Solid Phase Synthesis of a Functionalized Bis-Peptide Using “Safety Catch” Methodology
Instructor Prep
concepts
Student Protocol
1. Setup
- The reaction set-up for the solid-phase synthesis is a polypropylene filter cartridge or glass reactor that is connected via polypropylene tubing to an enclosed filtering flask under vacuum as shown in Figure 1. The reaction can be mixed by a magnetic stir bar or by bubbling nitrogen through the reactor.
- A gas manifold connected to an Argon cylinder equipped with a drying tube and oil bubbler is also recommended as it allows the reaction vessel to be contained under an inert atmosphere and allows the removal of reagents from sealed containers.
- All operations are performed in a fume hood and proper personal protective equipment (safety glasses, lab coat and nitrile gloves) is required.
2. Loading First Bis-Peptide Onto Resin
- Weigh 114 mg of HMBA-AM Resin (0.88 mmol / g loading, 100 μmol) into 8 mL reaction vessel and add magnetic stir bar. Cap the top of the vessel with a rubber septum and purge the tube with argon for at least 5 minutes.
- In the meantime, weigh 117.3 mg of compound 1 of Figure 3 (586.63 g / mol, 2eq) and 59.2 mg of 1-(mesitylene-2-sulfonyl)-3-nitro-1,2,4-triazole (MSNT, 296.0 g / mol, 2eq) into a 15 mL disposable centrifuge tube and dissolve in 2 mL anhydrous dichloromethane (DCM). Add 24 μL of 1-methylimidazole (NMI, 80.81 mL / mol, 3eq) to solution and mix until completely dissolved.
- Transfer the activated solution to the reaction vessel via syringe and allow to stir under argon overnight (~10 hours).
- Remove the septum and drain the reaction mixture. Wash the resin with DCM (5x) and dimethylformamide (DMF) (5x). Perform the “methyl red test” described in section 10.1 to assess the degree of resin loading. If the resin remains red during the methyl red test then steps 2.2 and 2.3 should be repeated. A yellow color, indicative of a negative methyl red test, is preferred; however, since any remaining hydroxyl groups will be capped in the next step, a slightly positive result (light orange resin color) may be acceptable.
3. Deprotection of First Bis-Peptide and Simultaneous Resin Capping
- Add 1 mL of DCM to the reaction vessel then add 1 mL 33% hydrogen bromide in acetic acid dropwise over 30 seconds (bubbling occurs) and allow to stir for 15 minutes. Drain and wash the resin with DCM (5x) then repeat process once more.
- Wash the resin with DCM (5x) then DMF (5x). Neutralize the resin by washing twice with a 5% v/v solution of N,N-diisopropylethylamine (DIPEA) in DMF then wash with DCM (5x) and DMF (5x) again. Perform the “methyl red test” and “chloranil test” discussed in section 10.1 and 10.2. Results should be negative for the methyl red test and positive for the chloranil test.
4. Coupling Boc/tBu-Protected Functionalized Bis-Amino Acid
- Reintroduce an inert atmosphere to the resin-containing reaction vessel by washing three times with anhydrous DCM then attach a septum and argon line. Purge and wash the vessel by adding 1-2 mL of anhydrous DCM and letting stir for 30 seconds then draining the vessel until the argon line bubbler begins to rise. Do this at least once more.
- Prepare a solution of 0.15 M functionalized bis-amino acid (3eq) and 245 mg of 1-hydroxy-7-azabenzotriazole (HOAt, 136.11 g / mol, 18eq) in 2 mL of 2:1 DCM:DMF in a flame dried test tube under argon atmosphere. Add 47 μL of diisopropylcarbodiimide (DIC, 156.6 mL / mol, 3eq) and stir for 90 minutes.
- Add 35 μL DIPEA (174.19 mL / mol, 2eq) in 666 μL anhydrous DMF to resin and allow to stir for 5 minutes.
- Transfer the pre-activated bis-amino acid solution to reaction vessel via syringe and allow to stir overnight.
- Drain the reaction mixture and wash twice with anhydrous DCM while under argon.
- To promote the closure of the diketopiperazine, add a 0.25 M solution of HOAt (136.11 g / mol, 10eq) and DIC (156.6 mL / mol, 10eq) in a 4 mL of 1:1 DCM:DMF and allow to stir under argon for 1 hour.
- Remove the septum and drain the reaction mixture. Wash the resin with DCM (5x) and DMF (5x). If desired, perform the “chloranil test” discussed in section 10.2.
5. Deprotection of Boc/tBu-Protected Functionalized Bis-Amino Acid
- Add 2 mL of a solution of 95:5 trifluororacetic acid (TFA):triisopropylsilane (TIPS) to the reaction vessel and allow it to stir for 1 hour. Drain and wash the resin for about 30 seconds with DCM (5x) then repeat process once more.
- Wash the resin with DCM (5x) then DMF (5x). Neutralize the resin by washing twice with a 5%v/v solution of DIPEA in DMF then wash DCM (5x) and DMF (5x) again. If desired, perform the “chloranil test” discussed in section 10.2.
6. Repeat steps 4 and 5 as desired to synthesize targeted bis-peptide.
7. Functionalizing the Bis-Peptide Prolidine End
- The prolidine end of the growing bis-peptide may be acylated independently or together through a diketopiperazine. Also, this end can be left protected, which will be cleaved latter, affording the free amino acid. If desired, perform the “chloranil test” discussed in section 10.2 to assess coupling efficiency.
8. Deprotection of Fmoc and Acylation of the Quaternary End of the Bis-Peptide
- A 2mL solution of 20% piperidine in DMF is added and the reaction is mixed for 20 minutes. Drain and wash the resin with DMF (5x) then repeat process once more.
- Wash the resin with DCM (5x) then DMF (5x).
- Prepare a 0.15 M solution of amino acid (3eq) in 2 mL of N-methylpyrrolidone (NMP) with 114 mg 2-(7-aza-1H-benzotriazole-1-yl)-1,1,3,3-tetramethyluronium hexafluorophosphate (HATU, 380.2 g / mol, 3eq) and 104.5 μL DIPEA (174.19 mL / mol, 6eq) and mix well. Add to reaction vessel and allow to stir for 6 hours.
- Wash the resin with DCM (5x) then DMF (5x).
9. Remove the Boc Group from the Resin Bound Amino Acid and Cleave from Resin
- Add 2 mL of a 1:1 TFA:DCM solution to the reaction vessel and allow to stir for 30 minutes. Drain and wash the resin with DCM (5x) then repeat process once more.
- Wash and drain the resin for 30 seconds with DCM (5x) then DMF (5x).
- Add 2 mL of a solution of 10% DIPEA in anhydrous DMF and allow to stir 24-48 hours.
- Collect reaction mixture into pre-weighed round bottom flask. Transfer 30 μL of this solution to 450 μL of THF in an LC-MS vial and submit for analysis. Wash the resin with additional aliquots of DMF and collect into the round bottom flask then remove solvent in vacuo.
10. Purification of Bis-Peptide
- Dissolve crude bis-peptide in a minimal amount of dimethyl sulfoxide (100-250 μL) and transfer into HPLC vial insert. Place insert into autosampler of semi-prepitive HPLC system (Hewlett Packard 1100 Series) equipped with an XTerra Prep MS C18 5 μm 7.8×150 mm column and a 100 μL injection loop.
- Perform multiple 50 μL injections of the sample using a gradient program of 5-95% acetonitrile in water with 0.1% formic acid over 30 minutes while monitoring at 274 nm. Collect the product peak in a pre-weighed disposable centrifuge tube and freeze dry using a lyophilizer. Caution should be taken with the first run as a slight shift in peak retention time compared to analytical LCMS is typically observed.
11. Assessment Methods
- METHYL RED TEST7: Remove ~1 mg of dry resin via disposable pipette and rinse into 4 mL reaction vessel. Add a solution of 20 mg methyl red, 50 μL N,N’-diisopropylcarbodiimide (DIC), and 5 mg 4-dimethylaminopyridine (DMAP) in 500 μL anhydrous DCM and allow to stir for 5-10 min. Drain and wash resin with DCM until the filtrate becomes colorless. Positive indication is the resin beads remaining orange or red.
- CHLORANIL TEST12: Transfer ~1 mg of dry resin into a small vial via disposable pipette. Add 3 drops of both a 0.8 mM chloranil in DMF solution and 2% acetaldehyde in DMF solution and let sit at room temperature for 5-10 minutes. Positive indication is the resin beads turning blue/purple.
- ACTIVATION TRAP TEST: Activated compounds during the synthesis can be assessed by transferring a small amount (5-10 μL) of the activated solution to a liquid chromatography-mass spectrometry (LC-MS) vial containing 50 μL of pyrrolidine. Mix by hand for a few seconds (solution should become yellow) then dilute with 450 μL of tetrahydrofuran (THF) and submit for LC-MS analysis.
- ANALYTICAL LC-MS: The final product and activated intermediates can be assessed using an HP 1200 series LC-MS system equipped with a Waters Xterra MS C18 3.5 μm 4.6 mm x 150 mm column and a gradient system of 5-95% acetonitrile in water with 0.1% formic acid over 30 minutes.
12. Representative Results
An example of both crude (Figure 4) and purified (Figure 5) LCMS traces are provided. Purified yields of approximately 10% are expected using the methods outlined above.
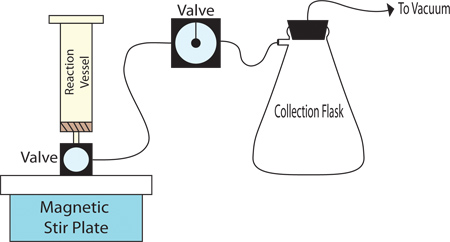
Figure 1. Diagram of Experimental Set-Up for Solid Phase Synthesis.
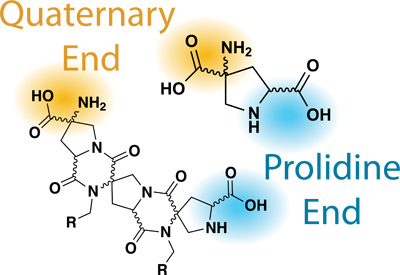
Figure 2. Relevant Nomenclature of Bis-Amino Acids/Bis-Peptides.
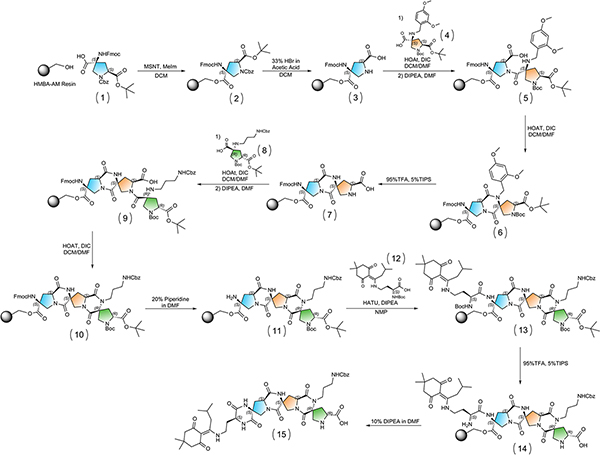
Figure 3. Overall Synthetic Scheme. Click here to view larger figure.
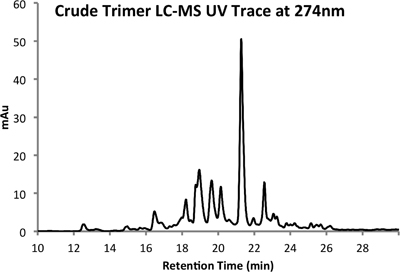
Figure 4a. HPLC Trace of Crude Product at 274 nm.
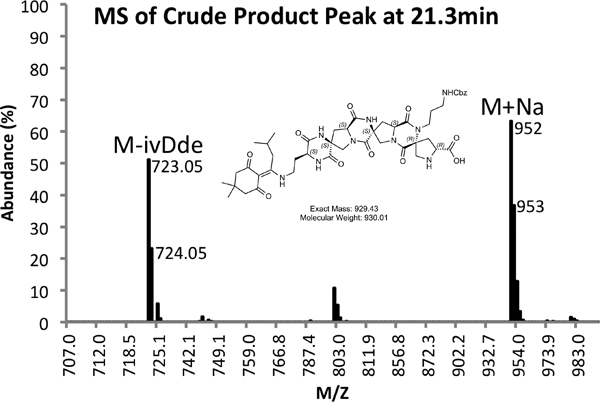
Figure 4b. MS Spectrum of Crude Product Peak.
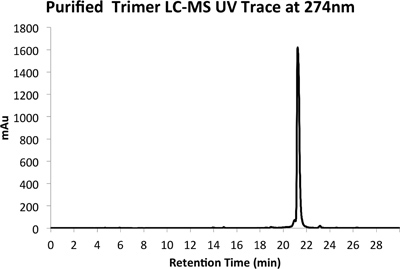
Figure 5a. HPLC Trace of Purified Product at 274 nm.
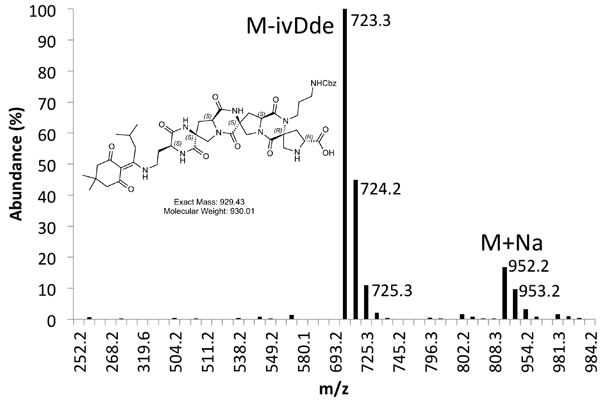
Figure 5b. MS Spectrum of Purified Product Peak.
Solid Phase Synthesis of a Functionalized Bis-Peptide Using “Safety Catch” Methodology
Learning Objectives
List of Materials
| Name | Company | Catalogue Number | Comments |
| HMBA-Am Resin | NovaBiochem | 855018 | |
| MSNT | NovaBiochem | 851011 | |
| NMI | Sigma-Aldrich | 336092 | Toxic, Corrosive |
| DCM | Sigma-Aldrich | D65100 | Carcinogenic |
| Anhydrous DCM | Acros | 34846 | Carcinogenic |
| 33% Hydrogen Bromide in Acetic Acid | Sigma-Aldrich | 248630 | Toxic, Corrosive, Fumes when open |
| DIPEA | Sigma-Aldrich | 387649 | Flammable, Toxic, Corrosive |
| DMF | Fisher Scientific | AC27960 | Flammable, Toxic |
| Anhydrous DMF | Acros | 34843 | Flammable, Toxic |
| HOAt | GenScript | C01568 | |
| DIC | Acros | BP590 | Flammable, Toxic, Corrosive |
| TFA | Sigma-Aldrich | T6508 | Toxic, Corrosive |
| TIPS | Acros | 21492 | Flammable, Toxic |
| Piperidine | Sigma-Aldrich | 104094 | Flammable, Toxic, Corrosive |
| HATU | GenScript | C01566 | Toxic |
| NMP | Acros | 36438 | Toxic |
| DMAP | NovaBiochem | 851055 | Toxic |
| Methyl Red | Sigma-Aldrich | 250198 | |
| THF | Sigma-Aldrich | 401757 | Flammable, Toxic, Peroxide Forming |
| Pyrrolidine | Sigma-Aldrich | P73803 | Flammable, Toxic, Corrosive |
| Dimethyl Sulfoxide | Fisher | D1281 | |
| SPPS Reaction Vessels | Grace | 211108 | |
| LCMS | Agilent | 1200 Series | |
| Semi-Prep LC | Hewlett Packard | 1100 Series | |
| Lyophilizer | Labconco | 7934027 | |
| Rotovapor | Buchi | R-210 Series | |
| Argon | Airgas | AR PP300CT |
Lab Prep
In 1962, R.B. Merrifield published the first procedure using solid-phase peptide synthesis as a novel route to efficiently synthesize peptides. This technique quickly proved advantageous over its solution-phase predecessor in both time and labor. Improvements concerning the nature of solid support, the protecting groups employed and the coupling methods employed over the last five decades have only increased the usefulness of Merrifield’s original system. Today, use of a Boc-based protection and base/nucleophile cleavable resin strategy or Fmoc-based protection and acidic cleavable resin strategy, pioneered by R.C. Sheppard, are most commonly used for the synthesis of peptides1.
Inspired by Merrifield’s solid supported strategy, we have developed a Boc/tert-butyl solid-phase synthesis strategy for the assembly of functionalized bis-peptides2, which is described herein. The use of solid-phase synthesis compared to solution-phase methodology is not only advantageous in both time and labor as described by Merrifield1, but also allows greater ease in the synthesis of bis-peptide libraries. The synthesis that we demonstrate here incorporates a final cleavage stage that uses a two-step “safety catch” mechanism to release the functionalized bis-peptide from the resin by diketopiperazine formation.
Bis-peptides are rigid, spiro-ladder oligomers of bis-amino acids that are able to position functionality in a predictable and designable way, controlled by the type and stereochemistry of the monomeric units and the connectivity between each monomer. Each bis-amino acid is a stereochemically pure, cyclic scaffold that contains two amino acids (a carboxylic acid with an α-amine)3,4. Our laboratory is currently investigating the potential of functional bis-peptides across a wide variety of fields including catalysis, protein-protein interactions and nanomaterials.
In 1962, R.B. Merrifield published the first procedure using solid-phase peptide synthesis as a novel route to efficiently synthesize peptides. This technique quickly proved advantageous over its solution-phase predecessor in both time and labor. Improvements concerning the nature of solid support, the protecting groups employed and the coupling methods employed over the last five decades have only increased the usefulness of Merrifield’s original system. Today, use of a Boc-based protection and base/nucleophile cleavable resin strategy or Fmoc-based protection and acidic cleavable resin strategy, pioneered by R.C. Sheppard, are most commonly used for the synthesis of peptides1.
Inspired by Merrifield’s solid supported strategy, we have developed a Boc/tert-butyl solid-phase synthesis strategy for the assembly of functionalized bis-peptides2, which is described herein. The use of solid-phase synthesis compared to solution-phase methodology is not only advantageous in both time and labor as described by Merrifield1, but also allows greater ease in the synthesis of bis-peptide libraries. The synthesis that we demonstrate here incorporates a final cleavage stage that uses a two-step “safety catch” mechanism to release the functionalized bis-peptide from the resin by diketopiperazine formation.
Bis-peptides are rigid, spiro-ladder oligomers of bis-amino acids that are able to position functionality in a predictable and designable way, controlled by the type and stereochemistry of the monomeric units and the connectivity between each monomer. Each bis-amino acid is a stereochemically pure, cyclic scaffold that contains two amino acids (a carboxylic acid with an α-amine)3,4. Our laboratory is currently investigating the potential of functional bis-peptides across a wide variety of fields including catalysis, protein-protein interactions and nanomaterials.
Procedure
In 1962, R.B. Merrifield published the first procedure using solid-phase peptide synthesis as a novel route to efficiently synthesize peptides. This technique quickly proved advantageous over its solution-phase predecessor in both time and labor. Improvements concerning the nature of solid support, the protecting groups employed and the coupling methods employed over the last five decades have only increased the usefulness of Merrifield’s original system. Today, use of a Boc-based protection and base/nucleophile cleavable resin strategy or Fmoc-based protection and acidic cleavable resin strategy, pioneered by R.C. Sheppard, are most commonly used for the synthesis of peptides1.
Inspired by Merrifield’s solid supported strategy, we have developed a Boc/tert-butyl solid-phase synthesis strategy for the assembly of functionalized bis-peptides2, which is described herein. The use of solid-phase synthesis compared to solution-phase methodology is not only advantageous in both time and labor as described by Merrifield1, but also allows greater ease in the synthesis of bis-peptide libraries. The synthesis that we demonstrate here incorporates a final cleavage stage that uses a two-step “safety catch” mechanism to release the functionalized bis-peptide from the resin by diketopiperazine formation.
Bis-peptides are rigid, spiro-ladder oligomers of bis-amino acids that are able to position functionality in a predictable and designable way, controlled by the type and stereochemistry of the monomeric units and the connectivity between each monomer. Each bis-amino acid is a stereochemically pure, cyclic scaffold that contains two amino acids (a carboxylic acid with an α-amine)3,4. Our laboratory is currently investigating the potential of functional bis-peptides across a wide variety of fields including catalysis, protein-protein interactions and nanomaterials.
