Creating Anatomically Accurate and Reproducible Intracranial Xenografts of Human Brain Tumors
Summary
The brain is a unique site with qualities that are not well represented by in vitro or ectopic analyses. Orthotopic mouse models with reproducible location and growth characteristics can be reliably created with intracranial injections using a stereotaxic fixation instrument and a low pressure syringe pump.
Abstract
Orthotopic tumor models are currently the best way to study the characteristics of a tumor type, with and without intervention, in the context of a live animal – particularly in sites with unique physiological and architectural qualities such as the brain. In vitro and ectopic models cannot account for features such as vasculature, blood brain barrier, metabolism, drug delivery and toxicity, and a host of other relevant factors. Orthotopic models have their limitations too, but with proper technique tumor cells of interest can be accurately engrafted into tissue that most closely mimics conditions in the human brain. By employing methods that deliver precisely measured volumes to accurately defined locations at a consistent rate and pressure, mouse models of human brain tumors with predictable growth rates can be reproducibly created and are suitable for reliable analysis of various interventions. The protocol described here focuses on the technical details of designing and preparing for an intracranial injection, performing the surgery, and ensuring successful and reproducible tumor growth and provides starting points for a variety of conditions that can be customized for a range of different brain tumor models.
Introduction
In vitro studies of brain tumor cells are invaluable for dissecting molecular mechanisms driving growth, survival, migration, and invasion of cancer cells; cultured cell experiments can define signaling pathways, suggest potential therapeutic targets, and characterize cellular response to drug treatment. But in vitro systems are far too simplistic to predict organismal response to pharmaceuticals; they lack the physiological reactions, immune responses, cell microenvironment, and overall heterogeneity of living animal systems. Genetically engineered models can be invaluable, when available, but molecular differences exist between species and murine cells may not recapitulate events in human processes, resulting in significant discrepancies when comparing animal models to clinical observations1. Mouse xenograft models involving subcutaneous (SQ) injection of human brain tumor cell lines under the skin of the flank are easy to perform and measure; they can be used to address effects of gene modification and drug administration/delivery, metabolism and toxicity. Significant drawbacks, however, limit the utility of SQ models. The microenvironment does not recapitulate that of a naturally occurring brain tumor: the interactions of various cell types and tissues; the local vasculature, and myriad other factors unique to the brain cannot be replicated. To more accurately reproduce the unique milieu of a naturally occurring brain tumor and test the effects of pharmaceutical interventions, a mouse orthotopic model should be utilized. Furthermore, orthotopic techniques may be used as part of a genetically engineered approach in which human primary non-cancerous cells (differentiated or progenitor) are genetically modified and injected into the relevant site of a mouse, with or without human stroma cells, resulting in tumorigenesis similar to that seen in humans1.
This article describes a methodology to precisely and reproducibly create brain tumors in mice. Using this technique, the user can accurately inject a small aliquot of suspended cells into a specified location of the fronto-parieto-temporal region of the mouse cerebral cortex. Mouse mortality is extremely low; in our hands, no mice have died from surgical complications after 185 procedures. Characteristics of the resultant tumor can be compared with that of typical human clinical tumors; for example: rapidity of growth, degree of necrosis, extent of invasion, heterogeneity of cell type, presence of mitotic cells, markers of proliferation and apoptosis, etc. Cell lines or disaggregated human tissue or tumor samples can then be evaluated based on their ability to simulate actual clinical presentation. Pharmaceuticals, selected based on their performance in cell culture, can be tested in the context of a functioning metabolism, circulatory system, and blood-brain barrier as they exist in an animal burdened with a tumor, all in a relevant architectural context. Furthermore, the cells chosen for injection may be genetically modified to investigate the impact of specific knockdowns, deletions, knock-ins, mutations, etc. on tumor growth and survival.
A number of publications document tumors studies using a variety of intracranial techniques. Yamada et al. did a detailed study of the injection of dye and of U87 cells and found that minimizing volume and injection rate produced the best tumor2. Brooks et al. found superior reproducibility and efficiency using a microprocessor-controlled injector rather than a manual method to deliver viral vectors; their conclusions regarding optimal injection parameters are applicable to cell delivery3. Shankavaram et al. showed that glioblastoma multiforme (GBM) cell lines injected orthotopically (using a manual method) into the brain recapitulated the gene expression profile of the clinical tumors more closely than either in vitro or SQ xenografts, supporting the use of intracranial models for preclinical studies4. Giannini et al. injected cells from human surgical specimens that had been sustained in the flanks of nude mice by serial passaging into the brains of additional mice, and showed that this approach preserved patient tumor gene alterations in the model5. Similar results were reported by Yi et al6. Using a stereotaxic setup, carefully defined injection site, and a slow and steady injection rate, they obtained reproducible brain tumors with consistent growth rates and high (100%) engraftment rate. The validity of this technique has therefore been well established; a literature search suggests that the applications of this technique are extensive. Carty et al. used intracranial injections to successfully deliver viral vectors expressing therapeutic genes into the frontal cortex of transgenic model of Alzheimer’s disease7. Thaci et al. described the use of intracranial injections to deliver therapeutic oncolytic adenovirus in a neural stem cell based carrier into nude mice already carrying orthotopically injected GBM tumors8. Clearly, intracranial injections are a versatile and effective tool for preclinical research. Earlier publications in The Journal of Visualized Experiments describe fundamental approaches9-11, but we take the concept of intracranial tumor injection and orthotopic modeling to a higher level of precision using easy-to-master technology.
Protocol
All described procedures were reviewed and approved by our institutional animal care and use committee.
1. Plan the Experiment
- Choose cells to be injected. Cells from a variety of sources are candidates for injection: adherent cell culture lines, genetically modified clones, neurosphere cells, primary cultures, or disaggregated tumors. The type of model desired will define the most appropriate site of injection.
- Determine cell number for injection. The number of cells required to form tumors varies with cell line and must be determined empirically; tumor growth rate depends heavily on cell type, cell number and tissue culture passage number. Cell numbers ranging from 1 x 104 to 2 x 105 or more per injection have produced tumors of varying growth rates.
- Limit the volume for injection. The cells should be suspended in the smallest volume of serum free media or phosphate buffered saline (PBS) that allows for smooth, easy passage through a 26 G needle. The smaller the volume actually delivered into the brain, the more precise and defined the resulting tumor; optimal results are obtained by injecting volumes ranging from 3 to 6 µl. Plan to prepare a final total volume of at least 50 µl of suspended cells, regardless of the number of injections planned, to allow accurate measurement, mixing and sampling.
- Choose appropriate mice for the model. Murine models of human tumors typically utilize young immune-compromised mice to avoid immune mediated graft rejection. It is not necessary for the work to take place in a biological safety cabinet if appropriate precautions are taken, including decontamination, disinfection, and sterilization of materials and equipment. The work shown here has been done in athymic nude mice, male and female, between 6 and 12 weeks of age. Mice that weigh less than 20 g can be difficult to position on a stereotaxic frame and may be more susceptible to hypothermia.
2. Assemble the Equipment
- Obtain and decontaminate a mouse stereotaxic frame, alignment console, and mouse gas anesthesia head holder, microsyringe pump, heat pad, anesthesia machine, fiber optic work lamp, and variable speed rotary drill. Clean and decontaminate all equipment.
- Program the injection parameters (including injection volume, flow rate, rate units, syringe type) and other variables necessary for the operation of the syringe pump. Optimize settings for each particular model.
NOTE: In these injections the settings were 3,000 nl samples at a rate of 400 nl/min (rate units “M”) using a 25 µl syringe (Device Type “E”) in the Non-Grouped (“N”) mode. - Clean a precision microsyringe with 26 G needle with several rinses of sterile deionized water (diH2O) and 70% ethanol (EtOH), doing a final rinse with diH2O. Some but not all models are designed to be autoclaved. Ensure that the plunger movement is smooth and free.
- Obtain anesthesia delivery equipment. Isoflurane is the preferred anesthetic as it is easy to regulate the depth and duration of anesthesia. Ensure that the stereotaxic unit is equipped with a mouse anesthesia gas attachment and that the necessary tubing and connectors are in place to deliver the gas mixture from the anesthesia machine to the mouse.
- Autoclave surgical tools, including fine tip scissors, two forceps/hemostats (with teeth) for suturing, medium sized scissors, medium- and fine-tipped tweezers, and at least two 1 mm dental drill bits. Keep instruments clean during the procedure with 70% EtOH, disinfectant, or a bead sterilizer.
- Obtain isoflurane, analgesic, ophthalmic ointment/lubricant, antibiotic ointment, saline, 30% hydrogen peroxide (H2O2), antiseptic, and bone wax (if desired) from a veterinary or medical supply. Prepare sterile diH2O and 70% EtOH. Disposables such as gauze pads, sterile dressings, sutures, ethanol swabs, cotton tipped swabs, sterile surgical gloves, surgical blades and more are itemized in the materials list. A fine tip permanent marker for marking the skull should be decontaminated and dedicated for surgical use.
3. Prepare Cells for Injection
- In the experiments shown here, select an immortalized human GBM cell line. Determine the number of cells (2 x 105) and the volume of suspension to be injected (3 µl) empirically; details may vary with cell type. Use a volume of at least 50 µl to facilitate accurate measuring, mixing, and injection.
- Trypsinize cells and resuspend in media or PBS. Perform the following steps just prior to cell injection:
- Remove media from cells by aspiration and rinse with 10 ml PBS per 100 mm plate; remove PBS.
- Add 1 ml trypsin to each 100 mm plate and incubate at RT just long enough to yield a single cell suspension.
- Stop trypsin activity with 5 ml Dulbecco’s Modified Eagle Medium (DMEM) + 10% Fetal Bovine Serum (FBS) per 100 mm plate.
- Pellet cells by centrifugation at 450 x g and resuspend in ~5 ml serum free DMEM (SF-DMEM) per 100 mm plate.
- Count viable cells using a hemacytometer or an automated cell counter.
- Adjust the live cell density to 2 x 105 cells/3 µl (or as determined by experimentation) by pelleting the cells and resuspending in SF-DMEM.
- Keep cells chilled on ice or a chill pack (do not allow cells to freeze). Mix gently by finger flicking.
4. Anesthetize and Prepare Mouse for Surgery
- Induce anesthesia using ~5% isoflurane (oxygen flow ~2 L/min) or as directed by veterinarian. Induction should take 2 to 3 min. Maintain mouse at an adequate depth of anesthesia with ~3% isoflurane.
- Check depth of anesthesia by toe pinch, adjust isoflurane if indicated and continue to monitor breathing and toe pinch response throughout the procedure. Cover mouse with gauze pads for warmth and monitor mouse regularly throughout the procedure, which may take 45 min to 1 hr, depending on the rate of injection.
- Position the mouse in stereotaxic frame by carefully sliding the palate bar over the tongue and into the mouth. Hook the front incisors into the tooth hole. Use ear bars to stabilize the head, with the ear-to-eye surface as near horizontal as possible. Do not force bars into ear canal. It is critical to position the head securely: check side-to-side and up-and-down motion gently with fingertips. Use the adjustable controls of the stereotaxic unit to optimize fit.
- Lubricate the eyes with ophthalmic ointment. Clean the skin between ears and eyes twice with alternating applications of a povidone-iodine antiseptic and 70% EtOH.
- Inject chosen analgesic subcutaneously (SQ) into flank (or as directed).
5. Perform Injection
- Determine the injection site. The injection site may vary with the size and strain of mouse and the type of cell. Cells injected too close to the ventricles may lead to cranio-spinal spread of disease. Cells injected too shallow may grow out through the needle track.
NOTE: In the experiments shown here, the target was defined as the frontal region of the cerebral cortex a site 2.5 mm lateral (right), 1.5 mm anterior, and 3.5 mm ventral with respect to the bregma was chosen (Figure 1), producing a midbrain tumor in mice ranging from 20 to 30 g. - Make a small incision (~8 mm long) using sterile technique through the skin of the head to expose the bregma and the site of injection. A diagonal cut, from a point between the midline axis and the right eye towards the right ear (Figure 2) allows access to both the bregma and the injection site.
- Retract the skin and use a sterile cotton swab to dry the surface of the skull. Blot the bone with another swab moistened with H2O2 to visualize bregma. The H2O2 will produce oxygen bubbles, leaving thin lines of white foam along the coronal and sagittal sutures that intersect at bregma.
- Lock an empty syringe with needle onto the micropump and maneuver the tip of the needle directly over the bregma; set coordinates on the alignment console to 0.0 mm lateral, 0.0 mm anterior/posterior, and 0.0 mm ventral/dorsal.
- Move the needle to 2.5 mm lateral (right) and 1.5 mm anterior with respect to bregma (or desired location) using the control knobs on the stereotaxic unit. Raise the syringe slightly and mark the exact location on the skull with a dedicated felt-tip pen. If the outer surface of the skull is not on a level plane with bregma (i.e., the dorsal/ventral coordinate no longer reads zero), reset the ventral/dorsal reading to 0.0 to ensure that the depth of the injection is not shifted.
- Drill a small hole into the skull with a hand-held rotary drill outfitted with a sterile dental tip at the appropriate site marked with the pen in step 5.5. Hold the drill at an angle to the skull and very gently touch the tip to the bone. Repeat as needed. Drilling almost but not completely through the bone, leaving the needle to actually penetrate this layer of the skull, results in the cleanest injection.
- Remove bone dust with a cotton swab moistened in PBS or saline. Return the syringe to the pump and lower the needle straight down to the burr hole to confirm location. If the burr hole is not centered with the needle, use the drill to enlarge the opening.
- Gently mix the cell suspension and draw the cells into the syringe using the pump controller. Avoid bubbles and clumps and wipe the needle with an alcohol swab to remove contaminating cells on the exterior surface. If the syringe/needle is clogged or frozen, remove and quickly clean with sterile water and 70% EtOH.
- Lower the needle to the level of the skull surface (0.0 mm ventral/dorsal). Then slowly (over approximately 1 min) lower the needle to pierce through the full thickness of the skull and penetrate the brain to a depth of 4 mm ventrall. Withdraw the needle slowly to 3.5 mm ventral. If the mouse head is held securely in the stereotaxic unit, there should be almost no movement of the skull.
- Confirm that the correct parameters are entered into the pump controller (Section 2.2) and press “RUN/STOP” (or as directed by product manual) to autoinject cells. Monitor the mouse and adjust isoflurane if indicated. Watch the syringe and ensure that the plunger is moving in the barrel. Frozen or clogged movement can result in a bent/broken plunger.
- Wait 1 to 2 min with the needle in the brain, then very slowly (over 3 to 4 min) withdraw needle from tissue. Total time to remove the needle after the injection is complete is 5 min. Use a cotton swab to blot the area around the burr hole; leave the edges of the skin open to allow the bone to dry.
- Remove syringe from the pump and quickly rinse 3x with sterile H2O, 3x with 70% EtOH, and 3x with sterile H2O (or as directed by maker). Wipe needle with EtOH and set aside.
- Apply sterile bone wax (equivalent to 1 or 2 µl) to the burr hole and use the wooden end of a sterile cotton swab to tamp the wax onto and into the bone. If the surface is dried adequately, it should stick.
- Spread sterile drape over the mouse and surrounding work area and suture the incision; three stitches should be sufficient. Alternatively, surgical adhesive may be used. Apply a topical antibiotic.
- Reduce isoflurane to 0% and remove the mouse from the stereotaxic unit, being careful to protect teeth. Check ears, mouth and tongue.
- Monitor the mouse closely following injection. Place mouse on a heat pad for 5 to 10 min; then transfer to cage (on heat pad) and observe until mouse wakes and is ambulatory. Do not leave the mouse unattended until it has regained enough consciousness to maintain sternal recumbence. Do not return mouse to the company of other animals until fully recovered. Consider the use of additional analgesic if signs of pain are noticed.
6. Finish and Monitor Recovery & Tumor Development
- Clean all instruments and equipment using 70% EtOH, a bead sterilizer or disinfectant between mice.
- Monitor the injected mice for two days after the procedure for signs of pain, infection or other complications. Inject analgesic at 24 and 48 hr (or as indicated) after procedure. Remove sutures at 7 to 10 days, if necessary.
- Evaluate the mice for clinical signs of disease, paralysis, decreased activity, weight loss, seizures, or general illness.
- Monitor the tumor development by the appropriate method [magnetic resonance imaging (MRI), In Vivo Imaging Systems (IVIS), etc.].
Representative Results
Reliable intracranial xenografts can be created with this described technique. Identifying the critical structures of the mouse skull (Figure 1) will allow for recognition of the bregma and guide the investigator to a precise and reproducible injection location. In these studies the U251 parental line, U251 cells transfected with luciferase (U251-Luc), or U87 immortalized human GBM tissue culture cells were suspended in 4 to 6 µl of SF-DMEM and injected 2.5 mm lateral (right), 1.5 mm anterior, and 3.5 mm ventral with respect to bregma (Figure 2).
The resulting tumors can be visualized and analyzed by magnetic resonance imaging (MRI, Figure 3), in vivo luminescent imaging (IVIS, Figure 4), or routine gross pathology techniques (H&E staining, Figure 5). Particular care must be taken and optimization performed to determine the appropriate cells and location of injection to represent timing, growth rate, and tumor model desired.
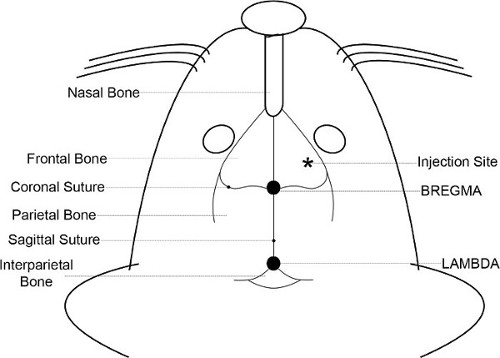
Figure 1. Skull anatomy. The anatomical features of the mouse head and skull are illustrated. The bregma, which is on the midline axis between the eyes and the ears at the intersection of the coronal and the sagittal sutures, is used to reproducibly locate the injection coordinates.
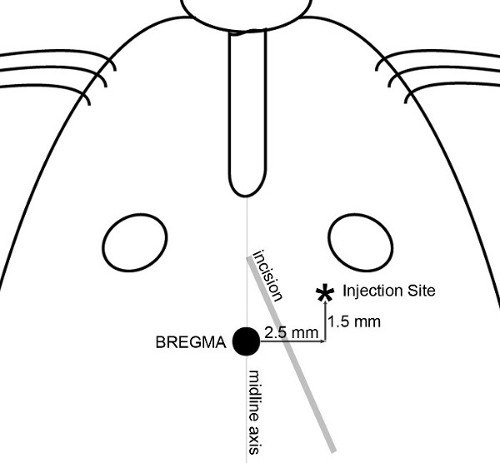
Figure 2. Incision and injection map. The features used to determine skin incision and precisely locate the injection site are illustrated. A diagonal incision is made to allow access to both bregma and the injection site. Application of 30% H2O2 to the surface of the skull helps to visualize skull sutures. Position a syringe with needle onto the micropump and maneuver the needle tip directly over the bregma. Set coordinates on the alignment console to zero; all skull measurements are then reproducibly made with respect to bregma.
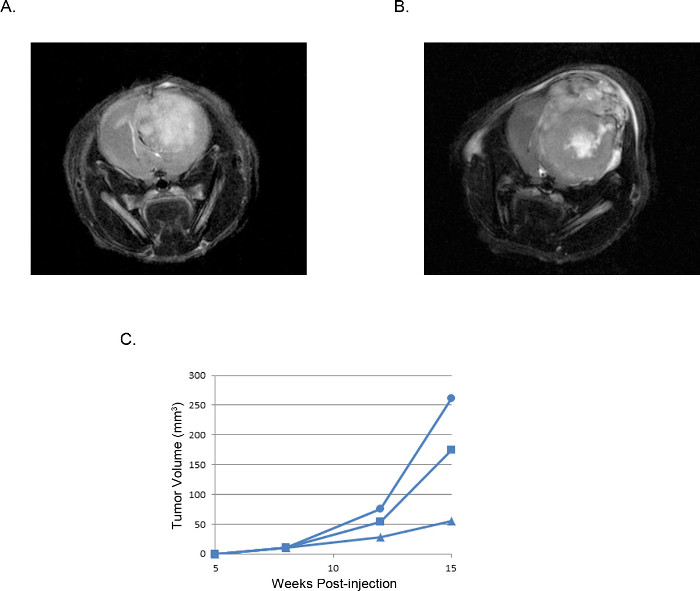
Figure 3. Representative intracranial xenograft tumors: MRI images. MRI T2 weighted images of a tumor derived from (A) 2 x 105 U87 cells compared with a tumor derived from (B) 2 x 105 U251 cells. (C) Tumor volumes calculated from MRI images of three individual mice injected with U251 cells plotted over time shows a reproducible window of tumor development and growth that is consistent in all experiments.
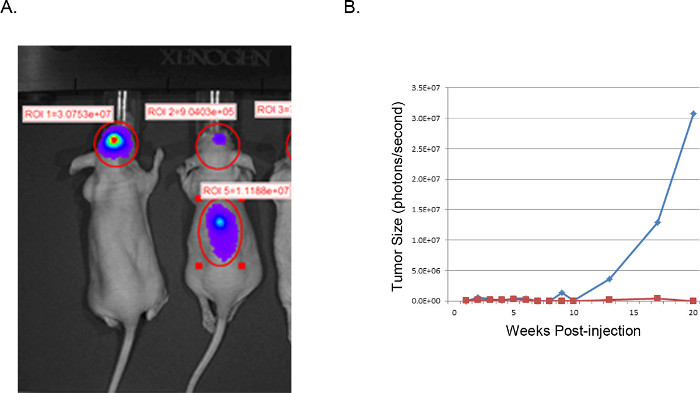
Figure 4. Representative xenograft tumors: IVIS images. IVIS image of U251-Luciferase transduced GBM cells injected intracranially into nude mice (A). The mouse on the left was successfully injected as described with U251-Luc cells and shows a very strong focalized signal (photons/sec) in the desired location. The mouse on the right demonstrates an unsuccessful result from improper injection location. H&E examination of the spine revealed tumor cell growth in the spinal column resulting from injection too close to midline with ventricular dissemination. (B) Tumor size is estimated from luciferase activity (photons/second) in the brain region of interest, plotted over time. The blue line corresponds to the mouse with a successful intracranial injection, while the red line corresponds to the mouse with tumor cell displacement to the spine.
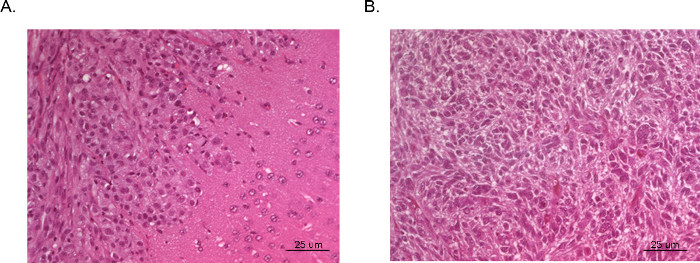
Figure 5. H&E of Intracranial xenograft tumors. Whole brains were collected from mice post sacrifice and fixed in formalin, mounted in paraffin, sectioned and stained with H&E. Tumor (A) was derived from U87 GBM cells and illustrates an area of dense tumor growth (left upper corner) and adjacent normal brain tissue with microscopic invasion of malignant cells (right lower corner). Tumor (B) was taken from a section of the center of a tumor derived from U251 GBM cells. The section very closely replicates the bizarre histopathology with multinucleated malignant cells seen in typical human GBM.
Discussion
Orthotopic mouse models of human brain cancer can be an excellent tool for assessing the effectiveness of clinical therapies, but care must be taken to optimize the placement of cells in brain tissue. Studies have shown that excessive aliquot volumes, suboptimal injection technique and hasty injection rates can lead to leakiness and the appearance of tumor cells in undesirable locations (ventricles, spinal cord, extradural regions, etc.) and high variation in tumor size2 (personal observations). An analysis of microprocessor-driven delivery of noncellular samples found that the use of a micropump produced more focalized delivery, less sample reflux and less variability than manual methods of injection, attributed to the smooth, uniform delivery and consistent pressures3. While it may be challenging to condense the needed number of cells in a volume of only 2 or 4 µl, the use of a stereotaxic instrument and alignment console equipped with a programmable micro-pump can yield reproducible and reliable tumors with similar growth rates. A xenograft model can never truly duplicate the microenvironment, initiation and development of a naturally occurring tumor, particularly with immune deficient hosts, but a well designed and implemented orthotopic model is the best alternative and is far superior to an ectopic model.
The major advantage of this protocol is the establishment of detectable tumors within a consistent time frame. The timing of tumor appearance is dependent on cell type and the number of cells injected, but is fairly predictable, and most tumors are established within a narrow window of time relative to the duration of tumor growth (i.e., time until endpoint). This enables the researcher to identify time points for data collection (such as MRI or IVIS) or intervention (such as drug treatment). Tumor growth rates vary with cell type, number of cells injected, and from mouse to mouse (much as they do in human patients), but are consistent from experiment to experiment.
While this protocol employs a technique that may require more instrumentation and time than less exact manual methods of injection and may not be applicable to large scale investigations, techniques for intracranial injections that allow for throughput of large numbers of animals (such as that described by Iwami et al.12) by definition involve rapid, high pressure injection and often involve hand-held instrumentation and manual measurement, which are subject to unsteadiness and uncertainty. These factors may be associated with leakage and off-target delivery2,3. The precision, reproducibility, and low mortality of this procedure will allow the investigator to design treatment experiments using fewer mice to obtain statistically significant results – a net savings.
One step is critical to the success of tumor implantation: the precise location of the injection. Cells injected too close to the ventricles may lead to CSF spread of disease through the ventricular system or into extracranial regions. Cells injected too shallow may grow out through the needle track. Predetermined coordinates are of little value if needle placement is sloppy. Take time to position the mouse head securely in the stereotaxic unit. Use the stereotaxic adjustment options and the ear bars to fit the equipment to the mouse, and ensure that the head is stably positioned and will not rock or twist during the procedure. Changes or variation if the injection site may affect tumor characteristics, including tumor take, growth, invasive potential, and access to drug delivery and oxygen supply.
Other parameters that have a profound impact on the resulting tumor include the injection rate, cell number and volume; all must be determined empirically. Minimize the volume for the number of cells required for tumor growth; use the slowest injection rate practical. Cell number and passage number also have major effects on tumor take and growth rate. The specialized equipment used here offer superior control, but the concepts of minimal volume, precise targeting, minimal and consistent injection rate, and slow needle withdrawal may be applied to a variety of techniques (including manual injection) and a variety of instruments.
This procedure is open to a variety of modifications: the injection site may be customized to recapitulate particular types of tumors. Adherent tissue culture cells, genetically modified clones, neurospheres, disaggregated mouse tumors, or human tissue fragments may be engrafted into a mouse brain. This technique may even be adapted for noncellular studies, including viral gene transfer3. Once a method for reliably producing tumors of the desired characteristics is established, experiments comparing the efficacy of therapies, drug treatments and combinations, and other options such as gene transfer may be performed.
Disclosures
The authors have nothing to disclose.
Acknowledgements
Dr. Keating is funded by DOD grant CA100335 and is a St. Baldrick’s Foundation Scholar.
Materials
| Name of Reagent/ Equipment | Company | Catalog Number | Comments/Description |
| Equipment | |||
| Small Animal Stereotaxic Instrument with Digital Display Console. | Kopf | Model 940 | |
| Mouse Gas Anesthesia Head Holder | Kopf | Model 923-B | |
| Mouse Ear Bars | Kopf | Medel 922 | |
| Fiber Optic Illuminator | Fisher | 12-562-36 | |
| UltraMicroPump III | WPI | UMP3 | |
| Micro4 microprocessor | WPI | UMC4 | |
| Variable speed hand-held rotary drill | Dremel | Model 300 | |
| Dental drill bit, 1.0 mm | Spoelting | 514554 | |
| Adaptor for dental drill bit: 3/32 inch collet | Dremel | 481 | |
| Heating pad | for mice | ||
| Isoflurane vaporizer system | for mice | ||
| Medical tubing and connectors | to connect isoflurane vaporizer with stereotaxic frame | ||
| Instruments | |||
| Precision 25 ul micro syringe | Hamilton | 7636-01 | Model 702, without needle |
| Microsyringe needles, 26s gauge | Hamilton | 7804-04 | RN, 25 mm point style 2 |
| Fine-tipped scissors (straight, sharp/sharp) | |||
| Medium-sized standard scissors | |||
| Standard serrated forceps | |||
| Serrated hemostats (2) | |||
| Fine-tipped forceps | |||
| Supplies | |||
| Sutures 5-0 vicryl P-3 13 mm (Ethicon) | MWI | J463G | |
| Surgical blades #10, stainless (Feather) | Fisher | 296#10 | |
| Isoflurane (Fluriso) | VetOne | NDC 13985-528-60 | Item #502017. Liquid inhalation anesthetic. federal law restricts this drug to use by or on the order of a licensed veterinarian. |
| Carprofen (Rimadyl Injectable 50 mg/mL) | Pfizer | NDC 61106-8507-01 | dilute in saline |
| Ophthalmic ointment (artificial tears) | Rugby | NDC 0536-6550-91 | |
| Topical antibiotic (AK-Poly-Bac ) | Akorn | NDC 17478-238-35 | |
| Povidone-iodine topical antiseptic, 10% (Betadine) | Betadine | NDC 67618-150-04 | |
| Hydrogen Peroxide, 30% | Fisher | H325-100 | for visualizing skull landmarks |
| Sterile saline | VetOne | NDC 13985-807-25 | for diluting solutions, cleaning tissue |
| Bone wax | WPI | Item #501771 | |
| Sterile drapes | McKesson | 25-517 | |
| Sterile surgical gloves | McKesson | (to fit) | |
| Sterile gauze pads, 2 x 2 | Fisherbrand | 22028556 | |
| Sterile gauze pads, 4 x 4 | Fisherbrand | 22-415-469 | |
| Alcohol prep pads (medium) | PDI | B603 | |
| Sterile cotton-tipped applicators | Fisherbrand | 23-400-114 | |
| Sterile 0.5 ml screw cap tube with caps for cells | USA Scientific | 1405-4700 | for cells |
| Individually wrapped sterile dispo pipettes | Fisher | BD 357575 | for needle cleaning solutions |
| BD insulin syringes with needles | Fisher | 329461 | for analgesic |
| 70% ethanol | for cleaning | ||
| Sterile di H2O | for cleaning | ||
| Microfuge tubes for cleaning solutions | for needle cleaning solutions | ||
| Felt tip pen (dedicated) | for marking skull |
References
- Heyer, J., Kwong, L. N., Lowe, S. W., Chin, L. Non-germline genetically engineered mouse models for translational cancer research. Nature reviews. Cancer. 10, 470-480 (2010).
- Yamada, S., et al. A method to accurately inject tumor cells into the caudate/putamen nuclei of the mouse brain. The Tokai journal of experimental and clinical medicine. 29, 167-173 (2004).
- Brooks, A. I., et al. Reproducible and efficient murine CNS gene delivery using a microprocessor-controlled injector. Journal of neuroscience. 80, 137-147 (1998).
- Shankavaram, U. T., et al. Molecular profiling indicates orthotopic xenograft of glioma cell lines simulate a subclass of human glioblastoma. Journal of cellular and molecular medicine. 16, 545-554 (2012).
- Giannini, C., et al. Patient tumor EGFR and PDGFRA gene amplifications retained in an invasive intracranial xenograft model of glioblastoma multiforme. Neuro-oncology. 7, 164-176 (2005).
- Yi, D., Hua, T. X., Lin, H. Y. EGFR gene overexpression retained in an invasive xenograft model by solid orthotopic transplantation of human glioblastoma multiforme into nude mice. Cancer investigation. 29, 229-239 (2011).
- Carty, N., et al. Intracranial injection of AAV expressing NEP but not IDE reduces amyloid pathology in APP+PS1 transgenic mice. PLos ONE. 8, e59626 (2013).
- Thaci, B., et al. Pharmacokinetic study of neural stem cell-based cell carrier for oncolytic virotherapy: targeted delivery of the therapeutic payload in an orthotopic brain tumor model. Cancer gene therapy. 19, 431-442 (2012).
- Ozawa, T., James, C. D. Establishing intracranial brain tumor xenografts with subsequent analysis of tumor growth and response to therapy using bioluminescence imaging. J. Vis. Exp. (41), (2010).
- Valadez, J. G., Sarangi, A., Lundberg, C. J., Cooper, M. K. Primary orthotopic glioma xenografts recapitulate infiltrative growth and isocitrate dehydrogenase I mutation. J. Vis. Exp. (83), (2014).
- Baumann, B. C., Dorsey, J. F., Benci, J. L., Joh, D. Y., Kao, G. D. Stereotactic intracranial implantation and in vivo bioluminescent imaging of tumor xenografts in a mouse model system of glioblastoma multiforme. J. Vis. Exp. (67), (2012).
- Iwami, K., et al. A novel method of intracranial injection via the postglenoid foramen for brain tumor mouse models. Journal of neurosurgery. 116, 630-635 (2012).

