Platelet-Rich Plasma Lysate for Treatment of Eye Surface Diseases
Instructor Prep
concepts
Student Protocol
PRP-L used for the quantitative assessment of growth factors were collected within a wider study on the characterization of PRP products for regenerative purposes, carried out at the AUSL-IRCCS di Reggio Emilia and approved by the Area Vasta Emilia Nord Ethical Committee on 10 January, 2019 (protocol number 2019/0003319). Donors gave their informed consent as per the Declaration of Helsinki. No ethical approval was necessary for collecting the aggregated, anonymous data of the Ocular Surface Disease Index (OSDI) questionnaire, which is routinely used by clinicians to monitor dry eye syndrome symptoms. Figure 1A shows an outline of the protocol followed, while the pictures in Figure 1B depict the main steps of the procedure.
1. Platelet-rich plasma (PRP) collection
- PRP apheresis
- For this protocol, select platelet donors according to Italian laws: platelet donors must be 18-65 years old, with normal pressure and blood count parameters and a platelet count not less than 180 x 109 platelets/L23. Eligible donors cannot take antiplatelet or anticoagulant drugs within 1 week before donation.
- Perform plasma-platelet-apheresis using an automated blood collection system, according to the manufacturers' instructions and national laws23, to obtain 1 unit of single-donor platelet-rich plasma (PRP). Collect PRP in Adenine Citrate Dextrose Solution A (ACD-A) anticoagulant solution.
NOTE: Platelet-apheresis is conducted with a continuous procedure; the time of collection is in a range between 40 min and 90 min. The amount of ACD delivered to the donors and the time of the procedures depend on the donor characteristics, e.g., hematocrit and needle gauge.
- PRP units' characteristics
NOTE: The following step is usually performed automatically by the automated blood collection system during the plasma-platelet-apheresis procedure. Please check the manufacturer's manual of instruction.- Resuspend the PRP units collected by apheresis in an adequate amount of preservative solution with the minimum amount of residual plasma, necessary to maintain pH > 6.4 during the whole storage time, to a mean final volume of 180 mL net of the anticoagulant solution (around 40 mL).
NOTE: According to Italian law, quality controls have to assess that the platelet (PLTs) count is at least 2.0 x 1011 PLTs/unit, while residual leucocytes have to be less than 1 x 106 cells/unit. - Store leucodepleted and irradiated PRP for a maximum of 5 days at 22 °C ± 2 °C on a platelet shaker before further manipulation23.
- Resuspend the PRP units collected by apheresis in an adequate amount of preservative solution with the minimum amount of residual plasma, necessary to maintain pH > 6.4 during the whole storage time, to a mean final volume of 180 mL net of the anticoagulant solution (around 40 mL).
- PRP dilution
- Immediately before starting PRP dilution, perform a PLT count with a hemocytometer using the sample collected from the main bag through a piercing spike.
NOTE: Perform the next steps in sterility under a class II biohazard hood. Wear personal protection equipment (lab coat, gloves, and goggles) during the procedure. - Dilute PRP with an adequate amount of sterile 0.9% NaCl to a final concentration of 0.32 x 109 ± 0.03 x 109 PLTs/mL, which simulates the average PLT concentration in peripheral blood.
- Taking advantage of a piercing spike for blood bags, split the diluted PRP into 300 mL empty collection bags to reach a net volume of 190 mL/bag.
- Use an aliquot of residual diluted PRP (usually 1 mL) to perform quality controls assessing possible microbial contaminations. Perform a sterility assay following the manufacturer's instructions in a microbiology laboratory (see Table of Materials).
NOTE: Use culture vials specific for aerobic blood cultures, which are able to perform the qualitative culture and recovery of aerobic microorganisms (mainly bacteria and yeast) from small volume blood specimens. - Store diluted PRP bags at −80 °C for a maximum of 2 months before thawing.
- Immediately before starting PRP dilution, perform a PLT count with a hemocytometer using the sample collected from the main bag through a piercing spike.
2. Platelet-rich plasma lysate (PRP-L) preparation
- Thawing
- Before starting the thawing procedure, ensure that a warm bath is set at 37 °C. Put the PRP bags into the warm bath and wait until completely thawed.
- PRP-L collection
- Centrifuge the PRP bags at 3000 x g for 30 min at room temperature.
NOTE: The next steps should be performed in sterility under a class II biohazard hood. - Exploiting the piercing spike of the transfer bag, connect the centrifuged bag with an empty sterile 300 mL transfer bag. Carefully, transfer the PRP-L supernatant, while avoiding debris, into the new bag. When possible, use a bag press.
- Seal the connection tube of the PRP-L unit with a bag sealer.
- Centrifuge the PRP bags at 3000 x g for 30 min at room temperature.
- PRP-L aliquotation
NOTE: A starting unit containing 190 mL PRP (see step 1.3.3.) is sufficient to fill two eye drops kits (for details on the specific medical devices used for the application and preservation of eye drops from blood components, see the Table of Materials). Eye drops kits should be opened under a class II hood with the whole string vials positioned above the pre-connected syringe and the central arrow of the stopcock pointing leftward to exclude the anti-bacterial filter.- Collect 30-60 mL of PRP-L with a sterile syringe and link the syringe to the Luer/lock connection on the filling line.
- According to the manufacturer's instructions, turn the stopcock by half of a turn to open the line between the PRP-L-containing syringe and the pre-connected syringe. Fill the pre-connected syringe with PRP-L.
- Disconnect the PRP-L syringe, close the tube cap of the luer/lock connection and rotate the stopcock to the original position. Use the eye drops kit syringe to fill the vials with PRP-L.
- Repeat the procedure from steps 2.3.1.-2.3.3. until all the applicator vials are filled. Ensure that each applicator is properly filled, then individually seal them with a bag sealer.
- Repeat the procedure with a new eye drops kit.
- Use a small aliquot of residual diluted PRP-L to assess possible microbial contamination (see step 1.3.4.).
NOTE: If the liquid accidentally reaches the anti-bacterial filter at the end of the string, the suction syringe may oppose resistance, hindering the filling. To continue the filling cycle, raise the end section of the string for about 5/6 aliquots from the anti-bacterial hydrophobic filter at the end of the string. In this position, use a new sterile syringe (of 30 mL volume) that has already been filled with air. Connect the female luer/lock of the anti-bacterial filter and press hard and repeatedly on the syringe's plunger to remove all residues of blood component and render the membrane of the anti-bacterial filter free of the liquid. Remove the syringe and fill the remaining vials.
- PRP-L storage
- Properly label each applicator and put them into a plastic bag. Label the plastic bag too, taking care to highlight the donor's blood group.
- Store at −80 °C for a maximum of 24 months before patient assignment, according to the Italian law23 and guidelines24.
3. PRP-L dispensation
- Perform patient assignment preferably by matching the PRP-L blood group. Deliver PRP-L applicator vials using a cool box and ensure that each applicator vial contains 1.45 mL of PRP-L, which corresponds to approximately 45 drops. Instruct the patient that applicator vials can be stored at the patients' homes for up to 1 month at −15 °C.
Platelet-Rich Plasma Lysate for Treatment of Eye Surface Diseases
Learning Objectives
The rationale for the use of serum-derived eye drops (which is the blood-based product most frequently used for the treatment of eye surface diseases) lies in their content of GFs, which are almost completely derived from circulating platelets. PRP contains a significantly higher number of platelets (and, consequently, of platelet-derived GFs) compared to peripheral blood serum, ranging between 0.15 x 109-0.45 x 109 PLTs/mL. According to Italian laws, the platelet count in PRP units should be at least 0.9 x 109-1 x 109 PLTs/mL. Therefore, to obtain a product that simulates the efficacy of serum eye drops, PRP should be diluted to the physiological platelet content before lysate preparation.
Nevertheless, since tissue repair is mainly driven by platelet-derived GFs, the PLT scount alone might be misleading for an effective therapy of eye surface diseases. In DED, which is the eye disease most commonly treated with blood-derived eye drops, tear film production and homeostasis are impaired. Platelet-based products for the treatment of DED, therefore, should also mimic the physiological content of tears.
To identify the most suitable PRP-L for treating eye surface diseases, described in step 1.3.2. of the present protocol, we preliminarily assessed different PRP dilutions, according to their PLTs content (between 0.7 x 109/mL and 0.3 x 109/mL), and some representative GFs from those that are known to be involved in eye tissue repair12,20,21.
Platelet count was performed with a hemocytometer, while GFs were assessed by means of a multiplex protein quantification assay. The assay was performed as previously described25 according to the manufacturer's instructions. GFs shown in this manuscript were selected for quantification after a preliminary screening of 36 GFs and GFRs performed on PRP lysate with a semi-quantitative protein array. Luminex quantification was performed on 3 out of the 36 screened GFs: EGF and PDGF (which turned out to be the most abundant ones in our PRP lysates) and TGFβ-1,2,3 isoforms (for which the content is important for the eye surface treatment21). EGF and PDGF content were measured as they may influence the efficacy of PRP-L22, while TGFβ isoforms were selected for their known role in immune signaling regualtion21.
Since protein arrays are part of another in vitro study on the characterization of different PRP26, those data are not presented in this manuscript.
We quantitatively assessed EGF, PDGF, and TGFβ in PRP lysates from two different donors (D1 and D2), previously diluted between 0.7 x 109-0.3 x 109 PLTs/mL in 0.9% NaCl. Figure 2 shows the results of the 0.3 x 109 PLTs/mL dilution, which turned out to be most similar to tear composition.
The 0.3 x 109 PLTs/mL dilution was selected based on literature data on tear composition. The EGF values were found to be quite low compared to the mean tear value but still in the range of normality27. Even PDGF, despite being highly variable between the two donors considered, was always comparable to the concentration found in normal tears20. Finally, TGFβ-1 was found to be the most abundant isoform in PRP-L, similar to tears21.
Once the most suitable PLT dilution to prepare apheresis PRP-L had been identified, the Transfusion Medicine Unit started distributing these products to patients affected by eye surface disorders in 2015. The ophthalmologists routinely collected the OSDI questionnaires to monitor DED symptoms; the OSDI test assesses quality of life measures, such as the perception of ocular irritation and how it affects the functioning related to vision. The questionnaire, created by the Outcomes Research Group at Allergan Inc. in 1995 and now accepted as a valid instrument to monitor DED, is submitted to patients and analyzed as previously described28,29.
Here, we show the aggregate results of OSDI tests of DED patients treated between January 2020 and January 2021 (n = 27). After a 6 month therapy with PRP-L, the OSDI scores decreased from 56 ± 21 to 45 ± 21, indicating an improvement in patients' quality of life (Figure 3).
Despite these data still being in the severe range and not relating to clinical outcomes of efficacy, they suggest that DED patients consider PRP-L a useful product that ameliorates ocular discomfort; this aspect should be further investigated in prospective clinical trials aimed at assessing its efficacy in treating ocular surface diseases.
In Table 1, we report a comparison of the present method of production with another method for preparing allogenic PRP-L for eye drops30 and for other purposes22. To our knowledge, Zhang's30 protocol and the current protocol are the only published methods to produce PRP-L for the eye surface. In both, PRP-L is obtained from apheresis; differences between the two protocols, mainly relating to the number of freeze and thaw cycles and centrifugation steps, should be compared in order to improve PRP-L production. Nevertheless, these methodological differences have not been proved to be detrimental to the regenerative capacity of PRP-L tested on other tissues22.
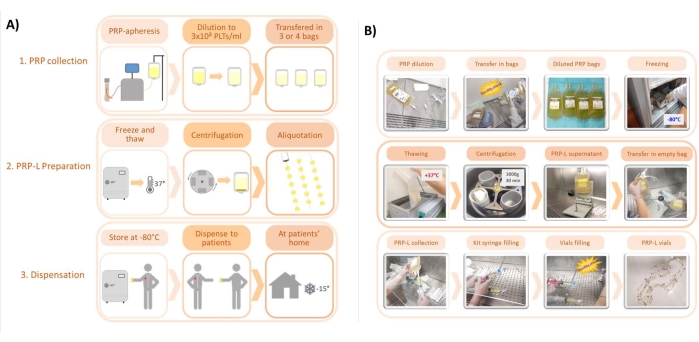
Figure 1: Main steps of the protocol for the preparation of PRP-L. (A) Scheme of the protocol, from PRP collection to PRP-L preparation and dispensation. (B) Representative pictures of the main steps of the protocols. Please click here to view a larger version of this figure.
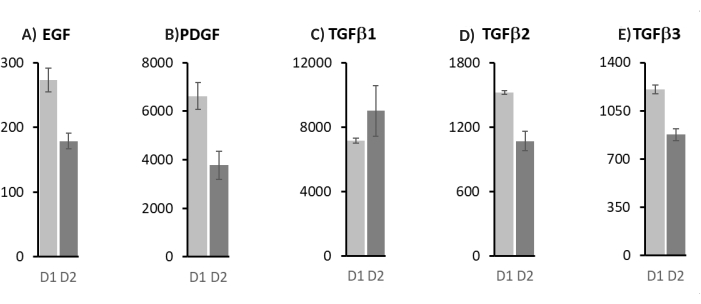
Figure 2: Luminex quantification of platelet-derived growth factors for the 0.3 x 109/mL dilution of PRP-L. (A) Epidermal growth factor (EGF); (B) platelet-derived growth factor (PDGF); (C) transforming growth factor-beta isoform 1 (TGFβ1); (D) transforming growth factor-beta isoform 2 (TGFβ2); (E) transforming growth factor-beta isoform 3 (TGFβ3). Values are expressed as pg/mL, mean ± standard deviation of three independent measurements. D1 and D2 are two different platelet donors. Please click here to view a larger version of this figure.
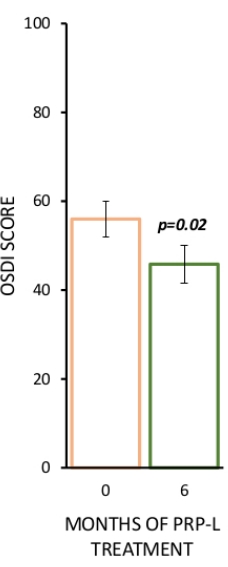
Figure 3: Aggregate OSDI scores of DED patients treated with PRP-L between January 2020 and January 2021 at the Ophthalmology Unit of the AUSL-IRCCS di Reggio Emilia. N = 27 patients. OSDI score aggregate results are represented as mean ± standard error, p-value was calculated with a paired t-test with data analysis software. Please click here to view a larger version of this figure.
| This article | PRP-L for the eye (in vitro study)29 | PRP-L for other purposes21 | |
| Source | PLTs apheresis | PLTs apheresis | Apheresis and whole blood |
| Freeze and thaw cycles | 1 (at -80 °C) | 2 (at -80 °C) | 1-3 (at -20 °C and -80 °C) |
| Storage temperature | at -80 °C | at -80 °C | at -20 °C and -80 °C |
| Centrifugation speed before storage | 3000 x g/30 min | 3500 x g/30 min | 400-3000 x g/6 min -30 min |
| Filtration before storage | No | Yes | No/Yes |
Table 1: Comparison of protocols to prepare allogenic PRP-L from platelet-based products collected by apheresis.
List of Materials
| Equipments | |||
| CompoSeal Mobilea II | Fresenius Kabi, Germany | bag sealer | |
| HeraSafe hood | Heraeus Instruments, Germany | Class II biohazard hood | |
| MCS+ 9000 Mobile Platelet Collection System | Haemonetics, Italy | automated plasma and multicomponent collection equipment for donating platelet, red cell, plasma, or combination blood components | |
| Platelet shaker, PF396i | Helmer, USA | Platelet shaker | |
| Raycell X-ray Blood Irradiator | MDS Nordion, Canada | X-ray Blood Irradiator | |
| ROTIXA 50RS | Hettich Zentrifugen, Germany | High speed entrifuge | |
| Sysmex XS-1000i | Sysmex Europe GMBH, Germany | haemocytometer for platelet count | |
| Warm bath, WB-M15 | Falc Instruments, Italy | Warm bath | |
| Materials | |||
| ACD-A anticoagulant solution A | Fenwal Inc., USA | DIN 00788139 | anticoagulant solution for platelet apheresis (1000 ml) |
| BD BACTEC Peds Plus/F Culture vials | BD Biosciences, USA | BD 442020 | Sterility assay |
| BD BACTEC Peds Plus/F Culture vials | BD Biosciences, USA | 442020 | At least 2 vials for sterility assay |
| BD Luer Lok Syringe | BD Plastipack, USA | 300865 | At least 4 sterile syringes (50 ml) |
| Bio-Plex Human Cancer Panel 1 | BioRad Laboratories, USA | 171AC500M | Standard panel for PDGF isoforms assessment |
| Bio-Plex Human Cancer Panel 2 | BioRad Laboratories, USA | 171AC600M | Standard panel for EGF assessment |
| Bio-Plex MAGPIX Multiplex Reader | BioRad Laboratories, USA | Magpix | This instrument allows multiple immunoassays using functionalized magnetic beads. |
| Bio-Plex Pro TGF-b Assay | BioRad Laboratories, USA | 10024984 | Set and standards for TGFb isoforms assessment |
| BioRet | ARIES s.r.l., Italy | A2DH0020 | At least 4 piercing spike for blood bags |
| Blood collection tube | BD Vacutainer, USA | 367835 | 1 tube, necessary to perform platelet counts |
| Eye drops kit. COL Medical Device for the application and preservation of eye drops from haemocomponents | Biomed Device s.r.l., Italy | COLC50 | Eye drops kit. At least 2 kits for each PRP unit collected |
| Human Cancer PDGF-AB/BB Set 1x96well | BioRad Laboratories, USA | 171BC511 | Set for PDGF isoforms assessment |
| Human Cancer2 EGF Set 1x96well | BioRad Laboratories, USA | 171BC603M | Set for EGF assessment |
| NaCl 0.9% sterile solution | Baxter S.p.A., Italy | B05BB01 | 1000 ml |
| OSDI Questionnaire | Allergan Inc., USA | OSDI | Ocular Surface Disease Index Questionnaire |
| Piercing spike | BioRet ARIES s.r.l., Italy | BS051004 | Spike |
| Platelet Additive Solution A+ T-PAS+ | TERUMO BCT Inc., Italy | 40842 | preservative solution for platelet concentrates (1000 ml) |
| Software Excel | Microsoft, USA | Excel | Data analysis software |
| Teruflex Transfer bag 1000 ml | TERUMO BCT Inc., Italy | BB*T100BM | 1 for PRP dilution |
| Teruflex Transfer bag 300 ml | TERUMO BCT Inc., Italy | BB*030CM | At least 6 for each PRP unit collected |
Lab Prep
Various ocular surface diseases are treated with blood-derived eye drops. Their use has been introduced in clinical practice because of their metabolite and growth factor content, which promotes eye surface regeneration. Blood-based eye drops can be prepared from different sources (i.e., whole blood or platelet apheresis donation), as well as with different protocols (e.g., different dilutions and freeze/thaw cycles). This variability hampers the standardization of clinical protocols and, consequently, the evaluation of their clinical efficacy. Detailing and sharing the methodological procedures may contribute to defining common guidelines. Over the last years, allogenic products have been diffusing as an alternative to the autologous treatments since they guarantee higher efficacy standards; among them, the platelet-rich plasma lysate (PRP-L) eye drops are prepared with simple manufacturing procedures. In the transfusion medicine unit at AUSL-IRCCS di Reggio Emilia, Italy, PRP-L is obtained from platelet-apheresis donation. This product is initially diluted to 0.3 x 109 platelets/mL (starting from an average concentration of 1 x 109 platelets/mL) in 0.9% NaCl. Diluted platelets are frozen/thawed and, subsequently, centrifuged to eliminate debris. The final volume is split into 1.45 mL aliquots and stored at −80 °C. Before being dispensed to patients, eye drops are tested for sterility. Patients may store platelet lysates at −15 °C for up to 1 month. The growth factor composition is also assessed from randomly selected aliquots, and the mean values are reported here.
Various ocular surface diseases are treated with blood-derived eye drops. Their use has been introduced in clinical practice because of their metabolite and growth factor content, which promotes eye surface regeneration. Blood-based eye drops can be prepared from different sources (i.e., whole blood or platelet apheresis donation), as well as with different protocols (e.g., different dilutions and freeze/thaw cycles). This variability hampers the standardization of clinical protocols and, consequently, the evaluation of their clinical efficacy. Detailing and sharing the methodological procedures may contribute to defining common guidelines. Over the last years, allogenic products have been diffusing as an alternative to the autologous treatments since they guarantee higher efficacy standards; among them, the platelet-rich plasma lysate (PRP-L) eye drops are prepared with simple manufacturing procedures. In the transfusion medicine unit at AUSL-IRCCS di Reggio Emilia, Italy, PRP-L is obtained from platelet-apheresis donation. This product is initially diluted to 0.3 x 109 platelets/mL (starting from an average concentration of 1 x 109 platelets/mL) in 0.9% NaCl. Diluted platelets are frozen/thawed and, subsequently, centrifuged to eliminate debris. The final volume is split into 1.45 mL aliquots and stored at −80 °C. Before being dispensed to patients, eye drops are tested for sterility. Patients may store platelet lysates at −15 °C for up to 1 month. The growth factor composition is also assessed from randomly selected aliquots, and the mean values are reported here.
Procedure
Various ocular surface diseases are treated with blood-derived eye drops. Their use has been introduced in clinical practice because of their metabolite and growth factor content, which promotes eye surface regeneration. Blood-based eye drops can be prepared from different sources (i.e., whole blood or platelet apheresis donation), as well as with different protocols (e.g., different dilutions and freeze/thaw cycles). This variability hampers the standardization of clinical protocols and, consequently, the evaluation of their clinical efficacy. Detailing and sharing the methodological procedures may contribute to defining common guidelines. Over the last years, allogenic products have been diffusing as an alternative to the autologous treatments since they guarantee higher efficacy standards; among them, the platelet-rich plasma lysate (PRP-L) eye drops are prepared with simple manufacturing procedures. In the transfusion medicine unit at AUSL-IRCCS di Reggio Emilia, Italy, PRP-L is obtained from platelet-apheresis donation. This product is initially diluted to 0.3 x 109 platelets/mL (starting from an average concentration of 1 x 109 platelets/mL) in 0.9% NaCl. Diluted platelets are frozen/thawed and, subsequently, centrifuged to eliminate debris. The final volume is split into 1.45 mL aliquots and stored at −80 °C. Before being dispensed to patients, eye drops are tested for sterility. Patients may store platelet lysates at −15 °C for up to 1 month. The growth factor composition is also assessed from randomly selected aliquots, and the mean values are reported here.
