Fabrication of Size-Controlled and Emulsion-Free Chitosan-Genipin Microgels for Tissue Engineering Applications
Summary
The present protocol describes a non-emulsion-based method for the fabrication of chitosan-genipin microgels. The size of these microgels can be precisely controlled, and they can display pH-dependent swelling, degrade in vivo, and be loaded with therapeutic molecules that release over time in a sustained manner, making them highly relevant for tissue engineering applications.
Abstract
Chitosan microgels are of significant interest in tissue engineering due to their wide range of applications, low cost, and immunogenicity. However, chitosan microgels are commonly fabricated using emulsion methods that require organic solvent rinses, which are toxic and harmful to the environment. The present protocol presents a rapid, non-cytotoxic, non-emulsion-based method for fabricating chitosan-genipin microgels without the need for organic solvent rinses. The microgels described herein can be fabricated with precise size control. They exhibit sustained release of biomolecules, making them highly relevant for tissue engineering, biomaterials, and regenerative medicine. Chitosan is crosslinked with genipin to form a hydrogel network, then passed through a syringe filter to produce the microgels. The microgels can be filtered to create a range of sizes, and they show pH-dependent swelling and degrade over time enzymatically. These microgels have been employed in a rat growth plate injury model and were demonstrated to promote increased cartilage tissue repair and to show complete degradation at 28 days in vivo. Due to their low cost, high convenience, and ease of fabrication with cytocompatible materials, these chitosan microgels present an exciting and unique technology in tissue engineering.
Introduction
The growth plate, also known as the physis, is the cartilage structure located at the end of long bones that mediates growth in children. If the growth plate becomes injured, repair tissue known as a "bony bar" can form, which interrupts normal growth and can cause growth defects or angular deformities. Epidemiological data have shown that 15%-30% of all childhood skeletal injuries are related to the growth plate. Bony bar formation occurs in up to 30% of these injuries, making growth plate injuries and their associated treatment a significant clinical manifestation issue1,2,3,4. When bony bar formation occurs, the most common treatment avenue involves resectioning the bony bar and inserting an interpositional material, such as silicon or adipose tissue5. However, patients that undergo bony bar resection surgery often have a poor prognosis for full recovery, as there is currently no treatment that can fully repair an injured growth plate6,7,8. In light of these shortcomings, there is a critical need for effective strategies for treating growth plate injuries, both in preventing the formation of a bony bar and regenerating healthy physeal cartilage tissue.
Hydrogel microparticles, or microgels, have recently gained interest as injectable scaffolds that can provide sustained release of therapeutics9. Due to their high tunability and biocompatibility, microgels are also well-suited for bioactive factor or cell encapsulation. Microgels can be made of various materials, ranging from synthetic polymers, such as polyethylene glycol (PEG), to natural polymers like alginate or chitosan10,11,12. Chitosan has been shown to have several beneficial effects for tissue engineering, such as its ability to destabilize the outer membrane of gram-negative bacteria, thereby offering inherent antimicrobial activity13,14. Additionally, chitosan is cost-effective, cell-interactive, and easily modified using its amine-containing structure. Chitosan-based microgels promise a biomaterial strategy for drug delivery and material signaling that can promote tissue regeneration while preventing bacterial infection. However, chitosan microgels are often fabricated with a wide range of techniques that require special equipment, emulsion techniques, or cytotoxic solvent rinses. For example, some studies have fabricated chitosan microgels with emulsion-based methods, but these protocols require solvent rinses and cytotoxic crosslinkers, potentially negating their translation to clinical settings15,16. Other studies have used microfluidics or electrospray approaches to fabricate chitosan microgels, which require special equipment, preparation, and training17,18. Chitosan microgels are also commonly made with a dropwise process of crosslinker into chitosan solution; however, this method is highly dependent on solution viscosity, polymer concentration, and flow rate, making it difficult to control the size and dispersity of the microgels19,20. Conversely, the method for microgel fabrication described herein requires no specialist equipment or solvent rinses, making these microgels viable for fabrication in nearly any lab or setting. Therefore, these microgels represent highly relevant biomaterials for a quick, cost-effective, and easy-to-produce drug delivery vehicle for many applications.
By modulating a microgel's composition and material characteristics, researchers can gain precise control over the cellular microenvironment, thus directing cell behavior in a material-dependent manner. Microgels can be employed on their own or combined with bulk biomaterial systems to impart specific functionalities, such as the extended release of bioactive factors or precise special signaling for native or exogenous cells. Biomaterials and microgels have emerged as attractive treatment avenues for growth plate injuries. Significant effort has been dedicated to developing alginate and chitosan-based biomaterials to treat growth plate injuries21,22,23,24,25. Due to the dynamic temporal nature of growth plate ossification and bone elongation, the mechanism of bony bar formation is not fully understood. Therefore, several animal models have been developed to better elucidate the mechanisms of endochondral ossification and bony bar formation, such as in rats, rabbits, and sheep26,27,28. One such model is a rat growth plate injury model, which uses a drill-hole defect in the rat tibia to produce a bony bar in a predictable and reproducible manner and mimics human injuries across all three zones of the growth plate29,30. Several biomaterial-based strategies for treating growth plate injuries have been tested using this model. Additionally, two different methods for fabricating chitosan microgels have been developed, which can be used as an injectable biomaterial system that releases therapeutics in a sustained manner10,31. These microgels have been employed in a rat physeal injury model, and they showed improved cartilage regeneration31 when releasing SDF-1a and TGF-b3. The techniques provided in this protocol describe methods developed to fabricate these chitosan microgels, which can then be employed in a wide variety of tissue engineering applications. For example, recent studies have used thermo- or magento-responsive chitosan microgels for controlled oncological drug delivery applications32,33.
Protocol
All animal procedures were approved by the University of Colorado Denver Institutional Animal Care and Use Committee. 6-week old male Sprague-Dawley rats were used for the present study. The rat growth plate injury model was created following a previously published report30.
1. Preparation of the chitosan polymer
- Obtain purified and lyophilized low molecular weight (LMW) chitosan from commercially available sources (see Table of Materials).
- Add 495 mL of double-distilled water (ddH2O) and a stir bar to a 1 L beaker. Add 5 g of chitosan (Step 1.1.) and mix well.
NOTE: Chitosan is only sparingly soluble in an aqueous solution at physiological pH, so the chitosan will not dissolve easily at this step. - Add 5 mL of glacial acetic acid to the above-prepared chitosan solution.
- Stir covered at 300 rpm for 18 h with the beaker set in a water bath held at 50 °C.
- Using a Büchner flask and funnel, filter the chitosan solution through decreasing sizes of filter paper: 22 µm, 8 µm, and 2.7 µm (see Table of Materials).
- Add the filtered chitosan solution to cellulose dialysis tubing (see Table of Materials) and allow to dialyze in ddH2O at room temperature for 4 days, changing the ddH2O every day.
NOTE: Use ultrapure ddH2O water for the last change. - Transfer the dialyzed chitosan solution to a beaker and adjust the pH to 8.0 using 1 M NaOH.
- Aliquot the chitosan into centrifuge tubes and centrifuge at 4000 x g for 5 min at room temperature.
- Decant the supernatant to a waste stream and resuspend the chitosan in ddH2O, repeating 2x.
- Freeze and then lyophilize the chitosan pellet.
- Each day, remove the lyophilized product and record the mass.
NOTE: When the mass of the lyophilized product is no longer changing, the product is fully dried and can be stored at -20 °C until ready for use.
- Each day, remove the lyophilized product and record the mass.
2. Fabrication of chitosan hydrogel
- Add 2 mL of 6% acetic acid and 120 mg of purified chitosan (Step 1) to a 10 mL Luer-lock syringe to form a 6% w/v chitosan solution.
- Connect the Luer-lock syringe to another identical syringe using a female-female Luer-lock connector and mix the solution back and forth for 30 s, or until the chitosan has fully dissolved in the acetic acid.
- Before crosslinking, add any therapeutic or bioactive agent to the chitosan solution (if needed). For the present study, 200 ng of SDF-1a and TGF-b3 (see Table of Materials) were added to the microgels.
NOTE: SDF-1a and TGF-b3 are bioactive agents relevant to growth plate tissue regeneration. SDF-1a promotes migration of mesenchymal stem cells to the defect site, and TGF-b3 serves as a chondrogenic factor to induce differentiation of these stem cells down the chondrogenic lineage31.
NOTE: Mix the chitosan again between the syringes to fully incorporate the therapeutic. - Prepare 100 mM of stock crosslinker solution of genipin (see Table of Materials) in 100% ethanol.
- Add 100 µL of the prepared genipin solution (Step 2.4.) to the chitosan-containing syringe, and again mix back and forth between syringes for 30 s.
- Extrude the mixture from the syringe onto a 35 mm Petri dish, cover it with paraffin film, and incubate it at 37 °C overnight in a humidified atmosphere.
NOTE: The solution will turn dark blue, indicating that the crosslinking reaction between chitosan and genipin has occurred, leading to the formation of chitosan microgel. - Filter the prepared chitosan microgel following the steps below.
- Gently break the hydrogel into smaller pieces using a spatula.
NOTE: The pieces should be small enough to be transferred to the back of a 10 mL syringe, ~1-2 cm in diameter. - Place a filter of desired mesh size into the back of a clean 10 mL syringe.
NOTE: The typical size range for microgels is between 50-200 µm. - Transfer the broken gel pieces into the syringe fitted with the filter and add 6 mL of ddH2O.
NOTE: The chitosan gel will swell significantly in the aqueous medium, so a large change in the gel volume is expected. - Connect the syringe via a Luer-lock connector to another clean 10 mL syringe.
- Force the gel + water mixture through the syringe with the filter to create microgels with a specified maximum diameter.
- After the first filtration, open the back of the syringe containing the filter and extrude the mixture back into this syringe.
- Replace the back of the syringe and force the mixture through the filter again.
- Repeat the filtration 5-6x or until there is little resistance through the filter.
- Gently break the hydrogel into smaller pieces using a spatula.
- Rinse and purify the filtered microgels.
- Transfer the filtered gel mixture to a 50 mL conical tube, bring the total volume up to 20 mL with ddH2O, and then vortex the mixture to ensure homogeneous dispersion.
- Centrifuge the microgels at 100 x g for 5 min at room temperature and decant the upper aqueous phase.
- Resuspend the microgels in 10 mL of 70% ethanol, vortex, and place under UV light for 1 h to sterilize.
- Centrifuge the microgels at 1,000 x g for 5 min at room temperature, discard the ethanol, and rinse 3x with ddH2O.
3. Preparation of microgels for in vitro or in vivo applications
NOTE: For the present study, cartilage regeneration in growth plate injuries was studied in a rat model. For details, see reference31.
- Resuspend the microgel pellets 1:1 in ddH2O. Microgels can be stored for up to 1 month suspended in ddH2O at 4 °C. If a bioactive agent is used, the microgels must be used immediately.
- Create the injury site in the animal following a previously published report30.
- Flush the injury site with saline and either keep the animal untreated (for control study) or inject the chitosan microgels only or the microgels loaded with the bioactive agents (Step 3.2.).
- Close the wound in the animal and administer post-surgical analgesics30.
- At days 7 or 28 post-surgery, euthanize the rat by CO2 overdose, excise the limbs and perform histology to assess the tissue repair at the injury site31.
Representative Results
Successful fabrication of chitosan microgels relies on the crosslinking reaction between genipin and chitosan, specifically involving the amines on the chitosan polymer chains. In contrast to other microgel fabrication techniques, this method does not require emulsions or solvent rinses and can be quickly and easily conducted with inexpensive equipment. A hallmark indicator for successful microgel fabrication is the distinct color change from off-white to dark blue after the chitosan and genipin have been mixed. The crosslinking reaction between genipin and amine-containing compounds, such as chitosan or other proteins, has been well-characterized in the literature34. In short, the crosslinking mechanism is considered to be a nucleophilic attack by the amino groups of chitosan, in which genipin acts as a dialdehyde with stable condensation products35. The short chains of stable, condensed genipin act as crosslinking bridges between the chitosan polymers. The crosslinking reaction causes the solution to turn a dark blue, likely due to oxygen radical-induced polymerization and dehydrogenation of intermediate compounds, which follows the ring-opening reaction from nucleophilic attack36.
Once the microgels have been filtered and resuspended in a 1:1 water dilution, they can be easily employed in various biomaterial applications. Work has recently been published using these emulsion-free chitosan microgels to promote cartilage regeneration in growth plate injuries. The microgels were fabricated as described herein and either kept empty or loaded with SDF-1a and TGF-b3, which are bioactive agents that are relevant in growth plate tissue regeneration, with SDF-1a promoting migration of mesenchymal stem cells to the defect site and TGF-b3 serving as a chondrogenic factor to induce differentiation of these stem cells down the chondrogenic lineage37,38. The release rate of the proteins was quantified in vitro via ELISA, and the release of these molecules was sustained over time31. Then, the microgels were injected into a growth plate injury in an in vivo rat model, and the injected microgels prevented early bony bar formation in vivo31. These injectable, cost-effective, and simple-to-produce chitosan microgels could easily be employed in many biomaterial applications.
Although this process for microgel fabrication has been optimized for simple setup and applications, several problems could still arise that researchers should be mindful of. Insufficient mixing of the polymer and crosslinking components is the most likely cause for different results during fabrication. The solid chitosan must be mixed vigorously between the syringes, and the resulting chitosan solution must be entirely homogenous before the genipin crosslinker is added. If the solution is not homogenous, the solid chitosan chunks remaining in the solution will form lumps, and uneven crosslinking will occur, preventing effective filtering and resulting in poly-dispersed microgels with significantly varying diameters. Another important factor to consider during fabrication is avoiding evaporation during the crosslinking period, which must be prevented with paraffin film or other evaporation-trapping techniques. If the chitosan hydrogel dries out, it will not swell during the water rinses, and it will not filter through the syringe. Lastly, the microgels must be suspended in excess water during the filtration process and stored in water at 4 °C when not in use. The microgels are not extrudable or injectable unless suspended in at least a 1:1 dilution of water.
Figure 1 shows a broad overview of the microgel fabrication process. The same process is depicted again in Figure 2, which shows photographs of the process, emphasizing the protocol stages that are difficult to understand from text alone. For example, Figure 2D shows how a wire mesh filter is inserted into the 10 mL syringe. Once fully seated against the top of the syringe, this wire mesh filter allows for quick and convenient filtration of the chitosan microgels without specialist equipment or solvents. Similarly, Figure 2E shows the flow of hydrated chitosan gel through the mesh filter, which is the basis for microgel fabrication. Figure 3 was adapted from our previous publication on these microgels and shows their pH-dependent swelling behavior and the differences in the size of the microgels dependent on the pore size of the mesh filter. Different mesh sizes can be ordered from the manufacturer, which allows for convenient control over the size of the microgels. This precise control over microgel size is highly important when designing drug delivery systems with well-defined therapeutic load release rates. Previous work on microgels also showed that they degrade significantly in the presence of lysozyme at 2-4 weeks31. Finally, Figure 4 shows histology images31 in a rat growth plate injury model treated with the chitosan microgels loaded with SDF-1a and TGF-b3.
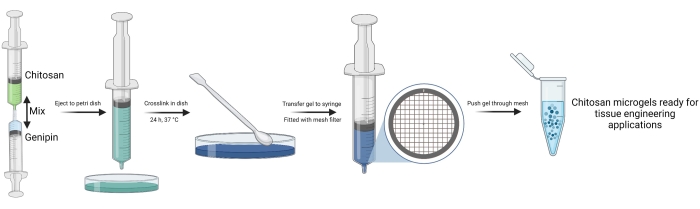
Figure 1: Schematic overview of chitosan microgel fabrication. The figure was created using biorender.com. Please click here to view a larger version of this figure.

Figure 2: Photographs of the microgel fabrication process. (A) Chitosan solution in syringes connected using Luer lock. (B) Extrusion of chitosan gel into a 35 mm Petri dish. (C) Retrieval of chitosan gel after crosslinking color change from off-white to dark blue. (D) Top-down view inside the syringe showing the wire mesh sieve fitted against the nozzle of the syringe. (E) Chitosan gel was pressed through a mesh filter to produce microgels. (F) Microgels were stored in a 1:1 dilution of ddH2O in a conical tube. Please click here to view a larger version of this figure.
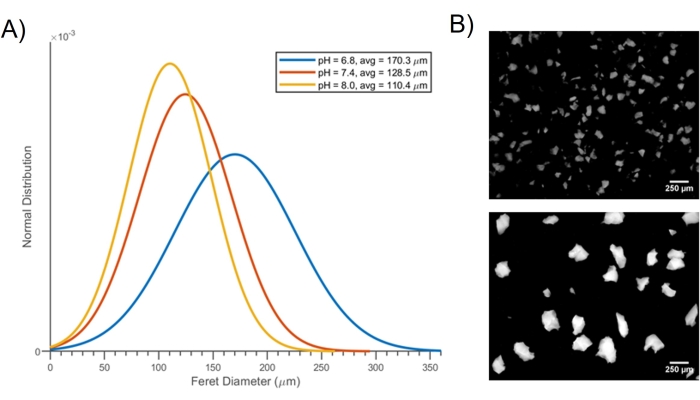
Figure 3: pH-dependent swelling behavior of the microgels. (A) Normal distribution graph of the Feret diameter showing swelling behavior of the microgels in response to pH changes. (B) Fluorescent images of microgels fabricated using No. 200 mesh (upper image: <75 µm sized microgels) and No. 100 mesh (lower image: 75-150 µm sized microgels). The figure is reprinted with permission from reference31. Please click here to view a larger version of this figure.
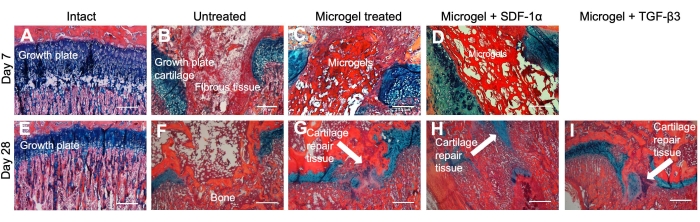
Figure 4: Histology images in a rat growth plate injury model treated with the chitosan microgels loaded with SDF-1a and TGF-b3. 10x histological images showing growth plate repair tissue of intact (A) and (E), untreated (B) and (F), microgel treated (C) and (G), microgel + SDF-1a treated (D) and (H), and microgel + TGF-b3 treated (I) limbs. No day 7 animals were treated with microgel + TGFb3. Alcian blue hematoxylin (ABH) stains the bone orange to red, the fibrous tissue pink, and the cartilage blue. The microgel appears as a dark red fibrous-like tissue. Scale bars = 500 µm. The figure is reprinted with permission from reference31. Please click here to view a larger version of this figure.
Discussion
Microgels have been widely researched in recent years due to their high level of applicability for various purposes, such as drug delivery or cell encapsulation9. The ease of manufacturing of micro-scale biomaterial constructs is of significant relevance in tissue engineering, as it allows researchers to develop hydrogel-based strategies at a specific size and time scale. However, most methods for fabricating chitosan microgels require expensive equipment and reagents, emulsions, or cytotoxic solvent rinses, which prevents their translation to clinical use15,16,17,18,19,20. These microgels excel in terms of their significant convenience of fabrication, requiring no emulsion techniques or solvent rinses. Additionally, these microgels retain the ideal properties for a tissue-engineered construct, such as pH-dependent swelling and drug loading, tuned degradation behavior, and sustained release of therapeutics.
The most critical step in fabricating these chitosan microgels is the filtration between syringes. These microgels start as bulk hydrogel and are filtered to a specific size range using wire mesh filters. Without filtering, the microgels' applicability, mechanical properties, and drug release characteristics would be significantly different. The filtering step allows for precise control over the size of the hydrogels, and it also allows for high throughput fabrication of microgels that exhibit pH-dependent swelling and sustained release of therapeutics.
A limitation of this process is that the filtering step did not lead to hydrogels with a perfectly spherical shape, which may be an important factor to consider for some applications. For this reason, the characteristic size of the microgels was described using Feret diameter (Figure 3), which is useful for quantifying irregularly-shaped particles39. Although the geometry of the microgels was not a perfect sphere, the average size of the particles was easy to control based on the mesh size of the syringe filter, and, for many applications, having perfectly spherical particles is not necessary. The microgels' polydispersity index (PDI) was quantified using the square-ratio of the standard deviation of Feret diameter to the average Feret diameter obtained from a large population of particles (n = 74). The PDI was calculated as 0.076 using the equation
PDI = (s/D)2
where s is the standard deviation of the average Feret diameter and D is the average Feret diameter40. Due to the filtering done during this process and the use of the Feret diameter for irregularly shaped particles, the polydispersity index of these particles was quite low, to the extent that they could be considered monodisperse.
For future research, several modifications to this protocol could be made to better fit the given research need. For example, only two proteins, SDF1-a and TGF-b3, have been studied for their controlled release with these microgels. Previous work has shown sustained release of these bioactive factors up to ~30 days in vitro. However, other relevant therapeutics, such as nanoparticles, RNA interfering (RNAi) molecules, other biologics, or small-molecule drugs could also be explored to quantify their release rate and efficacy when applied with this chitosan microgel technology. Another variable that could be investigated in the future is changing the size range of the microgels, which is done simply by changing the mesh size of the syringe filter. This could also have a significant impact on the release rate of therapeutics from the microgels, allowing for convenient control over release kinetics without altering the chemistry of crosslinking. Additionally, this protocol can be easily scaled-up using larger syringes and filters or vacuum filtration techniques to produce large quantities of chitosan microgels.
Disclosures
The authors have nothing to disclose.
Acknowledgements
Research reported in this publication was supported by the National Institute of Arthritis and Musculoskeletal and Skin Diseases of the National Institute of Health under award numbers R03AR068087 and R21AR071585 and by the Boettcher Foundation (#11219) to MDK. CBE was supported by NIH/NCATS Colorado CTSA Grant Number TL1 TR001081.
Materials
| Acetic acid | SigmaAldrich | AX0073 | |
| BD Luer-Lock Syringe | Fisher Scientific | 14-823-16E | |
| Büchner Funnel | Fisher Scientific | FB966F | 100 mm diameter |
| Chitosan (low molecular weight) | SigmaAldrich | 448869 | 75-80% deacetylation |
| Dialysis Membrane Tubing | Fisher Scientific | 08-670-5C | 3500 MWCO |
| Ethanol | SigmaAldrich | 493538 | |
| Genipin | SigmaAldrich | G4796 | |
| Heracell 150i Incubator | ThermoFisher | 50116047 | |
| Parafilm | Fisher Scientific | 13-374-12 | |
| Recombinant human SDF-1a | Peprotech | 300-28A | |
| Recombinant human TGF-b3 | Peprotech | 100-36E | |
| Whatman Filter Paper Grade 540 | SigmaAldrich | Z241547 | 8 mm pore size |
| Whatman Filter Paper Grade 541 | SigmaAldrich | WHA1541055 | 22 mm pore size |
| Whatman Filter paper Grade 542 | SigmaAldrich | WHA1542185 | 2.7 mm pore size |
| Wire Mesh Sieve | McMaster-Carr | 9317T86 | No. 100 Mesh |
References
- Mizuta, T., Benson, W. M., Foster, B. K., Morris, L. L. Statistical analysis of the incidence of physeal injuries. Journal of Pediatric Orthopaedics. 7 (5), 518-523 (1987).
- Mann, D. C., Rajmaira, S. Distribution of physeal and nonphyseal fractures in 2,650 long-bone fractures in children aged 0-16 years. Journal of Pediatric Orthopaedics. 10 (6), 713-716 (1990).
- Eid, A. M., Hafez, M. A. Traumatic injuries of the distal femoral physis. Retrospective study on 151 cases. Injury. 33 (3), 251-255 (2002).
- Barmada, A., Gaynor, T., Mubarak, S. J. Premature physeal closure following distal tibia physeal fractures: a new radiographic predictor. Journal of Pediatric Orthopaedics. 23 (6), 733-739 (2003).
- Shaw, N., et al. Regenerative medicine approaches for the treatment of pediatric physeal injuries. Tissue Engineering Part B: Reviews. 24 (2), 85-97 (2018).
- Dabash, S., Prabhakar, G., Potter, E., Thabet, A. M., Abdelgawad, A., Heinrich, S. Management of growth arrest: current practice and future directions. Journal of Clinical Orthopaedics and Trauma. 9, 58-66 (2018).
- Williamson, R. V., Staheli, L. T. Partial physeal growth arrest: treatment by bridge resection and fat interposition. Journal of Pediatric Orthopedics. 10 (6), 769-776 (1990).
- Escott, B. G., Kelley, S. P. Management of traumatic physeal growth arrest. Orthopaedics and Trauma. 26 (3), 200-211 (2012).
- Newsom, J. P., Payne, K. A., Krebs, M. D. Microgels: modular, tunable constructs for tissue regeneration. Acta Biomaterialia. 88, 32-41 (2019).
- Riederer, M. S., Requist, B. D., Payne, K. A., Way, J. D., Krebs, M. D. Injectable and microporous scaffold of densely-packed, growth factor-encapsulating chitosan microgels. Carbohydrate Polymers. 152, 792-801 (2016).
- Xin, S., Wyman, O. M., Alge, D. L. Assembly of PEG microgels into porous cell-instructive 3D scaffolds via thiol-ene click chemistry. Advanced Healthcare Materials. 7 (11), 1800160 (2018).
- Kim, P. -. H., et al. Injectable multifunctional microgel encapsulating outgrowth endothelial cells and growth factors for enhanced neovascularization. Journal of Controlled Release. 187, 1-13 (2014).
- Rabea, E. I., Badawy, M. E. -. T., Stevens, C. V., Smagghe, G., Steurbaut, W. Chitosan as antimicrobial agent: applications and mode of action. Biomacromolecules. 4 (6), 1457-1465 (2003).
- Sarmento, B., Goycoolea, F. M., Sosnik, A., das Neves, J. Chitosan and chitosan derivatives for biological applications: chemistry and functionalization. International Journal of Carbohydrate Chemistry. 2011, 1 (2011).
- Galdioli Pellá, M. C., et al. Chitosan hybrid microgels for oral drug delivery. Carbohydrate Polymers. 239, 116236 (2020).
- Echeverria, C., et al. One-pot synthesis of dual-stimuli responsive hybrid PNIPAAm-chitosan microgels. Materials & Design. 86, 745-751 (2015).
- Kim, M. Y., Kim, J. Chitosan microgels embedded with catalase nanozyme-loaded mesocellular silica foam for glucose-responsive drug delivery. ACS Biomaterials Science & Engineering. 3 (4), 572-578 (2017).
- Mora-Boza, A., et al. Microfluidics generation of chitosan microgels containing glycerylphytate crosslinker for in situ human mesenchymal stem cells encapsulation. Materials Science and Engineering: C. 120, 111716 (2021).
- Zhang, H., Mardyani, S., Chan, W. C. W., Kumacheva, E. Design of biocompatible chitosan microgels for targeted pH-mediated intracellular release of cancer therapeutics. Biomacromolecules. 7 (5), 1568-1572 (2006).
- Huang, P., et al. Effect of pH on the mechanical, interfacial, and emulsification properties of chitosan microgels. Food Hydrocolloids. 121, 106972 (2021).
- Fletcher, N. A., Krebs, M. D. Sustained delivery of anti-VEGF from injectable hydrogel systems provides a prolonged decrease of endothelial cell proliferation and angiogenesis in vitro. RSC Advances. 8 (16), 8999-9005 (2018).
- Fletcher, N. A., Babcock, L. R., Murray, E. A., Krebs, M. D. Controlled delivery of antibodies from injectable hydrogels. Materials Science and Engineering: C. 59, 801-806 (2016).
- Fletcher, N. A., Von Nieda, E. L., Krebs, M. D. Cell-interactive alginate-chitosan biopolymer systems with tunable mechanics and antibody release rates. Carbohydrate Polymers. 175, 765-772 (2017).
- Erickson, C. B., et al. In vivo degradation rate of alginate-chitosan hydrogels influences tissue repair following physeal injury. Journal of Biomedical Materials Research Part B: Applied Biomaterials. , 34580 (2020).
- Erickson, C. B., et al. Anti-VEGF antibody delivered locally reduces bony bar formation following physeal injury in rats. Journal of Orthopaedic Research. , 24907 (2020).
- Lee, M. A., Nissen, T. P., Otsuka, N. Y. Utilization of a murine model to investigate the molecular process of transphyseal bone formation. Journal of Pediatric Orthopaedics. 20 (6), 802-806 (2000).
- Planka, L., et al. Nanotechnology and mesenchymal stem cells with chondrocytes in prevention of partial growth plate arrest in pigs. Biomedical Papers. 156 (2), 128-134 (2012).
- Yu, Y., et al. Rabbit model of physeal injury for the evaluation of regenerative medicine approaches. Tissue Engineering Part C: Methods. 25 (12), 701-710 (2019).
- Xian, C. J., Zhou, F. H., McCarty, R. C., Foster, B. K. Intramembranous ossification mechanism for bone bridge formation at the growth plate cartilage injury site. Journal of Orthopaedic Research. 22 (2), 417-426 (2004).
- Erickson, C. B., Shaw, N., Hadley-Miller, N., Riederer, M. S., Krebs, M. D., Payne, K. A. A rat tibial growth plate injury model to characterize repair mechanisms and evaluate growth plate regeneration strategies. Journal of Visualized Experiments. (125), e55571 (2017).
- Erickson, C., Stager, M., Riederer, M., Payne, K. A., Krebs, M. Emulsion-free chitosan-genipin microgels for growth plate cartilage regeneration. Journal of Biomaterials Applications. 36 (2), 289-296 (2021).
- Yang, D., et al. Microfluidic synthesis of chitosan-coated magnetic alginate microparticles for controlled and sustained drug delivery. International Journal of Biological Macromolecules. 182, 639-647 (2021).
- Marsili, L., Dal Bo, M., Berti, F., Toffoli, G. Thermoresponsive chitosan-grafted-poly(N-vinylcaprolactam) microgels via ionotropic gelation for oncological applications. Pharmaceutics. 13 (10), 1654 (2021).
- Muzzarelli, R., El Mehtedi, M., Bottegoni, C., Aquili, A., Gigante, A. Genipin-crosslinked chitosan gels and scaffolds for tissue engineering and regeneration of cartilage and bone. Marine Drugs. 13 (12), 7314-7338 (2015).
- Muzzarelli, R. A. A. Genipin-crosslinked chitosan hydrogels as biomedical and pharmaceutical aids. Carbohydrate Polymers. 77 (1), 1-9 (2009).
- Butler, M. F., Ng, Y. -. F., Pudney, P. D. A. Mechanism and kinetics of the crosslinking reaction between biopolymers containing primary amine groups and genipin. Journal of Polymer Science Part A: Polymer Chemistry. 41 (24), 3941-3953 (2003).
- Marquez-Curtis, L. A., Janowska-Wieczorek, A. Enhancing the migration ability of mesenchymal stromal cells by targeting the SDF-1/CXCR4 axis. BioMed Research International. 2013, 1-15 (2013).
- Tang, Q. O., et al. TGF-β3: A potential biological therapy for enhancing chondrogenesis. Expert Opinion on Biological Therapy. 9 (6), 689-701 (2009).
- Hogg, R., Turek, M. L., Kaya, E. The role of particle shape in size analysis and the evaluation of comminution processes. Particulate Science and Technology. 22 (4), 355-366 (2004).
- Raval, N., Maheshwari, R., Kalyane, D., Youngren-Ortiz, S. R., Chougule, M. B., Tekade, R. K. Importance of physicochemical characterization of nanoparticles in pharmaceutical product development. Basic Fundamentals of Drug Delivery. , 369-400 (2019).

