Assessment of Aphidicidal Effect of Entomopathogenic Fungi against Parthenogenetic Insect, Mustard Aphid, Lipaphis erysimi (Kalt.)
Summary
This protocol presents an optimized detached-leaf bioassay system for evaluating the effectiveness of entomopathogenic fungi (EPF) against the mustard aphid (Lipaphis erysimi (Kalt.)), a parthenogenetic insect. The method outlines the data collection process during Petri dish experiments, enabling researchers to consistently measure the virulence of EPF against mustard aphids and other parthenogenetic insects.
Abstract
The mustard aphid (L. erysimi) is a pest that infests various cruciferous crops and transmits plant viruses. To achieve eco-friendly pest management, entomopathogenic fungi (EPF) are potential microbial control agents for controlling this pest. Therefore, virulence screening of EPF isolates under Petri dish conditions is necessary before field application. However, the mustard aphid is a parthenogenetic insect, making it difficult to record data during Petri dish experiments. A modified system for detached-leaf bioassays was developed to address this issue, using a micro-sprayer to inoculate conidia onto aphids and prevent drowning by facilitating air-drying after spore suspension. The system maintained high relative humidity throughout the observation period, and the leaf disc remained fresh for over ten days, allowing parthenogenetic reproduction of the aphids. To prevent offspring buildup, a process of daily removal using a painting brush was implemented. This protocol demonstrates a stable system for evaluating the virulence of EPF isolates against mustard aphids or other aphids, enabling the selection of potential isolates for aphid control.
Introduction
The mustard aphid (L. erysimi) is a notorious pest that infests a variety of cruciferous crops, causing significant economic losses1. While several systematic insecticides have been recommended to combat aphid infestations, the frequent use of these insecticides raises concerns about pesticide resistance2,3. Therefore, in terms of eco-friendly pest management, entomopathogenic fungi (EPF) could serve as a suitable alternative control strategy. EPF is an insect pathogen with the ability to infect hosts by penetrating their cuticles, making it a potent agent for controlling aphids and other plant-sucking insects4. Furthermore, EPF has proven to be a feasible and sustainable pest management technique, offering benefits such as plant pathogen antagonism and plant growth promotion5.
EPF can be obtained through insect-soil baiting or isolated from insect cadavers in the field6,7. However, before further use of fungal isolates, pathogenicity screening is necessary. Several studies have been conducted on the effectiveness of EPF against aphids, which are significant crop pests that can cause severe damage8,9. Mustard aphids, among various species of aphids, have been tested for susceptibility to several strains of Beauveria spp., Metarhizium spp., Lecanicillium spp., Paecilomyces spp., and even Alternaria, which is primarily known as a saprophytic and plant pathogenic fungus but has shown some lethal effects against mustard aphids10,11,12.
To evaluate the effectiveness of EPF against aphids under laboratory conditions, bioassays can be divided into two main parts: the inoculation chamber and fungal inoculation. The current protocol describes the construction of an inoculation chamber, where aphids can be maintained using various methods such as an excised leaf with a petiole wrapped in moist cotton, an excised leaf disc with a Petri dish lined with damped filter paper, direct maintenance on pot plants, or an excised leaf disc embedded in water agar within a Petri dish or container10,11,13. Common methods for fungal inoculation include conidia spraying, aphid immersion into a conidia suspension, leaf dipping into a conidia suspension, and plant endophyte inoculation11,14,15,16. While various inoculation methods exist, the bioassays should simulate field application conditions. For example, in the case of the leaf dipped method12,17, the efficiency of EPF can be evaluated, but since the aphids infest the fungus-loaded leaves, the dorsal side of the aphid, which is a preferential penetration site, does not usually get exposed to the fungus.
To evaluate the aphidicidal effect of EPF under laboratory conditions, this protocol suggests using the detached-leaf method described by Yokomi and Gottwald18 with some modifications, followed by conidia inoculation using a micro-sprayer. This method maintains approximately 100% humidity in the bioassay chamber for at least seven days without requiring additional replenishment of water18,19. Additionally, confining aphids to one surface ensures their exposure to conidia spraying and facilitates observations20. However, aphids may become stuck in the exposed agar surface while moving within the inoculation chamber. Furthermore, recording data in the Petri dish experiment with mustard aphids, which are parthenogenetic insects, can be challenging due to their rapid development and reproduction. It is difficult to distinguish between inoculated adults and their progeny without removal. The details of how to proceed with this step are seldom mentioned, and some inconsistent factors, such as leaf consumption area, need to be optimized.
This protocol demonstrates a stable system for screening the virulence of EPF isolates against mustard aphids, enabling the selection of potential isolates against various aphid species from an extensive EPF library. Field-collected aphids can be identified, and a sufficient laboratory population of mustard aphids can be established to evaluate the aphidicidal effect of various fungal isolates using an easy and feasible methodology with consistent outcomes. Aphids have developed multiple evolutionary mechanisms in response to intense and repeated anthropogenic pressures in agroecosystems, posing challenges to food security9. Therefore, this described method could be extended to evaluate potential EPF isolates against various aphid species.
Protocol
NOTE: The complete flowchart is shown in Figure 1.
1. Mustard aphid collection and maintenance
- Collection of mustard aphids
- Flip the leaves and visually check for infestation of mustard aphids on cruciferous crops in the field.
- Record the sampling site information (i.e., GPS) and host plant(s), and confirm the history of insecticide applications with the farmers.
- Use an insect aspirator or a fine painting brush (see Table of Materials) to collect about 50 mustard aphids into a 50 mL centrifuge tube from cruciferous crops in the field and bring the sample to the laboratory within 3 h.
- Prepare a temporary maintenance Petri dish with excised cruciferous leaves and water-infiltrated filter paper at the bottom.
- Place the five aphids on the temporary maintenance Petri dish under room temperature (25 ± 2 °C) with a relative humidity of 70% and 12:12 h (light:dark) photoperiod for 14 days to confirm that aphid spare natural enemy-free before further rearing.
NOTE: To ensure that field-collected aphids are free from natural enemies such as parasitoid wasps or entomopathogenic fungi, it is crucial to confirm the absence of these organisms before proceeding with further rearing.
- Maintainance of mustard aphids
NOTE: To maintain mustard aphids, use pesticide-free (including biopesticide) cruciferous leaves.- Provide a stable and sufficient supply of cruciferous leaves (about 25 cm2).
NOTE: Obtain the cruciferous leaves either from the market or grow the plants. Before long-term, massive rearing of mustard aphids, several different kinds of crucifer could be tested to find a suitable crucifer. Once the species of the cruciferous leaf is fixed, do not change it. In this study, Komatsuna (Brassica rapa var. perviridis) at 6-7-leaf stage was used (see Table of Materials). Additionally, dispose of the 2-3 oldest leaves. - Add water before the filter paper at the bottom is completely dry.
- Observe the population density of the mustard aphids daily. The population should grow to 2-3-fold after 7 days.
- When the number of aphids increases, cut the originally excised leaves with mustard aphids into 4-6 smaller pieces and put each small leaf piece with mustard aphids into one Petri dish with fresh excised leaf and water-infiltrated filter paper.
- Place the Petri dish in an incubator at 25 °C with a 12:12 h (light:dark) photoperiod and observe the population density of mustard aphids daily.
- Remove the original excised leaves after most of the aphids migrate to fresh excised leaves before the original leaves rot.
- Provide a stable and sufficient supply of cruciferous leaves (about 25 cm2).
2. Molecular identification of mustard aphid
NOTE: To confirm the species of field-collected mustard aphid, molecular identification was performed using two molecular markers: the sequence characterized amplified region (SCAR) based A05Le designed by Lu et al.21, and mustard aphid cytochrome oxidase subunit 1 (COI) region of the mustard aphid.
- Extraction of mustard aphid genomic DNA
- Collect approximately 50 aphids in a 1.5 mL centrifuge tube.
NOTE: Aphids used for molecular identification should be descendants of one single aphid, and various stages could also be pooled in the same tube for DNA extraction. - Homogenize the aphids with a pellet pestle and extract the DNA using Gene-Spin Genomic DNA Isolation Kit following the manufacturer's instructions (see Table of Materials).
- Elute the genomic DNA with 50 µL of preheated nuclease-free water.
- Collect approximately 50 aphids in a 1.5 mL centrifuge tube.
- PCR amplification and DNA sequencing
- Amplify the target DNA sequences from aphid genomic DNA using PCR Master Mix (2x) with primer pairs A05LeF/A05LeR and LeCO1F/LeCO1R (Table 1) with different PCR programs21.
NOTE: The PCR reaction with a total volume of 20 µL is composed of 2 µL aphid genomic DNA template, 1 µL forward and 1 µL reversed primers, 10 µL PCR Master Mix (see Table of Materials), and 6 µL ddH2O. - Perform the PCR in a thermal cycler (see Table of Materials) with the following program: 94 °C for 5 min, 25 cycles of 94 °C for 45 s, 61 °C (for A05LeF/A05LeR) and 58 °C (for LeCO1F/LeCO1R) for 45 s, 72 °C for 1 min, followed by a final extension of 72 °C for 5 min.
- Analyze the PCR product by 1% agarose gel electrophoresis to confirm the identity of the mustard aphid (Figure 2).
NOTE: If primer pair A05LeF/A05LeR successfully amplifies the correct size of the DNA fragment, it has convincingly identified the field-collected aphid as mustard aphid21. The primer pairs are listed in Table 2. - Purify the PCR product of mustard aphid COI amplicon using a commercially available gel/PCR kit following the manufacturer's instructions (see Table of Materials), and sequence the PCR product using a commercial sequencing service.
- Amplify the target DNA sequences from aphid genomic DNA using PCR Master Mix (2x) with primer pairs A05LeF/A05LeR and LeCO1F/LeCO1R (Table 1) with different PCR programs21.
- Sequence analysis
NOTE: According to Lu et al.21, the DNA bending of A05Le is a mustard aphid-specific DNA fragment; therefore, the A05Le PCR product does not need to be further sequenced.- Check the read quality by Chromas software (see Table of Materials) after obtaining the LeCO1 sequence.
- Trim upstream and downstream low-quality reads by opening a fasta (or txt) file and directly removing the low-quality reads.
- Submit the trimmed sequence to NCBI web BLAST (https://blast.ncbi.nlm.nih.gov/Blast.cgi) using default parameters.
NOTE: If the amplicon sizes of A05Le and LeCO1 primer sets are correct, theoretically, the sequence blast search against NCBI standard databases must match mustard aphid.
3. Preparation of Entomopathogenic fungi
NOTE: The EPF used in this study is listed in Table 1.
- Recovery of EPF from fungal library
- Thaw the preserved fungi stock (conidia suspended in 30% glycerin) from the -80 °C freezer.
- Transfer a small amount (approximately 10 µL) of the conidia suspension to 6 cm ¼ Sabouraud dextrose agar (SDA) plate (0.75 g Sabouraud dextrose broth with 1.5 g agar per 100 mL ddH2O) and spread it evenly using a cell spreader in a laminar flow hood.
- Seal the Petri dish with paraffin film, and incubate it in darkness at 25 °C for 10-14 days.
- Re-culture the fungal isolates by streaking fungal mass on a ¼ SDA plate and incubating fungi in darkness at 25 °C for 10-14 days before harvesting the conidia22.
NOTE: The fungal culture should be used approximately 10 days before inoculating the aphids. EPF culture that is more than 14 days old is not recommended for use in the virulence test.
- Preparation of conidia suspension
- Scrape the conidia from the 10-14 days old fungal culture on ¼ SDA plate by inoculating loop after adding 2-3 mL 0.03% Tween 80 (see Table of Materials).
- Collect the fungal suspension into a centrifuge tube.
- Vortex the solution at the highest speed, count the number of conidia using a hemocytometer under a light microscope, and adjust the concentration of the conidia suspension to 108 conidia/mL.
- Transfer the conidia suspension into a UV-sterilized micro-sprayer.
NOTE: Vortex the conidia suspension again before transferring it, as the conidia are prone to precipitate. The micro-sprayer should be exposed to ultraviolet radiation in a laminar flow hood for 30 min before use.
4. Virulence screening against mustard aphid
- Preparation of newly emerged adults
- Move apterous (without wing) and alate (winged) adults from the rearing Petri dish to freshly excised leaves to reproduce new progenies of the same age. Remove the adults after 24 h to avoid overcrowding and minimize the production of alated progenies23,24. Only apterous adults will be used in further experiments.
- Rear progenies to the adult stage and remove the progenies that do not develop into the adult stage after 48 h (Figure 3) to prevent age variation between aphids for the experiment. Additionally, remove alated adults.
- Preparation of inoculation chamber
NOTE: It is recommended to prepare 9 cm diameter cruciferous leaves first, so that the leaf disc can be embedded into the water agar before solidification.- Clean the cruciferous leaves with tap water, remove dirt and residue, and any other arthropod if present.
- Wipe away any excess water with a paper towel and check for any other arthropods on the surface of the cruciferous leaves using a stereomicroscope. Remove any arthropods if present.
- Cut a 9 cm diameter leaf disc either by directly pressing the bottom Petri dish on the leaf as a mold or by using scissors.
NOTE: If the leaves are smaller than 9 cm in diameter, combine a couple of excised leaves. - Prepare 1.5% water agar for each Petri dish by heating and dissolving 450 mg of agar (see Table of Materials) in 30 mL ddH2O using microwave.
NOTE: The amount of 1.5% water agar can be adjusted depending on the experiment scale. - Pour 30 mL of 1.5% water agar in the 9 cm Petri dish before solidification in the laminar flow hood, and then wait for the agar to cool down to ~40 °C until the surface of the agar is semi-solidified.
NOTE: Check if the surface is semi-solidified by gently horizontally shaking the Petri dish. - Place the leaf disc, abaxial surface up, on top of the water agar, embedding the leaf disc in the agar.
NOTE: Minimize the exposure of the agar surface. - After the water agar has solidified completely, close the cover of the Petri dish.
NOTE: Open the cover for water vaporization if lots of droplets are condensing on the cover immediately. - Place 20 apterous adults on the leaf disc.
NOTE: It is recommended to operate under a stereomicroscope to prevent damage to their mouthparts.
- EPF inoculation and observation
- Open the cover of the inoculation chamber and spray 0.3 mL of conidia suspension (from step 3.2) directly onto the aphids and leaf disc from about 15 cm above the leaf disc.
NOTE: Vortex the conidia suspension again before spraying. The spraying areas should be tested before the experiment as they can vary between different sprayers. In this case, 0.3 mL with 15 cm distance is recommended. The spraying area should cover the entire leaf disc, and a thin layer of conidia suspension should cover the leaf disc. If droplets converge into standing water on the leaf, the inoculation volume should be decreased. Clean the table with 70% ethanol before inoculation and between using different isolates. - Wait for the conidia suspension to dry, and then close the cover to prevent mustard aphids from drowning.
NOTE: Aphids seldom roam around during the experiment; however, ensuring that the aphids do not escape is still necessary. - Seal the inoculation chamber with paraffin film to maintain high relative humidity and place the inoculation chamber in an incubator at 25 °C with 12:12 h (light:dark) photoperiod.
- Open the cover of the inoculation chamber to count mortality under a stereomicroscope every 12 h for 5 days.
- Wipe out the honeydew adhering to the cover with a paper towel and remove the newly emerged aphids with a fine painting brush. Clean the cover with tap water every 24 h.
NOTE: The observation period might vary for different species of aphids based on adult longevity. - Keep the cadaver on the leaf disc for mycosis to confirm successful inoculation.
- Open the cover of the inoculation chamber and spray 0.3 mL of conidia suspension (from step 3.2) directly onto the aphids and leaf disc from about 15 cm above the leaf disc.
- Confirmation of conidia germination rate
NOTE: This step is optional. To confirm the conidia germination rate pairwise when performing the virulence screening to ensure the activity of conidia.- Drop one droplet of 5 µL conidia suspension on ¼ SDA for three replicates.
- After 18 h, calculate the germination rate with the light microscope. Count at least 100 spores randomly in a single droplet to confirm the fungal germination rate.
NOTE: Normally, the germination rate of the isolates used in the experiment should be higher than 90%.
5. Bioassay of selected EPF isolates
NOTE: EPF isolates that showed high virulence, which was selected from step 4, were subjected to a bioassay against mustard aphids using four concentrations of conidia suspensions (ranging from 104 to 107 conidia/mL).
- Preparation of conidia suspension
- Repeat step 3.1 to recover the selected EPF isolates.
- Repeat step 3.2 to prepare four concentrations of conidia suspension, including 104, 105,106, and 107 conidia/mL.
- Preparation of newly emerged adults
- Repeat step 4.1 to prepare newly emerged adults.
- Preparation of inoculation chamber
- Repeat step 4.1. to prepare the inoculation chamber.
- EPF inoculation and observation
- Repeat step 4.3. to perform the EPF inoculation and observation with the four concentrations of conidia suspension, respectively.
6. Statistical analysis
- Calculation of corrected mortality
- Calculate corrected mortality using Abbott's formula25, as shown below:
Corrected mortality (%) = [(MT− MC) × 100%] / (100− MC)
MT represents the mortality of the treatment group, and MC represents the mortality of the control group.
NOTE: The mortality of the control group should not be higher than 20%. - Open the SPSS software platform (see Table of Materials), create variables including "treatment" and "cor_mor" (representing corrected mortality), and enter the calculated results for the inoculation of different isolates at the same time point with the same inoculated concentration.
- Select Analyze, Compare Means and Proportions, Independent-Samples T Test in SPSS for independent t-test analysis.
- In SPSS, input treatment into the "Grouping Variable" box, and cor_mor into the "Test Variable(s)". Define groups by different fungal isolates. Press OK to analyze.
- Calculate corrected mortality using Abbott's formula25, as shown below:
- Calculation of median lethal time (LT50) and median lethal concentration (LC50)
- Open the SPSS, create variables "total", "response" and "duration"/"concentration", and input the recorded result into the spreadsheet.
- Select Analyze, Regression, Probit in SPSS to calculate LT50 and/or LC50.
NOTE: If cumulative mortality does not exceed 50% eventually, LT50 and/or LC50 cannot be estimated. - Input total into the "Total Observed" box, response into "Response Frequency" box, duration/concentration into "Covariate(s)" box. Press OK to analyze.
NOTE: Generally, press Options and set the "Significance level for use of heterogeneity factor" to 0.05.
Representative Results
The presented flowchart illustrates the stable condition of the mustard aphids from field collection to virulence screening. The maintenance of aphids from field collection ensured a stable increase in aphid colonies with an adequate food supply. The field-collected aphids were confirmed as mustard aphids through the use of molecular markers, including PCR amplicon size and LeCO1 sequencing. The virulence screening, conducted using the detached-leaf method, revealed a consistent survival rate for mustard aphids, with the control group exhibiting an 85% survival rate (Figure 4).
During the virulence screening, Cc-NCHU-213 demonstrated the fastest aphid-killing ability, resulting in 50% and 90% mortalities at 3 days and 4.5 days post-inoculation (d.p.i.), respectively (Figure 4). However, varying aphid-killing abilities were observed among the five B. bassiana isolates. Bb-NCHU-141, -143, and -153 exhibited slow aphid-killing abilities, with only 5% mortality at 3 d.p.i., even when excluding the effects of 0.03% Tween 80 spraying or other lethal factors (Figure 4). Hence, the corrected mortality formula was employed to normalize the control mortality. Most EPF isolates against mustard aphids exhibited corrected mortality rates higher than 70% within 5 d.p.i., except for Pl-NCHU-152 (Table 3). Among these EPF isolates, Bb-NCHU-286 demonstrated the highest mortality rate of 100% (Table 3). Additionally, EPF mycosis was observed on cadavers of mustard aphids infected with Metarhizium spp., Beauveria spp., Purpureocillium lilacinum, and Cordyceps cateniannulata during the virulence screening, indicating the effectiveness of this system (Figure 5).
Based on the results of the virulence screening, two EPF isolates, namely Mb-NCHU-197 and Cc-NCHU-213, exhibiting rapid insect-killing activity (40% and 50% mortality at 3 d.p.i., respectively), were selected for bioassay against mustard aphids. The results demonstrated significantly different corrected mortalities for Mb-NCHU-197 and Cc-NCHU-213 at 3 and 4 d.p.i. with an inoculation of 107 conidia/mL (Figure 6). In the LT50 assay, the treatment with 107 conidia/mL of Cc-NCHU-213 exhibited a significantly shorter duration compared to other treatments (Table 4). Furthermore, the LC50 value of Cc-NCHU-213 (9.32 × 104) was lower than that of Mb-NCHU-213 (2.30 × 105), indicating that Cc-NCHU-213 possesses greater virulence against mustard aphids (Table 5).
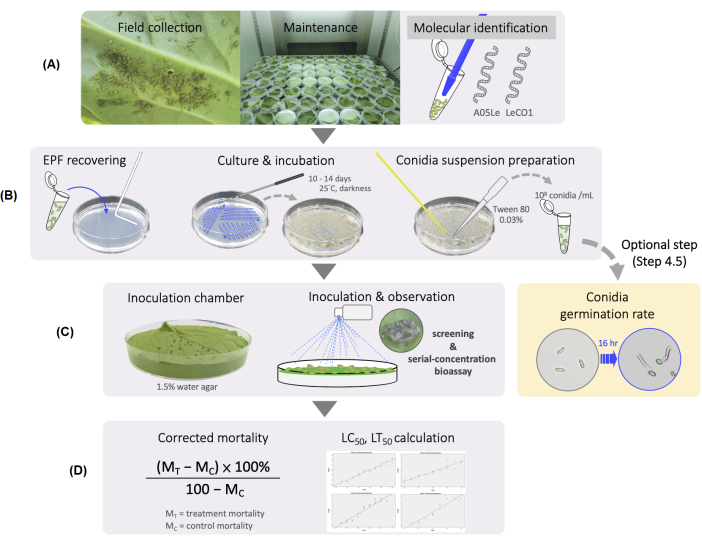
Figure 1: Experimental flowchart for screening EPF virulence against mustard aphids. (A) Establishment of a mustard aphid-rearing system. (B) Preparation of EPF. (C) Fungal inoculation. (D) Statistical analysis. Please click here to view a larger version of this figure.

Figure 2: Electrophoresis of mustard aphid genomic DNA amplified with A05Le and Le CO1 primer sets. Electrophoresis was performed on a 1% agarose gel. M = 100 bp DNA ladder; bp = base pairs.The red asterisk indicates the target bend of PCR amplification. Please click here to view a larger version of this figure.

Figure 3: Differences between apterous fourth-instar nymph and adult mustard aphids. (A) Fourth-instar nymph. (B) Adult. The tibiae of the hind legs of the fourth-instar nymph are whitish (marked with red arrow). Newly emerged aphids were removed by fine camel brush during virulence tests. Scale bar = 1 mm. Please click here to view a larger version of this figure.
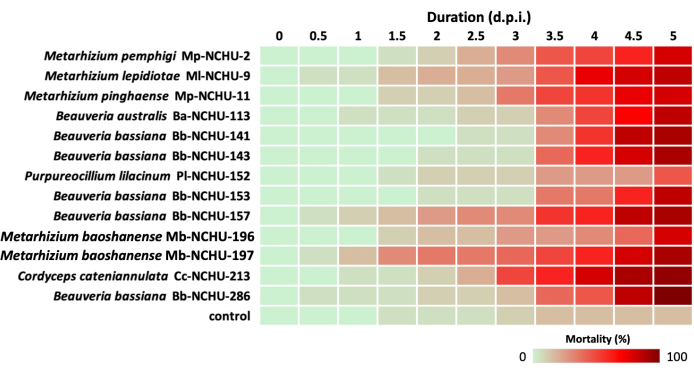
Figure 4: Mortality heat map of 13 EPF isolates against mustard aphids through the detached-leaf method. Please click here to view a larger version of this figure.

Figure 5: Observation of fungal mycosis of 13 EPF isolates. Mepe-NCHU-2 = Metarhizium pemphigi; Mp-NCHU-11 = Metarhizium pinghaense; Mb = Metarhizium baoshanense;Cc = Cordyceps cateniannulata; Ba = Beauveria australis;Bb = Beauveriabassiana; Pl = Purpureocillium lilacinum. Scale bar = 1 mm. Please click here to view a larger version of this figure.
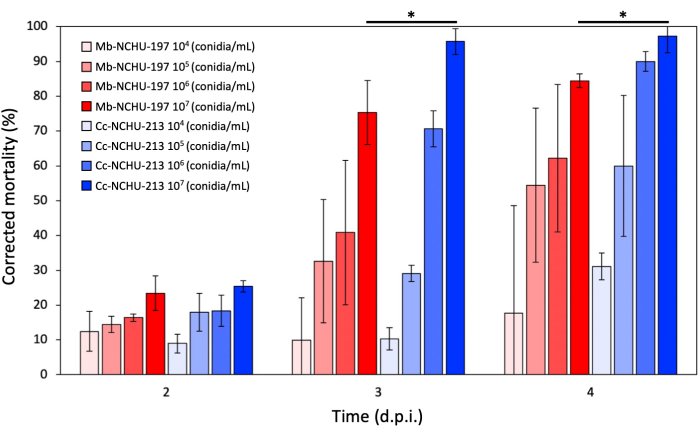
Figure 6: Corrected mortality of fungal isolates Mb-NCHU-197 and Cc-NCHU-213 against mustard aphid. The error bars represent the standard deviation (SD). The mortalities of Mb-NCHU-197 and Cc-NCHU-213 at the same time point with the same inoculated concentration were compared using independent t-test, and mortalities marked with an asterisk were found to be significantly different (p < 0.05). Please click here to view a larger version of this figure.
| Isolate | Species | Host or source* | Location |
| Mp-NCHU-2 | Metarhizium pemphigi | soil | Yilan |
| Ml-NCHU-9 | Metarhizium lepidiotae | soil | Yilan |
| Mp-NCHU-11 | Metarhizium pinghaense | soil | Yilan |
| Ba-NCHU-113 | Beauveria australis | soil | Taichung |
| Bb-NCHU-141 | Beauveria bassiana | Hypothenemus hampei | Chiayi |
| Bb-NCHU-143 | Beauveria bassiana | Hypothenemus hampei | Chiayi |
| Pl-NCHU-152 | Purpureocillium lilacinum | Tessaratoma papillosa | Chiayi |
| Bb-NCHU-153 | Beauveria bassiana | Rhynchophorus ferrugineus | Chunghua |
| Bb-NCHU-157 | Beauveria bassiana | Rhynchophorus ferrugineus | Chunghua |
| Mb-NCHU-196 | Metarhizium baoshanense | soil | Taichung |
| Mb-NCHU-197 | Metarhizium baoshanense | soil | Taichung |
| Cc-NCHU-213 | Cordyceps cateniannulata | soil | Taichung |
| Bb-NCHU-286 | Beauveria bassiana | Cerambycidae | Taichung |
Table 1: EPF isolates used in this study.
| Primer name | Sequence (5' – 3') | Product size (bp) | Reference | |
| A05Le | F-GGGTCTTGGATGGTGTGGTG | 953 | Lu et al. [21] | |
| R-AGGGGTCTTGTCGCCATTTT | ||||
| LeCO1 | F-CTTTTCCCATGATCAATTTT | 593 | This study | |
| R-ACGTAGTGGAAATGAGCAAC | ||||
Table 2: Primer pairs used for molecular identification of mustard aphids.
| Isolate | Species | Corrected mortality (%) |
| Mp-NCHU-2 | Metarhizium pemphigi | 76.47 |
| Ml-NCHU-9 | Metarhizium lepidiotae | 82.35 |
| Mp-NCHU-11 | Metarhizium pinghaense | 76.47 |
| Ba-NCHU-113 | Beauveria australis | 82.35 |
| Bb-NCHU-141 | Beauveria bassiana | 88.24 |
| Bb-NCHU-143 | Beauveria bassiana | 88.24 |
| Pl-NCHU-152 | Purpureocillium lilacinum | 35.29 |
| Bb-NCHU-153 | Beauveria bassiana | 82.35 |
| Bb-NCHU-157 | Beauveria bassiana | 88.24 |
| Mb-NCHU-196 | Metarhizium baoshanense | 76.47 |
| Mb-NCHU-197 | Metarhizium baoshanense | 88.24 |
| Cc-NCHU-213 | Cordyceps cateniannulata | 94.12 |
| Bb-NCHU-286 | Beauveria bassiana | 100.00 |
Table 3: Corrected mortality rates of 13 EPF isolates against mustard aphids at 5 days post inoculation (d.p.i.).
| Isolate | Species | Conc.(conidia/mL) | N* | LT50 (days)† | 95% confidence limits | Slope (SE) | X2 (df)‡ |
| Mb-NCHU-197 | Metarhizium baoshanense | 104 | 60 | 3.816 a | 3.61–4.05 | 0.60 (0.05) | 15.64 (28) |
| 105 | 60 | 3.112 bd | 2.95–3.28 | 0.72 (0.05) | 14.61 (28) | ||
| 106 | 60 | 2.908 b | 2.76–3.06 | 0.85 (0.06) | 15.04 (28) | ||
| 107 | 60 | 2.549 c | 2.40–2.69 | 0.90 (0.06) | 24.31 (28) | ||
| Cc-NCHU-213 | Cordyceps cateniannulata | 104 | 60 | 3.948 a | 3.71–4.23 | 0.53 (0.05) | 8.81 (28) |
| 105 | 60 | 3.237 d | 3.07–3.41 | 0.72 (0.05) | 22.86 (28) | ||
| 106 | 60 | 2.414 c | 2.28–2.54 | 1.08 (0.07) | 28.30 (28) | ||
| 107 | 60 | 2.132 e | 2.02–2.25 | 1.41 (0.10) | 28.96 (28) | ||
| *Number of insects observed. | |||||||
| †LT50 values marked with different letters were considered significantly different as their 95% confidence limits did not overlap. | |||||||
| ‡X2, Chi-square value in Pearson's goodness-of-fit test; df, degrees of freedom | |||||||
Table 4: LT50 values of fungal isolates Mb-NCHU-197 and Cc-NCHU-213 against mustard aphids under different conidia concentrations. *Number of insects observed. †LT50 values marked with different letters were considered significantly different as their 95% confidence limits did not overlap. ‡X2, Chi-square value in Pearson's goodness-of-fit test; df, degrees of freedom.
| Isolate | Species | N* | LC50 (conidia/mL)† | 95% confidence limits | Slpoe (SE) | X2 (df)‡ |
| Mb-NCHU-197 | Metarhizium baoshanense | 240 | 2.30 × 105 a | 8.63 × 104–5.70 × 105 | 0.43 (0.08) | 10.14 (10) |
| Cc-NCHU-213 | Cordyceps cateniannulata | 240 | 9.32 × 104 a | 4.97 × 104–1.62 × 105 | 0.76 (0.09) | 4.33 (10) |
| *Number of insects observed. | ||||||
| †LC50 values marked with different letters were considered significantly different as their 95% confidence limits did not overlap. | ||||||
| ‡X2, Chi-square value in Pearson's goodness-of-fit test; df, degrees of freedom | ||||||
Table 5: LC50 values of fungal isolates Mb-NCHU-197, and Cc-NCHU-213 against mustard aphids. *Number of insects observed. †LC50 values marked with different letters were considered significantly different as their 95% confidence limits did not overlap. ‡X2, Chi-square value in Pearson's goodness-of-fit test; df, degrees of freedom.
Discussion
Crucifers, a group of vegetables, are frequently infested by multiple aphid species, including mustard aphid (L. erysimi) and cabbage aphid (Brevicoryne brassicae)26. Both species have been reported in Taiwan27, and it is possible for them to coexist at the collection site. To distinguish closely related aphid species, this study employed a molecular identification technique using a multiplex primer set21. By designing a molecular marker from the mustard aphid COI gene fragment, we successfully identified the mustard aphid, thus confirming the reliability of the molecular marker A05Le for this purpose21.
The observations revealed that distinguishing between apterous fourth-instar nymphs and adult mustard aphids based solely on their size or morphology is challenging without experience. However, a crucial distinguishing feature is the tibia of the hind legs, which appears white in fourth-instar nymphs but not in adults (Figure 3). Previous studies on L. attenuatum, B. bassiana against cotton aphids, and M. brunneum against green peach aphids have demonstrated that molting can be a strategy to avoid infection28,29,30. Therefore, misidentifying aphid stages can lead to errors in virulence assessment, as molting can affect the effectiveness of EPF28,29,31. To address this issue and ensure greater accuracy and precision in virulence evaluation, exclude aphids with whitish portions on their hind leg tibiae that have not reached the adult stage unless intentionally using aphid nymphs. These aphids do not fully mature and may undergo molting, which could allow them to escape the infection process of EPF. Additionally, a time course of observation and data recording every 12 h is recommended. The 12 h interval is preferable for demonstrating differences between multiple promising isolates with similar aphid-killing abilities and facilitates the precise calculation of LT50. However, the time interval can be adjusted based on the different life cycles of other insect species or determined through a small-scale pretest.
Although the detached-leaf method may leave minimal honeydew on the leaf disc, most of the honeydew adheres to the Petri dish cover since the leaf disc is in close proximity. It may be wiped or washed away during the observation period. However, honeydew is undoubtedly present in the practical environment of crop cultivation when aphids invade32. The honeydew present in the inoculation chamber may somewhat simulate the situation when EPF is inoculated into aphids in the field. Aphid cuticles mostly contain honeydew, which serves as a source of nutrients for the fungus and may stimulate the germination of EPF16. Thus, the combination of EPF application and honeydew production by aphids can be advantageous.
To confirm that aphid death is due to infection, cadavers are typically transferred from the EPF inoculation chamber to damp filter paper or a culture medium for observing fungal outgrowth11,14,33,34. However, in this study, water agar was used in the inoculation chamber to maintain high humidity, allowing the aphid cadavers to remain on the leaves without needing to be moved for fungal outgrowth observation. Although the detached-leaf method provides information on the aphidicidal effect of EPF isolates, it still has limitations. The water agar used to maintain the leaf disc condition can cause aphids to become stuck while moving in the inoculation chamber. To address this issue, the percentage of water agar was increased to 3%, providing a firm enough surface for Russian wheat aphids (Diuraphis noxia) to walk on20. In this experiment, mustard aphids were able to move comfortably on water agar surfaces with concentrations ranging from 1.5% to 3%. However, as the observation period progressed, a few aphids became stuck upside down with their legs upward and had to be rescued back to the leaf disc. This may be due to the differences in aphid size or mobility, indicating that increasing the agar concentration alone may not solve the problem. Therefore, ensure that the leaf disc fully covers the agar surface by using a leaf that fits more snugly to the Petri dish size, which can help alleviate this issue. In comparison to the detached-leaf method described by Yokomi and Gottwald18, this study has improved upon one aspect by using a leaf size that is approximately a good fit for the Petri dish. This allows for relative quantification of food consumption and minimizes the exposed agar surface. Additionally, the leaf needs to be "embedded" in the semi-solidified water agar, whereas in the original reference, it was described as "placed" on the water agar18. Embedding the leaf disc into the water agar reduces the likelihood of aphids getting stuck to the agar surface and facilitates observation, as the aphids are confined to one side.
This protocol provides detailed instructions on how to proceed with the detached-leaf method and EPF inoculation, along with some modifications that have facilitated operations, observations, and consistent results. It also establishes a standardized method for selecting EPF isolates against plant-sucking pests under laboratory conditions. Furthermore, conducting a standard check with known effective or commercially available products can be performed for the future commercialization of EPF isolates.
Disclosures
The authors have nothing to disclose.
Acknowledgements
This research was supported by 109-2313-B-005 -048 -MY3 from the Ministry of Science and Technology (MOST).
Materials
| 10 μL Inoculating Loop | NEST Scientific | 718201 | |
| 100 bp DNA Ladder III | Geneaid | DL007 | |
| 2x SuperRed PCR Master Mix | Biotools | TE-SR01 | |
| 50 mL centrifuge tube | Bioman Scientific | ET5050-12 | |
| 6 cm Petri dish | Alpha Plus Scientific | 16021 | |
| 6 mm insect aspirator | MegaView Science | BA6001 | |
| 70 mm filter paper NO.1 | Toyo Roshi Kaisha | ||
| 70% ethanol | |||
| 9 cm Petri dish | Alpha Plus Scientific | 16001 | |
| Agar | Bioman Scientific | AGR001.1 | Microbiology grade |
| Agarose | Bioman Scientific | PB1200 | |
| BioGreen Safe DNA Gel Buffer | Bioman Scientific | SDB001T | |
| Chromas | Technelysium | ||
| GeneDoc | |||
| GenepHlow Gel/PCR Kit | Geneaid | DFH300 | https://www.geneaid.com/data/files/1605861013102532959.pdf |
| Gene-Spin Genomic DNA Isolation Kit | Protech Technology | PT-GD112-V3 | http://www.protech-bio.com/UserFiles/file/Gene-Spin%20Genomic%20DNA%20Kit.pdf |
| Hemocytometer | Paul Marienfeld | 640030 | |
| Komatsuna leaves (Brassica rapa var. perviridis) | Tai Cheng Farm | 1-010-300410 | |
| Microsprayer | |||
| MiniAmp Thermal Cycler | Thermo Fisher Scientific | A37834 | |
| Mustard aphid (Lipaphis erysimi) | |||
| Painting brush | Tian Cheng brush company | 4716608400352 | |
| Parafilm M | Bemis | PM-996 | |
| Pellet pestle | Bioman Scientific | GT100R | |
| Sabouraud Dextrose Broth | HiMedia | MH033-500G | |
| SPSS Statistics | IBM | ||
| TAE buffer 50x | Bioman Scientific | TAE501000 | |
| Tween 80 | PanReac AppliChem | 142050.1661 |
References
- Ghosh, S., Roy, A., Chatterjee, A., Sikdar, S. R. Effect of regional wind circulation and meteorological factors on long-range migration of mustard aphids over indo-gangetic plain. Scientific Reports. 9, 5626 (2019).
- Dhillon, M. K., Singh, N., Yadava, D. K. Preventable yield losses and management of mustard aphid, Lipaphis erysimi (Kaltenbach) in different cultivars of Brassica juncea(L.) Czern & Coss. Crop Protection. 161, 106070 (2022).
- Huang, F., Hao, Z., Yan, F. Influence of oilseed rape seed treatment with imidacloprid on survival, feeding behavior, and detoxifying enzymes of mustard aphid, lipaphis erysimi. Insects. 10 (5), 144 (2019).
- Mannino, M. C., Huarte-Bonnet, C., Davyt-Colo, B., Pedrini, N. Is the insect cuticle the only entry gate for fungal infection? insights into alternative modes of action of entomopathogenic fungi. Journal of Fungi. 5 (2), 33 (2019).
- Bamisile, B. S., Akutse, K. S., Siddiqui, J. A., Xu, Y. Model application of entomopathogenic fungi as alternatives to chemical pesticides: prospects, challenges, and insights for next-generation sustainable agriculture. Frontiers in Plant Science. 12, 741804 (2021).
- Scorsetti, A. C., Humber, R. A., Garcia, J. J., Lopez Lastra, C. C. Natural occurrence of entomopathogenic fungi (Zygomycetes: Entomophthorales) of aphid (Hemiptera: Aphididae) pests of horticultural crops in Argentina. Biocontrol. 52, 641-655 (2007).
- Liu, Y. C., Ni, N. T., Chang, J. C., Li, Y. H., Lee, M. R., Kim, J. S., et al. Isolation and selection of entomopathogenic fungi from soil samples and evaluation of fungal virulence against insect pests. Journal of Visualized Experiments. 175, e62882 (2021).
- Francis, F., Fingu-Mabola, J. C., Fekih, I. B. Direct and endophytic effects of fungal entomopathogens for sustainable aphid control: a review. Agriculture. 12 (12), 2081 (2022).
- Simon, J., Peccoud, J. Rapid evolution of aphid pests in agricultural environments. Current Opinion in Insect Science. 26, 17-24 (2018).
- Ujjan, A. A., Shahzad, S. Use of Entomopathogenic Fungi for the Control of Mustard Aphid (Lipaphis erysimi) on canola (Brassica napus L). Pakistan Journal of Botany. 44 (6), 2081-2086 (2012).
- Sajid, M., Bashir, N. H., Batool, Q., Munir, I., Bilal, M., Jamal, M. A., et al. In-vitro evaluation of biopesticides (Beauveria bassiana, Metarhizium anisopliae, Bacillus thuringiensis) against mustard aphid Lipaphis erysimi kalt. (Hemiptera: Aphididae). Journal of Entomology and Zoology Studies. 5 (6), 331-335 (2017).
- Paschapur, A. U., Subbanna, A. R. N. S., Singh, A. K., Jeevan, B., Stanley, J., Rajashekara, H., Mishra, K. K., Koti, P. S., Kant, L., Pattanayak, A. Alternaria alternata strain VLH1: a potential entomopathogenic fungus native to North Western Indian Himalayas. Egyptian Journal of Biological Pest Control. 32, 138 (2022).
- Miohammed, A. A. Lecanicillium muscarium and Adalia bipunctata combination for the control of black bean aphid, Aphis fabae. Biocontrol. 63, 277-287 (2018).
- Thaochan, N., Ngampongsai, A., Prabhakar, C. S., Hu, Q. Beauveria bassiana PSUB01 simultaneously displays biocontrol activity against Lipaphis erysimi (Kalt.) (Hemiptera: Aphididae) and promotes plant growth in Chinese kale under hydroponic growing conditions. Biocontrol Science and Technology. 31 (10), 997-1015 (2021).
- Mseddi, J., Farhat-Touzri, D. B., Azzouz, H. Selection and characterization of thermotolerant Beauveria bassiana isolates and with insecticidal activity against the cotton-melon aphid Aphis gossypii (Glover) (Hemiptera: Aphididae). Pest Management Science. 78 (6), 2183-2195 (2022).
- Butt, T. M., Ibrahim, L., Clark, S. J., Beckett, A. The germination behaviour of Metarhizium anisopliae on the surface of aphid and flea beetle cuticles. Mycological Research. 99 (8), 945-950 (1995).
- Ullah, S., Raza, A. B. M., Alkafafy, M., Sayed, S., Hamid, M. I., Majeed, M. Z., Riaz, M. A., Gaber, N. M., Asim, M. Isolation, identification and virulence of indigenous entomopathogenic fungal strains against the peach-potato aphid, Myzus persicae Sulzer (Hemiptera: Aphididae), and the fall armyworm, Spodoptera frugiperda (J.E. Smith) (Lepidoptera: Noctuidae). Egyptian Journal of Biological Pest Control. 32, 2 (2022).
- Yokomi, R. K., Gottwald, T. R. Virulence of Verticillium lecanii Isolates in Aphids Determined by Detached-leaf Bioassay. Journal of Inbertebrate Pathology. 51, 250-258 (1988).
- Vu, V. H., Hong, S. I., Kim, K. Selection of entomopathogenic fungi for aphid control. Journal of Bioscience and Bioengineering. 104 (6), 498-505 (2007).
- Vandenberg, J. D. Standardized bioassay and screening of beauveria bassiana and paecilomyces fumosoroseus against the russian wheat aphid (homoptera: aphididae). Journal of Economic Entomology. 89 (6), 1418-1423 (1996).
- Lu, W. N., Wu, Y. T., Kuo, M. H. Development of species-specific primers for the identification of aphids in Taiwan. Applied Entomology and Zoology. 43 (1), 91-96 (2008).
- Liu, Y. C., et al. Isolation and selection of entomopathogenic fungi from soil samples and evaluation of fungal virulence against insect pests. Journal of Visualized Experiments. 175, e62882 (2021).
- Menger, J., Beauzay, P., Chirumamilla, A., Dierks, C., Gavloski, J., Glogoza, P., et al. Implementation of a diagnostic-concentration bioassay for detection of susceptibility to pyrethroids in soybean aphid (hemiptera: aphididae). Journal of Economic Entomology. 113 (2), 932-939 (2020).
- Zhang, R., Chen, J., Jiang, L., Qiao, G. The genes expression difference between winged and wingless bird cherry-oat aphid Rhopalosiphum padi based on transcriptomic data. Scientific Reports. 9, 4754 (2019).
- Abbott, W. S. A method of computing the effectiveness of an insecticide. Journal of Economic Entomology. 18, 265-267 (1925).
- Liu, T. X., Sparks, A. N. . Aphids on Cruciferous Crops: Identification and Management. , 9-11 (2001).
- Kuo, M., Chianglin, H. Temperature dependent life table of brevicoryne brassicae (l.)(hemiptera: aphididae) on radish. Formosan Entomologist. 27, 293-302 (2007).
- Im, Y., Park, S., Lee, S. Y., Kim, J., Kim, J. J. Early-Stage defense mechanism of the cotton aphid aphis gossypii against infection with the insect-killing fungus beauveria bassiana JEF-544. Frontiers in Immunology. 13, 907088 (2022).
- Kim, J. J., Roberts, D. W. The relationship between conidial dose, moulting and insect developmental stage on the susceptibility of cotton aphid, Aphis gossypii, to conidia of Lecanicillium attenuatum, an entomopathogenic fungus. Biocontrol Science and Technology. 22 (3), 319-331 (2012).
- Reingold, V., Kottakota, C., Birnbaum, N., Goldenberg, M., Lebedev, G., Ghanim, M., et al. Intraspecies variation ofMetarhiziumbrunneumagainst the green peach aphid,Myzus persicae, provides insight into thecomplexity of disease progression. Pest Management Science. 77, 2557-2567 (2021).
- Ortiz-Urquiza, A., Keyhani, N. O. Action on the Surface: entomopathogenic fungi versus the insect cuticle. Insects. 4, 357-374 (2013).
- Knodel, J. J., Beauzay, P., Boetel, M., Prochaska, T., Chirumamilla, A. . 2022 North Dakota Field Crop Insect Management Guide. , (2021).
- Yeo, H., Pell, J. K., Alderson, P. G., Clark, S. J., Pye, B. J. Laboratory evaluation of temperature effects on the germination and growth of entomopathogenic fungi and on their pathogenicity to two aphid species. Pest Management Science. 59 (2), 156-165 (2003).
- Erdos, Z., Chandler, D., Bass, C., Raymond, B. Controlling insecticide resistant clones of the aphid, Myzus persicae, using the entomopathogenic fungus Akanthomyces muscarius: fitness cost of resistance under pathogen challenge. Pest Management Science. 77 (11), 5286-5293 (2021).

