Quantification of γH2AX Foci in Response to Ionising Radiation
Summary
Quantification of DNA double-strand streaks using γH2AX formation as a molecular marker has become an invaluable tool in radiation biology. Here we demonstrate the use of an immunofluorescence assay for quantification of γH2AX foci after exposure of cells to radiation.
Abstract
Protocol
Cell Preparation
- Human keratinocytes (FEP-1811) were grown in Keratinocyte-Serum Free Medium (K-SFM; Invitrogen) supplemented with epidermal growth factor, bovine pituitary extract and 20 μg/ml gentamicin, at 37°C and 5% CO2.
- A single cell suspension was prepared by detaching with trypsin-EDTA (0.05% v/v)
- Cells were seeded in 8-well Lab Tek II microchamber slides (10,000 cells/well) and slides were incubated for 3 days at 37°C and 5% CO2.
Irradiation
- Cells were irradiated on ice with 2 Gy using a 137Cs source (Gammacell 1000 Elite irradiator; Nordion International, ON, Canada; 20.6 seconds/Gy)
- Unirradiated control and 2 Gy irradiated cells were incubated for 1 hour at 37°C and 5% CO2.
Immunofluorescence staining
- Media was tipped off and cells were washed with 300μl of PBS (w/o Ca2+ or Mg2+) per well and were rotated on an orbital mixer for 5 minutes.
- The buffer was tipped off and 100μl of freshly prepared 4% (v/v) paraformaldehyde was added to each well and slides were incubated at room temperature for 10 minutes.
All incubations were performed in a humidified staining trough - Cells were then washed with PBS (w/o Ca2+ or Mg2+). Slides were placed in a Coplin jar and rotated on an orbital mixer for 5 minutes. This wash step was repeated a further two times.
- The buffer was tipped off and excess PBS was gently blotted.
- Cells were permeabilized using 100ml Triton X-100 (0.1% v/v) per well and a 10 minute incubation at room temperature.
- Cells were washed with PBS (w/o Ca2+ or Mg2+) as described in steps 3 and 4 above.
- Non-specific protein binding was blocked with 100ml of BSA (1% v/v) per well and 20 minute incubation at room temperature.
- Excess BSA was tipped off and 100μl of primary mouse monoclonal anti-phospho histone-H2AX antibody (diluted 1:500, in 1% BSA; Millipore), was added to each well for a 1 hour incubation at room temperature.
- Cells were washed with PBS (w/o Ca2+ or Mg2+) as described in steps 3 and 4 above and incubated with 100μl of secondary antibody (Alexa Fluor 488 goat anti-mouse IgG diluted 1:500, in 1% BSA; Invitrogen) per well for 45 minutes at room temperature in the dark. (Diluted antibody was kept in the dark throughout the procedure)
- Cells were washed with PBS (w/o Ca2+ or Mg2+) as described in steps 3 and 4 above. However, exposure to light was minimized using foil.
- Nuclear counterstaining was performed with 100ml TOPRO3 (diluted 1:500; Invitrogen) per well and a 10 minute incubation at room temperature
- Cells were washed with PBS (w/o Ca2+ or Mg2+) as described in step 10 above.
- The chambers were carefully removed from the slides, excess moisture was blotted and slides were allowed to air dry.
- One drop of ProLong GOLD anti-fade solution (Invitrogen) was added per well and slides were mounted (22×50 mm coverslip) and any excess liquid around the edges of slide was blotted.
- The slides were kept in the dark for a further 30 minutes at room temperature before sealing with nail polish.
- The slides were stored overnight at 4°C in the dark before analysis.
Microscopy / Analysis
- Zeiss LSM510 Meta Confocal Microscpe used to acquire images using the standard GFP (for γH2AX – Alexa Fluor 488 goat anti-mouse IgG) and far red lasers (for TOPRO-3). Typically, a 63 x oil immersion objective lens is used.
Images are acquired in a Z-series pattern with a step size of 0.5 μm. A step size of 0.5 μm was chosen to minimize loss of foci present in different planes in the nuclei. During analysis, individual planes are deconvoluted and stacked to produce a maximum projected image to minimize the overlap of foci (Top-hat filter applied). - Metamorph (Molecular Devices, USA) was used to analyse number of foci.
The program quantitates the number of foci in each cell after the threshold has been applied to exclude background. The information is logged in a Microsoft Excel spreadsheet for further analysis.
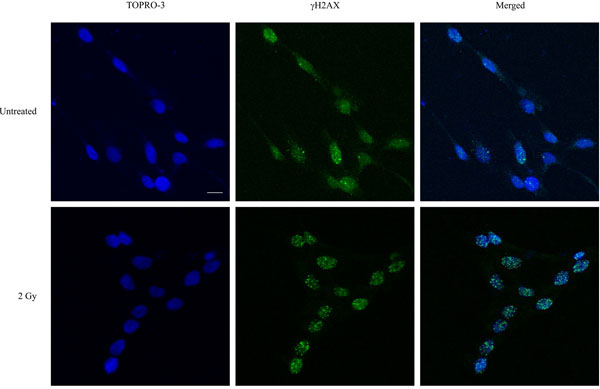
Figure 1. Immunofluorescence visualization of γH2AX foci (green) in untreated human keratinocytes and in cells irradiated with 2 Gy and incubated for a further 1 hour at 37°C, 5% CO2. DNA was stained with TOPRO-3 (blue). Images were acquired using a Zeiss LSM 510 Meta Confocal microscope. Bar = 10 μm.
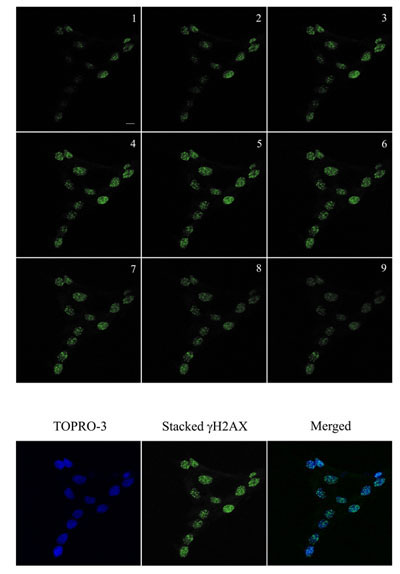
Figure 2. Immunofluorescence visualization of γH2AX foci (green) in human keratinocytes and in cells irradiated with 2 Gy and incubated for a further 1 hour at 37°C, 5% CO2. Images were acquired using a Zeiss LSM 510 Meta Confocal microscope using 0.5 μm Z-sectioning (1-9) to ensure all foci were acquired. The images was then stacked for quantitation using Metamorph. DNA was stained with TOPRO-3 (blue). The stacked γH2AX and blue images were stacked for visualization. Bar = 10 μm.
Discussion
Following exposure to ionising radiation (γ-rays), γH2AX foci form rapidly and foci numbers reach a maximum between 30-60 minutes2. Therefore, our 1 hour post-irradiation time point reflects initial DSB formation. We have used the clinically relevant radiation dose of 2 Gy for our experiment. However, the method can be used for radiation doses up to 4 Gy for detection of initial DSB formation; significant overlap of foci precludes accurate quantitation at higher doses. Higher radiation doses may be used for longer post-irradiation incubation times, as γH2AX foci are lost due to repair, resulting in quantifiable numbers. Typically, 4 hour and up to 24 hour post-irradiation incubation times are used for monitoring DNA repair. We have used the DNA stain TOPRO-3 due to limitations in the excitation lasers of our confocal microscope. More commonly, 4,6-diamidino-2-phenylindole dihydrochloride (DAPI) is used for nuclear counterstaining. Although we used a confocal microscope, epifluorescent microscopes with z-sectioning capacity are adequate. The immunofluorescence method is suitable for other adherent cancer and normal cell lines; we have tested T98G human glioblastoma and normal human endothelial cells and rat H9c2 embryonic ventricular myocytes.
Finally, quantitation of γH2AX is useful in the context of ionising radiation-induced DNA damage, for monitoring DNA damage – as illustrated by our experiment – and repair (radiation sensitivity). The assay is also useful for evaluating the efficacy of compounds that modulate cellular responses to radiation (i.e. radiation protectors and sensitizers). Further, γH2AX is emerging as a potential molecular marker in aging and disease, predominantly in cancer10.
Disclosures
The authors have nothing to disclose.
Acknowledgements
The support of the Australian Institute of Nuclear Science and Engineering is acknowledged. TCK was the recipient of AINSE awards. Epigenomic Medicine Lab is supported by the National Health and Medical Research Council of Australia (566559). LM is supported by Melbourne Research (University of Melbourne) and Biomedical Imaging CRC supplementary scholarships. The support of Monash Micro Imaging (Drs Stephen Cody and Iśka Carmichael) was invaluable for this work.
Materials
| Material Name | Type | Company | Catalogue Number | Comment |
|---|---|---|---|---|
| Keratinocyte-Serum Free Medium (K-SFM) | Media | Invitrogen | 17005042 | Keratinocyte media supplemented with human recombinant Epidermal Growth Factor 1-53 (EGF 1-53), and Bovine Pituitary Extract (BPE). |
| Lab Tek lI Chamber Slides (8-well) | Chamber Slides | Nunc Denmark | NUN154534 | |
| Coverslips (22x50mm) | Coverslips | Menzel-Gläser | CS2250100 | |
| Bovine Serum Albumin (BSA) | Sigma-Aldrich | A7906 | BSA (1%) is used to block any non-specific antibody binding. Primary and secondary antibodies are diluted in BSA. | |
| PBS (without Ca2+ and Mg2+) | Invitrogen | 17-517Q | ||
| 0.5% Trypsin-EDTA x10 | Invitrogen | 15400-054 | Trypsin-EDTA (0.05%) used to detach cells from culture flasks. | |
| Triton X-100 | Reagent | Sigma-Aldrich | T8787 | Triton X-100 (0.1%) used to permeabilise cells. |
| Paraformaldehyde | Reagent | Sigma-Aldrich | 158127 | Paraformaldehyde (4%) used to fix cells. |
| Mouse monoclonal anti-phospho histone-H2AX antibody | Primary Antibody | Millipore | 16193 | Dilution of primary antibody (1:500), in 1% BSA. |
| Alexa Fluor 488 goat anti-mouse IgG (H+L) | Secondary Antibody | Invitrogen | 11029 | Dilution of secondary antibody (1:500), in 1% BSA. |
| TOPRO3 | DNA Stain | Invitrogen | T3605 | DNA stain commonly used: 4,6-diamidino-2-phenylindole dihydrochloride (DAPI). Can only be used with microscopes with the appropriate excitation laser. |
| ProLong Gold | Anti-fade solution | Invitrogen | P36930 | Refractive index of 1.42 at 20°C. |
| Tissue Culture Flask, Vented Cap | Culture Flask | BD Falcon | 353112 | |
| Tissue Culture Dish (150x25mm) | Petridish | BD Falcon | 353025 | |
| Coplin Jar, glass | Grale Scientific P/L | 1771-OG | ||
| Staining Trough | Grale Scientific P/L | V1991.99 | ||
| Gammacell 1000 Elite Irradiator | Gamma Irradiator | Nordion International Inc. | ||
| Zeiss LSM 510 Meta Confocal | Confocal Microscope | |||
| Metamorph | Software for Imaging analysis | Molecular Devices, USA |
References
- Rogakou, E. P., Boon, C., Redon, C., Bonner, W. M. Megabase chromatin domains involved in DNA double-strand breaks in vivo. J. Cell Biol. 146, 905-916 (1999).
- Bonner, W. M. Gamma H2AX and cancer. Nature Reviews Cancer. 8 (12), 957-967 (2008).
- Savic, V. Formation of Dynamic [gamma]-H2AX Domains along Broken DNA Strands Is Distinctly Regulated by ATM and MDC1 and Dependent upon H2AX Densities in Chromatin. Molecular Cell. 34 (3), 298-310 (2009).
- Downs, J. Binding of chromatin-modifying activities to phosphorylated histone H2A at DNA damage sites. Mol Cell. 16 (6), 979-990 (2004).
- Chowdhury, D. [gamma]-H2AX Dephosphorylation by Protein Phosphatase 2A Facilitates DNA Double-Strand Break Repair. Molecular Cell. 20 (5), 801-809 (2005).
- Nakada, S., Chen, G., Gingras, A., Durocher, D. PP4 is a gamma H2AX phosphatase required for recovery from the DNA damage checkpoint. EMBO Rep. 9 (10), 1019-1026 (2008).
- Altaf, M., Auger, A., Covic, M., Côté, J. Connection between histone H2A variants and chromatin remodeling complexes. Biochem Cell Biol. 87 (1), 35-50 (2009).
- Kusch, T. Acetylation by Tip60 Is Required for Selective Histone Variant Exchange at DNA Lesions. Science. 306, 2084-2087 (2004).
- Hurlin, P. Progression of human papillomavirus type 18-immortalized human keratinocytes to a malignant phenotype. Proc Natl Acad Sci U S A. 88 (2), 570-574 (1991).
- Dickey, J. H2AX: functional roles and potential applications. Chromosoma. , (2009).

