaLStexhV pkUVa9VkWUzgaAr-iEALw_wcB |
| Xylazine HCl 100 mg/mL, Injectable Solution, 50 mL | Patterson Veterinary, Loveland, CO 80538 | 07-894-5244 | https://www.pattersonvet.com/ProductItem/078945244 |
| Yorkshire Pigs | Oak Hill Genetics, Ewing, IL, USA | 138274 | Female/Male Swine- Yorkshire/Landrace 81-100lbs |
References
- Judge, E. P., et al. Anatomy and bronchoscopy of the porcine lung. A model for translational respiratory medicine. Am J Respir Cell Mol Biol. 51 (3), 334-343 (2014).
- Anna Bassols, C. P., et al. The pig as an animal model for human pathologies: A proteomics perspective. Proteomics Clin. Appl. 8, 715-731 (2014).
- Hughes, G. C., Post, M. J., Simons, M., Annex, B. H. Translational physiology: porcine models of human coronary artery disease: implications for preclinical trials of therapeutic angiogenesis. J Appl Physiol. 94 (5), 1689-1701 (2003).
- Tobin, M. J. Respiratory monitoring. JAMA. 264 (2), 244-251 (1990).
- Magnusdottir, S. O., et al. Hyperoxia affects the lung tissue: A porcine histopathological and metabolite study using five hours of apneic oxygenation. Metabol Open. 4, 100018 (2019).
- Matute-Bello, G., Frevert, C. W., Martin, T. R. Animal models of acute lung injury. Am J Physiol Lung Cell Mol Physiol. 295 (3), 379-399 (2008).
- Alvis, B., et al. Respiratory non-invasive venous waveform analysis for assessment of respiratory distress in coronavirus disease 2019 patients: an observational study. Crit Care Explor. 3 (10), 0539 (2021).
- Hultkvist-Bengtsson, U., Martensson, L. Oleic acid-induced injuries in the guinea-pig. Effects of allopurinol on cell dynamics, erythrocyte-catalase and uric acid plasma levels. Clin Exp Pharmacol Physiol. 18 (3), 127-130 (1991).
- Jacobs, B. R., Brilli, R. J., Ballard, E. T., Passerini, D. J., Smith, D. J. Aerosolized soluble nitric oxide donor improves oxygenation and pulmonary hypertension in acute lung injury. Am J Respir Crit Care Med. 158 (5 Pt 1), 1536-1542 (1998).
- Lee, S. M., et al. microRNAs mediate oleic acid-induced acute lung injury in rats using an alternative injury mechanism. Mol Med Rep. 10 (1), 292-300 (2014).
- Rissel, R., et al. Bronchoalveolar lavage and oleic acid-injection in pigs as a double-hit model for acute respiratory distress syndrome (ARDS). J Vis Exp. 159, 61358 (2020).
- Helen Chum, C. P. Endotracheal intubation in swine. Lab Animal. 41, 309 (2012).
- Alessa, M. A., et al. Porcine as a training module for head and neck microvascular reconstruction. J Vis Exp. 139, 58104 (2018).
- Higgs, Z. C., Macafee, D. A., Braithwaite, B. D., Maxwell-Armstrong, C. A. The Seldinger technique: 50 years on. Lancet. 366 (9494), 1407-1409 (2005).
- Wise, E. S., et al. Hemodynamic parameters in the assessment of fluid status in a porcine hemorrhage and resuscitation model. Anesthesiology. 134 (4), 607-616 (2021).
- Riedinger, M. S., Shellock, F. G., Swan, H. J. Reading pulmonary artery and pulmonary capillary wedge pressure waveforms with respiratory variations. Heart Lung. 10 (4), 675-678 (1981).
- Holliman, C. J., Kenfield, K., Nutter, E., Saffle, J. R., Warden, G. D. Technique for acute suprapubic catheterization of urinary bladder in the pig. Am J Vet Res. 43 (6), 1056-1057 (1982).
- Borges, A. M., et al. Challenges and perspectives in porcine model of acute lung injury using oleic acid. Pulm Pharmacol Ther. 59, 101837 (2019).
- Mutch, W. A., et al. Biologically variable ventilation increases arterial oxygenation over that seen with positive end-expiratory pressure alone in a porcine model of acute respiratory distress syndrome. Crit Care Med. 28 (7), 2457-2464 (2000).
- Boker, A., et al. Improved arterial oxygenation with biologically variable or fractal ventilation using low tidal volumes in a porcine model of acute respiratory distress syndrome. Am J Respir Crit Care Med. 165 (4), 456-462 (2002).
- Josan, E., Pastis, N., Shaman, Z. Ultrasound guided pulmonary artery catheter insertion: An alternative to fluoroscopic guidance. Respir Med Case Rep. 38, 101678 (2022).
- Weinberg, L., et al. Video fluoroscopy for positioning of pulmonary artery catheters in patients undergoing cardiac surgery. J Cardiothorac Vasc Anesth. 29 (6), 1511-1516 (2015).
- NHLBI N. . Clinical network mechanical ventilation protocol summary. , (2023).
- Alvis, B., et al. Noninvasive Venous waveform analysis correlates with pulmonary capillary wedge pressure and predicts 30-day admission in patients with heart failure undergoing right heart catheterization. J Card Fail. 28 (12), 1692-1702 (2022).
- Alvis, B. D., et al. Non-invasive venous waveform analysis (NIVA) for monitoring blood loss in human blood donors and validation in a porcine hemorrhage model. J Clin Anesth. 61, 109664 (2020).
- Hocking, K. M., et al. Evaluation of common clinical and hemodynamic parameters to pulmonary capillary wedge pressures in patients undergoing right heart catheterization. Medical Research Archives: European Society of Medicine. 10, (2022).
- Sobey, J. H., et al. Non-invasive venous waveform analysis (NIVA) for volume assessment during complex cranial vault reconstruction: A proof-of-concept study in children. PLOS One. 15 (7), 0235933 (2020).
- Carreau, A., El Hafny-Rahbi, B., Matejuk, A., Grillon, C., Kieda, C. Why is the partial oxygen pressure of human tissues a crucial parameter? Small molecules and hypoxia. J Cell Mol Med. 15 (6), 1239-1253 (2011).
- Rogers, K. M., McCutcheon, K. Four steps to interpreting arterial blood gases. J Perioper Pract. 25 (3), 46-52 (2015).
- Brown, S. M., et al. Nonlinear Imputation of PaO2/FiO2 from SpO2/FiO2 among patients with acute respiratory distress syndrome. Chest. 150 (2), 307-313 (2016).
- Sileshi, B., et al. Peripheral venous waveform analysis for detecting early hemorrhage: a pilot study. Intensive Care Med. 41 (6), 1147-1148 (2015).
- Eecen, C. M. W., Kooter, A. J. J. Pulse oximetry: principles, limitations and practical applications. Ned Tijdschr Geneeskd. 165, 5891 (2021).
- Sinex, J. E. Pulse oximetry: principles and limitations. Am J Emerg Med. 17 (1), 59-67 (1999).
- Chan, E. D., Chan, M. M., Chan, M. M. Pulse oximetry: understanding its basic principles facilitates appreciation of its limitations. Respir Med. 107 (6), 789-799 (2013).

.
