Visualization of Surface-tethered Large DNA Molecules with a Fluorescent Protein DNA Binding Peptide
Instructor Prep
concepts
Student Protocol
1. DNA Biotinylation
- Biotinylation of blunt ended DNA using Terminal Transferase (TdT)
Note: Use T4 DNA (166 kbp), which is a blunt ended DNA.- Add 5 µl of 2.5 mM CoCl2, 5 µl of 10× reaction buffer, 0.5 µl of 10 mM biotin-11-dUTP, 0.5 µl of terminal transferase (10 units), and 0.5 µl of T4 DNA (0.5 µg/µl) to the reaction mixture. Make to a final volume of 50 µl by adding 38.5 µl of water.
- Incubate the reaction mixture at 37 °C for 1 hr.
Note: Extended reaction time may yield double-tethered DNA, i.e., it is possible that biotins are tagged at both ends. - Stop the reaction by adding 5 µl of 0.5 M EDTA, pH 8.
- Keep the tube at 4 °C.
- Biotinylation of sticky ended DNA Using DNA Ligase
Note: Use λ DNA (48.5 kbp), which is a sticky-end DNA.- Add 1 µl of T4 DNA ligase, 5 µl of 10× ligation buffer, 1 µl of 25 ng/µl λ phage DNA, and add 43 µl of water to make a final volume of 50 µl.
- Keep the tube at 4 °C.
2. Functionalized Surface Derivatization
Note: In order to tether an end of a DNA molecule on glass surface, a primary amine group is silanized on a coverslip followed by biotin-PEG coating as shown in Figure 1. This PEGylation process is important for single-molecule DNA imaging because it can significantly decrease random noise created by attachment of undesired molecules on the surface.
- Piranha Cleaning
Note: Piranha solutions react vigorously with organic materials, therefore it should be handled with care, following proper safety guidelines.- Place coverslips on a polytetrafluoroethylene (PTFE) rack, and hold them by PTFE thread seal tape lengthwise, half on the edge of the surfaces and half on the rack. After wrapping, leave a long piece (~ 5 cm) of tape for handling the rack during the cleaning procedure.
- Fill a 1 L glass beaker with 350 ml of H2SO4 and 150 ml of H2O2 to make piranha solution in a fume hood. Place the rack in piranha solution for 2 hr.
- Empty piranha solution from the beaker, and rinse coverslips thoroughly with deionized water until the pH of water in the beaker reaches neutral. Use pH paper strips.
- Sonicate the racks of cover slips in the beaker containing water, for 30 min. Empty water from the beaker just before amino silanization. Use 75 W of sonication power to clean and derivatize the glass substrates.
- Aminosilanization on Glass Surface
- Add 2 ml of N-[3-(trimethoxysilyl)propyl]ethylenediamine and 10 ml of glacial acetic acid to 200 ml of methyl alcohol in a clean polypropylene container for preparing the aminosilanization solution.
- Place cleaned coverslips in the polypropylene container. Shake them at 100 rpm and room temperature for 30 min.
- Sonicate the beaker with derivatized cover slips for 15 min at 75 W. Shake them at 100 rpm and room temperature for at least 30 min.
- Empty the solution from the beaker, and rinse coverslips carefully, once with methyl alcohol, and twice with ethyl alcohol. Store coverslips in ethyl alcohol and use them within two weeks.
- PEGylation of the coverslip
- Make 10 ml of 0.1 M sodium bicarbonate (NaHCO3). Filter the solution with a 0.22-µm syringe filter.
- Dissolve 2 mg of biotin-PEG-succinimidyl carbonate (biotin-PEG-SC) and 80 mg of PEG-succinimidyl valerate (mPEG-SVG) in 350 µl of NaHCO3. Use a light protection tube.
- Vortex the tube vigorously for 10 sec, and centrifuge it at 10,000 × g for 1 min to remove bubbles.
- Rinse a glass slide with acetone, followed by rinsing with ethyl alcohol. Allow the slide to air dry completely.
- Place a drop (50 µl) of PEG solution on the clean glass slide. Cover the droplet gently with an amino silanized cover slip, without generating any bubbles.
- Place slides for 3 hr to overnight in a dark, well-leveled humid chamber at room temperature. Use an empty pipette tip holder as the chamber, with water at the bottom. Seal residual PEG solution in the tube and keep it at 4 °C.
- Rinse the PEGylated coverslip thoroughly with deionized water. Store it in a dark and dry place until use.
3. Assembling a Flow Chamber
- Fabricate a punched acrylic holder, as in Figure 2. Ensure that the diameter of the tubing insert is 0.762 mm, and designate one hole is an inlet and the other is an outlet (Figure 2).
- Place double-sided sticky tape strips (width 5-6 mm) on an acrylic holder (Figure 2), aligning perpendicularly to an inlet and outlet hole, not perturbing any channel holes. Use these tape strips as multi-channel walls to make chambers. Scrub the tape using a pipet tip.
- Place a PEGylated coverslip on the top to make flow chambers, with the PEGylated side facedown.
- To prevent any solution from leaking, press the top of a coverslip over the area where double-sided tapes are placed. Add quick-dry epoxy glue to close the edges of the chamber at the top and at the bottom (yellow color in Figure 2).
- Connect a short piece (2.5 cm) of tubing (outer diameter, OD, 0.042") to a gas-tight syringe and seal the joint with epoxy glue.
- Connect a long flexible tubing (OD: 0.03") to the tubing linked to the syringe and seal the joint with epoxy glue.
- Fill the tubing linked to the syringe with DI water. Make sure there is no air bubble.
- Insert the tubing into the hole of the flow chamber sealed with epoxy glue.
- Place a yellow tip (200 µl tip) on the other hole as a reservoir.
4. Sample Loading into Flow Chamber
Note: Neutravidin can be replaced with other avidin proteins such as streptavidin. All of the reactions can be performed at room temperature, unless it is mentioned. Take sample solution in a yellow tip, and install the tip with the solution onto the hole of acrylic holder (Figure 2b).
- Set the flow rate of the syringe pump at 50 µl/min. Load 20 µl of avidin protein (25 µg/ml in T50 solution, Tris 10 mM, NaCl 50 mM, pH 8), and keep it for 10 min.
- Load 20 µl of biotinylated oligodeoxynucleotides (100 µM in 1× TE), and keep it for 10 min. If terminal transferase is used, skip the loading of oligodeoxynucleotides.
- Load 20 µl of DNA solution from Step 1 into the flow chamber at a flow rate of 10 µl/min, and keep it for 30 min.
- Wash the flow chamber with 1× TE, and load 40 µl of FP-DBP10 (~80 nM).
- Observe DNA under a fluorescent microscope with continuous flow of staining molecules in 1× TE on a 60X objective lens. Use 488 nm solid state laser for excitation of FP(eGFP)-DBP. For full stretch of DNA molecules, apply different flow rates according to the DNA lengths used. For example, apply 50 µl/min for 48.5 kbp of λ DNA, and 100 µl/min for 166 kbp of T4 DNA.
- For recycling of the acrylic holder, soak assembled flow chambers in a household detergent solution12. Remove tapes and epoxy with a razor blade and rub with hands to completely take them off. Unclog holes with syringe needles. Keep the holders in deionized water until further use.
Visualization of Surface-tethered Large DNA Molecules with a Fluorescent Protein DNA Binding Peptide
Learning Objectives
Figure 1 shows two different DNA tethering methods depending on terminal structures of the DNA molecule. Figure 1a illustrates how the sticky ended DNA molecules are hybridized with complementary biotinylated oligonucleotides, which are immobilized on the avidin-coated PEG surface. Figure 1b shows the addition of biotinylated ddNTP or dNTP to the 3' hydroxyl group of a blunt ended DNA by terminal transferase. We added flexible linkers between DNA molecules and biotin. This increased the ligation and avidin-biotin binding efficiency to provide sufficient space for the reactions. Figure 1c demonstrates the overall schematic representation for DNA tethering on the avidin-coated PEG surface.
Figure 2 depicts the assembly of the flow chamber along with an acrylic holder. Compared to glass/quartz slide glasses, a custom-made acrylic holder simplifies the preparation of the flow chamber for epi-fluorescent microscopy.12 The flow chamber enables instant changing of buffer conditions for enzyme reactions,6 and stretching DNA molecules.
Figure 3 demonstrates single-tethered DNA and T4 λDNA stained with FP-DBP. Microfluidic shear flows elongated single DNA molecules up to full contour lengths such as 16.5 μm (48.5 kb λ DNA x 0.34 nm) and 56.4 μm (166 kb T4 DNA x 0.34 nm). We were able to repeat DNA stretching and relaxation multiple times during an extended period (e.g., half-hour), since we utilized FP-DBP that does not cause photo-cleavage.
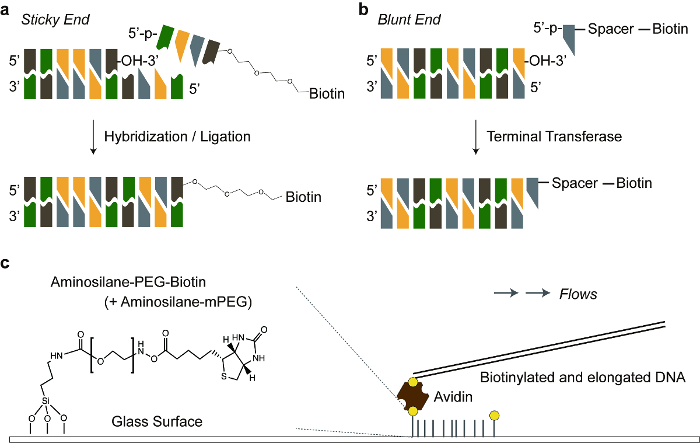
Figure 1. Schematic illustration of DNA tethering. a) A sticky ended DNA is hybridized with a biotinylated complementary oligonucleotide. Triethylene glycol is a flexible linker between the oligonucleotide and biotin. b) Biotinylated dNTP is added to a blunt end of double stranded DNA, by terminal transferase. Polycarbon spacer (11-atoms) is a flexible linker between dNTP and biotin. c) Biotinylated DNA is anchored to an avidin protein, which is linked to a biotinylated PEG covalently bonded to the glass surface. The ratio of biotinylated PEG to normal PEG was 0.025 (1:40). Please click here to view a larger version of this figure.
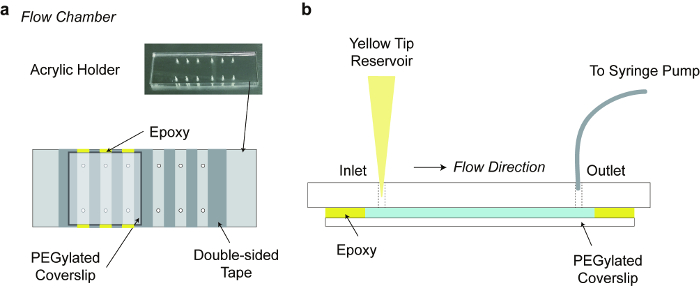
Figure 2. Flow chamber. a) Flow chamber consisting of an acrylic acid resin holder, strips of double-sided tapes and a PEGylated cover slip. The acrylic holder of dimensions 76 mm x 26 mm x 5 mm (L x W x H), was fabricated with laser cutting. b) Side view of the Flow chamber: punched holes are an inlet port on which a yellow tip is installed for a buffer reservoir, and an outlet port that is connected to a tubing controlled by a syringe-pump. Please click here to view a larger version of this figure.

Figure 3. Fluorescent microscopic images of stretched DNA molecules stained with FP-DBP. a) Bacteriophage λ DNA (48.5 kbp) tethered on the surface after ligation with biotinylated complementary oligonucleotides. b) Bacteriophage T4 DNA (166 kbp) tethered on the surface after adding biotin with terminal transferase. Indicated arrows show DNA molecules stretched up to their full contour lengths such as 16.5 µm for λ DNA and 56.4 µm for T4 DNA. Scale bars: 10 µm. Please click here to view a larger version of this figure.
List of Materials
| 1. DNA Biotinylation | |||
| 1.1) Biotinylation of blunt-end DNA using Terminal Transferase (TdT) | |||
| Terminal Transferase | New England Biolabs | M0315S | Provided with 10x reaction buffer, 2.5 mM Cobalt chloride |
| Biotin-11-dUTP | Invitrogen | R0081 | Biotin-ddNTP is also available |
| T4GT7 Phage DNA | Nippon Gene | 318-03971 | |
| Ethylenediaminetetraacetic acid | Sigma-Aldrich | E6758 | EDTA |
| 1.2) Biotinylation of sticky end DNA Using DNA Ligase | |||
| T4 DNA Ligase | New England Biolabs | M0202S | Provided with 10x reaction buffer |
| Lambda Phage DNA | Bioneer | D-2510 | Also available at New England Biolabs |
| 2. Functionalized Surface Derivatization | |||
| 2.1) Piranha Cleaning | |||
| Coverslip | Marienfeld-Superior | 0101050 | 22×22 mm, No. 1 Thickness |
| Teflon rack | Custom Fabrication | ||
| PTFE Thread Seal Tape | Han Yang Chemical Co. Ltd. | 3032292 | Teflon™ tape |
| Sulferic acid | Jin Chemical Co. Ltd. | S280823 | H2SO4, 95 % Purity |
| Hydrogen peroxide | Jin Chemical Co. Ltd. | H290423 | H2O2, 35 % in water |
| Sonicator | Daihan Scientific Co. Ltd. | WUC-A02H | Table-top Ultrasonic Cleaner |
| 2.2) Aminosilanization on Glass Surface | |||
| N-[3-(Trimethoxysilyl)propyl] ethylenediamine |
Sigma-Aldrich | 104884 | |
| Glacial Acetic Acid | Duksan Chemicals | 414 | 99 % Purity |
| Methyl Alcohol | Jin Chemical Co. Ltd. | M300318 | 99.9 % Purity |
| Polypropylene Container | Qorpak | PLC-04907 | |
| Ethyl Alcohol | Jin Chemical Co. Ltd. | A300202 | 99.9 % Purity |
| 2.3) PEGylation of the coverslip | |||
| Sodium Bicarbonate | Sigma-Aldrich | S5761 | |
| Syringe Filter | Sartorius | 16534———-K | |
| Biotin-PEG-SC | Laysan Bio | Biotin-PEG-SC-5000 | |
| mPEG-SVG | Laysan Bio | MPEG-SVA-5000 | |
| Acetone | Jin Chemical Co. Ltd. | A300129 | 99 % Purity |
| Microscope Slides | Marienfeld-Superior | 1000612 | ~76x26x1 mm |
| 3. Assembling a Flow Chamber | |||
| Acrylic Support | Custom Fabrication | ||
| Double-sided Tape | 3M | Transparent type | |
| Quick-dry Epoxy | 3M | ||
| Polyethylene Tubing | Cole-Parmer | 06417-11, 06417-21 | |
| Gas Tight 250 µl Syringe | Hamilton | 81165 | |
| Syringe Pump | New Era Pump Systems Inc. | NE-1000 | |
| 4. Sample Loading into Flow Chamber | |||
| Neutravidin | Thermo Scientific | 31000 | |
| Tris base | Sigma-Aldrich | T1503-5KG | Trizma base |
| Microscope | Olympus | IX70 | |
| EMCCD Camera | Q Imaging | Rolera EM-C2 | |
| Solid-state Laser (488 nm) | Oxxius | LBX488 | |
| Alconox | Alconox Inc. | ||
Lab Prep
Large DNA molecules tethered on the functionalized glass surface have been utilized in polymer physics and biochemistry particularly for investigating interactions between DNA and its binding proteins. Here, we report a method that uses fluorescent microscopy for visualizing large DNA molecules tethered on the surface. First, glass coverslips are biotinylated and passivated by coating with biotinylated polyethylene glycol, which specifically binds biotinylated DNA via avidin protein linkers and significantly reduces undesirable binding from non-specific interactions of proteins or DNA molecules on the surface. Second, the DNA molecules are biotinylated by two different methods depending on their terminals. The blunt ended DNA is tagged with biotinylated dUTP at its 3′ hydroxyl terminus, by terminal transferase, while the sticky ended DNA is hybridized with biotinylated complimentary oligonucleotides by DNA ligase. Finally, a microfluidic shear flow makes single DNA molecules stretch to their full contour lengths after being stained with fluorescent protein-DNA binding peptide (FP-DBP).
Large DNA molecules tethered on the functionalized glass surface have been utilized in polymer physics and biochemistry particularly for investigating interactions between DNA and its binding proteins. Here, we report a method that uses fluorescent microscopy for visualizing large DNA molecules tethered on the surface. First, glass coverslips are biotinylated and passivated by coating with biotinylated polyethylene glycol, which specifically binds biotinylated DNA via avidin protein linkers and significantly reduces undesirable binding from non-specific interactions of proteins or DNA molecules on the surface. Second, the DNA molecules are biotinylated by two different methods depending on their terminals. The blunt ended DNA is tagged with biotinylated dUTP at its 3′ hydroxyl terminus, by terminal transferase, while the sticky ended DNA is hybridized with biotinylated complimentary oligonucleotides by DNA ligase. Finally, a microfluidic shear flow makes single DNA molecules stretch to their full contour lengths after being stained with fluorescent protein-DNA binding peptide (FP-DBP).
Procedure
Large DNA molecules tethered on the functionalized glass surface have been utilized in polymer physics and biochemistry particularly for investigating interactions between DNA and its binding proteins. Here, we report a method that uses fluorescent microscopy for visualizing large DNA molecules tethered on the surface. First, glass coverslips are biotinylated and passivated by coating with biotinylated polyethylene glycol, which specifically binds biotinylated DNA via avidin protein linkers and significantly reduces undesirable binding from non-specific interactions of proteins or DNA molecules on the surface. Second, the DNA molecules are biotinylated by two different methods depending on their terminals. The blunt ended DNA is tagged with biotinylated dUTP at its 3′ hydroxyl terminus, by terminal transferase, while the sticky ended DNA is hybridized with biotinylated complimentary oligonucleotides by DNA ligase. Finally, a microfluidic shear flow makes single DNA molecules stretch to their full contour lengths after being stained with fluorescent protein-DNA binding peptide (FP-DBP).
