Intravital Two-Photon Microscopy of the Transplanted Mouse Lung
Summary
Here, we present a protocol to intravitally image the transplanted mouse left lung using two-photon microscopy. This represents a valuable tool for studying cellular dynamics and interactions in real-time following murine lung transplantation.
Abstract
Complications after lung transplantation are largely related to the host immune system responding to the graft. Such immune responses are regulated by crosstalk between donor and recipient cells. A better understanding of these processes relies on the use of preclinical animal models and is aided by an ability to study intra-graft immune cell trafficking in real-time. Intravital two-photon microscopy can be used to image tissues and organs for depths up to several hundred microns with minimal photodamage, which affords a great advantage over single-photon confocal microscopy. Selective use of transgenic mice with promoter-specific fluorescent protein expression and/or adoptive transfer of fluorescent dye-labeled cells during intravital two-photon microscopy allows for the dynamic study of single cells within their physiologic environment. Our group has developed a technique to stabilize mouse lungs, which has enabled us to image cellular dynamics in naïve lungs and orthotopically transplanted pulmonary grafts. This technique allows for detailed assessment of cellular behavior within the vasculature and in the interstitium, as well as for examination of interactions between various cell populations. This procedure can be readily learned and adapted to study immune mechanisms that regulate inflammatory and tolerogenic responses after lung transplantation. It can also be expanded to the study of other pathogenic pulmonary conditions.
Introduction
Lung transplantation is the final option for many patients suffering from end-stage pulmonary disease; however, long-term survival after lung transplantation is poor compared to other solid organ transplants. Survival at 5 years is only ~60%-70%1, compared to 80%-90% in hearts2 and 85%-90% in kidneys3. Many complications after lung transplantation, such as primary graft dysfunction, antibody-mediated rejection, and chronic lung allograft dysfunction, are due to the host immune response to the allograft. For example, our group has shown that neutrophils are rapidly recruited into the lung allograft following transplant-induced ischemia-reperfusion injury and form dynamic clusters surrounding blood monocytes4. Crosstalk between donor and recipient cells is responsible for deleterious alloimmune responses5,6,7, and the ability to study these dynamic cell interactions in a live animal model is invaluable.
Two-photon microscopy allows for high-resolution intravital imaging for depths up to several hundred microns with minimal photobleaching of tissues8,9. It is used in a variety of tissues and anatomic sites, including the neocortex10,11, skin12,13, and kidney14,15. More recently, it has been adapted to non-static organs such as the lung and heart4,16,17. In this protocol, we describe a technique to image stabilized, ventilated, and perfused pulmonary grafts following murine orthotopic left lung transplantation. A key benefit of the transplant model is the ability to genetically manipulate the donor and recipient separately. Individual cell populations can be visualized with transgenic mouse strains with knock-in fluorescent protein expression, adoptive transfer of fluorescently-labeled cells5, or intravenous injection of fluorescently-labeled antibodies to bind cell-specific markers4,16,17,18.
In order to stabilize the lung during imaging, this protocol involves gluing the lung to a coverglass, while other groups have described suction stabilization using a custom-made reversible vacuum device19. Our protocol has several advantages, including a larger area of imaging and relative ease of setup using commonly available materials in a microscopy lab (including coverglass and glue). Since this gluing technique constrains the lung at the upper surface, it is expected to decrease ventilatory motion and allow for deeper imaging. This intravital imaging technique allows for detailed observation of immune cell behavior and interactions in real-time, which contributes to the study of immune mechanisms that regulate inflammatory versus tolerogenic responses following lung transplantation.
Protocol
All animal handling procedures were conducted in compliance with the National Institutes of Health Care and Use of Laboratory Animals guidelines and approved by the Institutional Animal Care and Use Committee at Washington University School of Medicine.
1. Anesthesia and intubation
NOTE: Orthotopic mouse left lung transplant is performed, as previously described20,21. Lungs from 20-25 g C57BL/6 (B6) mice are transplanted into sex- and age-matched B6 recipients. B6.LysM-GFP reporter mice are used as recipients for select experiments to visualize neutrophil infiltration into lung grafts. Recipient mice can be imaged immediately following a lung transplant.
- Re-anesthetize mice with intraperitoneal administration of ketamine (80−100 mg/kg) and xylazine (8−10 mg/kg). Administer sustained-release buprenorphine (0.5-1.0 mg/kg) subcutaneously for additional pain control. Confirm adequate depth of anesthesia with a paw pinch.
NOTE: If time between lung transplantation and planned intravital imaging is less than two hours, can redose anesthetic with 50% of initial dose of ketamine. - Remove the hair from the entire left chest using an electric clipper and wipe the chest to remove the excess clipped hairs.
- Apply non-medicated ophthalmic ointment to the eyes of the mouse to prevent corneal drying.
- Re-intubate the mouse orotracheally with a 20 G angiocatheter, as previously described21.
- Ventilate with room air at a rate of 120 breaths/min and tidal volume of 0.5 cc.
- Deliver 1% isofluorane endotracheally during the entire procedure.
- To visualize blood vessels during imaging, inject 20 µL of 655 nm nontargeted quantum dots (q-dots) in 50 µL of phosphate-buffered saline intravenously at least 5 min prior to imaging.
2. Surgical preparation of left lung for imaging
- Place the mouse in a right lateral decubitus position (see Figure 1A).
- Disinfect the chest skin with a mixture of 0.75% iodine and 70% ethanol, three times.
- Re-open the left thoracotomy (from mouse lung transplant) through the third intercostal space (see Figure 1A).
- Remove a portion of the skin and soft tissue over the left thoracotomy site, measuring 1.5 cm x 1.5 cm (see Figure 1B).
- Prior to rib resection, first clamp the ribs for ~10 s to thrombose any microvasculature to achieve hemostasis (see Figure 1C, D).
- Resect portions of the 3rd to 5th ribs to create an imaging window large enough to extricate the lung (~0.8 cm craniocaudally and ~1.0 cm anteroposteriorly, Figure 1D, E).
- Stop any additional bleeding from the rib edges with electrocautery.
NOTE: It is critical to achieve hemostasis in the cut edge of the ribs to avoid excessive blood loss during the imaging period. - Place a small bump (4 cm x 4 cm gauze folded longitudinally 3 times) under the right chest (see Figure 1E).
- Using cotton swabs, elevate the lung graft out of the chest and place the lower aspect of the lung onto a 1 cm x 3 cm strip of saline-soaked gauze (see Figure 1F).
NOTE: This strip of saline-soaked gauze prevents sliding of the lung, maintains a moist environment for the lung, protects the lung from the sharp edges of the resected ribs, and separates the lung from heart motion. Once the circumferential imaging chamber is applied over the lung (Figure 1G, H, described in section 3), there should be minimal heat and fluid loss from the open thoracic cavity during the imaging period.
3. Imaging chamber preparation
NOTE: An imaging chamber is custom-built (see Figure 2A). This imaging chamber consists of a base plate and a top plate between which the mouse is placed and secured in place with spring-loaded bolts on either side (Figure 2B). The top plate contains a circular cutout measuring ~2 cm in diameter. A correspondingly sized black O ring is placed into this opening on the front side of the top plate, which will protect the objective lens (Figure 2C). A 24 mm x 50 mm coverglass is adhered to the back of the top plate using high vacuum grease, which will create a watertight seal. This coverglass will serve as the imaging window by adhering to the lung below and holding the imaging media (i.e., water) above. Ensure that no vacuum grease gets within the circular imaging window. The coverglass will be replaced for each new imaging experiment. The base plate is heated to 35-37 ˚C using a thermocouple temperature probe (Figure 2A).
- Place the intubated mouse onto the base plate of the imaging chamber with the open left chest facing up (see Figure 1G, H). Secure the mouse with silk tape over the face, front paws, and tail (see Figure 1H and Figure 2A).
- Apply glue to the bottom side of the cover glass attached to the top plate (grey circle in Figure 2C, bottom), leaving a 0.8-1.0 cm glue-free circle in the center.
NOTE: There is a tradeoff between the size of the glue-free circle and the amount of motion artifact; thus, it is recommended that the circle does not exceed 1.0 cm in diameter. - Lower the top plate until the coverglass makes contact with the lung, with the glue touching the lung surface (see Figure 1G).
NOTE: The left lung will be glued to the coverglass with a clean, glue-free circle in the middle, which is the area that will be imaged. - Hold the coverglass against the lung and inflate the lung for 1-2 s so that the area of glue adheres to the surrounding tissue. Accomplish lung inflation by occluding the outflow tubing from the mouse endotracheal tube to the ventilator.
NOTE: Do not inflate the lung for longer than 1-2 seconds, as this may cause excessive barotrauma. - Back off the top plate slightly to reduce pressure on the lung tissue. Ensure that the area of the lung within the imaging window does not move with ventilation.
- Secure both ends of the top plate by securing the thumb nut over each bolt (see Figure 2B).
4. Two-photon imaging
NOTE: A fixed-stage, upright microscope with a 20x or 25x >1.0 numerical aperture (NA) water immersion objective should be used for intravital microscopy. Below is the setup used in this study for a B6 to B6.LysM-GFP left lung transplant with 655-nm q-dot blood vessel-labeling. When applying this protocol, the setup of the microscope, lasers, and dichroic filters can be adapted based on the needs of the specific experiment and the fluorescent reporters used.
- Place the entire imaging chamber with the intubated recipient mouse (B6 left lung transplanted into B6.LysM-GFP with 655-nm q-dots) onto the microscope stage (see Figure 2A).
- Use dichroic filters of wavelengths 480 nm, 560 nm, and 635 nm, which will appropriately separate the signals from the GFP reporter, q-dots, and second harmonic generation (SHG) signal (445 nm) generated by collagen. These filters can be adapted based on the specific experiment.
NOTE: The SHG signal generated by collagen demarcates dark crater-like structures, which represent alveolar airspaces (see white arrows in Figure 3). - Set the titanium-sapphire femtosecond pulsed laser to 890 nm (within the near-infrared range) for excitation. Use the lowest possible laser power to achieve a sufficient signal.
NOTE: The laser settings should be adjusted for the specific experiment to maximize signal separation. - Apply ~1 mL of water to completely cover the coverglass on the top plate, which will be kept in place by the O-ring and the vacuum grease seal of the cover glass to the top plate (see Figure 1G and Figure 2C).
- Use the 20x >1.0 NA water immersion objective on the microscope. Ensure that the objective lens is adapted based on the specific experiment.
NOTE: Larger NA allows for more light collection, higher contrast, and more detailed images. Water immersion objectives are preferred as water or buffer is more compatible with biological tissues. - Lower the objective of the microscope into the water and focus on the surface of the lung.
- Activate the base plate heater and maintain the temperature at 35-37 ˚C.
NOTE: If there are concerns about the inability to maintain tissue temperature or adequate perfusion to the lung, two additional resisters can be attached to warm the top plate in addition to the base plate (see Supplementary Figure 1). - Acquire image stacks with acquisition software of choice.
NOTE: Ensure that the room is completely dark during acquisition, which may involve the use of blackout curtains/blinds and strategic covering of all light sources.
5. Video acquisition
NOTE: The following parameters can be adapted based on the specific experiment. Steps 5.1-5.3 describe the specific parameters used for the B6 to B6.Lysm-GFP murine left lung transplant, which can be used as a reference guide.
- Using a video-rate bidirectional scanner, set the x-y scan dimensions to 512 x 512 pixels.
- Using an xyz stepper unit, acquire time-lapse recordings of a 21-step z-stack with 10-frame averaging per step. Each stack takes about 20 s to acquire, which includes the time required for frame-averaging, the stepper motor to move and check its position, and a 100 ms delay.
NOTE: The bidirectional scanner on the microscope used in this study operates at a video rate of 30 frames/s. This may differ depending on the specific microscope setup. - Acquire 80 consecutive z-stacks to obtain time-lapse imaging of leukocyte migration within the tissue parenchyma. This takes about ~27 min at 20 s/stack.
NOTE: The stack size, frame-averaging, laser power, and total length of the time-lapse recording should be minimized to prevent excessive laser exposure to the tissue. If fluorescent reporters used are bright (i.e., GFP) and laser power is low, then repeating a 21-step z-stack with a 10-frame average, 512 x 512-pixel scan, at 20 s intervals for ~27 min is well tolerated by the mouse. If longer periods of imaging are desired, the acquisition time per stack should be increased to keep the total laser exposure the same (i.e., 1 min per stack over ~1.5 h). These settings will need to be optimized based on the fluorescent reporters used. - Euthanize the mouse at the end of image acquisition. To euthanize, anesthetize the mouse with ketamine (80−100 mg/kg) and xylazine (8−10 mg/kg), followed by subsequent cervical dislocation. Typical imaging preparations will easily maintain mouse viability for up to 2 h.
NOTE: To extend this period, additional fluid supplementation, anesthesia with isoflurane, and attention to temperature regulation may be required. - Complete video rendering with Imaris. Other imaging software (such as ImageJ) can also be used.
NOTE: Images may be processed post-acquisition using contrast enhancement, smoothing, and channel arithmetics to remove channel crosstalk.
Representative Results
After 1 h of cold ischemic storage at 4 ˚C, we orthotopically transplanted the left lung from a B6 mouse into a B6.LysM-GFP mouse4, and then intravital two-photon imaging was performed, as described above. We performed imaging at two time points post-transplant – 2 h (Figure 3A) and 24 h (Figure 3B). Blood vessels are labeled in red by the q-dots injected immediately prior to imaging. Additionally, we can visualize monocytes that have taken up q-dots at 24 h4. Green-labeled recipient-derived neutrophils can be seen infiltrating the lung graft in the corresponding video at 24 h post-transplant (Supplementary Video 1). Alveoli can be visualized as dark craters due to SHG from collagen (white arrows in Figure 3). We observe that at 24 h post-transplant, there are more neutrophils in the lung compared to at 2 h post-transplant (Figure 3).
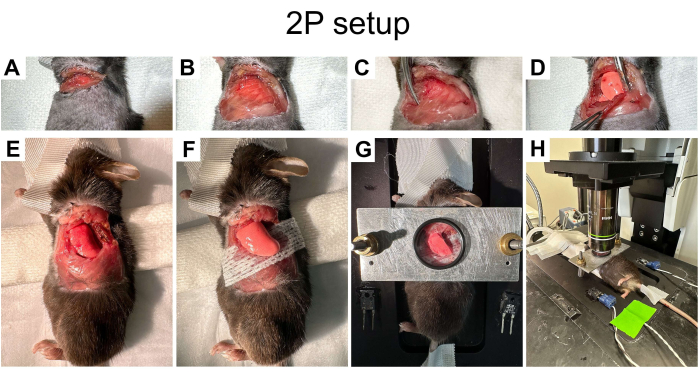
Figure 1: Preparation of mouse for two-photon imaging. (A) Skin re-opened over the left thoracotomy. (B) Skin and soft tissue excised over the left chest. (C,D) Ribs clamped for 10 s prior to dividing. (E) Left 3rd-5th ribs resected to create a window measuring ~0.8 cm craniocaudally x 1.0 cm anteroposteriorly. (F) Left lung elevated out of the left chest and placed on a strip of gauze. (G) An imaging chamber with an imaging window overlies the left lung. (H)The imaging chamber placed onto the microscope stage. Please click here to view a larger version of this figure.
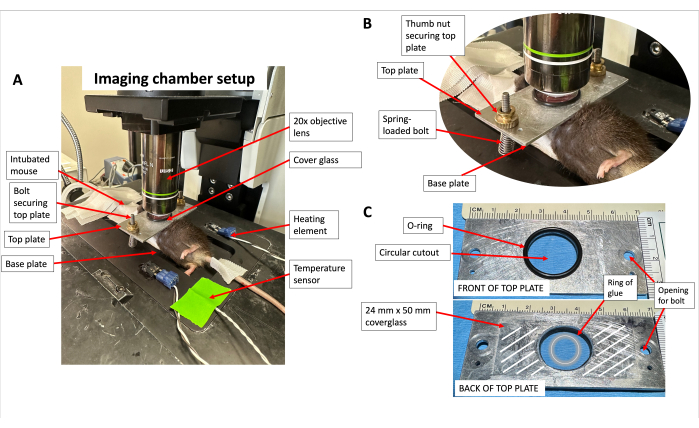
Figure 2: Imaging chamber setup. (A) The imaging chamber placed onto the microscope stage. (B) Magnified view of spring-loaded bolt mechanism to secure top plate over left lung. (C) Front and back of the top plate with O-ring and coverglass applied. White lines represent the area where vacuum grease is applied to adhere coverglass to the top plate. The grey circle represents a ring of glue to apply to the cover glass. Please click here to view a larger version of this figure.
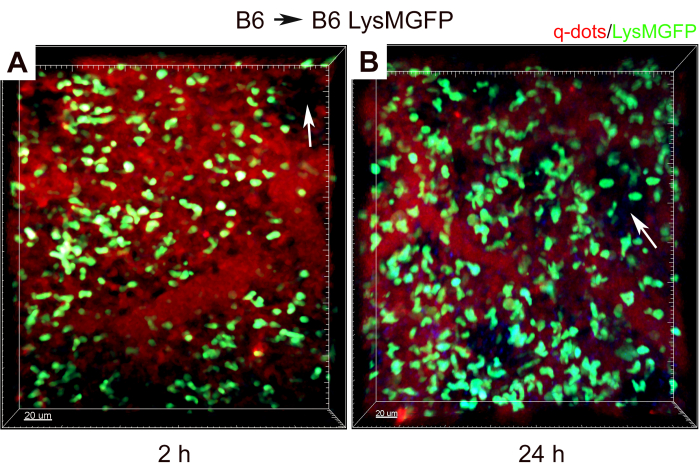
Figure 3: Two-photon intravital imaging of mouse left lung transplant from B6 to B6.LysM-GFP mouse with 1 h cold ischemic storage. Imaging is performed at (A) 2 h and (B) 24 h after transplantation. Red represents q-dots. Green represents neutrophils from B6.LysM-GFP recipient. Blue represents SHG from collagen-rich alveoli. White arrows show alveolar airspaces. The scale bar represents 20 µm. Please click here to view a larger version of this figure.
Supplementary Figure 1: Top plate with two resisters attached to provide additional heating. Please click here to download this File.
Supplementary Video 1: Two-photon time-lapse imaging of transplanted mouse left lung from B6 to B6.LysM-GFP mouse with 1 h of cold ischemic storage, performed at 24 h after transplantation. Red represents q-dots. Green represents neutrophils from LysM-GFP recipient. Blue represents the second harmonic generation from collagen-rich alveoli. The scale bar represents 20 µm. Please click here to download this File.
Discussion
Two-photon excitation was first described in her doctoral thesis by Maria Göppert-Mayer in 1931, who later won the Nobel Prize in Physics for describing the nuclear shell structure22,23. Traditional fluorescence microscopy relies on single-photon excitation, with excitation wavelengths that are shorter and higher energy than emission wavelengths. In contrast to single-photon microscopy, two-photon microscopy involves simultaneous excitation by two photons, each with longer wavelengths than that of the emitted fluorescence signal24,25,26. Because two individual photons need to co-localize to excite a single focal point in the tissue, two-photon excitation is spatially to a small sample volume, allowing for optical sectioning24,25,26 and there is less photobleaching above and below the plane containing the focal point27. The probability of simultaneous excitation by two photons is very low, so a femtosecond pulsed laser is needed to achieve the excitation intensity required for a fluorescence emission signal25,26,27. The excitation laser is in the near-infrared range (700-1100 nm), an advantage of which is less scatter, which allows the light to penetrate more deeply into the tissue28. The depth of imaging is dependent on the oscillation speed and power of the laser24. Within the lung, imaging depths of ~500-600 µm can be achieved17. Thus, the combination of imaging depth and low photobleaching makes two-photon microscopy highly suitable for intravital imaging of organs.
Two-photon microscopy has been used in multiple different tissues and anatomic sites, such as the skin12,13, lymph nodes29, venous endothelium30, brain cortex10,11, kidney14,15, and liver31,32. For example, in mouse dermis, intravital two-photon imaging has been used to describe neutrophil swarming following sterile physical injury12. In a model of venous thrombosis, blood monocytes and neutrophils were visualized crawling along the endothelium of the inferior vena cava30. Human microglia have been successfully imaged in vivo after being xenotransplanted into a human brain organoid model33. With continual advancements in two-photon microscopy, intravital imaging is possible at greater depths and even in non-static organs. For example, depths up to 1000 µm have been imaged in the neocortex in mice using a Ti:Al2O3 regenerative amplifier34.
One of the challenges of intravital imaging in thoracic organs is image stabilization. Due to motion from the beating heart and lung respirations, motion artifacts can significantly reduce image quality. Several methods of stabilization for heart and lung imaging have been developed, including gluing the organ4,7, suction-stabilization19,35,36,37, and a posteriori motion correction38. Our technique of imaging the lung relies on gluing the lung surface to a stabilized coverglass. We keep the lung perfused, ventilated, and normothermic, which simulates the conditions found in vivo. Compared to the suction-stabilization method described by Looney et al.19, this protocol has several advantages. Firstly, we do not require the creation of a specialized suction device. Instead, our imaging chamber and top plate can be constructed with materials readily available in a microscopy lab. Additionally, compared to the suction device (which has an internal diameter of 4 mm), the gluing technique used in this study allows for a larger surface area of imaging (up to 10 mm). With circumferential gluing, the area of the lung within the circle of glue is adequately constrained, and ventilatory motion is minimized. Compared to the suction technique, the cover glass approach prevents convex deformation of the lung surface, which could affect subpleural capillary blood flow. Disadvantages of our system include the inability to remove the coverslip without compromising the pleuroparenchymal junction. Thus, it is not possible to image the lung a second time with the glue technique used here, whereas the suction technique allows for serial imaging. Additionally, the use of a thoracotomy necessitates the loss of the normal negative intrathoracic pressure, which, combined with positive pressure ventilation, is not physiologic. Additional limitations limitations of two-photon imaging are discussed below.
While the chest is open during intravital imaging, it is critical to maintain as close to a physiologic environment as possible. The base plate on which the mouse lies is maintained at a temperature of 35-37 ˚C. With only the base plate heated, we have found that the mouse core temperature (measured via a rectal probe) is well-regulated over several hours. In our experience, cell motility and behavior are only visibly affected when the temperature drops below 32 ˚C, which is unlikely to occur with this setup. If there is a difficulty encountered in maintaining tissue normothermia or lung perfusion during the initial setup of this imaging chamber, two additional resisters can be attached to warm the top plate in addition to the base plate (Supplementary Figure 1). Once the imaging chamber is optimized, it is not necessary to routinely monitor the internal temperature of the mouse during each imaging experiment. Additionally, we have found that the combination of saline-soaked gauze placed over the open thoracic cavity and the occlusive ring of glue around the area of the lung to be imaged are effective at minimizing fluid losses during imaging.
An important technical consideration is achieving good hemostasis during rib resection when creating the thoracic imaging window. Parts of the left 3rd through 5th ribs are resected, and ongoing hemorrhage from these cut ribs will compromise the survival of the mouse during imaging. To combat this, we recommend first clamping the ribs proximally to where they are to be resected for 10 s to thrombose the microvasculature (see Figure 1C,D). Then, following the resection of the ribs, the cut edges of the ribs should be liberally coagulated with electrocautery. Finally, it is critical to place the left lung on top of the strip of saline-soaked gauze (see Figure 1F) during imaging to prevent the sharp rib edges from puncturing the lung. With this combination of technical considerations, we can capture clear videos of immune cell trafficking within the lung parenchyma with minimal motion artifact for several hours without lung injury or loss of normal perfusion patterns.
Two-photon microscopy has been used in lungs to study models of infectious disease and sterile inflammation. One group used two-photon imaging to study resident memory B cell motility and migration following repeat challenges of mouse lungs with influenza virus39. Additionally, two-photon microscopy has been used in skin, kidney, and pancreatic islet transplantation40,41,42,43. With a transplant model, the donor and recipient can be altered genetically to study their respective cell populations. To our knowledge, our group was the first to perform intravital two-photon imaging in murine lung transplants4. We observed rapid neutrophil recruitment and clustering following transplant-mediated ischemia-reperfusion injury, which was reduced with pharmacologic depletion of blood monocytes4. In subsequent work, we have shown that recipient spleen-derived monocytes facilitate neutrophil extravasation through the production of IL-1B44. Intravital imaging in these studies allowed us to uncover upstream regulators of neutrophil extravasation as possible therapeutic targets to reduce the post-transplant inflammatory response45. Additionally, we have also performed intravital imaging in beating mouse hearts following heterotopic transplantation16. We visualized neutrophil recruitment to the myocardium and found that ferroptotic cell death of cardiac graft cells initiates inflammation after heart transplantation46. We applied a similar stabilization technique to transplanted aortic arches to visualize monocytic trafficking into atherosclerotic plaques47. These studies demonstrated the feasibility and utility of intravital imaging of non-static transplanted thoracic organs.
Finally, it is worth discussing methods to visualize different cell types and the lung parenchyma. Two main methods are used to label cells – 1) transgenic mouse strains with knock-in mutations of fluorescent proteins and 2) fluorescent-labeled antibodies to stain cells. Commonly used transgenic mouse strains to identify immune cells by two-photon microscopy include LysM-GFP for neutrophils48, CX3Cr1-GFP for various interstitial macrophage populations49,50, CCR2-GFP for classical monocytes51, CD19-tdTomato for B cells52, and Foxp3-GFP for regulatory T cells5. Notably, CX3Cr1 alone is not sufficient to unambiguously identify cells, but this can be aided by cell morphology and location consistent with that of interstitial macrophages. Alveolar macrophages can be imaged without labeling due to their autofluorescence53,54, distinctive morphology, and behavior (i.e., lack of motility at the time resolution of time-lapse recordings). Compared to eosinophils, which are also autofluorescent, alveolar macrophages are larger and considerably brighter with 890 nm excitation54. Alternatively, alveolar macrophages can also be labeled based on their phagocytic function via intrapulmonary administration of dye aggregates54.
Visualization of immune cells in transplant recipients can also be accomplished by intravascular adoptive transfer of fluorescently labeled cells or by injection of fluorescently labeled antibodies against cell-specific surface markers18,55. Another inherent benefit of two-photon microscopy is SHG by certain birefringent structures within tissue, such as collagen56. This phenomenon is particularly useful for lung imaging because of the high collagen content in alveolar sacs, which acts as an intrinsic indicator of the alveolar structure without additional fluorescent dyes. Further, alveoli are also circumscribed by alveolar capillaries labeled by 655 nm q-dots. Finally, blood vessels can be labeled with intravenous injection of q-dots (as described in this protocol) or rhodamine B dextran. When designing an intravital two-photon microscopy experiment, we recommend the use of no more than four different emission channels to minimize the risk of bleed-through and poor differentiation between channels.
One of the main limitations of our glue stabilization technique described is the inability to remove the coverglass from the lung without potential tissue damage and residual glue on the lung surface. Thus, the lung can only be imaged once rather than at multiple time points over the span of multiple days. To address this limitation, one group has described a semi-permanent thoracic imaging window, which allows for serial imaging of the lung57. However, in our experience, this approach does not allow for adequate lung stabilization for prolonged periods of imaging. This technique affords the ability to image the lung for multiple hours, which provides key insights into innate and adaptive immune responses following transplantation.
Other limitations of two-photon imaging include the expensive upfront cost of setup and expertise required to maintain the system. The scan fields are relatively small compared to immunofluorescent imaging. Thermal and photodamage can be significant without careful acquisition parameter optimization. The two-photon excitation point spread function is broad, which somewhat limits resolution, especially in the z dimension. Additionally, two-photon imaging requires cells of interest to be fluorescently labeled, and some cells cannot be unambiguously labeled with a single cell marker. The number of signals that can be imaged simultaneously is often limited to four or fewer. Finally, anesthesia, surgery, and imaging may induce additional inflammation and physiologic changes on top of the lung transplant that can confound experimental results and interpretations despite best efforts to maintain a physiologic environment. Future directions include the development of minimally invasive methods of intravital imaging (such as through a smaller thoracotomy or bronchoscopic approach).
In conclusion, intravital two-photon microscopy is an invaluable tool in studying multiple disease processes. Here, we describe a reliable technique of lung immobilization and two-photon imaging, which allows for intravital observation of cell trafficking and dynamic interactions between different cell populations after murine lung transplantation. This technique has broadened our understanding of the post-transplant immune landscape and will continue to be an indispensable tool in the study of lung transplantation and other pulmonary disease processes.
Disclosures
The authors have nothing to disclose.
Acknowledgements
This work is supported by grants from NIH 1P01AI11650 and the Foundation for Barnes-Jewish Hospital. We thank the In Vivo Imaging Core at Washington University School of Medicine.
Materials
| 0.75% povidone-iodine | Aplicare | NDC 52380-0126-2 | For disinfectant |
| 1-inch 20G IV catheter | Terumo | SROX2025CA | For endotracheal tube (ETT) |
| 1-inch silk tape | Durapore | 3M ID 7100057168 | To secure mouse in position |
| 20x water immersion long objective lens | Olympus | N20X-PFH | |
| 3M Vetbond glue | Medi-Vet.com | 10872 | To glue coverglass to lung |
| 655 nm non-targeted quantum dots | ThermoFisher | Q21021MP | For labeling of blood vessels |
| 70% ethanol | Sigma Aldrich | EX0281 | For disinfectant |
| Argent High Temp Fine Tip Cautery Pen | McKesson | 231 | |
| Black O ring (2 cm) | Hardware store | N/A | For custom-built imaging chamber |
| Bolt (2) | Hardware store | N/A | For custom-built imaging chamber |
| Brass thumb nut (2) | Hardware store | N/A | For custom-built imaging chamber |
| Buprenorphine 1.3 mg/mL | Fidelis Animal Health | NDC 86084-100-30 | For pain control |
| Chameleon titanium-sapphire femtosecond pulsed laser | Coherent | N/A | |
| Cover glass (24 mm x 50 mm) | Thomas Scientific | 1202F63 | For custom-built imaging chamber |
| Curved mosquito clamp (1) | Fine Science Tools | 13009-12 | |
| Dual channel heater controller | Warner Instruments | TC-344B | |
| Fine scissors (1) | Fine Science Tools | 15040-11 | |
| Fixed-stage upright microscope | Olympus | BX51WI | |
| Gauze (cut to 1 cm x 3 cm) | McKesson | 476709 | To place under left lung |
| High vacuum grease | Dow Corning | N/A | To adhere coverglass onto top plate |
| Isoflurane 1% | Sigma Aldrich | 26675-46-7 | For anesthesia |
| Ketamine hydrochloride 100 mg/mL | Vedco | NDC 50989-996-06 | For anesthesia |
| Metal sheet (3 cm x 7 cm) | Hardware store | N/A | For custom-built imaging chamber |
| Pointed cotton-tipped applicators | Solon | 56225 | To manipulate lung and for blunt dissection |
| Power Pro Ultra clipper | Oster | 078400-020-001 | |
| Puralube Vet eye ointment | Medi-Vet.com | 11897 | To prevent eye dessiccation |
| Small animal ventilator | Harvard Apparatus | 55-0000 | |
| Straight forceps (1) | Fine Science Tools | 91113-10 | |
| Three channel shutter driver | Uniblitz | VMM-D3 | Resonant scanner |
| x.y.z optical stepper motor | Prior Scientific | OptiScan II | |
| Xylazine 20 mg/mL | Akorn | NDC 59399-110-20 | For pain control |
References
- Chambers, D. C., et al. The International Thoracic Organ Transplant Registry of the International Society for Heart and Lung Transplantation: Thirty-eighth adult lung transplantation report – 2021; Focus on recipient characteristics. J Heart Lung Transplant. 40 (10), 1060-1072 (2021).
- Khush, K. K., et al. The International Thoracic Organ Transplant Registry of the International Society for Heart and Lung Transplantation: 37th adult heart transplantation report-2020; focus on deceased donor characteristics. J Heart Lung Transplant. 39 (10), 1003-1015 (2020).
- Wang, J. H., Skeans, M. A., Israni, A. K. Current status of kidney transplant outcomes: Dying to survive. Adv Chronic Kidney Dis. 23 (5), 281-286 (2016).
- Kreisel, D., et al. In vivo two-photon imaging reveals monocyte-dependent neutrophil extravasation during pulmonary inflammation. Proc Natl Acad Sci U S A. 107 (42), 18073-18078 (2010).
- Li, W., et al. Bronchus-associated lymphoid tissue-resident Foxp3+ T lymphocytes prevent antibody-mediated lung rejection. J Clin Invest. 129 (2), 556-568 (2019).
- Kurihara, C., et al. Crosstalk between nonclassical monocytes and alveolar macrophages mediates transplant ischemia-reperfusion injury through classical monocyte recruitment. JCI Insight. 6 (6), e147282 (2021).
- Spahn, J. H., et al. DAP12 expression in lung macrophages mediates ischemia/reperfusion injury by promoting neutrophil extravasation. J Immunol. 194 (8), 4039-4048 (2015).
- Miller, M. J., Wei, S. H., Parker, I., Cahalan, M. D. Two-photon imaging of lymphocyte motility and antigen response in intact lymph node. Science. 296 (5574), 1869-1873 (2002).
- Cahalan, M. D., Parker, I., Wei, S. H., Miller, M. J. Two-photon tissue imaging: seeing the immune system in a fresh light. Nat Rev Immunol. 2 (11), 872-880 (2002).
- Svoboda, K., Helmchen, F., Denk, W., Tank, D. W. Spread of dendritic excitation in layer 2/3 pyramidal neurons in rat barrel cortex in vivo. Nat Neurosci. 2 (1), 65-73 (1999).
- Helmchen, F., Svoboda, K., Denk, W., Tank, D. W. In vivo dendritic calcium dynamics in deep-layer cortical pyramidal neurons. Nat Neurosci. 2 (11), 989-996 (1999).
- Ng, L. G., et al. Visualizing the neutrophil response to sterile tissue injury in mouse dermis reveals a three-phase cascade of events. J Invest Dermatol. 131 (10), 2058-2068 (2011).
- Lammermann, T., et al. Neutrophil swarms require LTB4 and integrins at sites of cell death in vivo. Nature. 498 (7454), 371-375 (2013).
- Ashworth, S. L., Sandoval, R. M., Tanner, G. A., Molitoris, B. A. Two-photon microscopy: visualization of kidney dynamics. Kidney Int. 72 (4), 416-421 (2007).
- Zhang, K., et al. In vivo two-photon microscopy reveals the contribution of Sox9(+) cell to kidney regeneration in a mouse model with extracellular vesicle treatment. J Biol Chem. 295 (34), 12203-12213 (2020).
- Li, W., et al. Intravital 2-photon imaging of leukocyte trafficking in beating heart. J Clin Invest. 122 (7), 2499-2508 (2012).
- Nava, R. G., et al. Two-photon microscopy in pulmonary research. Semin Immunopathol. 32 (3), 297-304 (2010).
- Li, W., et al. Necroptosis triggers spatially restricted neutrophil-mediated vascular damage during lung ischemia reperfusion injury. Proc Natl Acad Sci U S A. 119 (10), e2111537119 (2022).
- Looney, M. R., et al. Stabilized imaging of immune surveillance in the mouse lung. Nat Methods. 8 (1), 91-96 (2011).
- Krupnick, A. S., et al. Orthotopic mouse lung transplantation as experimental methodology to study transplant and tumor biology. Nat Protoc. 4 (1), 86-93 (2009).
- Okazaki, M., et al. A mouse model of orthotopic vascularized aerated lung transplantation. Am J Transplant. 7 (6), 1672-1679 (2007).
- Grzybowski, A., Pietrzak, K. Maria Goeppert-Mayer (1906-1972): two-photon effect on dermatology. Clin Dermatol. 31 (2), 221-225 (2013).
- Göppert-Mayer, M. Uber Elementarakte mit zwei Quantensprüngen. Ann Phys. 401 (3), 273-294 (1931).
- Helmchen, F., Denk, W. Deep tissue two-photon microscopy. Nat Methods. 2 (12), 932-940 (2005).
- Denk, W., Strickler, J. H., Webb, W. W. Two-photon laser scanning fluorescence microscopy. Science. 248 (4951), 73-76 (1990).
- Diaspro, A., et al. Multi-photon excitation microscopy. Biomed Eng Online. 5, 36 (2006).
- Williams, R. M., Piston, D. W., Webb, W. W. Two-photon molecular excitation provides intrinsic 3-dimensional resolution for laser-based microscopy and microphotochemistry. FASEB J. 8 (11), 804-813 (1994).
- Denk, W., et al. Anatomical and functional imaging of neurons using 2-photon laser scanning microscopy. J Neurosci Methods. 54 (2), 151-162 (1994).
- Chtanova, T., et al. Dynamics of neutrophil migration in lymph nodes during infection. Immunity. 29 (3), 487-496 (2008).
- von Bruhl, M. L., et al. Monocytes, neutrophils, and platelets cooperate to initiate and propagate venous thrombosis in mice in vivo. J Exp Med. 209 (4), 819-835 (2012).
- Stanciu, S. G., et al. Experimenting liver fibrosis diagnostic by two photon excitation microscopy and Bag-of-Features image classification. Sci Rep. 4, 4636 (2014).
- Hsiao, C. Y., et al. Improved second harmonic generation and two-photon excitation fluorescence microscopy-based quantitative assessments of liver fibrosis through auto-correction and optimal sampling. Quant Imaging Med Surg. 11 (1), 351-361 (2021).
- Schafer, S. T., et al. An in vivo neuroimmune organoid model to study human microglia phenotypes. Cell. 186 (10), 2111-2126 (2023).
- Theer, P., Hasan, M. T., Denk, W. Two-photon imaging to a depth of 1000 microm in living brains by use of a Ti:Al2O3 regenerative amplifier. Opt Lett. 28 (12), 1022-1024 (2003).
- Headley, M. B., et al. Visualization of immediate immune responses to pioneer metastatic cells in the lung. Nature. 531 (7595), 513-517 (2016).
- Presson, R. G., et al. Two-photon imaging within the murine thorax without respiratory and cardiac motion artifact. Am J Pathol. 179 (1), 75-82 (2011).
- Ueki, H., Wang, I. H., Zhao, D., Gunzer, M., Kawaoka, Y. Multicolor two-photon imaging of in vivo cellular pathophysiology upon influenza virus infection using the two-photon IMPRESS. Nat Protoc. 15 (3), 1041-1065 (2020).
- Fiole, D., et al. Two-photon intravital imaging of lungs during anthrax infection reveals long-lasting macrophage-dendritic cell contacts. Infect Immun. 82 (2), 864-872 (2014).
- MacLean, A. J., et al. Secondary influenza challenge triggers resident memory B cell migration and rapid relocation to boost antibody secretion at infected sites. Immunity. 55 (4), 718-733 (2022).
- Abdulreda, M. H., et al. High-resolution, noninvasive longitudinal live imaging of immune responses. Proc Natl Acad Sci U S A. 108 (31), 12863-12868 (2011).
- Camirand, G. New perspectives in transplantation through intravital microscopy imaging. Curr Opin Organ Transplant. 18 (1), 6-12 (2013).
- Camirand, G., et al. Multiphoton intravital microscopy of the transplanted mouse kidney. Am J Transplant. 11 (10), 2067-2074 (2011).
- Celli, S., Albert, M. L., Bousso, P. Visualizing the innate and adaptive immune responses underlying allograft rejection by two-photon microscopy. Nat Med. 17 (6), 744-749 (2011).
- Hsiao, H. M., et al. Spleen-derived classical monocytes mediate lung ischemia-reperfusion injury through IL-1beta. J Clin Invest. 128 (7), 2833-2847 (2018).
- Li, W., et al. Resolvin D1 prevents injurious neutrophil swarming in transplanted lungs. Proc Natl Acad Sci U S A. 120 (31), e2302938120 (2023).
- Li, W., et al. Ferroptotic cell death and TLR4/Trif signaling initiate neutrophil recruitment after heart transplantation. J Clin Invest. 129 (6), 2293-2304 (2019).
- Li, W., et al. Visualization of monocytic cells in regressing atherosclerotic plaques by intravital 2-photon and positron emission tomography-based imaging-Brief report. Arterioscler Thromb Vasc Biol. 38 (5), 1030-1036 (2018).
- Faust, N., Varas, F., Kelly, L. M., Heck, S., Graf, T. Insertion of enhanced green fluorescent protein into the lysozyme gene creates mice with green fluorescent granulocytes and macrophages. Blood. 96 (2), 719-726 (2000).
- Garcia, J. A., Cardona, S. M., Cardona, A. E. Analyses of microglia effector function using CX3CR1-GFP knock-in mice. Methods Mol Biol. 1041, 307-317 (2013).
- Kim, Y. M., Jeong, S., Choe, Y. H., Hyun, Y. M. Two-photon intravital imaging of leukocyte migration during inflammation in the respiratory system. Acute Crit Care. 34 (2), 101-107 (2019).
- Bajpai, G., et al. Tissue resident CCR2- and CCR2+ cardiac macrophages differentially orchestrate monocyte recruitment and fate specification following myocardial injury. Circ Res. 124 (2), 263-278 (2019).
- Adamo, L., et al. Myocardial B cells are a subset of circulating lymphocytes with delayed transit through the heart. JCI Insight. 5 (3), e134700 (2020).
- Lohmeyer, J., Friedrich, J., Rosseau, S., Pralle, H., Seeger, W. Multiparameter flow cytometric analysis of inflammatory cells contained in bronchoalveolar lavage fluid. J Immunol Methods. 172 (1), 59-70 (1994).
- Neupane, A. S., et al. Patrolling alveolar macrophages conceal bacteria from the immune system to maintain homeostasis. Cell. 183 (1), 110-125 (2020).
- McDonald, B., et al. Intravascular danger signals guide neutrophils to sites of sterile inflammation. Science. 330 (6002), 362-366 (2010).
- Zipfel, W. R., et al. Live tissue intrinsic emission microscopy using multiphoton-excited native fluorescence and second harmonic generation. Proc Natl Acad Sci U S A. 100 (12), 7075-7080 (2003).
- Borriello, L., Traub, B., Coste, A., Oktay, M. H., Entenberg, D. A Permanent Window for Investigating Cancer Metastasis to the Lung. J Vis Exp. (173), e62761 (2021).

.
