Comparison of Two Representative Methods for Differentiation of Human Induced Pluripotent Stem Cells into Mesenchymal Stromal Cells
Summary
This protocol describes and compares two representative methods for differentiating hiPSCs into mesenchymal stromal cells (MSCs). The monolayer method is characterized by lower cost, simpler operation, and easier osteogenic differentiation. The embryoid bodies (EBs) method is characterized by lower time consumption.
Abstract
Mesenchymal stromal cells (MSCs) are adult pluripotent stem cells which have been widely used in regenerative medicine. As somatic tissue-derived MSCs are restricted by limited donation, quality variations, and biosafety, the past 10 years have seen a great rise in efforts to generate MSCs from human induced pluripotent stem cells (hiPSCs). Past and recent efforts in the differentiation of hiPSCs into MSCs have been centered around two culture methodologies: (1) the formation of embryoid bodies (EBs) and (2) the use of monolayer culture. This protocol describes these two representative methods in deriving MSC from hiPSCs. Each method presents its advantages and disadvantages, including time, cost, cell proliferation ability, the expression of MSC markers, and their capability of differentiation in vitro. This protocol demonstrates that both methods can derive mature and functional MSCs from hiPSCs. The monolayer method is characterized by lower cost, simpler operation, and easier osteogenic differentiation, while the EB method is characterized by lower time consumption.
Introduction
Mesenchymal stromal cells (MSCs) are mesoderm-derived adult pluripotent stem cells1. MSCs are present in almost all connective tissues2. Since MSCs were first discovered in the 1970s and successfully isolated from bone marrow in 1987 by Friedenstein et al.3,4,5, a variety of human somatic (including fetal and adult) tissues have been used for isolating MSCs such as bone, cartilage, tendon, muscle, adipose tissue, and hematopoietic-supporting stroma1,2,6,7. MSCs demonstrate high proliferative capabilities and plasticity to differentiate into many somatic cell lineages and could migrate to injured and inflamed tissues2,8,9. These properties make MSCs a potential candidate for regenerative medicine10. However, somatic tissue-derived MSCs (st-MSCs) are restricted by limited donation, limited cell proliferative capacity, quality variations, and biosafety concern for possible transmission of pathogens, if any, from the donors11,12.
Human induced pluripotent stem cells (hiPSCs) are derived from adult cells reprogramming with transcription factors (Oct4, Sox2, Klf4, and c-Myc), which have similar functions as embryonic stem cells13,14. They can self-renew and possess the potential of differentiating into any type of somatic cells, including MSCs. Compared with st-MSCs, iPSC-MSCs has the advantage of unlimited supply, lower cost, higher purity, convenience in quality control, easy for scale production and gene modification15,16,17.
Due to these advantages of iPSC-MSCs, a variety of methods driving MSC from iPSC have been reported. These differentiation methods have been centered around two culture methodologies: (1) the formation of embryoid bodies (EBs) and (2) the use of monolayer cultures11,18,19,20. Herein, a representative approach for each of the two methodologies was characterized. Furthermore, comparisons between two representative approaches based on time, cost, proliferative ability, expression of MSC biomarkers, and differentiation capability in vitro were also accessed.
Protocol
1. hiPSCs maintenance
- Thawing of hiPSC
- Take out the cells from the liquid nitrogen and quickly thaw cells in a 37 °C water bath. Transfer the thawing cells to a 15 mL tube prepared with 3 mL of iPSC maintenance medium (Table of Materials). Gently mix the medium.
- Centrifuge at 300 x g for 5 min. Remove the supernatant, and gently resuspend the cells in 1 mL of iPSC maintenance medium with 10 µM Y-27632 (pipette the cells up and down 2-3 times).
- Transfer the cells suspension into a 6-well tissue culture plate coated with growth factor reduced (GFR)-extracellular matrix gel (1:100) and 2 mL of iPSC maintenance medium with 10 µM Y-27632 added in advance (about 4 x 104 cells/cm2).
- Culture the cells for 5-6 days (80%-90% confluence) at 37 °C, 5% CO2 with iPSC maintenance media change every day.
- Passage of hiPSC
- Remove the iPSC maintenance medium from the 6-well plate. Wash the hiPSCs once with DPBS.
- Add 700-800 µL of 0.48 mM EDTA solution and incubate for 1 min at room temperature (RT), then remove the digestion solution. Continue to incubate at 37 °C temperature for 3-5 min.
- When cells are digested into sheets (do not digest cells into single cells), add 1 mL of iPSC maintenance medium with 10 µM Y-27632 to terminate digestion. Carefully pipette the cells up and down 2-3 times.
- Transfer the cell suspension into a 6-well tissue culture plate coated with growth factor reduced (GFR)-extracellular matrix gel (1:100) and 2 mL of iPSC maintenance medium with 10 µM Y-27632 added in advance.
NOTE: The passaging ratio ranges from 1:6 to 1:20 (about 4 x 104 cells/cm2), and the mean aggregation size is approximately 50-200 µm. - Culture the cells until 80%-90% confluency (5-6 days) at 37 °C, 5% CO2 with iPSC maintenance media change every day.
2. MSCs differentiation from hiPSCs via EB formation
NOTE: The method is derived from previous literature21,22,23,24. An overview of the method is illustrated in Figure 1. The characteristics of the method are summarized in Table 1.
- Preparation of the medium
- Prepare MSC differentiation medium by supplementing α-MEM with 1% (v/v) GlutaMAX, 10% (v/v) FBS, 10 ng/mL FGF2, and 5 ng/mL TGFβ.
- Prepare MSC maintenance medium by supplementing α-MEM with 1% (v/v) GlutMAX, 10% (v/v) FBS, and 1 ng/mL FGF2.
- Preparation of hiPSCs
- Culture hiPSCs lines (passage at least 2-3 times after thawing) in iPSC maintenance medium on a 6-well tissue culture plate coated with growth factor reduced (GFR)-extracellular matrix gel (1:100) at 37 °C, 5% CO2 until 80%-90% confluency.
- Day 0: EB formation
- Remove the iPSC maintenance medium from the 6-well plate. Wash the hiPSCs once with DPBS.
- Add 700-800 µL of 0.48 mM EDTA solution and incubate for 1 min at RT, then remove the digestion solution. Continue to incubate at 37 °C temperature for 3-5 min.
- When cells are digested into sheets (do not digest cells into single cells), add 2 mL of iPSCs maintenance medium (containing 10 µM Rock inhibitor and 1:100 GFR- extracellular matrix gel) to terminate digestion.
- Transfer the cells to a 6-well low attachment plate. Seed the cells in a ratio of 2 wells of tissue culture plate to 1 well of low attachment plate. Incubate the cells in a shaker (60 rpm/min) at 37 °C, 5% CO2 for 24 h to form spherical EBs.
- Day 1-Day 7: EB differentiation
- Transfer the EBs to the centrifuge tube with a Pasteur pipette (without destroying EBs) and let the EBs naturally sediment for 5-10 min at RT. Then, remove the supernatant.
- Transfer the EBs to a 6-well low attachment plate with 2 mL of MSC differentiation medium. Culture the EBs on the shaker at 37 °C, 5% CO2 for 7 days with medium change once.
- Day 8-Day 17: EB inoculation
- Transfer the EBs to the centrifuge tube with a Pasteur pipette (without destroying EBs) and let the EBs naturally sediment for 5-10 min. Then, remove the supernatant.
- Transfer the EBs to a GFR-extracellular matrix gel-coated 6-well plate with 2 mL of MSC maintenance medium. Culture until 90% confluency (~10 days) with regular media change after cell adhesion.
- Day 18-Day 27: MSCs maturation and expansion
- Treat the EB-derived culture with a dissociation solution at 37 °C for 5-10 min (digestion time varies due to the presence of different cell types).
- When part of the cells are digested into single cells, add 2 mL of the MSC maintenance medium to terminate digestion and transfer the cell suspension to a centrifuge tube. Wash the remaining undigested cells once with DPBS and digest again. Repeat several times until all cells are digested into single cells. Centrifuge the cell suspension at 250 x g for 5 min, then remove the supernatant.
- Seed the cells on a Gelatin-coated culture plate (about 2 x 105 cells/cm2). Culture until 90% confluency in MSC maintenance medium with medium change regularly. Designate this generation of cells as passage 0 (P0) at this point.
- In order to make MSC pure and mature, continue to passage the cells twice at a 1:3 split ratio in approximately 6 days. Most of the cells present fibroblast-like morphology (spindle-shaped).
3. MSCs differentiation from hiPSCs via monolayer culture
NOTE: The method is derived from previous literature25,26,27,28. An overview of this method is illustrated in Figure 1. The characteristics of the method are summarized in Table 1.
- Preparation of the medium
- Prepare the MSC maintenance medium by supplementing α-MEM with 1% (v/v) GlutMAX, 10% (v/v) FBS, and 1 ng/mL FGF2.
- Preparation of hiPSCs
- Culture hiPSCs lines (passage at least 2-3 times after thawing) in the iPSC maintenance medium on a 6-well tissue culture plate coated with growth factor reduced (GFR)-extracellular matrix gel (1:100) at 37 °C, 5% CO2 until 50%-60% confluency.
- Day 0-Day 13: Differentiation by direct monolayer cultures
- Remove the iPSC maintenance medium from the 6-well plate.
- Directly add 2 mL of the MSC maintenance medium and culture for 14 days at 37 °C, 5% CO2, with the media change every day.
- Day 14-Day 35: MSCs maturation by repeated passage
- Treat the monolayer cultures with a dissociation solution at 37 °C for 5-10 min (digestion time varies due to the presence of different cell types).
- When the cells are digested into single cells, add 2 mL of MSC maintenance medium to terminate digestion and transfer the cell suspension to centrifuge tube. Wash the remaining undigested cells once with DPBS and digest again. Repeat several times until all cells are digested into single cells. Centrifuge the cell suspension at 250 x g for 5 min, then remove the supernatant.
- Seed the cells on a gelatin-coated culture dish (about 2 x 105 cells/cm2). Culture until 90% confluency in MSC maintenance medium with media change regularly. Designate this generation of cells as passage 0 (P0) at this point.
- In order to make MSC pure and mature, continue to passage the cells 6 times at a 1:3 split ratio in approximately 18 days. Most of the cells present fibroblast-like morphology (spindle-shaped).
4. Surface antigens analysis of hiPSC-driving MSCs by flow cytometry
NOTE: Similar to the surface antigens of bone marrow-derived MSCs, hiPSCs-driving MSC express CD105, CD73 and CD90, but do not express CD45, CD3429. In addition, hiPSCs can be used as negative control cells. Surface antigens analysis of hiPSC-driving MSCs and hiPSCs by flow cytometry are shown in Figure 2.
- Treat the hiPSC-driving MSCs with a dissociation solution at 37 °C for 2-3 min. Treat the hiPSCs with 0.48 mM EDTA dissociation reagent and incubate for 1 min at RT, then remove the dissociation reagent. Continue to incubate at 37 °C temperature for 3-5 min.
- When the cells are digested into single cells, add medium (MSC maintenance medium to MSCs and iPSC maintenance medium to iPSCs) to terminate digestion.
- Centrifuge the cell suspension at 350 x g for 5 min, then remove the supernatant.
- Wash the cells once with cold DPBS, centrifuge at 350 x g for 5 min, and then remove the supernatant.
- Count and resuspend the cells in cold 10% FBS-DPBS (v/v) solution at 10 x 106 cells/mL and distribute 100 µL/tube of cell suspension (1 x 106 cells/tube) into 1.5 mL centrifuge tubes.
- Add 5 µL of human Fc receptor-blocking solution to 100 µL of cell suspension. Incubate for 5-10 min at RT to perform nonspecific blocking.
- Centrifuge at 350 x g for 5 min and remove the supernatant. Wash the cells twice with a cold 2% FBS-DPBS (v/v) solution.
- Resuspend the cells in cold 100 µL of 2% FBS-DPBS (v/v) solution.
- Add 5 µL (1 test) of anti-CD34-FITC and 5 µL (1 test) of anti-CD45-APC to the 100 µL cell
Suspension. Add 5 µL (1 test) of anti-CD73-APC, 5 µL (1 test) of anti-CD90-FITC and 5 µL (1 test) of anti-CD105-PE to another 100 µL cell suspension tube. Add 5 µL of human IgG1 isotype control FITC, human IgG1 isotype control PE and human IgG1 isotype control APC to the third 100 µL cell suspension tube. Incubate for 15-20 min on ice in the dark. Meanwhile, prepare single-staining beads. - Wash the cells twice with a cold 2% FBS-DPBS (v/v) solution.
- Add 300 µL of cold 2% FBS-DPBS (v/v) solution to resuspend the cells, and filter via 200 mesh screen filter.
- Analyze the samples using flow cytometry. Analyze the cell suspension stained with isotype control antibodies and set gate. Then analyze cell suspension stained with target antibodies.
5. Osteogenic differentiation of hiPSC-driving MSCs
NOTE: The hiPSC-driving MSCs possess osteogenic differentiation potential (Figure 3A, B). The protocol for osteogenic differentiation is given below.
- Prepare osteogenic induction medium by supplementing α-MEM with 10% FBS, 100 nM dexamethasone, 10 mM beta-glycerophosphate, and 100 µM ascorbic acid.
- Seed 1 x 105 MSCs into a gelatin-coated 48-well plate and culture until it is 60%-70% confluent.
- Remove the medium and add 0.3 mL of osteogenic induction medium. Culture the cells in an osteogenic induction medium for 14 days at 37 °C, 5% CO2, with media change every 3 days.
- Remove the medium and wash with DPBS once.
- Add 0.3 mL of 4% PFA and incubate at RT for 30 min.
- Remove PFA. Rinse the cells with 0.3 mL of DPBS for 10 min at RT and repeat 3 times.
- Remove the DPBS, add 0.2 mL of Alizarin red staining solution, and incubate for 30 min at RT.
- Remove the staining solution, rinse the cells with 0.3 mL DPBS for 10 min at RT, and repeat 3 times.
- Observe the staining of calcium deposition under a light microscope.
6. Adipogenic differentiation of hiPSC-driving MSCs
NOTE: The hiPSC-driving MSCs possess adipogenic differentiation potential (Figure 3C, D). The protocol for adipogenic differentiation is given below.
- Preparare adipogenic induction medium by supplementing α-MEM with 10% FBS, 1 µM dexamethasone, 1 µM IBMX, 10 µg/mL insulin, and 100 µM indomethacin. Preparare adipogenic maintenance medium by supplementing α-MEM with 10% FBS, and 10 µg/mL insulin.
- Seed 1 x 105 MSCs into a gelatin-coated 48-well plate and culture until the cell density reaches 90%.
- Remove the medium and add 0.3 mL of adipogenic induction medium. Continue to culture the cells in the adipogenic induction medium for 4 days at 37 °C, 5% CO2.
- Remove the medium and add 0.3 mL of adipogenic maintenance medium. Continue to culture the cells in the adipogenic maintenance medium for 3 days at 37 °C, 5% CO2.
- Repeat steps 6.3 and 6.4 2-3 times until adipocyte differentiation and maturation.
- Remove the medium and wash with DPBS once.
- Add 0.3 mL of 4% PFA and incubate at RT for 10 min. Remove the PFA and rinse 2 times with DPBS.
- Apply Oli Red O stain per the manufacturer's instruction and observe the lipid droplets under a light microscope.
7. Chondrogenic differentiation of hiPSC-driving MSCs
NOTE: The hiPSC-driving MSCs possess chondrogenic differentiation potential (Figure 3E, F). The protocol for chondrogenic differentiation is given below.
- Prepare chondroblast induction medium by supplementing α-MEM with 10% FBS, 100 nM dexamethasone, 1% Insulin-Transferrin-Selenium (ITS), 10 µM ascorbic acid, 1 mM sodium pyruvate, 50 µg/mL proline, 0.02 nM transforming growth factor β3 (TGFβ3), and 0.5 µg/mL bone morphogenetic protein 6 (BMP-6).
- Collect 2.5 x 105 hiPSC-driving MSCs and centrifuge the cells at 150 x g for 5 min, then remove the supernatant. Suspend the cells in 1 mL of α-MEM and then centrifuge at 150 x g for 5 min.
- Remove the supernatant and suspend the cells in 0.5 mL of chondrogenic induction medium. Centrifuge the cells at 150 x g for 5 min.
- Directly unscrew the lid and culture for 21 days with chondrogenic induction media change every 3 days at 37 °C, 5% CO2. Flick the centrifuge tube gently to suspend the chondrogenic pellets (without destroying chondrogenic pellets) every day during chondrogenic differentiation.
- Remove the supernatant and wash with 0.5 mL of PBS twice.
- Add 0.3 mL of 4% PFA and fix for 24 h.
- Paraffin embed the chondrogenic pellets, and then section them (3 µm). Stain the sections in toluidine blue (1%) for 30 min.
- Observe the extracellular chondrocyte matrix under a light microscope.
Representative Results
Following the protocol (Figure 1A), hiPSCs were differentiated into MSCs via the EB formation and monolayer culture methods. During differentiation, the cells showed different representative morphologies (Figure 1B,C).
As shown in Figure 1B, the hiPSCs colonies display typical compact morphology before differentiation with a clear border composed of tightly packed cells. Uniform spherical EBs formed after hiPSCs dissociating and culturing for 24 h on the shaker. During day 1 to day 7 of culture in MSCs differentiation medium, the smooth edge of EB became rough, and the volume of EB grew big. From day 8 to day 17, after transferring the EBs to a GFR-extracellular matrix gel-coated 6-well plate, EBs gradually adhered to the plate, and many adherent monolayer cells spread around the EBs. When the cells reached 90% confluency on day 18, the cells were digested and seeded on a gelatin-coated culture plate. On day 19, the cell adhered and showed a polygonal shape. Consecutively, the cells were passaged twice when they were 90% confluent. The derived MSCs gradually matured and showed a typical spindle shape, and the colony grew in a swirl.
As shown in Figure 1C, the volume of the cells increased and spread around the colony after replacing the iPSC maintenance medium with the MSC maintenance medium for 24 h. While cultured in an MSC maintenance medium, the cells gradually proliferated and formed multilayer adherent cells. On day 14, the cells were digested and seeded on a gelatin-coated culture plate. On day 15, the cell adhered and showed a polygonal shape. Consecutively, the cells were passaged 6 times when they were 90% confluent. The derived MSCs gradually matured and showed a typical spindle shape, and the colony grew in a swirl.
The surface antigens of hiPSCs and hiPSC-driving MSCs were analyzed by flow cytometry (Figure 2). As shown in Figure 2C, hiPSCs were positive for CD90 and negative for CD34, CD45, CD73, and CD105. After differentiating hiPSCs into MSCs via both methods, driving MSCs were positive for CD90, CD73 and CD105, and negative for CD34, and CD45 (Figure 2A,B).
The differentiation capability of hiPSC-driving MSCs was investigated by osteogenic, adipogenic, and chondrogenic differentiation. As shown in Figure 3, both hiPSC-driving MSCs of the two methods differentiated into osteoblast, adipocyte, and chondrocyte. The hiPSC-driving MSCs of the monolayer method formed more calcium deposits than the EB method (Figure 3B). The two methods had no significant difference in adipogenic differentiation and chondrogenic differentiation ability (Figure 3D,F).
The proliferation ability of hiPSC-driving MSCs was examined by continuous passage culture. As shown in Figure 3G, the hiPSC-driving MSCs of both methods can be passaged for more than 20 passages and still maintain a rapid proliferation ability.
The comparison between the two approaches shown in Table 1 is based on differentiation time, cost, cell proliferation ability, MSC markers' expression, and their differentiation capability in vitro.
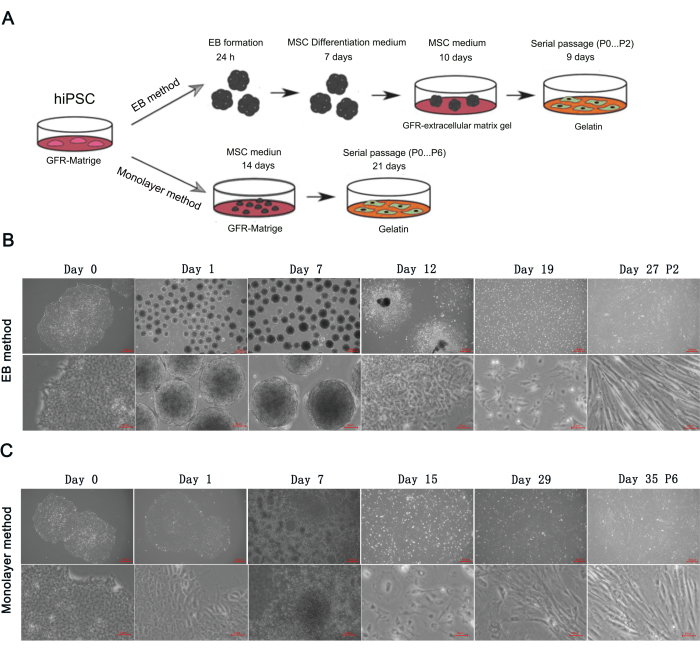
Figure 1: Differentiating hiPSCs into MSCs via EB formation method and monolayer culture method. (A) Schematic showing the differentiation of hiPSCs into MSCs via EB formation and monolayer culture. Representation morphology of cells at key phages during MSCs derivation from hiPSCs via (B) EB formation and (C) monolayer culture. Scale bars: 300 µm and 50 µm. Please click here to view a larger version of this figure.
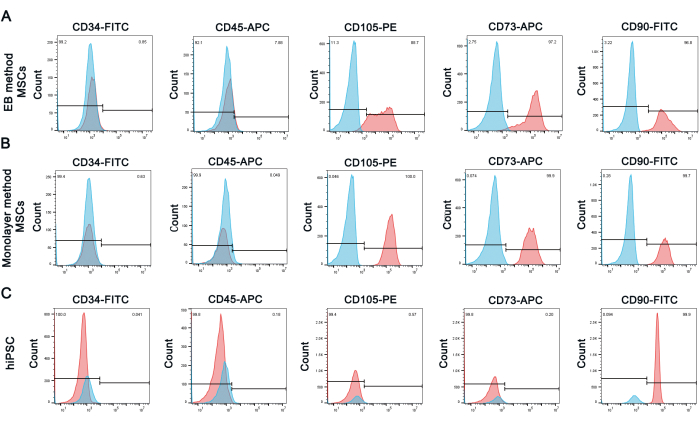
Figure 2: hiPSC-driving MSCs surface antigens analysis by flow cytometry. Expression percentages of MSC surface antigens in iPSC-driving MSCs via (A) EB formation and (B) monolayer culture. (C) hiPSCs were used as the negative control. MSCs negative markers: CD34, CD45; MSCs positive markers: CD73, CD90, and CD105. hiPSCs negative markers: CD34, CD45, CD73 and CD105; hiPSCs positive markers: CD90. Please click here to view a larger version of this figure.
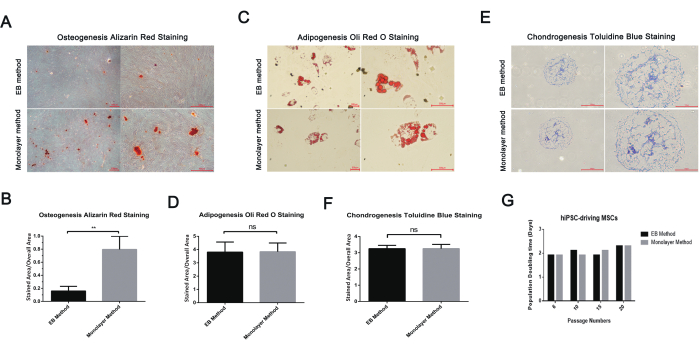
Figure 3: Three-line differentiation and proliferation ability of hiPSC-driving MSCs. (A) Alizarin red staining of calcium deposits of hiPSC-driving MSCs in osteogenic differentiation medium for 2 weeks. Scale bars: 300 µm. (B) Quantification of Alizarin red S staining by ImageJ analysis. (C) Oli red O staining of lipid droplets of hiPSC-driving MSCs in adipogenic differentiation medium for 2 weeks. Scale bars: 300 µm. (D) Quantification of Oli Red O staining by ImageJ analysis. (E) Toluidine blue staining of extracellular chondrocyte matrix. Scale bars: 300 µm and 150 µm. (F) Quantification of toluidine blue staining by ImageJ analysis. (G) hiPSC-driving MSCs population doubling time calculation. Please click here to view a larger version of this figure.
| Comparison | EB formation method | Monolayer cultures method |
| Differentiation time | 27 days | 35 days |
| Cost | High | Low |
| Proliferation Speed | Fast | Fast |
| Proliferation ability | ≥20 passage | ≥20 passage |
| Expression of MSC markers | CD73/CD90/CD105 positive,CD34/CD45 nagative | CD73/CD90/CD105 positive,CD34/CD45 nagative |
| Capability of differentiation | Adipogenic differentiation, chondrogenic differentiation and osteogenic differentiation capability | Adipogenic differentiation, chondrogenic differentiation and stronger osteogenic differentiation capability |
| Operation | Complicated | Simple |
Table 1: Characteristics of the two methods for differentiating hiPSCs into MSCs.
Discussion
In this protocol, two representative methods of differentiating hiPSCs into MSCs were examined20,21,22,23,24,25,26,27,28,30. Both methods were capable of derivating MSCs from hiPSCs. The hiPSC-derived MSCs were confirmed by cell morphology (Figure 1), surface antigens (Figure 2), and their ability to differentiate (Figure 3).
Both methods shared the same MSC maintenance medium, whereas the EB method also needed an MSC differentiation medium with TGF-β and FGF2. In addition, the EB method required low attachment of 6 well plates and a special shaker that can be placed in a carbon dioxide incubator. Therefore, the cost of the EB method was higher than that of the monolayer method18,20. The EB method seemed to be more complicated than the monolayer method. However, the differentiation time of the EB method was shorter, which avoided multiple passages to enrich MSCs.
We found that the tolerance to ambient conditions of the EB method was stronger than that of the monolayer method. Both methods were influenced by iPSC status, human interference, and culture environment11,20. However, we found that the success ratio of the EB method was much higher than that of the monolayer method. The key stage of the EB method was the EB differentiation stage from day 1 to day 7. During days 1-7, if EBs gradually become smaller or broken with a lot of floating dead cells in the culture medium or form vacuolate EBs, these unqualified EBs have difficulty adhering to the gelatin plate and little, if any, adherent cells crawling out of the EBs. From day 1 to day 7, qualified EBs were uniform and would be slightly enlarged, smooth edges of which gradually become rough with cells protruding. After being transferred to the gelatin plate, the EBs adhered to the plate quickly, and many adherent monolayer cells spread around the EBs. If EBs are qualified during days 1-7, subsequent differentiation is easy to achieve. In contrast, the monolayer method could fail at any stage. From day 1 to day 14, adherent cells may fall off in patches, indicating a failure of differentiation. Normally, adherent cells proliferate and form multilayer adherent cells. After day 14, the monolayer method would still fail because few cells are attached to the dish. During days 0-13 of the monolayer method, we observed various cell morphology in the dish. However, the cell morphology of the EB method on days 8-17 was homogeneous. Compared with the monolayer method, we suspect that the EB method is more similar to the embryonic development process and is a more controllable programmed differentiation method.
Both methods could be used for more than 20 passages and still maintain a rapid proliferation ability11. The calcium deposits of osteogenic differentiation in the monolayer method were more than those of the EB method. Stem cell-specific serum must be used to avoid the effect of cytokines in the serum on differentiation. We observed that the cells would stop proliferating if the cell density was too low. So, after reaching confluency, the cells must be passaged at a 1:3 split ratio. No antibiotics were used during the differentiation; therefore, strict observance of good laboratory practices (GLP) is mandatory for this experimental setup.
In conclusion, there were advantages and disadvantages for each method tested in this protocol. Both methods can generate MSC from hiPSCs, the choice is based on the user's requirements.
Disclosures
The authors have nothing to disclose.
Acknowledgements
We are extremely grateful to all members of the Mao and Hu Lab, past and present, for the interesting discussions and great contributions to the project. We are thankful to the National Clinical Research Center for Child Health for the great support. This study was financially supported by the National Natural Science Foundation of China (U20A20351 to Jianhua Mao, 82200784 to Lidan Hu), the Natural Science Foundation of Zhejiang Province of China (No. LQ22C070004 to Lidan Hu).
Materials
| Alizarin red staining kit | Beyotime Biotechnology | C0148S | |
| Anti-human-CD105 (PE) | Biolegend | 323206 | |
| Anti-human-CD34 (FITC) | Biolegend | 343503 | |
| Anti-human-CD45 (APC) | Biolegend | 304011 | |
| Anti-human-CD73( APC) | Biolegend | 344006 | |
| Anti-human-CD90 (FITC) | Biolegend | 328108 | |
| Ascorbic acid | Solarbio | A8100 | |
| BMP-6 | Novoprotein | C012 | |
| Carbon dioxide level shaker | Crystal | CO-06UC6 | |
| Compensation Beads | BioLegend | 424601 | |
| CryoStor CS10 | STEMCELL Technology | 07959 | |
| Dexamethasone | Beyotime Biotechnology | ST1254 | |
| DMEM/F12 medium | Servicebio | G4610 | |
| Fetal bovine serum | HAKATA | HS-FBS-500 | |
| FGF2 | Stemcell | 78003.1 | |
| Gelatin | Sigma-Aldrich | G2500-100G | |
| GlutaMAX | Gibco | 35050061 | |
| human IgG1 isotype control APC | BioLegend | 403505 | |
| human IgG1 isotype control FITC | BioLegend | 403507 | |
| human IgG1 isotype control PE | BioLegend | 403503 | |
| Human TGF-β1 | Stemcell | 78067 | |
| Human TruStain FcX | BioLegend | 422301 | |
| IBMX | Beyotime Biotechnology | ST1398 | |
| Indomethacin | Solarbio | SI9020 | |
| Insulin | Beyotime Biotechnology | P3376 | |
| iPSC maintenance medium | STEMCELL Technology | 85850 | |
| ITS Media Supplement | Beyotime Biotechnology | C0341-10mL | |
| Matrigel, growth factor reduced | BD Corning | 354230 | |
| Oli Red O staining kit | Beyotime Biotechnology | C0158S | |
| Proline | Solarbio | P0011 | |
| Sodium pyruvate | ThermoFisher | 11360-070 | |
| TGFβ3 | Novoprotein | CJ44 | |
| Toluidine blue staining kit | Solarbio | G2543 | |
| TrypLE Express Enzyme(1x) | Gibco | 12604013 | |
| Ultra-Low Attachment 6 Well Plate | Costar | 3471 | |
| Versene | Gibco | 15040-66 | |
| Y-27632 | Stemcell | 72304 | |
| α-MEM | Hyclone | SH30265 | |
| β-glycerophosphate | Solarbio | G8100 |
References
- Weng, Z., et al. Mesenchymal stem/stromal cell senescence: Hallmarks, mechanisms, and combating strategies. Stem Cells Translational Medicine. 11 (4), 356-371 (2022).
- Soliman, H., et al. Multipotent stromal cells: One name, multiple identities. Cell Stem Cell. 28 (10), 1690-1707 (2021).
- Friedenstein, A. J., Chailakhyan, R. K., Gerasimov, U. V. Bone marrow osteogenic stem cells: in vitro cultivation and transplantation in diffusion chambers. Cell and Tissue Kinetics. 20 (3), 263-272 (1987).
- Friedenstein, A. J., et al. Precursors for fibroblasts in different populations of hematopoietic cells as detected by the in vitro colony assay method. Experimental Hematology. 2 (2), 83-92 (1974).
- Friedenstein, A. J., Gorskaja, J. F., Kulagina, N. N. Fibroblast precursors in normal and irradiated mouse hematopoietic organs. Experimental Hematology. 4 (5), 267-274 (1976).
- El Agha, E., et al. Mesenchymal stem cells in fibrotic disease. Cell Stem Cell. 21 (2), 166-177 (2017).
- Mushahary, D., Spittler, A., Kasper, C., Weber, V., Charwat, V. Isolation, cultivation, and characterization of human mesenchymal stem cells. Cytometry A. 93 (1), 19-31 (2018).
- Ullah, M., Liu, D. D., Thakor, A. S. Mesenchymal stromal cell homing: Mechanisms and strategies for improvement. iScience. 15, 421-438 (2019).
- Regmi, S., et al. Enhanced viability and function of mesenchymal stromal cell spheroids is mediated via autophagy induction. Autophagy. 17 (10), 2991-3010 (2021).
- Hoang, D. M., et al. Stem cell-based therapy for human diseases. Signal Transduction and Targeted Therapy. 7 (1), 272 (2022).
- Jiang, B., et al. Concise review: Mesenchymal stem cells derived from human pluripotent cells, an unlimited and quality-controllable source for therapeutic applications. Stem Cells. 37 (5), 572-581 (2019).
- Soontararak, S., et al. Mesenchymal stem cells (MSC) derived from induced pluripotent stem cells (iPSC) equivalent to adipose-derived MSC in promoting intestinal healing and microbiome normalization in mouse inflammatory bowel disease model. Stem Cells Translational Medicine. 7 (6), 456-467 (2018).
- Di Baldassarre, A., Cimetta, E., Bollini, S., Gaggi, G., Ghinassi, B. Human-induced pluripotent stem cell technology and cardiomyocyte generation: Progress and clinical applications. Cells. 7 (6), 48 (2018).
- Shi, Y., Inoue, H., Wu, J. C., Yamanaka, S. Induced pluripotent stem cell technology: a decade of progress. Nature Reviews. Drug Discovery. 16 (2), 115-130 (2017).
- Levy, O., et al. Shattering barriers toward clinically meaningful MSC therapies. Science Advances. 6 (30), eaba6884 (2020).
- Zhao, C., Ikeya, M. Generation and applications of induced pluripotent stem cell-derived mesenchymal stem cells. Stem Cells International. 2018, 9601623 (2018).
- Path, G., Perakakis, N., Mantzoros, C. S., Seufert, J. Stem cells in the treatment of diabetes mellitus – Focus on mesenchymal stem cells. Metabolism. 90, 1-15 (2019).
- Zhou, Y., et al. One-step derivation of functional mesenchymal stem cells from human pluripotent stem cells. Bio-Protocol. 8 (22), e3080 (2018).
- Hua, Z., et al. Low-intensity pulsed ultrasound promotes osteogenic potential of iPSC-derived MSCs but fails to simplify the iPSC-EB-MSC differentiation process. Frontiers in Bioengineering and Biotechnology. 10, 841778 (2022).
- Dupuis, V., Oltra, E. Methods to produce induced pluripotent stem cell-derived mesenchymal stem cells: Mesenchymal stem cells from induced pluripotent stem cells. World Journal of Stem Cells. 13 (8), 1094-1111 (2021).
- Zhang, W., et al. Aging stem cells. A Werner syndrome stem cell model unveils heterochromatin alterations as a driver of human aging. Science. 348 (6239), 1160-1163 (2015).
- Liu, G. H., et al. Modelling Fanconi anemia pathogenesis and therapeutics using integration-free patient-derived iPSCs. Nature Communications. 5, 4330 (2014).
- Kubben, N., et al. Repression of the antioxidant NRF2 pathway in premature aging. Cell. 165 (6), 1361-1374 (2016).
- Duan, S., et al. PTEN deficiency reprogrammes human neural stem cells towards a glioblastoma stem cell-like phenotype. Nature Communications. 6, 10068 (2015).
- Zhang, J., et al. Exosomes released from human induced pluripotent stem cells-derived MSCs facilitate cutaneous wound healing by promoting collagen synthesis and angiogenesis. Journal of Translational Medicine. 13, 49 (2015).
- Hu, G. W., et al. Exosomes secreted by human-induced pluripotent stem cell-derived mesenchymal stem cells attenuate limb ischemia by promoting angiogenesis in mice. Stem Cell Research & Therapy. 6 (1), 10 (2015).
- Kang, R., et al. Mesenchymal stem cells derived from human induced pluripotent stem cells retain adequate osteogenicity and chondrogenicity but less adipogenicity. Stem Cell Research & Therapy. 6 (1), 144 (2015).
- Wang, L. T., et al. Differentiation of mesenchymal stem cells from human induced pluripotent stem cells results in downregulation of c-Myc and DNA replication pathways with immunomodulation toward CD4 and CD8 cells. Stem Cells. 36 (6), 903-914 (2018).
- Dominici, M., et al. Minimal criteria for defining multipotent mesenchymal stromal cells. The International Society for Cellular Therapy position statement. Cytotherapy. 8 (4), 315-317 (2006).
- Kim, S., Kim, T. M. Generation of mesenchymal stem-like cells for producing extracellular vesicles. World Journal of Stem Cells. 11 (5), 270-280 (2019).

