Construction and Characterization of a Novel Vocal Fold Bioreactor
Instructor Prep
concepts
Student Protocol
1. Bioreactor Assembly (Video 1)
- Make an aluminum mold (circular die + spacer pin) with pre-determined inner and outer dimensions (Figure 1).
- Using the mold from step 1.1, fabricate a silicone membrane (diameter: 42 mm, thickness: 1.5 mm, Figure 1) with an entrenched sleeve (diameter: 12 mm, thickness ~0.25 mm, shaped by the spacer pin in Figure 1) in the middle using a commercially available silicone elastomer kit.
- Make a pair of acrylic blocks (Figure 2 (4, 5)) with a circular opening (diameter: 24 mm) in the middle, engrave the top (1.8 cm thick) and bottom (0.9 cm thick) blocks with matching ridges and grooves.10
- Sandwich the silicone membrane between the paired acrylic blocks. Secure the assembly with four corner screws using a micro torque screwdriver set to a constant force (35 cN·m). As a result, a water-tight, 24 mm wide and 18 mm deep vibration chamber is created (Figure 2C).
- Mount a 3” extended range mini-woofer (Figure 2D, 8 Ω/20 W) underneath the vibration chamber through another set of corner screws on the bottom acrylic block. At this point, an individual vibration module is assembled.
- Replicate seven additional vibration modules. Affix four of them to one of two stationary aluminum bars (40 cm x 10 cm x 2.5 cm) by placing the speaker bases in evenly spaced circular holes (diameter: 7 cm, thickness: 2 cm) cut into the bars. Stabilize each speaker by inserting a screw through the side of the aluminum bar into each circular hole.
- Individually control the speakers by a speaker selector. Connect individual speakers to the selector by attaching wires to the positive and negative inputs on the speaker body then to the corresponding outputs on the selector. The speaker selector allows the signal from the function generator, after passing through a power amplifier, to reach all eight speakers at once (Figure 2E).
- Place the two chamber arrays, the speaker selector and associated electronics in an anti-humidity enclosure. House the entire assembly in a commercial cell culture incubator.
- Feed the main cables (through a medical grade PVC tubing) connecting the power amplifier and speaker selector through the filter assembly at the back of the incubator.
2. Scaffold Fabrication and Characterization
- Dissolve PCL pellets in chloroform at a concentration of 15 wt%. Load the solution into a 10 ml syringe capped with a 21 G blunt-ended needle.
- Lock the syringe onto a programmable syringe pump and set the flow rate at 1 ml/hr.
- Place the aluminum foil-covered collector across from the needle horizontally, with a needle tip-to-collector distance of ~18 cm.
- Clamp the positive alligator clip to the middle of the needle, and the ground alligator clip to the aluminum collector, then set the voltage on the high voltage power supply at 15 kV. CAUTION: high voltage, keep distance from the needle.
- Sequentially turn on the syringe pump and power supply; quickly clean/remove the residual polymer solution surrounding the tip of the needle using a dry paper towel before stable fiber jets and Taylor cone19 are formed.
- Allow the fibers to accumulate on the Al collector to a thickness of ~250-300 µm (~7 hr under the current spinning conditions). Store the resultant scaffolds in a vacuum desiccator for 1-2 days to remove any residual solvent.
- Image the scaffolds, sputter coated with gold, using a Scanning Electron Microscope to show consistent fiber morphology.10
3. Bioreactor Assembly and Characterization
- Punch a cylindrical disk (diameter: 8 mm) with four arms (length: 2 mm) out of the as-spun PCL mat (Figure 2A) by first using a 12 mm diameter biopsy punch to cut the outer diameter of the disk. Then use a second, 8 mm biopsy punch to make four 2 mm long notches evenly spaced around the circular blade to score where the arms are to be cut. After scoring with the 8 mm punch, use a scalpel blade to cut the edges of the arms outward. Insert the scaffold into the groove of the silicone membrane via the extended arms (Video 1). Flatten the inserted scaffold by gently pressing the surface using flathead tweezers.
- Attach a small piece of thin Al foil (8 mm x 2 mm, orthogonal shape, Figure 2B) to the PCL scaffold to aid laser reflection.
- Secure the assembled silicone membrane/PCL scaffold (as detailed in step 1.4) in the vibration chamber. Add 1.5 ml water in the chamber in order to hydrate the PCL scaffold before vibration.
- Using the function generator, introduce vibration signals (e.g., 200 Hz sinusoidal waves with a peak-to-peak voltage, Vpp, of 0.1 V) to the sandwiched acrylic chamber. Use a voltmeter to accurately measure the voltage at each speaker input. Note: the Vpp readout from the function generator will differ from the eventual voltage delivered to the speaker.
- Assemble the single-point LDV and secure the fiber-optic laser sensor head to a pan-tilt head tripod. Angle the sensor head so that it is pointing perpendicular to the tabletop. Connect the LDV sensor head to the data acquisition module via coaxial cable then the module to the laptop via USB.
- Focus the laser beam perpendicularly at various predetermined points on the silicone membrane (Figure 2B and Figure 3).
- Using the data acquisition software, record the mid-membrane displacement. Click “Acquisition Settings” from the “Options” menu; then change the measurement mode to “FFT”. Next, click the “Continuous Measurement” in the main toolbar then click the peak that forms at the chosen frequency (Figure 6D) to record displacement.
- Plot the normal mid-membrane displacement (w0) as a function of the relative position across the substrate. Construct a 3D colormap of the surface vibratory profile using Origin 8.5 data analysis software.
4. Vibratory Cell Culture
- Sub-culture human bone marrow-derived MSCs in T150 tissue culture flasks at an initial seeding density of 4,000-5,000 cells/cm2 in MSC maintenance media.
- After 7-8 days of cell culture (to ~85% confluency), trypsinize the cells with a cell dissociation reagent such as accutase, count using a hemocytometer, centrifuge (440 x g for 5 min), and re-suspend the cell pellet in fresh MSC maintenance media at a concentration of 4.5 x 106 cells/ml.
- Immerse the PCL scaffold in 70% ethanol O/N. After the solvent is evaporated, expose both sides of the scaffold to germicidal UV light (254 nm) for 5-8 min.
- Soak the PCL scaffold in a 20 µg/ml fibronectin solution at 37 °C for 1 hr. Insert the fibronectin-coated scaffold into the silicone membrane. Assemble the bioreactor as detailed in step 1.
- Distribute 40 µl of the cell suspension evenly on the secured PCL scaffold. Allow the cells to attach for 1-1.5 hr before adding an additional 1.5 ml fresh media to the vibration chamber.
- Culture the MSC-laden PCL scaffold statically for 3 days and refresh the media upon completion of the static culture.
- Impose selected vibration regimes to the cellular constructs. Note: As an example, cells are subjected to a 1-hr-on-1-hr-off (OF) vibration at 200 Hz with a w0 of ~40 μm for 12 hr per day for up to 7 days. Constructs subjected to vibratory stimulations are designated as Vib samples and those cultured statically in identical vibration chambers serve as static controls (Stat).
5. Biological Evaluations
- Collect 200 µl cell culture media from each chamber every other day (day 1, 3, 5, and 7) and pool the aliquots from the same sample together (800 µl each).
- Quantify the cellular production of matrix metalloproteinase-1 (MMP1) and HA using an MMP1 ELISA Development kit and a hyaluronan competitive ELISA kit, respectively, following the manufacturers’ procedure. Meanwhile, assay the production of soluble elastin precursor following the previously reported ELISA procedure.8
- Upon completion of the last vibration cycle at day 7, quickly remove the cellular constructs from the vibration chambers using sharp tweezers and briefly rinse them with cold phosphate buffered saline (PBS, 4 °C).
- For the live/dead staining, incubate the constructs with propidium iodide (1:2,000 in PBS) and Syto-13 (1:1,000 in PBS) simultaneously for 5 min at RT. Image the stained constructs with a multiphoton confocal microscope.
- Separately, snap-freeze the PBS-rinsed cellular constructs on dry ice and extract the total cellular RNA following a previously reported protocol for gene analysis.9
- Verify the quantity and quality of the extracted RNA using a UV-Vis spectrophotometer. RNA samples with A260/A280 and A260/A230 ratios of 1.8-2.2 are used for subsequent qPCR analysis.
- Reverse transcribe the RNA (500 ng/sample) into cDNA using a commercially available reverse transcription kit.
- Perform the PCR reaction on a real-time sequence detection system using a commercially available PCR master mix following the previously detailed procedure.8
- Analyze the qPCR results using commercial qPCR data analysis software. To ensure the reliability of the data analysis, multiple reference genes (YWHAZ, TBP, PPIA) are employed as internal controls, and the variance of specific primer efficiencies is taken into account.8
Construction and Characterization of a Novel Vocal Fold Bioreactor
Learning Objectives
The PCL scaffolds fabricated by electrospinning contain micron sized interstitial pores and randomly entangled fibers with an average diameter of 4.7 µm (Figure 4A). At a higher magnification, nanoscale grooves and pores are visible on individual fibers (Figure 4B). Coating of the scaffolds with fibronectin improves hydrophilicity and facilitates the initial cell adhesion/spreading on the PCL scaffold (unpublished observation).
Sinusoidal waveforms with desired frequency (f, 100-300 Hz) and voltage (Vpp: 0-0.125 V) are introduced to the speaker underneath each vibration chamber, and the air confined between the bottom of the silicone membrane and the paper cone of a mini-woofer is driven into oscillation. The air oscillation is delivered to the PCL scaffold with or without cells. LDV is used to analyze the vibration characteristics of the scaffold at a given f and Vpp, taking into account the refractive index of water (1.33).20 Figure 5 shows the normal displacement (w0) at the center of the PCL scaffold as a function of Vpp and f. The vibration frequencies are chosen to reflect fundamental human speaking frequencies.21 There is a linear relationship between w0 and Vpp in the range of 0-0.125 V for all the frequencies tested. At a given Vpp, w0 decreases as f increases from 100 to 300 Hz.
A specific vibration condition (f = 200 Hz, Vpp = 0.1 V) is selected for further analysis. The velocity profile as a function of time (Figure 6A) shows that the sinusoidal signal introduced to the speaker is captured by the PCL scaffold with high fidelity. The center of the PCL scaffold oscillates longitudinally with a peak velocity of 52 mm/sec, a peak acceleration of 66 m/sec2 (~6.7g) and a normal displacement of ~40 µm. The harmonic signals at 100, 300, 400 and 500 Hz are at least an order of magnitude lower than those at the fundamental frequency (200 Hz). However, if the Vpp value is too high (0.15 V), multiple harmonic peaks of comparable intensity to the fundamental frequency are detected (Figure 7). The vibration profile across the scaffold is created by monitoring the normal displacement from a total of 73 representative points on the radial directions of the PCL surface (Figure 3). The 3D colormap (Figure 8) demonstrates that the vibration detected on the surface of the membrane is axisymmetrical relative to the center and its resting positions. The normal displacement is found to decrease monotonically from the center to the edge where the membrane is secured.
MSCs cultured on PCL scaffolds are cultured under selected vibration conditions. Cells subjected to a 7-day OF stimulation maintain similar viability and proliferation properties as the static controls (Figure 9), confirming that the fibrous scaffolds were cytocompatible and the vibration applied resulted in no loss of viability. Cellular responses to vibratory stimulations are examined at the mRNA levels in terms of the expression of essential vocal fold ECM proteins, such as elastin (ELN), hyaluronan synthase-1 (HAS1), Col3A1 and MMP1 (Figure 10). The OF vibration leads to a 2.3 fold increase in ELN expression at day 7, relative to the static controls. The vibratory stimulations also increased Col3A1 expression moderately. It is noteworthy that the expression of major ECM remodeling enzymes, HAS1 and MMP1, is significantly augmented by the vibration signals. Specifically, the dynamic treatment resulted in a fold increase of ~1.7 and ~16.3 for HAS1 and MMP1 expression (both p < 0.05), respectively, over their static controls at day 7. Overall, the inductive effect of the vibrations on HAS1 and MMP1 was enhanced from day 3 to day 7.
To further substantiate the qPCR results, the cellular production of HA, soluble elastin and MMP1 is quantified by ELISA at the translational level (Figure 11). The dynamically cultured cells produce 4.2 ± 0.1 μg/mg (per dry scaffold weight) soluble elastin after 7 days of vibrations, whereas the static controls only accumulate 2.7 ± 0.2 μg/mg) elastin. On average, 7-day OF vibrations result in 2.2- and 4.7-fold increase in HA and MMP1 secretion, relative to the corresponding static controls.
Video 1: A 3D simulation showing the assembly of a J2 bioreactor. The video was created by Autodesk 3ds Max Design (Courtesy of Congfei Xie).

Figure 1. Photograph illustrating the dimension of the Al mold used for fabricating the silicone membrane with entrenched sleeve. Ø: diameter, d: thickness/depth, h: height. Please click here to view a larger version of this figure.

Figure 2. Flowchart showing the bioreactor assembly. (A) A photograph of a four-arm shaped PCL scaffold; (B) A photograph showing the PCL scaffold secured in the vibration chamber and the vibrometer laser focused on the bottom of the chamber; (C) A cross-sectional view of the vibration chamber. 1: PCL scaffold (red); 2: silicone membrane (cyan); 3: Al stationary bar; 4: top acrylic block; 5: bottom acrylic block; 6: mini-woofer; (D) side view of the vibration module; (E) A photograph showing the entire assembly. This figure has been modified from Tong et al.10 Copyright 2013, Mary Ann Liebert, Inc. Please click here to view a larger version of this figure.
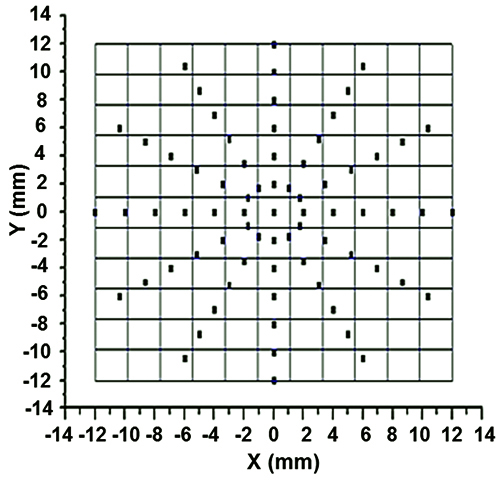
Figure 3. Illustration of radially marked silicone membrane for single point LDV measurements. This figure has been modified from Tong et al.10 Copyright 2013, Mary Ann Liebert, Inc.

Figure 4. SEM images of PCL scaffolds at different magnifications. (A) 600X, (B) 7,000X. This figure has been modified from Tong et al.10 Copyright 2013, Mary Ann Liebert, Inc. Please click here to view a larger version of this figure.
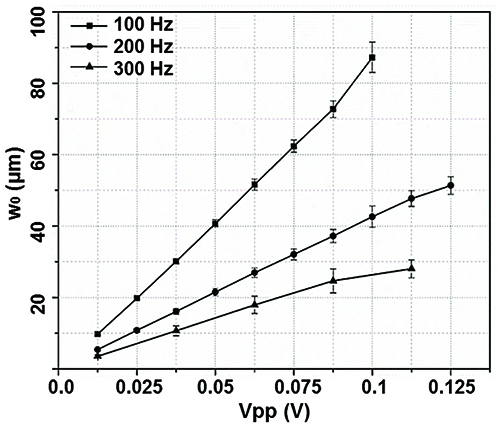
Figure 5. The normal displacement at the center of the silicone membrane (w0) as a function of the applied frequency (100, 200 and 300 Hz) and the driving voltage (Vpp = 0-0.125 V). This figure has been modified from Tong et al.10 Copyright 2013, Mary Ann Liebert, Inc.

Figure 6. Vibration characteristics detected at the center of the silicone membrane with f = 200 Hz and a Vpp = 0.1 V. (A) Velocity profile as a function of time. (B) Velocity profile as a function of frequency. (C) Acceleration as a function of frequency. (D) Normal displacement (w0) as a function of frequency. This figure has been modified from Tong et al.10. Copyright 2013, Mary Ann Liebert, Inc. Please click here to view a larger version of this figure.
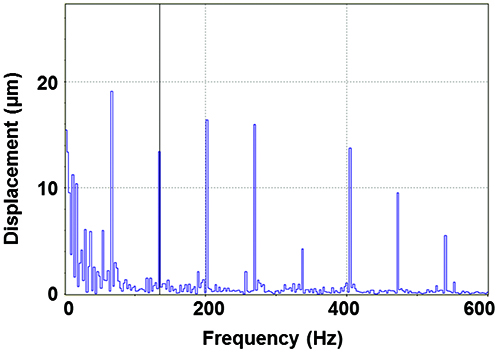
Figure 7. Normal displacement detected at the center of the membrane (w0) as a function of frequency when a 200 Hz sinusoidal wave is introduced to the mini-woofer at a Vpp = 0.15 V.
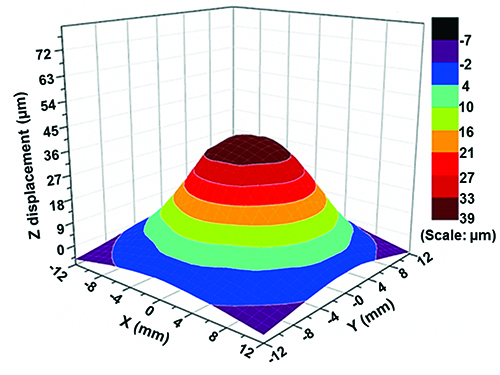
Figure 8. 3D colormap constructed by surface gridding using the normal displacement data collected from all locations marked on the PCL scaffold. This figure has been modified from Tong et al.10 Copyright 2013, Mary Ann Liebert, Inc.

Figure 9. Cell viability, visualized by live/dead staining, after 7 days of vibrations. This figure has been modified from Tong et al.10 Copyright 2013, Mary Ann Liebert, Inc. Please click here to view a larger version of this figure.
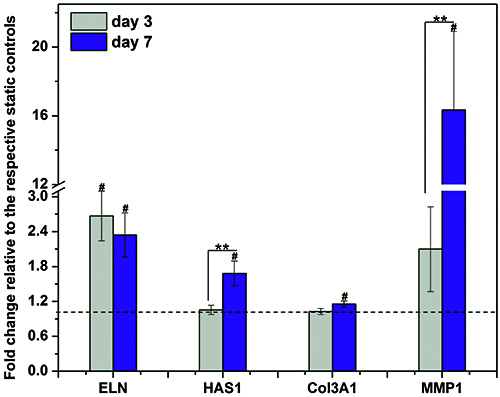
Figure 10. Cellular responses to the vibratory stimulations in terms of the expression of vocal fold relevant, ECM genes. The relative gene expression (fold change) is normalized to the respective static controls at day 3 and day 7 (dashed baseline). **: significant difference (p < 0.05) between day 3 and 7, #: significantly changed (p < 0.05) relative to the baseline. Data represents mean ± standard error of the mean (S.E.M, n=4). Two-tailed student’s t-test is used for statistical analysis, with p < 0.05 being considered as significantly difference (same as below). This figure has been modified from Tong et al.10 Copyright 2013, Mary Ann Liebert, Inc.
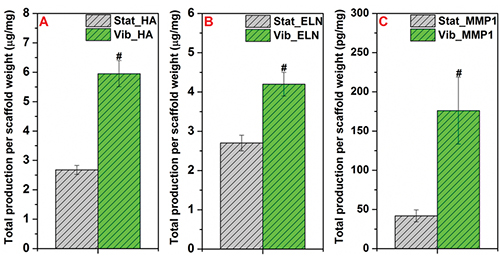
Figure 11. Biochemical quantification of HA (A), soluble ELN (B) and MMP1 (C) produced by MSCs cultured on the PCL scaffold under Stat and Vib conditions for 7 days. Total amount of ECM molecules per dry scaffold weight (mg) is represented as mean ± S.E.M, n = 4 from the representative trial. #: significantly enhanced (p < 0.05) compared to the Stat controls. This figure has been modified from Tong et al.10 Copyright 2013, Mary Ann Liebert, Inc.
List of Materials
| silicone elastomer kit | Dow Corning | Sylgard 184 | cure the membrane at 100 C for 2 hr |
| PCL | Sigma Aldrich | 440744-500G | Mn ~ 80 kDa, dissolve overnight |
| chloroform | Sigma Aldrich | C7559-5VL | |
| human bone marrow-derived MSCs | Lonza | PT-2501 | received with passage 2 |
| MSC maintenance media | Lonza | PT-3001 | 10% FBS in basal media supplemented with L-glutamine, gentamicin and amphotericin |
| Accutase cell dissociation reagent | Life Technologies | A11105-01 | |
| ethanol | Sigma Aldrich | E7023-500ML | |
| fibronectin | Sigma Aldrich | F2006-1MG | |
| MMP1 DuoSet ELISA kit | R&D systems | DY901 | |
| HA ELISA kit | Echelon Biosciences | K-1200 | |
| PBS | Life Technologies | 14190-136 | |
| propidium iodide | Life Technologies | P1304MP | |
| Syto-13 | Life Technologies | S7575 | |
| QuantiTect reverse transcription kit | Qiagen | 205311 | |
| SYBR Green PCR master mix | Life Technologies | 4309155 | |
| replacement speaker | DAYTON audio (via Parts Express) |
DS90-8 | paper cone, full range (80-13000 Hz), 85dB |
| Ergo Micro torque screwdriver | Mountz | # 020377 | torque range: 20-120 cN.m |
| stereo speaker selector | RadioShack | 40-244 | maximum power handling 50 W |
| function generator | Agilent | 33220A | frequency range 1 µHz- 20 MHz |
| power amplifier | PYLE audio | PylePro PT2400 | frequency response: 10 Hz-50 kHz, two speaker channels |
| cell culture incubator | Thermo Fisher | Steri-Cult 3307 | |
| syringe pump | New Era Pump Systems | NE-300 | |
| High voltage power supply | Spellman | CZE 1000R | output voltage: 0-30 kV |
| scanning electron microscope | JEOL-USA | JSM-7400F | |
| desk gold sputter coater | Denton Vacuum | DSK00V-0013 | |
| Doppler laser vibrometer | Polytec | PDV-100 | non-contact velocity measurement (0-22 kHz) |
| PCR sequence detection system | Applied Biosystems | ABI7300 | |
| multiphoton confocal microscope | Zeiss | Zeiss 510Meta NLO | |
| UV-VIS Spectrophotometer | NanoDrop Products via Thermo Scientific |
ND-2000 | |
| VibSoft Data Acquisition Software | Polytec | acquisition bandwidth up to 40 MHz | |
| Origin 8.5 data analysis software | OriginLab | ||
| qbasePlus qPCR data analysis software | Biogazelle | V2.3 | |
| aluminium alloy | McMaster-Carr | Alloy 6061 | |
| acrylic blocks | McMaster-Carr | ||
| polycarbonate anti-humidity chamber | McMaster-Carr | Impact-Resistant Polycarbonate | |
| screws | McMaster-Carr | ||
| electronic cable/wire | |||
| medical grade PVC tubing | US Plastic Corp. | Tygon S-50-HL | clear, biocompatible |
| 10 mL syringe | Becton Dickinson | 309604 | |
| 21 G blunt ended needle | Small Parts | NE-213PL-25 | 1-1/2" length |
| Alligator clip adapters | RadioShack | 270-354 | fully insulated |
| 8 mm biopsy punch | Sklar Surgical Instruments | 96-1152 | sterile, disposable |
| 12 mm biopsy punch | Acuderm (via Fisher Scientific) | NC9998681 | |
| tissue culture flasks | Corning | cell culture treated |
Lab Prep
In vitro engineering of mechanically active tissues requires the presentation of physiologically relevant mechanical conditions to cultured cells. To emulate the dynamic environment of vocal folds, a novel vocal fold bioreactor capable of producing vibratory stimulations at fundamental phonation frequencies is constructed and characterized. The device is composed of a function generator, a power amplifier, a speaker selector and parallel vibration chambers. Individual vibration chambers are created by sandwiching a custom-made silicone membrane between a pair of acrylic blocks. The silicone membrane not only serves as the bottom of the chamber but also provides a mechanism for securing the cell-laden scaffold. Vibration signals, generated by a speaker mounted underneath the bottom acrylic block, are transmitted to the membrane aerodynamically by the oscillating air. Eight identical vibration modules, fixed on two stationary metal bars, are housed in an anti-humidity chamber for long-term operation in a cell culture incubator. The vibration characteristics of the vocal fold bioreactor are analyzed non-destructively using a Laser Doppler Vibrometer (LDV). The utility of the dynamic culture device is demonstrated by culturing cellular constructs in the presence of 200-Hz sinusoidal vibrations with a mid-membrane displacement of 40 µm. Mesenchymal stem cells cultured in the bioreactor respond to the vibratory signals by altering the synthesis and degradation of vocal fold-relevant, extracellular matrix components. The novel bioreactor system presented herein offers an excellent in vitro platform for studying vibration-induced mechanotransduction and for the engineering of functional vocal fold tissues.
In vitro engineering of mechanically active tissues requires the presentation of physiologically relevant mechanical conditions to cultured cells. To emulate the dynamic environment of vocal folds, a novel vocal fold bioreactor capable of producing vibratory stimulations at fundamental phonation frequencies is constructed and characterized. The device is composed of a function generator, a power amplifier, a speaker selector and parallel vibration chambers. Individual vibration chambers are created by sandwiching a custom-made silicone membrane between a pair of acrylic blocks. The silicone membrane not only serves as the bottom of the chamber but also provides a mechanism for securing the cell-laden scaffold. Vibration signals, generated by a speaker mounted underneath the bottom acrylic block, are transmitted to the membrane aerodynamically by the oscillating air. Eight identical vibration modules, fixed on two stationary metal bars, are housed in an anti-humidity chamber for long-term operation in a cell culture incubator. The vibration characteristics of the vocal fold bioreactor are analyzed non-destructively using a Laser Doppler Vibrometer (LDV). The utility of the dynamic culture device is demonstrated by culturing cellular constructs in the presence of 200-Hz sinusoidal vibrations with a mid-membrane displacement of 40 µm. Mesenchymal stem cells cultured in the bioreactor respond to the vibratory signals by altering the synthesis and degradation of vocal fold-relevant, extracellular matrix components. The novel bioreactor system presented herein offers an excellent in vitro platform for studying vibration-induced mechanotransduction and for the engineering of functional vocal fold tissues.
Procedure
In vitro engineering of mechanically active tissues requires the presentation of physiologically relevant mechanical conditions to cultured cells. To emulate the dynamic environment of vocal folds, a novel vocal fold bioreactor capable of producing vibratory stimulations at fundamental phonation frequencies is constructed and characterized. The device is composed of a function generator, a power amplifier, a speaker selector and parallel vibration chambers. Individual vibration chambers are created by sandwiching a custom-made silicone membrane between a pair of acrylic blocks. The silicone membrane not only serves as the bottom of the chamber but also provides a mechanism for securing the cell-laden scaffold. Vibration signals, generated by a speaker mounted underneath the bottom acrylic block, are transmitted to the membrane aerodynamically by the oscillating air. Eight identical vibration modules, fixed on two stationary metal bars, are housed in an anti-humidity chamber for long-term operation in a cell culture incubator. The vibration characteristics of the vocal fold bioreactor are analyzed non-destructively using a Laser Doppler Vibrometer (LDV). The utility of the dynamic culture device is demonstrated by culturing cellular constructs in the presence of 200-Hz sinusoidal vibrations with a mid-membrane displacement of 40 µm. Mesenchymal stem cells cultured in the bioreactor respond to the vibratory signals by altering the synthesis and degradation of vocal fold-relevant, extracellular matrix components. The novel bioreactor system presented herein offers an excellent in vitro platform for studying vibration-induced mechanotransduction and for the engineering of functional vocal fold tissues.
