Hyperglycemic Clamp and Hypoglycemic Clamp in Conscious Mice
Summary
A hyperglycemic clamp is used for measuring insulin release with a maintained higher blood glucose concentration. A hypoglycemic clamp is for measuring glucose production induced by counter-regulatory responses. Both methods use the same surgical procedure. Here, we present a clamp technique to assess systemic glucose metabolism.
Abstract
Diabetes mellitus (DM) is caused by insufficient insulin release from the pancreatic β-cells (Type1 DM) and insulin sensitivity in muscles, liver, and adipose tissues (Type2 DM). Insulin injection treats DM patients but leads to hypoglycemia as a side effect. Cortisol and catecholamines are released to activate glucose production from the liver to recover hypoglycemia, called counter-regulatory responses (CRR). In DM research using rodent models, glucose tolerance tests and 2-deoxy-glucose injection are used to measure insulin release and CRR, respectively. However, blood glucose concentrations change persistently during experiments, causing difficulties in assessing net insulin release and CRR. This article describes a method in which blood glucose is kept at 250 mg/dL or 50 mg/dL in conscious mice to compare the release of insulin and CRR hormones, respectively.
Polyethylene tubing is implanted in the mice’s carotid artery and jugular vein, and the mice are allowed to recover from the surgery. The jugular vein tubing is connected to a Hamilton syringe with a syringe pump to enable insulin or glucose infusion at a constant and variable rate. The carotid artery tubing is for blood collection. For the hyperglycemic clamp, 30% glucose is infused into the vein, and blood glucose levels are measured from the arterial blood every 5 min or 10 min. The infusion rate of 30% glucose is increased until the blood glucose level becomes 250 mg/dL. Blood is collected to measure insulin concentrations. For hypoglycemic clamp, 10 mU/kg/min insulin is infused together with 30% glucose, whose infusion rate is variable to maintain 50 mg/dL of blood glucose level. Blood is collected to measure counter-regulatory hormones when both glucose infusion and blood glucose reach a steady state. Both hyperglycemic and hypoglycemic clamps have the same surgical procedure and experimental setups. Thus, this method is useful for researchers of systemic glucose metabolism.
Introduction
Glucose is an important source of energy for cells, and a lack of glucose can lead to a variety of symptoms and complications. In the event of low glucose (hypoglycemia, generally less than 70 mg/dL in fasting blood glucose level, but should not be determined by a single value1), the most common symptoms include weakness, confusion, sweating, and headache. It can also disrupt cerebral function and increase the risk of cardiovascular events and mortality2. Conversely, hyperglycemia is a medical condition in which the plasma glucose concentration exceeds normal levels (generally > 126 mg/dL in fasting blood glucose level3). This can occur in individuals with diabetes who have either a deficit in insulin production or utilization. Hyperglycemia can lead to diabetic ketoacidosis, which occurs when the body cannot use glucose for energy but instead breaks down fatty acids for fuel. The hyperglycemic hyperosmolar state also increases mortality4. Long-term hyperglycemia can cause damage to blood vessels, nerves, and organs, leading to the development of several chronic complications such as cardiovascular disease, retinopathies, and kidney diseases. Thus, the blood glucose concentration must be maintained in a tight range between 100 mg/dL and 120 mg/dL.
Blood glucose is regulated by the balance between glucose input and output in a one-compartment model (Figure 1A). Glucose input includes absorbed glucose from food and glucose production from the liver, kidneys, and small intestine. Glucose output comprises glucose uptake in tissues and glucose disposal from the kidneys. Both the amount of glucose input and output are regulated by endocrine hormones. For example, glucagon, corticosterone, and catecholamines, known as counter-regulatory hormones, are released when blood glucose levels decrease5. They stimulate the breakdown of glycogen and the synthesis of glucose, mainly from the liver; these processes are known as glycogenolysis and gluconeogenesis, respectively. Hyperglycemia increases insulin release from pancreatic β-cells and stimulates glucose uptake in the muscles, adipose tissues, and heart6,7,8,9. Exercise increases insulin-independent glucose uptake10. The sympathetic nervous system increases glucose uptake in muscles and brown adipose tissue6,11. To measure the ability to regulate glucose metabolism in peripheral tissues, researchers typically use the glucose tolerance test (GTT) and the insulin tolerance test (ITT) (Figure 1B,C). In GTT, two factors must be considered: insulin release and insulin sensitivity (Figure 1B). However, the glucose concentration curve during the 120 min test is different in each mouse, which may affect different amounts of hormone release. In ITT, blood glucose is regulated by both insulin sensitivity and the release of counter-regulatory hormones. Therefore, it is difficult to determine the precise meaning of glucose metabolism, insulin release, and insulin sensitivity in GTT and ITT, in situations where blood glucose levels are not constant.
To overcome these problems, it is desirable to keep blood glucose at a constant level (or "clamp"). In hyperglycemic clamp, glucose is infused into the bloodstream to raise blood glucose levels to a specific level and then maintained at that level for a period of time. The amount of infused glucose is adjusted based on measurements of blood glucose levels every 5-10 min to maintain a steady state. This technique is particularly useful for understanding the parameters of insulin secretion at a clamped glucose level. Hypoglycemic clamp is a method to maintain low blood glucose levels by infusing insulin. Glucose is infused at a variable rate to maintain a specific blood glucose level. If the mouse cannot recover from hypoglycemia, more glucose should be infused.
Although there are many advantages to performing hyperglycemic and hypoglycemic clamps, the surgical and experimental procedures are considered technically difficult. Thus, few research groups have been able to do them. We aimed to describe these methods for researchers with financial and workforce constraints to start these experiments at a lower budget.
Protocol
All procedures were approved by the Institutional Animal Care and Use Committee (IACUC) at Kumamoto University.
NOTE: For pain relief, ibuprofen was given in drinking water (0.11 mg/mL) for 48 h, and buprenorphine (0.05-0.1 mg/kg i.p.) was given 30 min before surgery. Sterile conditions include gloves, masks, and autoclaved instruments sterilized with ethylene oxide between animals. The surgery was performed on a heating pad set at 37 °C and covered by a new lab mat for each animal. Before the surgery, the surgical area was cleaned with a betadine solution and alcohol. All surgical instruments were sterilized with an autoclave (for no more than two surgeries). Before making the incision, mice were checked to ensure they were fully anesthetized. The depth of anesthesia for each mouse was assessed prior to and during surgery by a toe pinch. The acclimatization period was no more than 5 min each time. Follow the instructions of the IACUC at the respective institution.
1. Preparation of tubings for the jugular vein and carotid artery
- Sterilize all tubings, polypropylene supplies (e.g., pipet tips), and sutures with an autoclave or ethylene oxide. For the jugular vein, connect 8 cm of tubing1 (see Materials) and 3 cm of tubing2 using glue (Figure 2A). Put 2 mm of tubing1 on the top because tubing 2 (polyethylene) is too hard and may damage blood vessels (Tubing1.1).
- Extension tube for the jugular vein (Tubing1.2) to infuse glucose and insulin
- Prepare two 30 cm tubes and one 10 cm tube with tubing2 (Figure 2A). Cut off the sharp end of a 20 μL pipette tip (or anything that has a narrow tip end with OD < 0.5 mm to connect with tubing 1.1) and place three tubing2 (30 cm, 30 cm, and 10 cm) together in it. Seal with adhesive glue.
- Place 5 cm of tubing1 at the other end of tubing2.
- For carotid artery (Tubing1.3): Stretch tubing2 to make it thinner. Connect 8 cm of tubing1 and 3 cm of the stretched tubing2 using glue (Figure 2A). Make a mark at 9 mm from the tip.
- Needle with non-sharp end that connects tubing to the syringe: Make a scratch near the sharp end of the needle (23 G) with a metal file, gently bend it back and forth a few times with pliers, and break it. Do the same cut in the middle of the needle to make a piece of metal connector (Figure 2A).
- Extension tube for the artery to draw blood (Tubing set1.4): Connect 10 cm of tubing1 with the metal connector and the non-sharp needle that was made in step 1.4.
NOTE: All tubings and sutures are sterilized with an autoclave or ethylene oxide
2. Surgery
- Anesthetize a mouse with isoflurane (1.5-2.0%) or ketamine/xylazine (ketamine 10 mg/mL, xylazine 1 mg/mL in 0.9% sterile saline, 0.1 mL/10 g of body weight (BW) via intraperitoneal [ip] injection). Keep the mouse on the warm pad (37 °C) to reduce physical stress. Wait for deep anesthesia for 5-10 min. Confirm the depth of anesthesia by pedal reflex, respiration, and heart rate, and response to stimuli by toe pinching. During surgery, surgical tape should be used to secure the nose cap to the operating table for continuous inhalation of anesthesia. Apply ophthalmic ointment to eyes to prevent dryness while under anesthesia.
- Fill heparinized saline (100 U/mL) in tubing1.1 and 1.3 and connect them with a 1 mL syringe with a non-sharp needle (Figure 2B). Close the end of tubing3 (see Table of Materials) by melting it with a soldering iron, which will be used as caps for tubing1.1 and tubing1.3.
- Shave and wipe the first (interscapular on the back) and second (neck in the front) incision area with three cycles of 10% betadine followed by a preoperative preparation with sterile 70% alcohol. Make a small vertical midline incision 5 mm cephalic to the sternum, blunt dissect the tissues, and expose the artery. Separate the vagus nerve from the artery. This reduces the negative effects of removing the vagus nerve on glucose metabolism.
NOTE: The step from the ventral incision to the step where the catheter is inserted into the vein and secured with silk sutures is performed under a microscope. - Place two silk sutures under the artery. Stop blood flow by tying one suture tightly on the cranial side (Figure 2C-1) and the other loosely at the caudal side (Figure 2C-2), which is enough to stop blood flow but enough to open again later. Place one more suture under the artery (Figure 2C-3).
- Cut the artery near Figure 2C-1 with spring scissors and place tubing1.3 into the artery. Make a loose tie on both the artery and tubing (Figure 2C-3, do not tie firmly. The tubing will be inserted deeper into the artery). Open the knot on the caudal side (Figure 2C-2) to insert the tube until the 9 mm mark reaches the knot in the middle (Figure 2C-3). Securely tie all ligatures and flush with heparinized sterile saline.
- Expose the right jugular vein from the same incision as the right carotid artery for the jugular catheter. Isolate the cranial end and ligate it with silk suture (Figure 2D-1, Table of Materials). Place another piece of suture at the caudal end of the exposed vein (Figure 2D-2). Cut the vein near the mark Figure 2D-1 with spring scissors.
- Insert the catheter (not too deep to prevent penetrating vessels), tie it, and confirm visually that it samples blood. Flush with heparinized sterile saline (0.2 mL) and visually confirm that no blood remains in the catheter.
- Place the mouse on the new sterile surgical drape to prevent infection from the first wound. Turn over the mouse, wipe with three cycles of betadine followed by a preoperative preparation with sterile alcohol, and make a small incision between the shoulder blades.
- Pass a needle holder under the skin from the incision on the back to the ventral side. Clamp the catheters with the needle holder, pass them under the skin, and bring them back. Clean the incision site and close the ventral incisions with a synthetic suture (diameter 0.15-0.2 mm). Clamp the venous catheter with micro serrefine at the incision site between the shoulder blades.
- Cut the catheter 1 cm above the clamp, flush it with heparinized saline, and close it with a cap (step 2.4). Follow the same procedure for the arterial catheter. Close the dorsal incision with a synthetic suture (diameter 0.15-0.2 mm).
- Place the mouse in a warm, clean cage (Figure 2E). Perform postoperative care daily.
3. Recovery
- Single-house the mouse because another mouse may bite the catheter in the group housing.
- To reduce stress by social isolation, keep mice with environmental enrichments (e.g., shelters). Perform postoperative care daily. To relieve pain, distress, and discomfort, provide postoperative care, including analgesia (ibuprofen in drinking water (0.11 mg/mL).
- Observe mice for signs of infection, such as festering, lethargy, or pain at the incision site. Most healthy mice begin walking and feeding about 2 h after surgery. A hunched posture, ruffled fur, and decreased food intake may indicate pain. If these signs are observed, immediately euthanize the mice by decapitating them under deep anesthesia or carbon dioxide asphyxiation.
- Blood will enter the catheter in the artery and form a clot. To maintain catheter lines, remove the clot daily, following steps 3-6.
- Fill a 1 mL syringe and 23 G needle (non-sharp end) with heparinized saline (100 U/mL). Using an induction chamber, anesthetize the mouse lightly with isoflurane (1.0%-1.5%). Then, remove the mouse from the chamber and perform clot removal under isoflurane anesthesia with a nose cap.
- Clamp the tubing for the artery on the back of the mouse with micro serrefine, remove the cap, and extract the blood and clots. Flush the catheter with heparinized saline using another 1 mL syringe with a 23 G needle (non-sharp end) and re-cap it. Clean the venous line like the arterial line in the case of severe clotting.
- Do the same procedure to clean up the catheter once a day for 3-5 days.
- Check the body weight of the mouse. If the body weight reduces by more than 10% from the day of the surgery, apply supportive care and try to improve the normal body condition score, and use the mouse for another experiment.
NOTE: The weight loss of animals was less than 10% in the experiments in this study. Weight loss will strongly affect systemic glucose metabolism. Thus, it is recommended to wait for the body weight recovery or remove from the clamp experiment.
4. Set up the pump system (for hypoglycemic clamp)
- Prepare 1 U/mL insulin in 0.1% BSA saline, 30% glucose in saline, and heparinized saline.
- Measure the body weight of the mouse. Calculate the volume for 1 U/mL insulin to make the insulin infusate (10 mU/kg/min). Infuse 1.7 μL/min of insulin solution into the mice in the clamp experiment. To make 300 μL of insulin infusate, the required volume for 1 U/mL insulin (μL) is 2.647 (μL/g) x Body weight (g). A volume of 300 μL of insulin infusate is enough to finish the clamp experiment on 1 mouse. Table 1 shows an example of making insulin infusate.
- Fill insulin infusate and 30% glucose in each Hamilton syringe, connect them with tubing 1.2, and set the syringe on the syringe pump (Figure 3A). Fill each solution in tubing 1.2. Fill saline in a 1 mL syringe and tubing set 1.4 (Figure 2A).
- Anesthetize the mouse with isoflurane (1.0%-1.5%) and connect tubing1.2 to the venous catheter and tubing set1.4 to the arterial catheter (Figure 3A). Use cellophane tape to hold the tubes together.
- Place the mouse in an empty 500 mL beaker.
NOTE: For hyperglycemic clamp, use saline instead of 1 U/mL insulin.
5. Hypoglycemic clamp
- Measure blood glucose levels every 5-10 min, as shown in Figure 3B, and collect blood samples for hormone measurements at -15 min, 10 min, 20 min, 40 min, 60 min, 80 min, 100 min, and 120 min. Follow step 5.2 to measure blood glucose.
NOTE: It is not necessary to measure blood glucose at all time points, but it is recommended to measure it at least every 10 min. Measure blood glucose every 5 min when its level and glucose infusion rate are not steady. - Clamp the top end of the tubing set1.4 and connect a new syringe, extract 50 μL of blood, and place it in a 1.5 mL tube for washing. Measure blood glucose level.
NOTE: This is the blood diluted by the saline solution in the tube because of catheter washing after the previous sampling. A 50 μL volume is enough to replace diluted blood with pure blood. - Store the diluted blood (red blood cells). Wash the pooled blood (~500 μL) with saline (see steps 5.11-5.12) and return to the body to prevent hypoxia.
- The catheter is connected to the artery with high blood pressure, so when the clamp is loosened, blood will flow out and be used to measure glucose with a handy glucose meter. Infuse 50 μL of saline to keep enough amount of blood fluid.
- For blood sampling, extract an additional 50 μL of blood and place it in a 1.5 mL tube on ice. Infuse 100 μL (50 μL of replacement + 50 μL of sampling) of saline.
- After 0 min of blood glucose measurement, start the insulin syringe pump. First, infuse 30 mU/kg/min in bolus for 2 min (5.1 μL /min), then 10 mU/kg/min (1.7 μL /min) infusion for the rest of the duration.
- Measure blood glucose level and change the glucose infusion rate every 5-10 min for 120 min.
- Create a steady state where the glucose infusion rate does not change, and the blood glucose level is 50 mg/dL.
- After collecting the blood sample at t = 120 min, infuse a euthanasia agent into the jugular vein and collect tissues for RNA or protein analysis.
- Centrifuge blood at 1000 x g for 5 min and measure hormone concentration such as insulin or c-peptide using an ELISA kit according to the manufacturer's protocol.
- For washing blood, centrifuge the blood at 1000 x g for 3 min. Remove the supernatant, add 500 μL of heparinized saline, and perform pipetting.
- Repeat the wash again and place the washed red blood cells in 1 mL syringe for the artery. The blood cells will be reinfused automatically when 50 or 100 μL of saline is infused in steps 5.2 and 5.3. Monitor hematocrit carefully to prevent hypoxia.
NOTE: For blood glucose measurement, a commercial handy glucose meter was used.
6. Hyperglycemic clamp
- Follow the steps as the hypoglycemic clamp technique, but without insulin infusion and with blood glucose adjusted to 250-300 mg/dL. Measure blood glucose levels every 5-10 min, as shown in Figure 3B, and collect blood samples for hormone measurements at -15 min, 10 min, 20 min, 40 min, 60 min, 80 min, 100 min, and 120 min.
- Connect a new syringe to the tubing set1.4, extract 50 μL of blood, and place it in a 1.5 mL tube for washing.
- For blood sampling, extract an additional 50 μL of blood and place it in a 1.5 mL tube on ice. Infuse 100 μL (50 μL of replacement + 50 μL of sampling) of saline.
- After 0 min of blood glucose measurement, start the glucose syringe pump. First, infuse 30 g/kg/min in bolus for 2 min (5.1 μL /min), then 10 g/kg/min (1.7 μL /min) for the rest of the duration.
- Measure the blood glucose level and change the glucose infusion rate every 5 -10 min for 120 min. Create a steady state where the glucose infusion rate does not change, and the blood glucose level is 250-300 mg/dL, a clamped hyperglycemic state.
Representative Results
The hypoglycemic clamp study was performed in male C57BL/6N mice (8 weeks old, more than 25 g BW) 3 h fasted at the start of the experiment (Figure 4A,B). The initial blood glucose level was 136 mg/dL (t = -15 min). If it is less than 90 mg/dL, it may be either because the surgery did not go well, or the arterial catheter was inserted too deep, or blood clots have entered the blood flow. The mouse condition after surgery affects the energy metabolism in the mouse. Physiological glucose metabolism cannot be measured under poor health conditions. C57BL is more prone to clotting after surgery; mice that have a higher body weight, such as FVB or ICR mice, are less likely to lose weight due to clot washing after surgery. Thus, it's better for beginners to use FVB mice for practicing clamp procedures. C57BL mice require more intensive care. The 9 mm catheter insertion into the artery has not given us any trouble in collecting blood, even in FVB and ICR mice. Insulin was started at t = 0 min, and blood glucose levels decreased (Figure 4A). The GIR had not been stable between t = 0 min and t = 70 min. Subsequently, it became a steady state after 80 min.
The hyperglycemic clamp study was also performed in male C57BL/6N mice (8 weeks old, more than 25 g BW) 3 h fasted at the start of the experiment (Figure 4C,D). After measuring blood glucose and collecting blood samples at t = -15 and t = -5 min, glucose was infused from t = 0 min. The blood glucose level became 250 mg/dL at t = 40 min. Steady state continued from t = 30 min to the end.
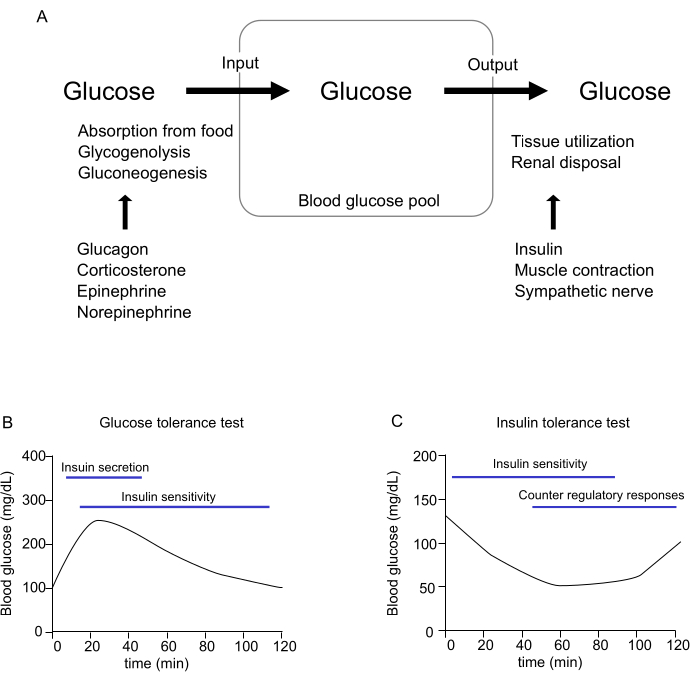
Figure 1: Regulation of blood glucose levels. (A) Conceptual diagram illustrating the regulation of blood glucose in the body. (B,C) Regulation of glucose metabolism in (B) glucose tolerance test (GTT) and (C) insulin tolerance test (ITT). Several factors regulate blood glucose levels. Please click here to view a larger version of this figure.
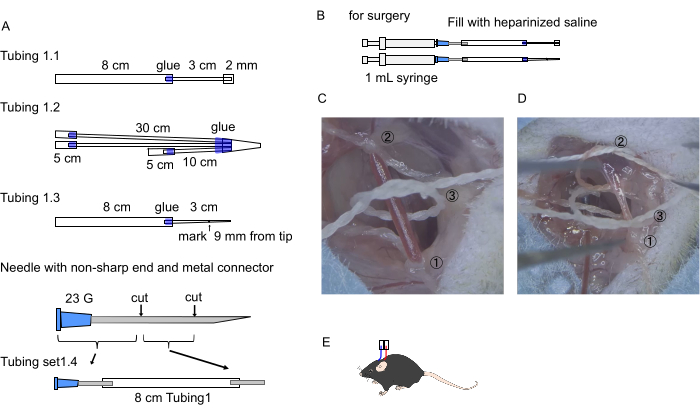
Figure 2: Preparation of tubings and steps of the surgery. (A) Images of tubings for carotid artery and jugular vein. Tubing numbers match the protocol step numbers. (B) Method of preparing the tubings for surgery. (C, D) The numbered positions where the silk threads are ligated to insert the catheter into (C) artery and (D) vein. Positions of the silk threads in the cranial (C-(1)) and caudal side of the artery (C-(2)), and one more in the middle of them (C-(3)) (procedure 2-4). Positions of the silk threads in the cranial (D-(1)) and caudal side of the vein (D-(2)), and one more in the middle of them (D-(3)) (procedure 2-6). (E) Diagram of arterial and venous cannulas exiting the back Please click here to view a larger version of this figure.
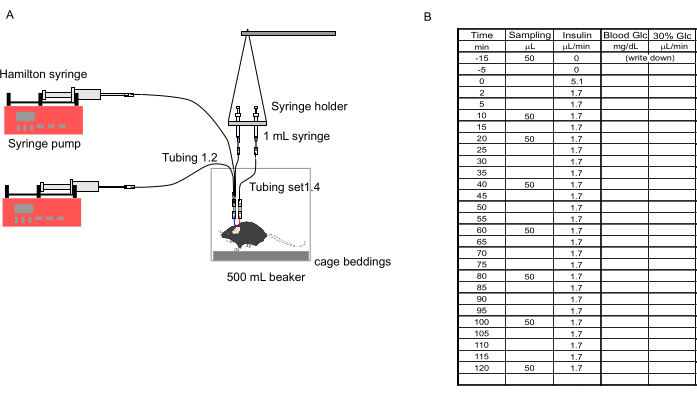
Figure 3: Setting of the clamp. (A) Diagram showing the setup of tubings, pumps, syringes, and the mouse. (B) Sheet to record blood glucose levels and glucose infusion rate. 50 μL of blood is collected at t = -15 min, 10 min, 20 min, 40 min, 60 min, 80 min, 100 min, and 120 min. The insulin infusion rate was 5.1 μL/min from t = 0 to 2 min and changed to 1.7 at t = 2 min. Blood glucose level was measured every 10 min. The blood glucose was measured every 5 min when its level was not stable. The glucose infusion rate was changed each time blood glucose was measured. Please click here to view a larger version of this figure.
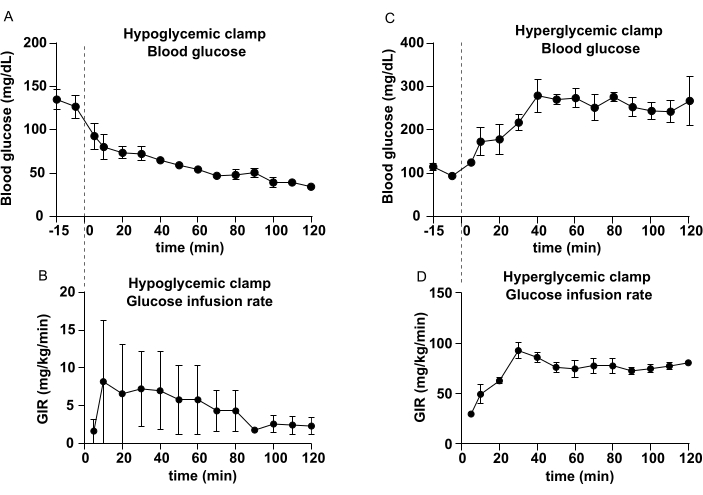
Figure 4: Representative results of hypoglycemic clamp and hyperglycemic clamp. (A) Blood glucose levels and (B) glucose infusion rate of hypoglycemic clamp. (C) Blood glucose levels and (D) glucose infusion rate of the hyperglycemic clamp. Data represent mean ± SEM. Please click here to view a larger version of this figure.
| Solution | Calculation | For 20 g mouse |
| 1 U/mL insulin | (2.647 x body weight) μL | 52.9 μL |
| 0.1% BSA saline | 300 μL – volume of insulin (μL) | 247.1 μL |
Table 1: An example calculation for preparing insulin infusate.
Discussion
The method described here is a simple one that can be done with pipette tips, syringes, and other items found in ordinary laboratories. Although researchers may need to purchase additional tubes and pumps, expensive equipment is not needed. Thus, this protocol of catheterization and clamp is easier to start compared to previous reports12,13,14.
The clamp technique was developed around 1970 and has been used in mice and humans15. It is a useful method for accurately measuring glucose metabolism and is said to be the gold standard. However, it is not a common technique used by many researchers. The hyperinsulinemic-euglycemic clamp technique has been reported in mice13 and rats14, but the method of catheterization here is different, and readers can choose the easier one for their experiments. One of the purposes of this paper is to reduce the hurdle for starting an experiment. Therefore, we provided detailed information on the materials of handmade catheters, surgical procedures, and an example of experimental time course. These are informative for the researcher who attempts to perform clamp in the first time.
Insulin secretion in obesity and diabetes is stage-dependent. Many reports suggest insulin secretion is increased in obesity to reduce blood glucose in the insulin-resistant state16, but β-cell function will be damaged in Type 2 DM17,18. In fact, the number and area of pancreatic islets and insulin secretion have been reported to be increased in obesity mouse models, such as mice fed with a high-fat diet19 or leptin-deficient mice20. In these mouse models, which have an obvious phenotype, differences can be determined by examining blood insulin levels 15-30 min after glucose administration in the GTT. However, in some cases, it is not easy to determine the differences in insulin secretion. For example, if transgenic (Tg) mice have a blood glucose level of 500 mg/dL while a WT mouse has 300 mg/dL and both mice have the same blood insulin level, can we say insulin secretion decreases in Tg? In this case, we cannot compare insulin secretion ability unless blood glucose levels are the same using the method introduced here. This is one of the reasons why there is no established theory as to when β-cell function begins to deteriorate in the transition from obesity to diabetes. We can also measure insulin secretion by primary culture of the pancreas21 or ex vivo22. However, it will ruin the effect of the central nervous system on insulin release because the innervation of the vagus nerve will be removed. Post-absorptive insulin secretion is well-known, but the brain and autonomic nervous system also regulate insulin release23. The experiment to analyze the latter should be performed in an unanesthetized, unrestrained, painless blood collection. This is also why the vagus nerve has to be separated from the carotid artery in step 2.3.
Diabetes mellitus has been reported to cause hyperglycemia due to insulin resistance and increased secretions of glucagon24 and other counter-regulatory hormones25. In addition, repeated episodes of hypoglycemia in humans with diabetes due to failures of insulin dosage or other causes can lead to a condition called recurrent hypoglycemia, in which patients are prone to hypoglycemia26. It has been suggested that the rate of fall in glycemia in hypoglycemic clamp affects peripheral or central detection of the hypoglycemia27. Slow onset hypoglycemia may be appropriate to study the role of glucose sensors in portal-mesenteric veins, while a very rapid decrease in blood glucose may be for the study of brain glucose sensors27. 1 U/mL Insulin is used in lean C57BL mice. But, a higher insulin concentration will be needed in obese mice because they have insulin resistance, and 1 U/mL is not enough to decrease blood glucose levels.
In the one-compartment model of the blood glucose pool (Figure 1A), the amount of absorbed glucose may affect the rate of glucose input14. Thus, the rate of glucose production, one of the main objectives of measuring the hyperinsulinemic-euglycemic clamp, can be affected by the duration of fasting. However, long fasting time may increase the release of counter-regulatory hormones. Hence, researchers set fasting time according to their analysis purpose. Another clamp method includes blood sampling from the tail, which is simple because only an intravenous cannula needs to be inserted14. However, blood is collected in a restraint, which causes a moderate amount of restraint stress and an increase in plasma catecholamines and other stress hormones14. In addition, it is preferable to measure hormone concentrations in the blood flow in the center of the body rather than in the blood at the extremities. Therefore, blood sampling from arteries in free-moving mice is the best for measuring glucose metabolism physiologically. The mice do not move around, and swivel is not needed when they are acclimated to the experimental environment. However, it is recommended to use a swivel to prevent entangling infusion and sampling lines. Tubing1.2 and tubing set1.4 (Figure 3A) are not good for using a swivel. The system should be improved if the researcher is required to use a swivel. Reinfusion of blood cells does not influence the established steady state of blood glucose and insulin. The present method can also be applied to metabolomics studies using isotopes. For example, if 13C-glucose is continuously infused into the vein, the systemic metabolic turnover rate and intracellular intermediate metabolites can be measured28. Thus, this is a useful method to analyze glucose metabolism.
Disclosures
The authors have nothing to disclose.
Acknowledgements
This work was supported by the Leading Initiative for Excellent Young Researchers (from MEXT); a Grant-in-Aid for Scientific Research (B) (Grant Number JP21H02352); Japan Agency for Medical Research and Development (AMED-RPIME, Grant Number JP21gm6510009h0001, JP22gm6510009h9901); the Uehara Memorial Foundation; Astellas Foundation for Research on Metabolic Disorders; Suzuken Memorial Foundation, Akiyama Life Science Foundation, and Narishige Neuroscience Research Foundation. We also thank Nur Farehan Asgar, Ph.D, for editing a draft of this manuscript.
Materials
| Adhesive glue | Henkel AG & Co. KGaA | LOCTITE 454 | |
| ELISA kit (C-peptide) | Morinaga Institute of Bilogical Science Inc | M1304 | Mouse C-peptide ELISA Kit |
| ELISA kit (insulin) | FUJIFILM Wako Pure Chemical Corporation | 633-03411 | LBIS Mouse Insulin ELISA Kit (U-type) |
| Handy glucose meter | Nipro Co. | 11-777 | Free Style Freedom Lite |
| Insulin (100U/ml) | Eli Lilly & Co. | 428021014 | Humulin R (100U/ml) |
| Mouse | Japan SLC Inc. | C57BL/6NCrSlc | C57BL |
| Suture | Natsume seisakusho | C-23S-560 No.2 | Sterilized |
| Syringe Pump | Pump Systems Inc. | NE-1000 | |
| Synthetic suture | VÖMEL | HR-17 | |
| Tubing1 | AS ONE Corporation | 9-869-01 | LABORAN(R) Silicone Tube |
| Tubing2 | Fisher Scientific | 427400 | BD Intramedic PE Tubing |
| Tubing3 | IGARASHI IKA KOGYO CO., LTD. | size5 | Polyethylene tubing size5 |
References
- Seaquist, E. R., et al. Hypoglycemia and diabetes: A report of a workgroup of the american diabetes association and the endocrine society. Journal of Clinical Endocrinology and Metabolism. 98 (5), 1845-1859 (2013).
- Amiel, S. A., et al. Hypoglycaemia, cardiovascular disease, and mortality in diabetes: epidemiology, pathogenesis, and management. The Lancet Diabetes and Endocrinology. 7 (5), 385-396 (2019).
- . Leanne Riley Mean fasting blood glucose Available from: https://www.who.int/data/gho/indicator-metadata-registry/imr-details/2380 (2022)
- Umpierrez, G., Korytkowski, M. Diabetic emergencies-ketoacidosis, hyperglycaemic hyperosmolar state and hypoglycaemia. Nature Reviews Endocrinology. 12 (4), 222-232 (2016).
- Sprague, J. E., Arbeláez, A. M. Glucose counterregulatory responses to hypoglycemia. Pediatric Endocrinology Reviews. 9 (1), 463-473 (2011).
- Toda, C., et al. Distinct effects of leptin and a melanocortin receptor agonist injected into medial hypothalamic nuclei on glucose uptake in peripheral tissues. Diabetes. 58 (12), 2757-2765 (2009).
- Toda, C., et al. Extracellular signal-regulated kinase in the ventromedial hypothalamus mediates leptin-Induced glucose uptake in red-type skeletal muscle. Diabetes. 62 (7), 2295-2307 (2013).
- Toda, C., Kim, J. D., Impellizzeri, D., Cuzzocrea, S., Liu, Z. -. W., Diano, S. UCP2 regulates mitochondrial fission and ventromedial nucleus control of glucose responsiveness. Cell. 164 (5), 872-883 (2016).
- Lee, M. L., et al. Prostaglandin in the ventromedial hypothalamus regulates peripheral glucose metabolism. Nature Communications. 12 (1), 2330 (2021).
- Jessen, N., Goodyear, L. J. Contraction signaling to glucose transport in skeletal muscle. Journal of Applied Physiology. 99 (1), 330-337 (2005).
- Shiuchi, T., et al. Induction of glucose uptake in skeletal muscle by central leptin is mediated by muscle β2-adrenergic receptor but not by AMPK. Scientific Reports. 7 (1), 15141 (2017).
- Ayala, J. E., et al. Hyperinsulinemic-euglycemic clamps in conscious, unrestrained mice. Journal of Visualized Experiments: JoVE. 57, e3188 (2011).
- Hughey, C. C., Hittel, D. S., Johnsen, V. L., Shearer, J. Hyperinsulinemic-euglycemic clamp in the conscious rat. Journal of Visualized Experiments: JoVE. 48, e2432 (2010).
- Ayala, J. E., Bracy, D. P., McGuinness, O. P., Wasserman, D. H. Considerations in the design of hyperinsulinemic-euglycemic clamps in the conscious mouse. Diabetes. 55 (2), 390-397 (2006).
- DeFronzo, R. A., Soman, V., Sherwin, R. S., Hendler, R., Felig, P. Insulin binding to monocytes and insulin action in human obesity, starvation, and refeeding. Journal of Clinical Investigation. 62 (1), 204-213 (1978).
- Czech, M. P. Insulin action and resistance in obesity and type 2 diabetes. Nature Medicine. 23 (7), 804-814 (2017).
- Saisho, Y. β-cell dysfunction: Its critical role in prevention and management of type 2 diabetes. World Journal of Diabetes. 6 (1), 109 (2015).
- Mittendorfer, B., Patterson, B. W., Smith, G. I., Yoshino, M., Klein, S. β Cell function and plasma insulin clearance in people with obesity and different glycemic status. Journal of Clinical Investigation. 132 (3), 154068 (2022).
- Nchienzia, H., et al. Hedgehog interacting protein (Hhip) regulates insulin secretion in mice fed high fat diets. Scientific reports. 9 (1), 11183 (2019).
- Tomita, T., Doull, V., Pollock, H. G., Krizsan, D. Pancreatic islets of obese hyperglycemic mice (ob/ob). Pancreas. 7 (3), 367-375 (1992).
- Uchida, K., et al. Lack of TRPM2 impaired insulin secretion and glucose metabolisms in mice. Diabetes. 60 (1), 119-126 (2011).
- Zhu, Y. X., Zhou, Y. C., Zhang, Y., Sun, P., Chang, X. A., Han, X. Protocol for in vivo and ex vivo assessments of glucose-stimulated insulin secretion in mouse islet β cells. STAR Protocols. 2 (3), 100728 (2021).
- Moullé, V. S. Autonomic control of pancreatic beta cells: What is known on the regulation of insulin secretion and beta-cell proliferation in rodents and humans. Peptides. 148, 170709 (2022).
- Honzawa, N., Fujimoto, K., Kitamura, T. Cell autonomous dysfunction and insulin resistance in pancreatic α cells. International Journal of Molecular Sciences. 20 (15), 3699 (2019).
- Siddiqui, A., Madhu, S. V., Sharma, S. B., Desai, N. G. Endocrine stress responses and risk of type 2 diabetes mellitus. Stress. 18 (5), 498-506 (2015).
- Chan, O., Sherwin, R. Influence of VMH fuel sensing on hypoglycemic responses. Trends in Endocrinology & Metabolism. 24 (12), 616-624 (2013).
- Donovan, C. M., Watts, A. G. Peripheral and central glucose sensing in hypoglycemic detection. Physiology. 29 (5), 314-324 (2014).
- TeSlaa, T., et al. The source of glycolytic intermediates in mammalian tissues. Cell Metabolism. 33 (2), 367-378.e5 (2021).

