Experimental Approaches to Study Mitochondrial Localization and Function of a Nuclear Cell Cycle Kinase, Cdk1
Instructor Prep
concepts
Student Protocol
1. Isolation of Mitochondria from Cultured Cells
- Preparation of Isolation Buffer for Cells (IBc) Buffer
- Prepare 0.1 M Tris/MOPS (tris(hydroxymethyl)aminomethane/3-(N-morpholino) propanesulfonic acid): Dissolve 12.1 g of Tris in 800 ml of distilled water, adjust pH to 7.4 using MOPS powder, add distilled water to a total volume of 1 L and store at 4 oC.
- Prepare 0.1 M EGTA (Ethylene glycol bis(2-aminoethyl ether)tetraacetic acid)/Tris: Dissolve 38.1 g of EGTA in 800 ml of distilled water, adjust pH to 7.4 using Tris powder, add distilled water to a total volume of 1 L and store at 4 oC.
- Prepare 1 M Sucrose: Dissolve 34.23 g of sucrose in 100 ml of distilled water, make 20 ml aliquots, and store at -20 oC.
- Prepare IBc buffer: Prepare 100 ml of IBc Buffer by adding 10 ml of 0.1 M Tris/MOPS and 1 ml of 0.1 M EGTA/Tris to 20 ml of 1 M sucrose. Add distilled water to a total volume of 100 ml. Adjust pH to 7.4.
- Preparation of Cell Lysis Buffer
- Prepare the lysis buffer containing 20 mM Tris-HCl (pH 7.5), 150 mM NaCl, 1 mM EDTA (Ethylene diamino tetraacetic acid), 1 mM EGTA, 1% Triton-X-100, 2.5 mM Sodium pyrophosphate, 1 mM β-glycerophosphate, 1 mM sodium orthovanadate. Add proteinase inhibitors 1 µg/ml Leupeptin and 1 mM PMSF (phenylmethylsulfonyl fluoride) right before use.
- Isolation of Mitochondria
- Harvest 3 x 107 cells with ice-cold 1x PBS (Phosphate Buffered Saline), pH 7.4 and centrifuge cells at 600 x g for 10 min at 4 oC. Discard the supernatant and re-suspend the pellet in 5 ml ice-cold IBc buffer.
- Homogenize the cells using a glass/glass tissue grinder for about 10 min. Transfer the homogenate to a 15 ml tube and centrifuge at 600 x g for 10 min at 4 oC. For functional mitochondria, use glass/Teflon coupling as it is less damaging to mitochondria than the glass/glass tissue grinder.
- Collect the supernatant into 1.5 ml tubes and centrifuge at 7,000 x g for 10 min at 4 oC. Transfer the supernatant to a 1.5 ml tube and save as cytosol protein.
- Wash the pellet (mitochondria) twice with 200 µl ice-cold IBc buffer and centrifuge at 7,000 x g for 10 min at 4 oC.
- Discard the supernatant and re-suspend the pellet in the cell lysis buffer and use it immediately or store at -80 oC for future use. If functional mitochondria are needed, re-suspend the pellet in the remaining buffer after discarding the supernatant. Diluting the mitochondrial fraction further may result in the loss of function of mitochondria. Store on ice and use the preparation in 1 – 3 hr for best results.
- Sonicate the resuspended pellet for thirty 3-sec bursts in an ice bath, centrifuge at 10,000 x g for 5 min, and save the supernatant as the mitochondrial fraction.
2. Co-immunostaining of Cdk1, CyclinB1 and COXIV, a Mitochondrial Resident Protein
- Grow 5 x 104 cells on coverslips in 24-well plates O/N or up to 70% confluence. Aspirate the medium and wash the cells twice with 500 µl ice-cold 1 x PBS, pH 7.4.
- Fix cells with 500 µl ice-cold 4% paraformaldehyde at RT for 10 min. Aspirate the fixing solution and wash the cells with 500 µl 1 x PBS 3 times, 5 min each with gentle shaking at RT.
- Permeabilize the cells with 0.2% Triton X-100 in 500 µl of PBS for 5 min at RT. Aspirate the solution and wash the cells 3 times, 5 min each with 500 µl PBS. Add the blocking solution (500 µl, 1% BSA (Bovine Serum Albumin) in PBS containing 1% Tween 20) for 30 min at RT.
- Dilute the primary antibodies 1:250 (v:v) in 500 µl of blocking solution and incubate the cells with desired primary antibodies raised in different species to avoid cross signaling (CyclinB1 (mouse) and COXIV (rabbit) or Cdk1 (mouse) and COXIV (rabbit) at RT for 30 – 60 min or O/N at 4 oC.
- Wash the coverslips with 500 µl of 1 x PBS 3 times, 5 min each and then incubate with secondary antibody diluted 1:1,000 in 500 µl blocking solution at RT for 1 hr followed by washing with 500 µl PBS 3 times, 5 min each.
- Mount the coverslips with 20 µl of anti-fade mounting solution and seal the slides with nail polish. Image the slides using a fluorescent microscope right away or keep in the dark at 4 oC for 1 – 2 weeks.
3. Sodium Carbonate Extraction of Intact Mitochondria
- Grow approximately 20 x 107 cells, harvest, wash once with PBS and isolate mitochondria as described in section 1. Divide mitochondrial isolates into two parts before the last centrifugation to pellet the mitochondria (before step 1.3.4); save half to be used as the total mitochondria (step 3.2), use the other half for sodium carbonate extraction (following steps 3.3-3.6) to separate soluble and membrane bound proteins.
- Lyse the total mitochondria pellet with 30 µl of cell lysis buffer and store the lysate at -80 oC until used in immunoblotting.
- Add 250 µl of 0.1 M Na2CO3 (Sodium carbonate), pH 11.0, to the other half of the mitochondrial pellet and incubate on ice for 30 min.
- Centrifuge at 100,000 x g for 20 min. Collect the supernatant and proceed to step 3.5. Lyse the pellet using 30 µl of cell lysis buffer and sonicate as in step 1.3.6. Store at -80 oC until used in immunoblotting.
- Add an equal volume of 20% freshly made trichloroacetic acid (TCA) to the supernatant to precipitate the proteins and keep on ice for 30 min.
- Centrifuge at 15,000 x g for 10 min. Discard the supernatant, dissolve the pellet in 80 µl of cell lysis buffer and store at -80 oC until used in immunoblotting.
4. Separation of Inner and Outer Membranes of Mitochondria (Isolation of Mitoplasts)
- Grow approximately 20 x 107 cells, harvest, wash once with PBS and isolate mitochondria as described in section 1. Separate the mitochondrial fractions into 10 equal portions before the last centrifugation to pellet the mitochondria (before step 1.3.4).
- Dissolve each pellet in 30 µl of the indicated concentration of hypotonic sucrose buffer (1 mM EDTA, 10 mM MOPS/KOH (Potassium hydroxide), pH 7.2; sucrose concentration ranging from 25 – 200 mM) with or without 50 µg/ml trypsin (see Table 1) and incubate 30 min on ice.
- Add 3 µl of 10 mM PMSF (to reach a final concentration of 1 mM PMSF) to the trypsin containing vials to stop the trypsin digestion and incubate on ice for 10 min.
- Centrifuge at 14,000 x g at 4 oC for 10 min. Transfer supernatant into a new tube, lyse the pellet in 30 µl of cell lysis buffer, sonicate as in step 1.3.6, and store at -80 oC.
5. Construction of Mitochondria-targeted GFP/RFP-tagged CyclinB1/Cdk1 Vectors and Confirmation of Their Mitochondrial Localization
- Clone the mitochondria targeting sequence (MTS; derived from the precursor of human cytochrome c oxidase subunit 8A (COX8) between nucleotides 76 – 161) in frame into the N-terminus of GFP or RFP at NheI and BamHI sites of pEGFP-N1 or pERFP-N1 vectors using standard molecular cloning techniques.
- When designing PCR primers for CyclinB1 and Cdk1 genes, add BamHI restriction enzyme recognition sites (bold) to the 5' of the PCR primers.
Forward Cdk1 BamH1 5' CAG TGG ATC CAA TGG AAG ATT ATA CCA AAA T
Reverse Cdk1 BamH1 5' CTG TGG ATC CTG CAT CTT CTT AAT CTG ATT
Forward CyclinB1 BamH1 5' ATA TGG ATC CAA TGG CGC TCC GAG TCA
Reverse CyclinB1 BamH1 5' ATA TGG ATC CTG CAC CTT TGC CAC AGC C - Amplify the Cdk1 and CyclinB1 genes using these primers following standard techniques. Digest the PCR products with 1 µl of BamHI restriction enzyme at 37 °C for 2 hr. Run the digestion products on a 1% agarose gel. Using a razor blade, cut the correct -sized DNA fragments out and purify the DNA from the gel using a gel extraction kit.
- Digest 1 µg of MTS-pEGFP-N1 and MTS-pERFP-N1 plasmids with 1 µl of BamHI enzyme at 37 °C for 2 hr. Add 1 µl of Calf-Intestinal Alkaline Phosphatase (CIP) for 30 min at 37 °C. Run the digestion products on a 1% agarose gel and purify linear plasmids from gel as in Step 5.3.
- Set up a ligation reaction by adding 1 µl of plasmid from Step 5.4 and 5 µl of Cdk1 or CyclinB1 from Step 5.3 to 3 µl of ligase buffer, 0.5 µl of T4 ligase, and 20.5 µl of dH2O. Incubate O/N at 4 °C.
- Transform Escherichia coli DH5α competent cells with 10 µL of ligation mixture following standard techniques and grow the bacteria on 10 mg/ml kanamycin-containing LB agar plates O/N at 37 °C to obtain colonies.
- Pick out a colony from O/N grown plates using a sterile pipette tip, insert the tip into a 5 ml of kanamycin-LB-containing tube and incubate O/N. Isolate the plasmid using miniprep kits according to the manufacturer's protocol, and sequence the plasmid using standard techniques.
- Transfect exponentially growing MCF10A cells (1.5 x 104 cells seeded on 96-well plates) with MTS-Cdk1-RFP or MTS-CyclinB1-GFP plasmids from Step 5.7 by preparing a plasmid/transfection reagent ratio of 1:2 (w:v, 100 ng: 0.2 µl) in 100 µl serum- and antibiotic-free medium.
- Incubate the cells in plasmid/reagent mix for 48 hr at 37 °C in a CO2 incubator with humidity control (5% CO2, 95% relative humidity).
- Prepare 1 mM stock solution of mitochondrial red and green fluorescent dyes by dissolving 50 µg of powder in 100 µl of DMSO. Dilute the stock solutions 1:400 in 1x PBS to obtain 2.5 µM working solutions. The stock solutions can be stored at -20 °C for a year.
- Mix 2 µl of 2.5 µM of red dye with 98 µl of culture medium to prepare a 50 nM final concentration.
- Replace the cell culture medium of CyclinB1-GFP bearing MCF10A cells (generated in step 5.8) with red dye containing medium for 2 min. Repeat the same procedure with green dye for Cdk1-RFP bearing MCF10A cells.
- Wash twice with 100 µl 1 x PBS to get rid of the excess staining solution, add 100 µl 1x PBS to the 96-well plate.
- Visualize the mitochondria and CyclinB1-GFP (excitation by 488 nm and emission at 494 – 536 nm) and Cdk1-RFP (excitation by 560 nm and emission at 565 – 620 nm) on 96-well plates under fluorescent microscope at 40X magnification.
6. Identification of Differentially Phosphorylated Proteins via 2D-DIGE
- Sample Preparation
- Isolate mitochondria and lyse mitochondrial fractions as in Step 1.3. Add an equal volume of 20% TCA (trichloroacetic acid in water) to each sample and vortex for 15 sec. Incubate samples on ice for 30 min as the proteins precipitate out of solution.
- Centrifuge samples at 9,300 x g at 4 °C for 5 min, remove supernatant, and add 60 µl cold acetone (100%) followed by centrifugation at 9,300 x g at 4°C for 5 min.
- Repeat the acetone wash step (Step 6.1.2) one more time, remove supernatant and re-suspend the samples in 50 µl DIGE labeling buffer (7 M urea, 2 M thiourea, 4% CHAPS, 30 mM Tris, pH 8.5).
- Prepare the Fluorescent Dyes
- Prepare stock solution: Dissolve dyes (5 nmol) in 5 µl of fresh dimethyl formamide (DMF) to a final concentration of 1 nmol/µl. Store the stock dye solution at -20 oC until use for a few months.
- Prepare working solution: Allow dyes to set at RT for 5 min. Dilute 1 µl of dye with 4 µl of DMF to a final concentration of 200 pmol/µl. Keep on ice during use. Note: The working solution can be kept at -20 oC for 3 weeks.
- Label Proteins
- Mix 1 µl of working dye solution per 12.5 µg protein. Scale up as needed. For pooled standards, add 6 µl of Cy2 working solution to 300 µg pooled protein.
- Vortex for 30 sec and centrifuge at 4 °C for 30 sec at 12,000 x g. Incubate on ice in the dark for 30 min. Stop the labeling by adding 1 µl of 10 mM lysine per 1 µl of working solution used. Mix well and incubate on ice in the dark for 10 min.
- Pool individually labeled samples and mix with rehydration buffer (7 M urea, 2 M thiourea, 4% CHAPS, 1.2% destreak, 1% pharmalytes, bromophenol blue). Total volume will be 125 µl.
- Vortex samples for 30 sec and allow samples to set at RT for 30 min. Load 125 µl of samples onto 7 cm pH 4 – 9, isoelectric focusing (IEF) strips.
- First Dimension Electrophoresis
- Place the strip holders (coffins) on the electrode plate, making sure the strip holders are completely clean and dry. Pipette 125 µl samples into a coffin, spreading evenly across the entire coffin.
- Using tweezers, pick up the IEF strip, carefully remove the plastic protective cover, and place it (gel side down) into coffin/rehydration buffer. Avoid trapping air bubbles. Slowly fill coffin (1 – 1.5 ml) with mineral oil and place the coffin covers onto the coffins.
- Begin isoelectric focusing following the protocol in Table 2:
Note: Once the run is complete, the strips may be stored in plastic tubes at -80 oC or directly moved on to equilibration and the second dimension.
- Second Dimension – Sodium Dodecyl Sulfate-Polyacrylamide Gel Electrophoresis (SDS-PAGE)
- Equilibration
- Thaw SDS equilibration buffer (8 ml per strip, 6 M urea, 30% glycerol, 2% SDS). Weigh out dithiothreitol (DTT; 10 mg DTT per 1 ml of SDS equilibration buffer). Dissolve DTT into 4 ml SDS equilibration buffer.
- Weigh out IAA (iodoacetamide, 25 mg IAA per 1 ml of SDS equilibration buffer). Dissolve IAA into 4 ml SDS equilibration buffer.
- Melt 1 ml sealing agarose solution in a water bath for later use.
- If the strips were frozen, allow them to thaw completely. Place strips gel side up into the reswelling tray. Add 4 ml of SDS equilibration buffer with DTT to each strip and incubate for 15 min with gentle shaking.
- Pour off all the SDS equilibration buffer with DTT. Add 4 ml of SDS equilibration buffer with IAA to each strip and incubate for another 15 min with gentle shaking. After equilibration with the IAA buffer, pour of the SDS equilibration buffer.
- SDS-PAGE
- Unwrap mini gels, remove the comb and rinse the gel and the wells with ddH2O. Remove the white tape at the bottom of the gel before running. Orient the strip on the gel correctly, gel orientation is critical for spot cutting.
- Place the gel flat on counter with the small plate up and gel top facing experimenter. Lay the strip onto the tall plate with anodic end (++ end with the barcode) facing the left side of the gel. Using a ruler, slowly push the strip down onto gel surface. Avoid trapping air bubbles between the strip and the gel. Stand the gel up in a glass plate stand.
- Add 1 ml of hot sealing agarose solution to the opening of the cassette. Make sure all air bubbles are pressed out using a flat ruler. Assemble the apparatus and run the gel at a constant 125 V for 90 min.
- When the gel finishes running, place the gel on a biomolecular imager with 2 ml ddH2O and scan the gel in fluorescence acquisition mode with 635 nm excitation laser and 665 nm emission filter.
- Phosphoprotein Staining
- Fix the gel with 50% methanol and 10% acetic acid mixture (10 ml) for 30 min twice. Wash with 10 ml of water for 10 min three times and stain with 10 ml phosphoprotein stain for 60 – 90 min, followed by destaining with 10 ml destain solution for 30 min three times.
- Wash with 10 ml water 5 min twice and image the gel with a laser gel scanner at 532 nm/560 nm Excitation/Emission.
- Protein Digestion and Identification
- Excise all of the differentially expressed protein spots from the gel into microplate wells using a robotic spot-picker according to manufacturer's specifications, and add 100 mM ammonium bicarbonate (2 ml) for 1 hr at RT to destain the gel pieces. Dehydrate with 2 ml 100% acetonitrile wash twice and dry in a vacuum concentrator for 30 min.
- Rehydrate the gel pieces with 100 µl of 13 ng/µl modified porcine trypsin in 50 mM ammonium bicarbonate for 16 hr at 37 °C. Collect the supernatants and further extract the proteins with 100 µl of 5% trifluoroacetic acid in 50% acetonitrile for 30 min.
- Concentrate the peptides down to 5 µl by vacuum centrifuge concentrator and analyze with Matrix Assisted Laser Desorption Ionization – Time of Flight – tandem Mass Spectrometry analysis (MALDI TOF-MS/MS)17.
- Equilibration
7. In Vitro Kinase Assay
- Preparation of Substrates
- Sub-clone Complex I (CI) subunits (NDUFV1, NDUFV3, NDUFS2, NDUFB6, and NDUFA12), which are identified as differentially phosphorylated Cdk1 targets, into pGEX-5X-1 vector to generate Glutathione-S-transferase (GST)-tagged bacterial expression plasmids11. Purify CI subunits using the high affinity glutathione columns following the below protocol.
- Culture GST-tagged CI subunit containing Escherichia coli strain BL-21 in Luria-Bertani (LB) broth with 50 µg/ml of ampicillin in a shaker at 37 oC until an optical density (OD) of 0.6 was reached. Add isopropyl-b-D-thio-galactopyranoside (IPTG) to the culture at a final concentration of 0.1 mM, and incubate for an additional 3 hr.
- Centrifuge at 600 x g for 10 min to collect the bacteria and re-suspend in 5 ml of 1 x PBS buffer containing 5 mM of DTT, 1 x proteinase inhibitor cocktail, and 0.1% lysozyme. Lyse the cells by sonication for twenty 5 s bursts, and remove the cell debris by centrifugation at 600 x g for 10 min.
- Collect the supernatant and incubate with glutathione-agarose 4B beads at a volume ratio of 4:1 (supernatant: bead) for 1 hr at RT.
- Elute the GST-fusion proteins from the beads with 3 ml of glutathione elution buffer (10 mM reduced glutathione in 50 mM Tris-HCl, pH 8.0) and subject the eluted proteins to dialysis in molecular porous membrane tubing (molecular weight cut-off 12-14 kiloDaltons) with 1 x PBS /1 mM EDTA. Store the purified proteins at – 80 oC.
- Sub-clone Complex I (CI) subunits (NDUFV1, NDUFV3, NDUFS2, NDUFB6, and NDUFA12), which are identified as differentially phosphorylated Cdk1 targets, into pGEX-5X-1 vector to generate Glutathione-S-transferase (GST)-tagged bacterial expression plasmids11. Purify CI subunits using the high affinity glutathione columns following the below protocol.
- Cdk1 Kinase Assay
- Prior to starting the kinase assay, set heating blocks to 30 °C and 100 °C. Thaw commercial CyclinB1/Cdk1 enzyme complex and substrates on ice.
- Prepare the reaction mix on ice. Since 32P ATP isotope is involved, prepare all reaction mixtures in 1.5 ml sample tubes with screw caps containing an O ring to prevent spread of radioactivity.
Caution: Follow the radioactive material protection rules. Use an 8 mm thick Plexiglas shielding and work at least at an arm's length. Wipe down any contaminated area with water. - Prepare a 30 µl reaction buffer containing 1 x Cdk1 buffer (50 mM Tris-HCl, 10 mM MgCl2, 2 mM DTT, 1 mM EGTA, 0.01% Brij 35, pH 7.5), 0.6 mM cold ATP, 0.1 µCi 32P ATP, 2 units of CyclinB1/Cdk1 and 6 µg of substrate protein. Adjust the volume to 30 µl with ddH2O. For positive control reaction, replace the substrate with 0.01 mM Histone H1, a well-known CyclinB1/Cdk1 substrate.
- For negative control reactions, (1) replace the substrate with GST protein only and (2) replace the substrate with 0.01 mM Histone H1 + 0.5 µM RO-3306, a selective Cdk1 inhibitor.
- Mix well with pipette and centrifuge to bring all the liquid to bottom of tube, minimizing risk of contamination. Incubate at 30 °C for 60 min.
- Add 6 µl of 5x SDS sample buffer to stop the reaction and denature at 100 oC for 10 min. Run samples on 10% SDS-PAGE gel at 160 V for 2 hr, or until dye front has reached the end of the gel. Transfer gel onto a chromatography paper and wrap with plastic wrap.
Caution: All liquid in contact with radioisotopes should be treated as radioactive waste and disposed of in a 5-gallon poly carboy provided by the environmental health and safety services as per institutional guidelines. - Expose the gel to x-ray film for 1 day or up to 1 week at -20 oC depending on the specific activity of radioisotope used and the enzyme.
8. Site-directed Mutagenesis to Generate Dominant Negative Cdk1 (D146N)
- Design mutagenesis primers; two primers, complementary to each other, containing the mutant site (G will be replaced by A nucleotide to code for N instead of D amino acid) flanked by 20 bases on each side11.
- Prepare a 50 µl Polymerase Chain Reaction (PCR) mixture containing 1x reaction buffer, 2.5 mM dNTPs, 10 µM of primers (both forward and reverse), 40 ng of the template, and ddH2O. Add 2.5 U of DNA polymerase into the reaction.
- Run the following PCR program: 95 °C 3 min, 95 °C 30 sec, 55 °C 30 min, 68 °C 6 min; 16 cycles (Note: 68 °C incubation time is determined by the size of the DNA. 1 min/kb is required for DNA ≤ 10 kb. For larger DNA, 2 min/kb plus 10 sec per cycle). Follow by 68 °C 7 min and hold at 4 °C until ready to use.
- Add 2 µl of Dpn I restriction enzyme (10 U/µl) and 5.7 µl Dpn I buffer, mix well with gentle tap with finger and incubate the reaction at 37 oC for 1 hr.
- Transfer 10 µl Dpn I-treated PCR products into 50 µl of DH5α competent cells for transformation. Incubate on ice for 30 min, followed by 45 sec heat-shock at 42 oC and 5 min on ice. Add 500 µl of LB and shake at 37 oC for 1 hr before plating onto LB-agar plates. Incubate O/N at 37 oC.
- Pick a colony from the O/N grown agar plates using a sterile yellow pipette tip, drop the tip into a tube containing 5 ml of LB, and grow O/N in a 37 oC shaker. Isolate plasmid using a miniprep kit and sequence the plasmid to confirm the mutation according to manufacturer's protocol.
9. Determination of Cell Cycle Phase Lengths with EdU Incorporation Assay
- Cell Sorting
- Transfect 2 x 105 cells with indicated vectors (MTS-GFP/RFP, MTS-CyclinB1-GFP/Cdk1-wt-RFP or MTS-Cdk1-dn-RFP) in a 6-well plate using 1:2 (w:v, 2.5 µg: 5 µl) ratio of DNA:Transfection Reagent mixture prepared in 2.5 ml serum- and antibiotic free culture media for 48 hr. Live sort cells stably expressing the GFP-tagged Cdk1 and RFP-tagged CyclinB1 proteins via flow cytometer.
- Wash cells with 1 x PBS, add 100 µl of trypsin into the dish and incubate at 37 oC for 5 min. Add 2 ml of culture media to collect the cells and pass them through a 70 µm filter to generate a single cell suspension. Wash the single cell suspension with 500 µl of 1% Fetal Calf Serum in PBS (FCS/PBS) before loading them onto flow cytometer.
- Set up the sorting parameters following established protocols18 gating for double positive cells stained with both GFP and RFP. Collect the sorted cells coming out of the cytometer into a tube containing cell culture medium and use these cells for analysis of cell cycle progression with EdU pulse-chase labeling as in protocol 9.2.
- Transfect 2 x 105 cells with indicated vectors (MTS-GFP/RFP, MTS-CyclinB1-GFP/Cdk1-wt-RFP or MTS-Cdk1-dn-RFP) in a 6-well plate using 1:2 (w:v, 2.5 µg: 5 µl) ratio of DNA:Transfection Reagent mixture prepared in 2.5 ml serum- and antibiotic free culture media for 48 hr. Live sort cells stably expressing the GFP-tagged Cdk1 and RFP-tagged CyclinB1 proteins via flow cytometer.
- Measurement of Cell Cycle Length Using the EdU Labeling Flow Cytometry Assay
- Seed sorted cells in 6-well plates at a density of 2.5 x 105 cells per well and culture O/N at 37 oC in a CO2 incubator. Add EdU to the culture medium at a final concentration of 25 µM. Incubate at 37 oC in a CO2 incubator for 1 hr.
- Wash cells with 1% BSA in 500 µl PBS and collect them in a 1.5 ml tube at 2 hr intervals for a total of 10 – 12 hr.
- Centrifuge cells at 350 x g for 5 min, discard the supernatant. Dislodge the pellet by adding 100 µl of fixing solution (provided by the manufacturer within the EdU labeling kit) for 15 min at RT and mix well.
- Wash cells with 1 ml of 1% BSA in PBS three times. Fix the cells again with 0.5 ml of 70% ethanol O/N at 4 oC.
Note: Ethanol fixing is critical to quench the internal GFP/RFP fluorescence due to the recombinant-tagged protein expression. - Centrifuge the cells again at 350 x g for 5 min and wash the pellet with 1 ml of 1% BSA in PBS once. Re-suspend the cells in 1 ml of permeabilization buffer (0.1% Triton-X-100, 1% BSA, 0.2 mg/ml RNase A in PBS) for 20 min at RT.
- Wash cells with 1 ml of 1% BSA in PBS once. Add 0.5 mL of reaction cocktail into each tube and mix well. Incubate reaction mixture at RT for 30 min in the dark.
- Wash with 1 ml of 1% BSA in PBS once and stain DNA with 50 µg/ml propidium Iodide (PI) in 1 ml of 1% BSA in PBS.
- Analyze the cells by flow cytometry to follow the EdU-positive population. Present scatter dot plot of EdU-labeled cells stained for DNA content (PI staining, X-axis) and EdU (Alexa 647 staining, Y-axis).
- Use APC channel for Alexa647-EdU utilizing a 670/30 band pass filter with all light present less than 685 nm hitting that filter; and phycoerythrin (PE) channel for PI with a 581/15 band pass filter in front of it with all light present less than 600 nm hitting that filter.
- Insert the first tube containing cells into the flow cytometry and use a standard gating strategy for acquisition: Plot FSC-Area X SSC-Area for morphology followed by PI (PE channel) X Alexa647-EdU (APC channel) for cell staining. Record data for all tubes one by one acquiring 10,000 events per sample.
- Determine the cell cycle phase distribution of the cells and phase lengths using a flow cytometry data analysis software following established protocols11,30.
Experimental Approaches to Study Mitochondrial Localization and Function of a Nuclear Cell Cycle Kinase, Cdk1
Learning Objectives
Sub-mitochondrial localization of CyclinB1 and Cdk1
Sodium carbonate extraction is used to determine whether a protein is located inside the mitochondria or on the outside surface, namely outer membrane. Once a protein is shown to localize inside the mitochondria, further determination of sub-mitochondrial localization can be made via mitoplasting combined with protease digestion. To specify the sub-mitochondrial localization of CyclinB1 or Cdk1, mitoplasts were isolated by diluting mitochondria in hypotonic buffers with decreasing concentrations of the osmotic sucrose from 200 mM to 25 mM. The outer membrane begins to rupture at 150 mM of sucrose, while the inner membrane remains intact until the final concentration at 25 mM of sucrose (Figure 1A). In combination with mitoplasting, protease protection assay can be performed using trypsin to digest exposed proteins following outer membrane rupture. This will result in digestion of intermembrane space proteins. If the protein of interest is protected from trypsin digestion, this indicates mitochondrial matrix localization of the protein. In this representative figure, mitochondrial matrix protein Hsp60, and intermembrane space protein Timm13 were used as sub-mitochondrial localization markers. Similar with Hsp60 but unlike Timm13, CyclinB1 and Cdk1 were protected from trypsin digestion, indicating their mitochondrial matrix localization (Figure 1B).
Mitochondrial Expression of MTS- and GFP-tagged CyclinB1 and Cdk1 Proteins
MTS is cloned in frame at the N-terminus of CyclinB1 or Cdk1 genes, which has GFP or RFP tags at their C-terminus. The resultant recombinant protein is mitochondria-targeted GFP- or RFP-tagged CyclinB1 or Cdk1. The list of the constructs generated and used in this study is shown in the figure. Using these constructs, overexpression of CyclinB1 and/or Cdk1 in the mitochondria was achieved, shown here by western blotting of the isolated mitochondrial fractions (Figure 2).
Potential Mitochondrial Targets of CyclinB1/Cdk1 Determined by 2D-DIGE
Cdk1 belongs to the serine/threonine (S/T) kinase family catalyzing the transfer of a phosphate from ATP to proline (P)-oriented S or T residues. A point mutation that replaces an aspartate (D) residue with asparagine (N) at position 146 of Cdk1 (D146N) generates a dominant negative (dn) Cdk1 mutant19. To study the function of mitochondrial Cdk1, a mitochondria-targeted Cdk1-dn protein was generated by constructing a plasmid (pERFP-N1-MTS-Cdk1-dn) containing a 29 amino acid-long mitochondrial targeting sequence (MTS) derived from the subunit VIII of the human cytochrome C oxidase linked to RFP-tagged dn-Cdk1. pERFP-N1-MTS producing mitochondria-targeted ERFP protein was used as an empty vector control. Mitochondrial phosphoproteins in G2/M cells transfected with both constructs were profiled by 2D gel analysis with pH 4-10 gel strips. Compared with empty vector transfectants (Figure 3, upper panel), a group of mitochondrial phosphoproteins was apparently absent or decreased in the Cdk1-dn transfectants (Figure 3, lower panel). Mass spectrometry analysis of the spots detected determined the identity of the proteins phosphorylated by Cdk1 in the mitochondria.
Cell Cycle Progression and Determination of Phase Lengths with EdU Pulse-chase Assay
To investigate the progression of cell cycle when mitochondrial CyclinB1/Cdk1 levels are increased, a pulse-chase labeling experiment using a thymidine analogue, ethynyl deoxyuridine (EdU) was performed to label the population of cells undergoing DNA synthesis20. This method allows the visualization of cell cycle captured over a 22 hr window by tracking the EdU-positive population when cells progress through S and G2/M phases and accumulate in G1 phase. The results show that labeled S phase cells progressed through G2/M phase and appeared in G1 phase as fast as 4 h in cells expressing wild type mitochondrial CyclinB1/Cdk1, as compared to 6 h in cells transfected with a vector control or mutant CyclinB1/Cdk1 (Figure 4A), indicating that enhancement of mitochondrial CyclinB1/Cdk1 accelerates cell cycle progression.
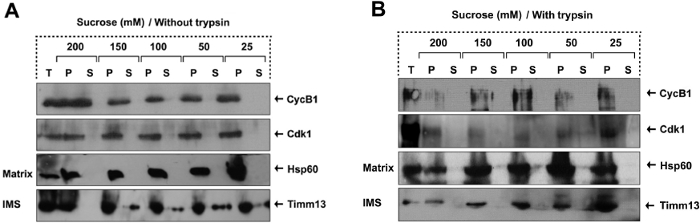
Figure 1. Mitochondrial CyclinB1/Cdk1 Localizes in the Matrix. (A-B) Sub-mitochondrial localization of CyclinB1 and Cdk1 detected by mitoplasting and protease protection assay, figure has been modified from Wang et al., 201411. The total (T), pellet (P), and supernatant (S) fractions were subjected to western blotting analysis with indicated antibodies. TIMM13 (an inter-space protein), and HSP60 (a matrix protein). Please click here to view a larger version of this figure.
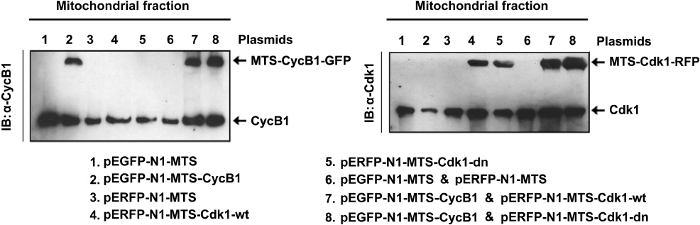
Figure 2. Expression of Mitochondrial Cdk1 Constructs. Western blotting of mitochondrial fractions isolated from cells transfected with mitochondria-targeted CyclinB1 and/or wild type or dominant negative mutant Cdk1 (plasmids are indicated on the bottom11. pEGFP-N1-MTS and the pERFP-N1-MTS vectors were empty vector controls for MTS-CyclinB1 and MTS-Cdk1 respectively). Please click here to view a larger version of this figure.
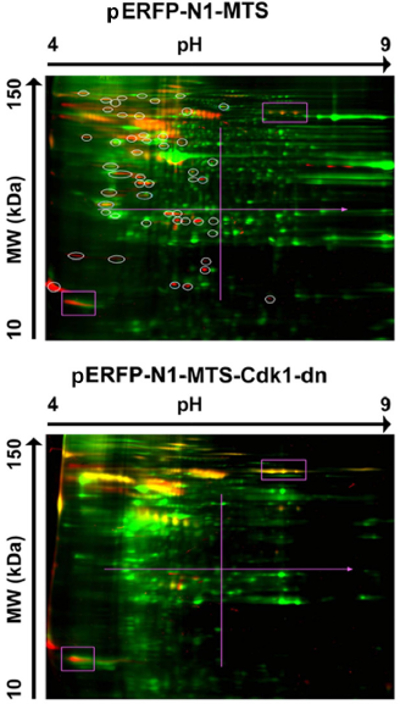
Figure 3. Potential Mitochondrial Substrates of Cdk1. Mitochondrial proteins extracted from G2/M-peaked cells transfected with mitochondria-targeted empty vector (pERFP-N1-MTS, upper panel) or mutant Cdk1 (pERFP-N1-MTS-Cdk1-dn, lower panel) were labeled with Cy5 (green), separated by 2-D gel and phosphorylated proteins were stained with phosphoprotein dye (red). This figure has been modified from Wang et al. 201411. Please click here to view a larger version of this figure.
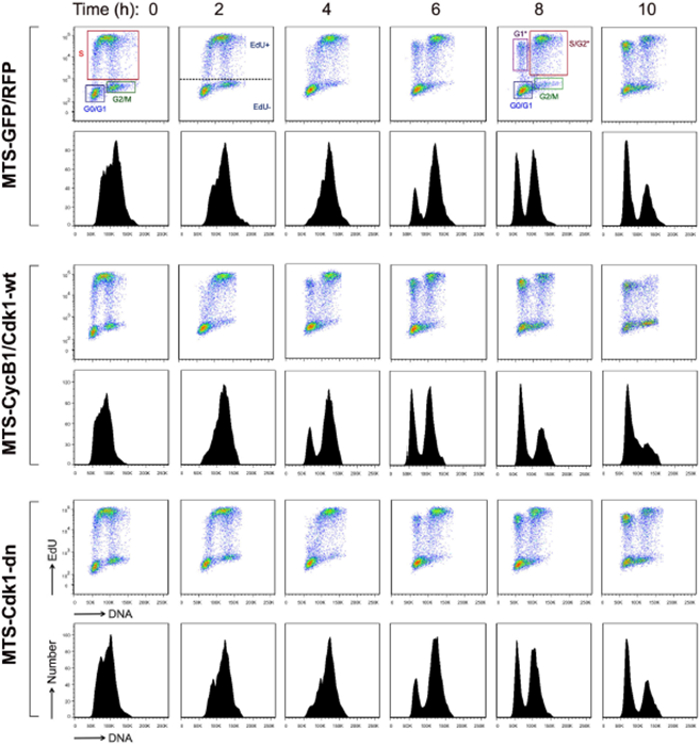
Figure 4. Mitochondrial Cdk1 Enhances G2/M Transition and Overall Cycle Progression.
Cell cycle analysis with EdU pulse-chase labeling. Scatter plot histograms of EdU-labeled cells were drawn for DNA content (X-axis) and EdU (Y-axis). The lower figures in each panel show the mean fluorescence intensity of the EdU labeled nuclei. The time points were indicated in h after the EdU pulse11. For all time points, gates displaying the following populations were drawn: G0/G1, S, and G2/M. For 6, 8, and 10-hr time points, EdU- labeled G1*, S/G2*, and G2/M* populations are shown. This figure has been modified from Wang et al., 201411. Please click here to view a larger version of this figure.
| Sucrose Concentrations Used | |||||
| No trypsin | 25 mM | 50 mM | 100 mM | 150 mM | 200 mM |
| + trypsin | 25 mM | 50 mM | 100 mM | 150 mM | 200 mM |
Table 1. Hypotonic Sucrose Buffers Used for Step 4.2
| Step 1 | 30 V | 12 hr | Step and Hold |
| Step 2 | 300 V | 0.5 hr | Step and Hold |
| Step 3 | 1,000 V | 0.5 hr | Gradient |
| Step 4 | 5,000 V | 1.33 hr | Gradient |
| Step 5 | 5,000 V | 20,000 V hr | Step and Hold |
Table 2. Isoelectric Protocol Used for Step 6.4.5
List of Materials
| 32P ATP | PerkinElmer | BLU002001MC | |
| Anti-mouse secondary antibody | Invitrogen | A-11003 | Alexa-546 conjugated |
| Anti-rabbit secondary antibody | Invitrogen | A11029 | Alexa-488 conjugated |
| ATP | Research Organics | 1166A | For in vitro kinase assay |
| Cdk1 antibody | Cell Signaling Technology | 9112 | |
| Cdk1 kinase buffer | New England Biolabs | P6020S | |
| Click-iT EdU Alexa Fluor 488 Imaging Kit | Life Technologies | C10337 | For cell cycle analysis with EdU labeling |
| COX IV antibody | Cell Signaling Technology | 4844S | For mitochondrial immunostaining |
| Cyclin B1 antibody | Santa Cruz Biotech | sc-752 | |
| CyclinB1/Cdk1 enzyme complex | New England Biolabs | P6020S | Avoid freeze/thaw |
| CyDye DIGE Fluor Labeling Kit | GE Healthcare Life Sciences | 25-8009-83 | |
| DIGE Gel and DIGE Buffer Kit | GE Healthcare Life Sciences | 28-9480-26 AA | |
| Dimethylformamide | Sigma Aldrich | 319937 | DMF |
| Dithiothreitol | Bio-Rad | 161-0611 | DTT |
| dNTP | EMD Millipore | 71004 | For site-directed mutagenesis |
| Dpn I enzyme | Stratagene | 200519-53 | For site-directed mutagenesis |
| Dry Strip cover fluid | GE Healthcare Life Sciences | 17-1335-01 | Used as mineral oil |
| EDTA | J.T. Baker | 4040-03 | |
| EGTA | Acros Organics | 409910250 | |
| Eppendorf Vacufuge Concentrator | Fisher Scientific | 07-748-13 | Used as vacuum centrifuge concentrator |
| Fluoromount G | Southern Biotech | 0100-01 | Anti-fade mounting solution |
| Fortessa Flow Cytometer | BD Biosciences | 649908 | For cell cycle analysis with EdU labeling |
| Histone H1 | Calbiochem | 382150 | For in vitro kinase assay |
| QIAquick Gel Extraction Kit | Qiagen | 28704 | For purifying DNA fragments from agarose gels |
| Immobiline DryStrip Gels | GE Healthcare Life Sciences | 18-1016-61 | IEF (isoelectric focusing) strips |
| Immobilized Glutathione | Thermo Scientific | 15160 | Glutathione-agarose beads |
| Iodoacetamide | Sigma Aldrich | I1149 | IAA |
| IPGphor 3 Isoelectric Focusing Unit | GE Healthcare Life Sciences | 11-0033-64 | IPGphor strip holders |
| Isopropyl-b-D-thio-galactopyranoside | RPI Corp | 156000-5.0 | IPTG |
| Leupeptin | Sigma Aldrich | L9783 | For cell lysis buffer |
| Lipofectamine 2000 | Life Technologies | 11668027 | Transfection reagent |
| Lysine | Sigma Aldrich | L5501 | For CyDye labeling |
| Lysozyme | EMD Chemicals | 5960 | |
| Mitoctracker Red/Green | Invitrogen | M7512/M7514 | Mitochondrial fluorescent dyes |
| MOPS | EMD Chemicals | 6310 | |
| pEGFP-N1 | Clonetech | 6085-1 | GFP-expressing vector |
| Pfu | Stratagene | 600-255-52 | |
| pGEX-5X-1 | GE Healthcare Life Sciences | 28-9545-53 | GST-expressing vector |
| Phenylmethylsulfonyl fluoride | Shelton Scientific | IB01090 | PMSF |
| Phosphate buffered saline | Life Technologies | 14040 | PBS |
| Spectra/Por 4 dialysis tubing | Spectrum Labs | 132700 | as porous membrane tubing for dialysis |
| Pro-Q Diamond Phosphoprotein Gel Stain | Life Technologies | P-33300 | For staining phosphoproteins on 2D gels |
| Proteinase inhibitor cocktail | Calbiochem | 539134 | For cell lysis buffer |
| QuikChange site-directed mutagenesis kit | Stratagene | 200519-5 | |
| QIAprep Spin Miniprep Kit | Qiagen | 27104 | MiniPrep Plasmid Isolation Kit |
| RO-3306 | Alexis Biochemicals | 270-463-M001 | Cdk1 inhibitor |
| Rotenone | MP Biomedicals | 150154 | Complex I inhibitor |
| Sodium carbonate | Fisher Scientific | S93359 | |
| Sodium chloride | EMD Chemicals | SX0420-5 | For cell lysis buffer |
| Sodium orthovanadate | MP Biomedicals | 159664 | For cell lysis buffer |
| Sodium pyrophosphate decahydrate | Alfa Aesar | 33385 | For cell lysis buffer |
| Sodium β-glycerophosphate | Alfa Aesar | L03425 | For cell lysis buffer |
| SpectraMax M2e | Molecular Devices | M2E | Microplate reader |
| Sucrose | Fisher Scientific | 57-50-1 | |
| Tissue Grinder pestle | Kimble Chase | 885301-0007 | For mitochondria isolation |
| Tissue Grinder tube | Kimble Chase | 885303-0007 | For mitochondria isolation |
| Trichloroacetic acid solution | Sigma Aldrich | T0699 | TCA |
| Tris | MP Biomedicals | 103133 | |
| Triton-x-100 | Teknova | T1105 | |
| Trypsin | Calbiochem | 650211 | |
| Typhoon Imager | GE Healthcare Life Sciences | 28-9558-09 | Laser gel scanner fro 2D-DIGE |
| Ubiquinone | Sigma Aldrich | C7956 |
Lab Prep
Although mitochondria possess their own transcriptional machinery, merely 1% of mitochondrial proteins are synthesized inside the organelle. The nuclear-encoded proteins are transported into mitochondria guided by their mitochondria targeting sequences (MTS); however, a majority of mitochondrial localized proteins lack an identifiable MTS. Nevertheless, the fact that MTS can instruct proteins to go into the mitochondria provides a valuable tool for studying mitochondrial functions of normally nuclear and/or cytoplasmic proteins. We have recently identified the cell cycle kinase CyclinB1/Cdk1 complex in the mitochondria. To specifically study the mitochondrial functions of this complex, mitochondrial overexpression and knock-down of this complex without interfering with its nuclear or cytoplasmic functions were essential. By tagging CyclinB1/Cdk1 with MTS, we were able to achieve mitochondrial overexpression of this complex to study its mitochondrial targets as well as functions. Via tagging dominant-negative Cdk1 with MTS, inhibition of Cdk1 activity was accomplished particularly in the mitochondria. Potential mitochondrial targets of CyclinB1/Cdk1 complex were identified using a gel-based proteomics approach. Unlike traditional 2D gel analysis, we employed 2-dimensional difference gel electrophoresis (2D-DIGE) technology followed by phosphoprotein staining to fluorescently label differentially phosphorylated proteins in mitochondrial Cdk1 expressing cells. Identification of phosphoprotein spots that were altered in wild type versus dominant negative Cdk1 bearing mitochondria revealed the identity of mitochondrial targets of Cdk1. Finally, to determine the effect of CyclinB1/Cdk1 mitochondrial localization in cell cycle progression, a cell proliferation assay using a synthetic thymidine analogue EdU (5-ethynyl-2′-deoxyuridine) was used to monitor the cells as they go through the cell cycle and replicate their DNA. Altogether, we demonstrated a variety of approaches available to study mitochondrial localization and activity of a cell cycle kinase. These are advanced, yet easy to follow methods that will be beneficial to many cell biology researchers.
Although mitochondria possess their own transcriptional machinery, merely 1% of mitochondrial proteins are synthesized inside the organelle. The nuclear-encoded proteins are transported into mitochondria guided by their mitochondria targeting sequences (MTS); however, a majority of mitochondrial localized proteins lack an identifiable MTS. Nevertheless, the fact that MTS can instruct proteins to go into the mitochondria provides a valuable tool for studying mitochondrial functions of normally nuclear and/or cytoplasmic proteins. We have recently identified the cell cycle kinase CyclinB1/Cdk1 complex in the mitochondria. To specifically study the mitochondrial functions of this complex, mitochondrial overexpression and knock-down of this complex without interfering with its nuclear or cytoplasmic functions were essential. By tagging CyclinB1/Cdk1 with MTS, we were able to achieve mitochondrial overexpression of this complex to study its mitochondrial targets as well as functions. Via tagging dominant-negative Cdk1 with MTS, inhibition of Cdk1 activity was accomplished particularly in the mitochondria. Potential mitochondrial targets of CyclinB1/Cdk1 complex were identified using a gel-based proteomics approach. Unlike traditional 2D gel analysis, we employed 2-dimensional difference gel electrophoresis (2D-DIGE) technology followed by phosphoprotein staining to fluorescently label differentially phosphorylated proteins in mitochondrial Cdk1 expressing cells. Identification of phosphoprotein spots that were altered in wild type versus dominant negative Cdk1 bearing mitochondria revealed the identity of mitochondrial targets of Cdk1. Finally, to determine the effect of CyclinB1/Cdk1 mitochondrial localization in cell cycle progression, a cell proliferation assay using a synthetic thymidine analogue EdU (5-ethynyl-2′-deoxyuridine) was used to monitor the cells as they go through the cell cycle and replicate their DNA. Altogether, we demonstrated a variety of approaches available to study mitochondrial localization and activity of a cell cycle kinase. These are advanced, yet easy to follow methods that will be beneficial to many cell biology researchers.
Procedure
Although mitochondria possess their own transcriptional machinery, merely 1% of mitochondrial proteins are synthesized inside the organelle. The nuclear-encoded proteins are transported into mitochondria guided by their mitochondria targeting sequences (MTS); however, a majority of mitochondrial localized proteins lack an identifiable MTS. Nevertheless, the fact that MTS can instruct proteins to go into the mitochondria provides a valuable tool for studying mitochondrial functions of normally nuclear and/or cytoplasmic proteins. We have recently identified the cell cycle kinase CyclinB1/Cdk1 complex in the mitochondria. To specifically study the mitochondrial functions of this complex, mitochondrial overexpression and knock-down of this complex without interfering with its nuclear or cytoplasmic functions were essential. By tagging CyclinB1/Cdk1 with MTS, we were able to achieve mitochondrial overexpression of this complex to study its mitochondrial targets as well as functions. Via tagging dominant-negative Cdk1 with MTS, inhibition of Cdk1 activity was accomplished particularly in the mitochondria. Potential mitochondrial targets of CyclinB1/Cdk1 complex were identified using a gel-based proteomics approach. Unlike traditional 2D gel analysis, we employed 2-dimensional difference gel electrophoresis (2D-DIGE) technology followed by phosphoprotein staining to fluorescently label differentially phosphorylated proteins in mitochondrial Cdk1 expressing cells. Identification of phosphoprotein spots that were altered in wild type versus dominant negative Cdk1 bearing mitochondria revealed the identity of mitochondrial targets of Cdk1. Finally, to determine the effect of CyclinB1/Cdk1 mitochondrial localization in cell cycle progression, a cell proliferation assay using a synthetic thymidine analogue EdU (5-ethynyl-2′-deoxyuridine) was used to monitor the cells as they go through the cell cycle and replicate their DNA. Altogether, we demonstrated a variety of approaches available to study mitochondrial localization and activity of a cell cycle kinase. These are advanced, yet easy to follow methods that will be beneficial to many cell biology researchers.
