Generating Primary Fibroblast Cultures from Mouse Ear and Tail Tissues
Instructor Prep
concepts
Student Protocol
Mice were housed in pathogen-free conditions in compliance with the institutional guidelines until euthanization (The Institutional Animal Care and Use Committee (IACUC) guidelines at the National University of Singapore and the National Advisory Committee for Laboratory Animal Research (NACLAR) guidelines).
1. Mice
- Order one mouse of the appropriate genetic background. This protocol is based on tissue derived from one C57BL/6 mouse.
2. Preparation of Complete Medium
- Prepare complete medium by adding the following components to Roswell Park Memorial Institute (RPMI) 1640 medium: 10% fetal calf serum (FCS), 50 µM 2-mercaptoethanol, 100 µM asparagine, 2 mM glutamine, 1% penicillin-streptomycin solution.
3. Preparation of Enzyme Solutions
- Prepare collagenase D solution in a 15 ml conical bottom tube.
- Weigh 10 mg of collagenase D. Dissolve collagenase D in 4 ml complete medium.
- Prepare pronase solution in a 1.7 ml microcentrifuge tube.
- Weigh 10 mg of pronase.
- Add 5 µl of 1 M Tris buffer (pH 8.0). Add 1 µl of 0.5 M EDTA (pH 8.0).
- Top up with 494 µl of sterile water.
- Incubate the pronase solution at 37 °C in a water bath for 30 min.
4. Preparation of Collagenase D-pronase Mix (≤2 Tails)
Note: Perform the subsequent steps in a sterile cell culture hood.
- Add 250 µl of pronase solution to 4 ml of collagenase D solution.
- Pass the collagenase D-pronase mixture through a 0.2 µm syringe filter into a sterile 15 ml conical bottom tube.
5. Extraction of Fibroblasts from Ear and Tail Tissues
- Euthanize mice according to the appropriate institutional guidelines.
- Place autoclaved surgical instruments (scissors and forceps) in the cell culture hood.
- Add 10 ml complete medium into two 10 cm cell culture dishes each.
- Cut ears (~1 cm radius) and 5 cm of tail (from the tip of the tail) of a mouse with scissors and incubate for 5 min in 40 ml 70% ethanol in a sterile 50 ml conical bottom tube.
- Air-dry ears and tail by placing them in an open 10 cm cell culture dish in the hood for 5 min. Once dried, transfer ear and tail pieces to two culture dishes labelled "ears" and "tail" containing 10 ml complete medium as described in step 5.3.
- Remove hair from the ears and tail using scissors.
- Cut ears and tail into pieces smaller than 3 mm in size using scissors.
- Transfer the cut tissues into 1.8 ml cryotube vials labelled "ears" and "tail" and add sufficient collagenase D-pronase solution for the volume to reach the 1.8 ml mark on the vial.
- Place the cryotube vials horizontally on a shaker and shake the samples at 200 rpm for 90 min at 37 °C.
- After 90 min incubation, remove the cryotube vials from the shaker and place the vials in the hood.
- Add 10 ml complete medium into two 10 cm cell culture dishes, labelled "ears" and "tail" each, and put a 70 µm cell strainer in each dish.
- Place the digested ear and tail tissues in the 70 µm cell strainer in the accordingly labelled dishes prepared in step 5.11 and forcefully grind the tissues using a 10 ml syringe plunger for >5 min. Shake the cell strainer occasionally in the medium to wash cells out of the cell strainer.
- Pipette the cell suspension from each dish into two 15 ml conical bottom tubes labelled "ears" and "tail". Wash the dish and strainer with additional 10 ml complete medium and add the medium to the appropriate 15 ml conical bottom tubes.
- Spin down the cell suspension for 7 min at ~580 x g and 4 °C using a refrigerated cell centrifuge.
- Remove supernatant, add 10 ml complete medium to the cell pellet in the 15 ml conical bottom tube and resuspend the cells.
- Repeat step 5.14 and 5.15.
6. Culturing of Cell Mixture
- Remove supernatant. Ensure that the cell pellet remains undisturbed.
- Re-suspend cells in 10 ml complete medium and add the respective mixture to two 10 cm cell culture dishes labelled "ears" and "tail".
- Add 10 µl amphotericin B solution (stock solution: 250 µg/ml) to the culture.
- Incubate cells at 37 °C in a humidified 5% CO2 incubator.
- On the third day, replace the medium with 10 ml fresh complete medium containing 10 µl of amphotericin B to remove debris (Figures 1 and 2).
7. Sub-culture of Fibroblasts
- When culture reaches around 70% confluency (day 3-4 of culture) remove the medium and wash the cells with 5 ml sterile 1x phosphate buffered saline (PBS).
- Remove PBS and add 2 ml sterile 1x trypsin-EDTA solution to the cells.
- Incubate the cells for 5 min at 37 °C in a humidified 5% CO2 incubator.
- After 5 min, gently tap the culture dish and add 6 ml complete medium to the cells.
- Transfer the cell suspension to a 15 ml conical bottom tube and spin the tube for 5 min at ~450 x g and 4 °C using a refrigerated cell centrifuge.
- Remove supernatant, and gently re-suspend the cell pellet in 5 ml complete medium.
- Seed 2 x 105 cells in a 10 cm cell culture dish and incubate the cells at 37 °C in a humidified 5% CO2 incubator for 3-4 days before repeating steps 7.1 to 7.6.
8. Preparation of Ears for Shipment
Note: Perform the subsequent steps in a sterile cell culture hood.
- Euthanize mice according to the appropriate institutional guidelines.
- Place autoclaved scissors and forceps into the cell culture hood.
- Add 50 ml complete medium into a 50 ml conical bottom tube.
- Cut ears (~1 cm radius) of a mouse with scissors.
- Transfer the ears into the 50 ml conical bottom tube (as prepared in step 3) using forceps.
- Seal the 50 ml conical bottom tube with parafilm before shipping at RT in an appropriate box.
Generating Primary Fibroblast Cultures from Mouse Ear and Tail Tissues
Learning Objectives
Extraction of fibroblasts from tissue results in a significant amount of tissue debris (Figure 1). In contrast to tissue debris, fibroblasts adhere to tissue culture plastic surfaces between day 1 and 3 of culture. The medium of fibroblast cultures can be safely changed on day 3 of culture, which should significantly decrease the levels of debris present in the culture (Figure 2). Fibroblasts display an elongated morphology and a clearly visible cytoplasm (Figures 1 and 2). Mitotic cells should be present from day 3 of culture onwards and cells should reach 70-80% of cell confluency within 3-4 days of culture. The yield from ear and tail tissues in a 10 cm culture dish ranges from 4 to 5 x 105 (ears) and 5 to 6 x 105 cells (tail) on day 3 of culture. Following the third day of fibroblasts isolation, the cells can be passaged and seeded at 2 x 105 cells per 10 cm culture dish.
Extraction of fibroblasts from ears stored at RT for 10 days should result in 70-80% cell confluency within 5 to 6 days of culture (Figure 3). Seeding the cells at 2 x 105 cells per 10 cm culture dish after day 5 should also give rise to approximately 1 x 106 cells within 3-4 days of culture. Long-term storage does not affect the time it takes for fibroblasts to enter senescence in our experience (data not shown).
To verify the identity of cells after 3 days of culture, cells can be labelled for the fibroblasts marker vimentin14. Using the above protocol, we routinely obtain pure fibroblast cultures as indicated by vimentin staining (Figure 4).
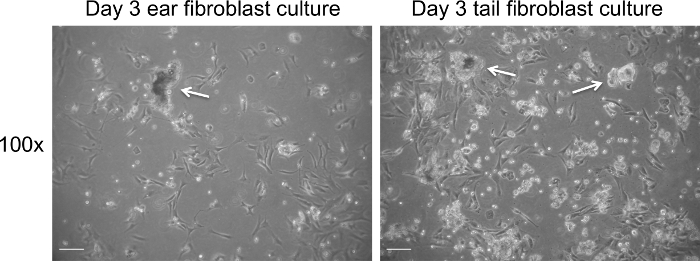
Figure 1. Fibroblast culture on day 3 post extraction prior to change of medium. Representative bright field images of cell debris (white arrows) present on day 3 of culture. Images were captured at 100× magnification using a light microscope. The scale bar represents 100 µm. Please click here to view a larger version of this figure.
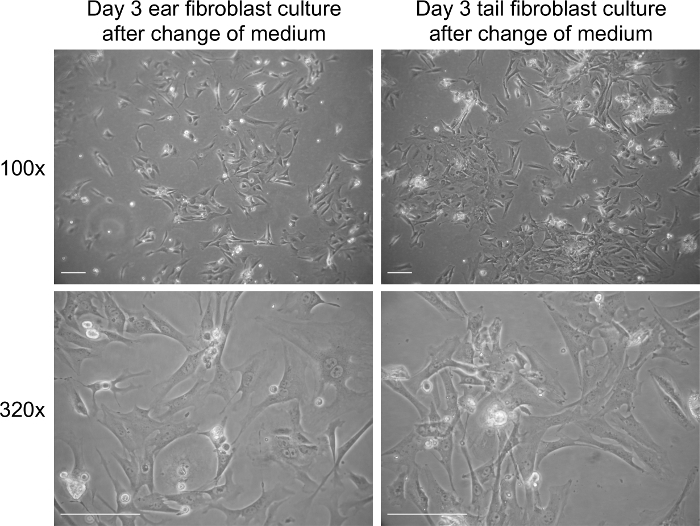
Figure 2. Fibroblast culture on day 3 post extraction after addition of new medium. Representative bright field images of fibroblasts on day 3 in culture. Images were captured at 100× and 320× magnification using a light microscope. The scale bar represents 100 µm. Please click here to view a larger version of this figure.
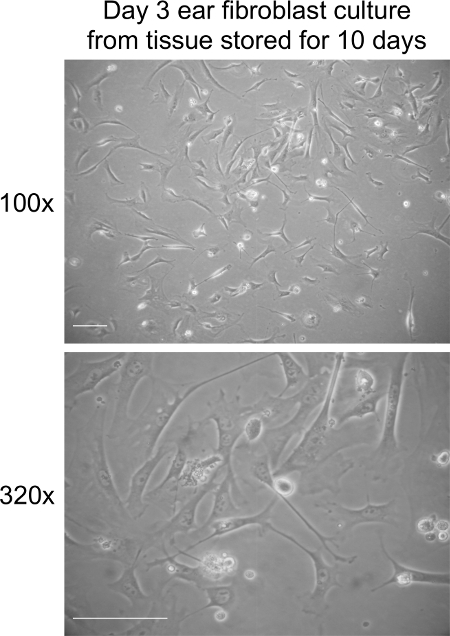
Figure 3. Fibroblast culture on day 3 post extraction from ear tissues stored at RT for 10 days. Representative bright field images of fibroblasts at day 3 in culture. Images were captured at 100× and 320× magnification using a light microscope. The scale bar represents 100 µm. Please click here to view a larger version of this figure.
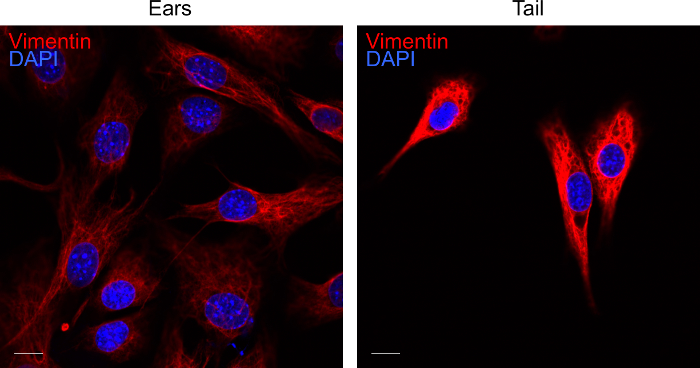
Figure 4. Labelling of fibroblast cultures for vimentin. Representative confocal image of ear and tail fibroblasts on day 3 of culture. Fibroblasts extracted from the tissues were labelled for vimentin (red) and DAPI (blue). The fluorescent images were acquired by confocal microscopy. The scale bar represents 10 µm. Please click here to view a larger version of this figure.
List of Materials
| RPMI-1640 | HyClone | SH30027.01 | |
| Fetal Calf Serum | HyClone | SV30160.03 | |
| 2-mercaptoethanol | Sigma-Aldrich | M3148 | |
| Asparagine | Sigma-Aldrich | A4159 | |
| Glutamine | Sigma-Aldrich | G8540 | |
| Penicillin/Streptomycin | HyClone | SV30010 | |
| Ethanol | Merck Millipore | 107017 | Absolute for analysis |
| Collagenase D | Roche Diagnostics | 11088866001 | From Clostridium histolyticum, lyophilized, non-sterile |
| Pronase protease | Merck Millipore | 53702 | From Streptomyces griseus |
| Tris buffer (pH 8) | 1st BASE | 1415 | Ultra pure grade |
| 0.5M EDTA (pH 8) | 1st BASE | BUF-1053 | Biotechnology grade |
| 10X Phosphate Buffered Saline (PBS) | 1st BASE | BUF-2040-10X4L | Ultra pure grade |
| Trypsin-EDTA solution 10X | Sigma-Aldrich | 59418C-100ML | 0.5% trypsin, 0.2% EDTA, trypsin gamma irradiated by SER-TAIN process, without phenol red, in saline |
| Amphotericin B | Sigma-Aldrich | A2492-20ml | 250 μg/ml in deionized water, sterile-filtered |
| Scissors | Aesculap | ||
| Forcep | Aesculap | AE-BD312R | |
| 0.2 μM syringe filter | Sartorius Stedim | 16534 | |
| 70 μM cell strainer | SPL | 93070 | |
| Syringe plunger | Terumo | SS+10L | |
| Cryovial tube | NUNC | 368362 | |
| 1.7 ml microcentrifuge tube | Axygen | MCT-175-C | |
| 10 cm cell culture dish | Greiner | 664160 | Cell culture treated dish |
| 15 ml conical bottom tube | Greiner | 188271 | |
| 50 ml conical bottom tube | Greiner | 227261 | |
| Water bath | GFL | 1002 | |
| Centrifuge | Eppendorf | 5810R | |
| Incubation shaker | Sartorius Stedim | Certomat-BS1 | |
| Zeiss Axiovert 25 light microscope | Carl Zeiss AG |
Lab Prep
Primary cells are derived directly from tissue and are thought to be more representative of the physiological state of cells in vivo than established cell lines. However, primary cell cultures usually have a finite life span and need to be frequently re-established. Fibroblasts are an easily accessible source of primary cells. Here, we discuss a simple and quick experimental procedure to establish primary fibroblast cultures from ears and tails of mice. The protocol can be used to establish primary fibroblast cultures from ears stored at RT for up to 10 days. When the protocol is carefully followed, contaminations are unlikely to occur despite the use of non-sterile tissue stored for extended time in some cases. Fibroblasts proliferate rapidly in culture and can be expanded to substantial numbers before undergoing replicative senescence.
Primary cells are derived directly from tissue and are thought to be more representative of the physiological state of cells in vivo than established cell lines. However, primary cell cultures usually have a finite life span and need to be frequently re-established. Fibroblasts are an easily accessible source of primary cells. Here, we discuss a simple and quick experimental procedure to establish primary fibroblast cultures from ears and tails of mice. The protocol can be used to establish primary fibroblast cultures from ears stored at RT for up to 10 days. When the protocol is carefully followed, contaminations are unlikely to occur despite the use of non-sterile tissue stored for extended time in some cases. Fibroblasts proliferate rapidly in culture and can be expanded to substantial numbers before undergoing replicative senescence.
Procedure
Primary cells are derived directly from tissue and are thought to be more representative of the physiological state of cells in vivo than established cell lines. However, primary cell cultures usually have a finite life span and need to be frequently re-established. Fibroblasts are an easily accessible source of primary cells. Here, we discuss a simple and quick experimental procedure to establish primary fibroblast cultures from ears and tails of mice. The protocol can be used to establish primary fibroblast cultures from ears stored at RT for up to 10 days. When the protocol is carefully followed, contaminations are unlikely to occur despite the use of non-sterile tissue stored for extended time in some cases. Fibroblasts proliferate rapidly in culture and can be expanded to substantial numbers before undergoing replicative senescence.
