Using Deuterium Oxide as a Non-Invasive, Non-Lethal Tool for Assessing Body Composition and Water Consumption in Mammals
Instructor Prep
concepts
Student Protocol
All experiments described here were approved by the University of Missouri Animal Care and Use Committee and conducted under the Missouri Department of Conservation (MDC) Wildlife Scientific Collection permit (Permit #16409 and #17649).
1. Preparation of sterile, isotonic, salinated D2O stock solution
- Make a 50 mL stock solution of 9.0 g/L salinated D2O.
- Weigh 450 mg of pharmaceutical grade NaCl and transfer all NaCl into a 100 mL, sterilized beaker. Record the exact amount of NaCl to 4 decimal places in the laboratory notebook.
- Using a sterile graduated cylinder, measure 50 g of ≥ 99.8% deuterium oxide and transfer to the sterile beaker containing the NaCl. Record the exact amount of deuterium oxide to 4 decimal places in the laboratory notebook or spreadsheet.
- Filter 10 mL of isosmotic strength NaCl (9.0 g/L) through a non-pyrogenic sterile disk filter with submicron pores (0.2 µm).
- Attach a 20 G needle to the non-pyrogenic sterile disk filter with submicron pores (0.2 µm) fitted with a 10 mL syringe barrel. Insert into the septum of a 100 mL sterile empty vial.
- Attach a vacuum tube to a 22 G needle and insert the needle into the septum of the 100 mL sterile empty vial.
- Pour 10 mL of the stock solution into the syringe barrel. Slowly turn on the vacuum until the D2O stock solution begins to slowly filter into the sterile vial. Continue to pour the D2O stock solution into the syringe barrel until all 50 mL is filtered.
NOTE: The stock solution may need to be diluted or concentrated depending on the dose required. The dose of D2O will vary based upon the species and the sensitivity of the analytical method. For cats, the working solution was used to administer a dose of 0.7 g/kg D2O. The stock solution described above minimizes the amount of NaCl solution introduced subcutaneously to the animal while still allowing accurate measurement of the dose. For small mammals such as bats, this concentration must be diluted to a working solution such as 0.1600 g/mL. This concentration allows the dose of 0.75 g/kg D2O to be accurately measured and administered in approximately 100 µL or less NaCl solution.
2. Preparation of sterile, isotonic, salinated D2O stock working solution for bats
- Weigh a 10 mL empty sterile vial and record weight to nearest 4 decimal places. Tare scale.
- Use a 1.0 mL syringe to transfer 0.65 mL of the D2O stock solution to the tared, 10 mL empty sterile vial. Record weight of D2O to 4 decimal places. Tare scale.
- Calculate the volume of D2O in the 10 mL empty vial. Use the following equation.
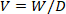
where W is recorded weight and D is the density of 99.8% D2O (1.107 g/mL). - Use the calculated volume and known weight of the D2O to determine the volume of isotonic saline required to make ~0.1600 g/mL working solution.
- Insert into the septum of the 10 mL sterile vial, the 22 G needle (attached to the vacuum tube). Insert into the septum of the 10 mL sterile vial, the 20 G needle (attached to a 0.22 µm syringe filter fitted with a 10 mL syringe barrel).
- Pour the calculated mass/volume of isotonic NaCl into the syringe barrel and turn on the vacuum to allow a slow drip into the sterile 10 mL vial.
- Record the weight of the vial and ensure a ~0.1600 g/mL working solution is created.
3. Determination of body composition of big brown bats (Eptesicus fucsus) with D2O
NOTE: The stock solution of D2O used in the protocol is 0.1598 g/mL. Before collecting blood, ensure that removing up to 200 µL of blood will be ≤ 10% of the total blood volume of the bat and is within the Institutional Animal Care and Use Committee's (IACUC) established guidelines for blood collection. All animals should be fasted or abdomen palpated to ensure an empty stomach. A recent meal could alter the animal's weight resulting in confounded results since calculations for determining body fat rely upon the body mass of the animal.
- Anesthetize a big brown bat.
- Use 5.0% isoflurane for induction. Maintain a stable plane of anesthesia using 0.5%−3.0% isoflurane.
- Determine proper anesthesia depth by testing the pedal withdrawal reflex (pinching the bat's toes). The bat should not respond to the sensation and the respiratory rate should remain slow and stable. Adjust isoflurane as needed to maintain a stable plane of anesthesia.
- Record isoflurane level, heart rate, respiratory rate, and other information as required by IACUC.
- Weigh the big brown bat and record the weight to 4 decimal places.
- Clean the uropatagium (tail membrane) over the interfemoral vein with an alcohol prep pad and allow to dry. Apply a thin layer of petroleum jelly over the interfemoral vein.
- Use a 29 G needle to puncture the dorsal portion of the interfemoral vein and collect 100 µL of blood using plastic sodium heparin capillary tubes. Ensure adequate mixing of the whole blood with the sodium heparin by gently rolling each tube after collection and label the tube.
- Using a DXA machine calibrated for small mammals, obtain three DXA scans of the bat10.
- Determine the mass (in g) of D2O to inject by multiplying the bat weight in kg by the D2O dose of 0.75 g/kg. Determine the volume of the calculated D2O dose (V) by dividing the weight of the D2O dose by the concentration of the working solution.
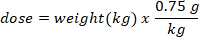
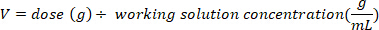
- Use an insulin syringe with a 29 G needle attached to draw up the volume of D2O calculated. Weigh the D2O, insulin syringe, and needle. Record to 4 decimal places.
- Inject the D2O subcutaneously over the dorsal hip region of the anesthetized bat.
- Allow bat to recover from anesthesia and record the time of injection.
- Immediately after injection, weigh the now empty insulin syringe with the 29 G needle attached. Record the weight to 4 decimal places.
- Determine the dose of D2O injected by subtracting the post-injection weight of the insulin syringe from the pre-injection D2O filled insulin syringe. Record to 4 decimal places.
- Within 30 min post blood collection, use a hematocrit centrifuge to spin each capillary tube for 5 min. If the hematocrit centrifuge allows multiple speeds, set to 10,000 x g.
- Use a sharp scissors to cut the plastic capillary tube between the whole blood and plasma. Use a 200 µL pipette to expel the plasma directly into a labelled, 500 µL storage tube.
- After the equilibration period, collect another 100 µL of blood from the interfemoral vein.
NOTE: The equilibration period will vary by species and if the bats go into torpor. For big brown bats, typically 2 h is sufficient for the equilibration period. - Separate plasma into a second labelled, 500 µL microcentrifuge screw top tube by repeating step 3.13. Store samples at -20 °C or colder until analysis.
4. Fourier-transform infrared spectrophotometry analysis
- Set the temperature of a sand bath to 60 °C to facilitate distillation (allow separation of water and D2O from other blood components).
- Pipette 50 µL of each plasma sample and standard onto the inside of a 1.5 mL conical microcentrifuge tube cap. Including standards containing known concentrations of D2O as quality control.
NOTE: Ideally each animal will have three replicates per sample and the average of the three replicates reported. Due to the limited sample volume and the volume of sample required for the FT-IR equipment utilized by the authors, no replicates were performed for the bat samples. If any sample contains less than 50 µL of plasma, pipette the sample amount onto the conical microcentrifuge tube cap and record the volume. - Keep the microcentrifuge cap upside down and screw the 1.5 mL conical microcentrifuge tube onto the cap. Place the inverted (upside-down) tube with the cap in contact with the sand in the sand bath for a minimum of 12 h (overnight).
- After 12 h, remove the cap and replace with a new, clean cap. Pulse the microcentrifuge tube for 10 s in a centrifuge.
- Create the following standards: 0 ppm (0 mg D2O in 1 L distilled water), 293 ppm (293 mg D2O in 1 L distilled water), 585 ppm (585 mg D2O in 1 L distilled water), 878 ppm (878 mg D2O in 1 L distilled water), and 1170 ppm D2O (1170 mg D2O in 1 L distilled water).
NOTE: The values above are suggested for a standard curve. Alternative values such as 250 ppm, 500 ppm, 750 ppm, and so forth can be used. - Install a liquid transmission cell into the Fourier-transform infrared spectrophotometry (FTIR) spectrometer (Table of Materials). Fill the cell with methanol and connect the injection port. Slowly fill the cell with background water while carefully removing the methanol syringe to reduce the risk of air bubbles. Attach tubing to the output port to allow removal of the samples post-analysis.
- Prepare the FTIR spectrometer software (Table of Materials) for analysis of D2O in water. The parameter settings for the spectrometer software used in this protocol are listed in Table 1.
- Collect a background sample using the diluent, 0.22 µm-filtered, distilled water. This should be the same water used for the standards.
- Inject 40 µL of the 0 ppm D2O and record the spectra. Save the spectra as a comma separated values (CSV) file.
- Continue to inject and save the spectra of all standards to create a standard curve.
- Repeat the background and standard curve every 60−90 min.
- Inject 40 µL of each distilled sample into the liquid transmission cell and save the spectra.
NOTE: Alter the injection volume of the standards and distilled samples based upon the volume of liquid transmission cell. Use a smaller volume liquid transmission cell if the sample volume is below 40 µL or dilute 1:1 with background distilled water. - Determine the concentration of D2O of each sample from the FTIR spectra using a spreadsheet program as described by Jennings et al.11 or the spectral software. When replicates are performed, use the average concentration to calculate the body composition.
5. Calculation of body composition
- Convert the deuterium enrichment (ppm) to atom percent concentration for each sample using the following equation12:
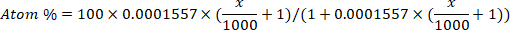
where x is the measured deuterium enrichment (ppm) of the sample and 0.0001557 is the mole fraction of deuterium reported in Vienna Standard Mean Ocean Water (VSMOW)13. - Calculate total body water for each sample using the following equation4,12,14:
 )
)
where E is the measured enrichment (atom%) of deuterium in the sample after background correction, B is the injection mass in g, and 0.998 is the concentration of injected D2O.
NOTE: Deuterium exchange with labile hydrogen causes a 2% overestimation of total body water mass. Total body water should be corrected by reducing the total body water mass estimate by 2% of the body weight. - Estimate the fat-free mass (lean body mass and all other non-fat components) of each bat using the following equation:
NOTE: Use the conventionally accepted value of 0.732 for the fractional moisture content of lean body mass for healthy, euhydrated, non-lactating bats. The fractional moisture content of fat-free mass can change in lactating big browns based upon the post-partum week15. For other species, use the values published in the literature or determine the fractional moisture content of lean body mass prior to performing calculations of the lean body mass. - Estimate the body fat mass using the following equation:
- Convert the body fat mass in g to percent body fat mass using the following equation:
6. Determination of water composition in a carnivore (Felis catus, domestic cat)
- Prepare the stock solution as described in section 1.
- Weigh each cat to the nearest 3 decimal places and record weight. Calculate the dose for each cat as described in step 3.6 using a D2O dose of 0.70 g/kg.
- Prepare each dose as described in steps 3.7−3.8. using a 3 mL or 5 mL syringe with a 22 G needle instead of an insulin syringe.
- Collect 500 µL of whole blood and subsequently administer subcutaneously the 0.7 g/kg D2O. Centrifuge whole blood at 2,000 x g for 15 min and store plasma in 1.5 mL microcentrifuge screw top tubes at -20°C until analysis.
- Collect 500 µL of whole blood 4 h post-injection. Centrifuge whole blood at 2,000 x g for 15 min and store plasma in 1.5 mL microcentrifuge screw top tubes at -20 °C until analysis.
- Collect 500 µL of whole blood 14 days post-injection. Centrifuge whole blood at 2,000 x g for 15 min and store plasma in 1.5 mL microcentrifuge screw top tubes at -20 °C until analysis.
NOTE: The number of days between blood collection can be based upon the experimental needs and the post-injection period in which D2O can be detected above the background levels. Fourteen days was the length of the dietary treatment blocks from Hooper et al.9. - Perform FT-IR analysis according to section 4 and calculate the body composition according to section 5 of this protocol.
- Calculate the water consumption in mL/day using the following equations:
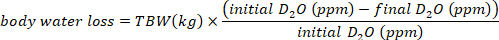
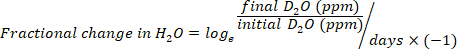
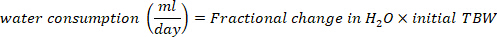
where TBW is total body water, initial D2O and final D2O are the concentrations measured in ppm in the post-injection D2O samples.
Using Deuterium Oxide as a Non-Invasive, Non-Lethal Tool for Assessing Body Composition and Water Consumption in Mammals
Learning Objectives
The deuterium oxide dilution technique can be used to assess the body composition of a variety of species. To demonstrate the adaptability, we are reporting the first use of the deuterium oxide dilution technique in a North American insectivorous bat species, Eptesicus fuscus, the big brown bat for representative results. A timing plateau was completed by taking pre- and post-D2O injection blood samples as should be done with any species where the equilibration period is unknown. It was determined that two hours post-injection in non-torpid bats was adequate for equilibration. With the equilibration time known, the total body water, lean body mass, and body fat mass for 13 wild-caught big brown bats and 8 captive big brown bats were determined (Table 2). An additional 2 wild-caught big brown bats and 5 captive big brown bats were determined to have a negative body fat mass. A negative body fat mass is calculated due to one or more of the following reasons: not receiving the entire dose of deuterium oxide, becoming torpid during the equilibration phase, having abnormally large fat masses and minimal lean mass, or bats having under 3%−5% body fat as determine by DXA (Table 3).
White-nose syndrome has caused many bat species to decline, so the technique was compared to the body fat measured using DXA. Figure 1 shows the percentage of body fat determined by the D2O dilution technique and DXA (n = 19). The two techniques were well correlated with a Pearson's r = 0.897 (Figure 2) and were not statistically different (one-way analysis of variance (ANOVA), F-value = 0.366, p = 0.549). The body fat showed strong correlations between body fat and body weight (Figure 3). The D2O dilution technique did not consistently over or underestimate the body fat mass.
The deuterium oxide method has been previously validated in cats16. Table 4 shows an example of the total body water, lean body mass, and body fat mass of a single cat9. Hooper et al.9 was the first to report the use of deuterium oxide dilution to measure the water consumption of socially housed animals with the daily water consumption of the cats during each dietary block of the experiment, as shown in Figure 4.
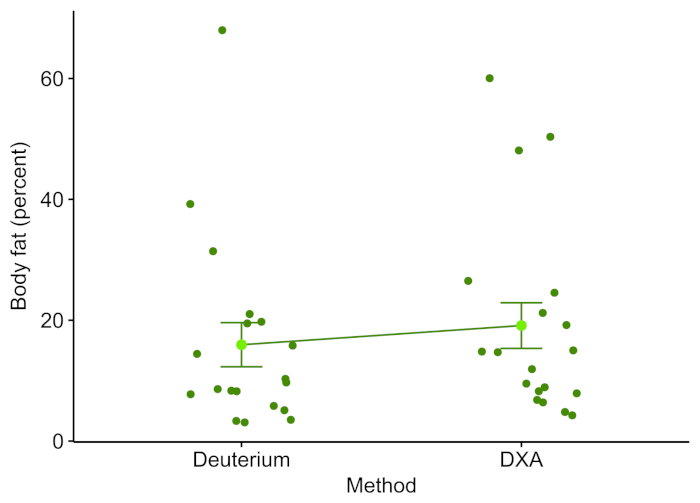
Figure 1: Deuterium oxide and DXA line plot. Each point represents the body fat percentage of an individual bat as determined by DXA or deuterium oxide. The mean is the light green point with error bars indicating the standard error of the mean. Please click here to view a larger version of this figure.
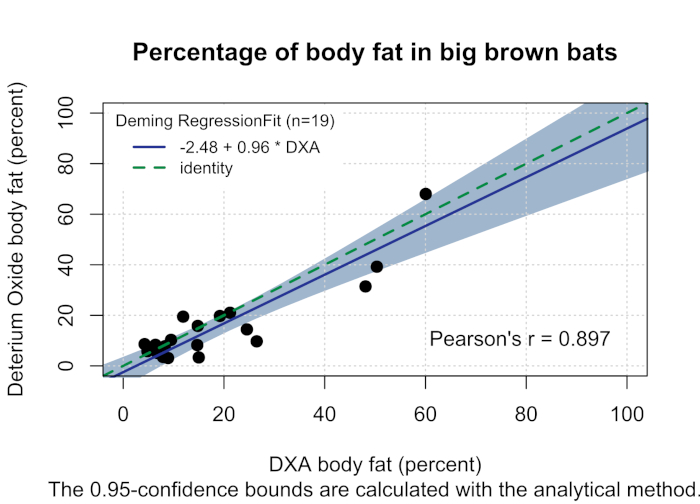
Figure 2: Percentage of body fat in big brown bats. Deming regression (solid blue line, Pearson's r = 0.897) comparing the percentage of body fat determined by DXA (x-axis, the reference method) and the percentage of body fat determined by deuterium oxide (y-axis, the test method) in big brown bats with 95% confidence intervals designated by gray shading. The green dash identity line drawn represents the regression line when the methods are equal. Please click here to view a larger version of this figure.
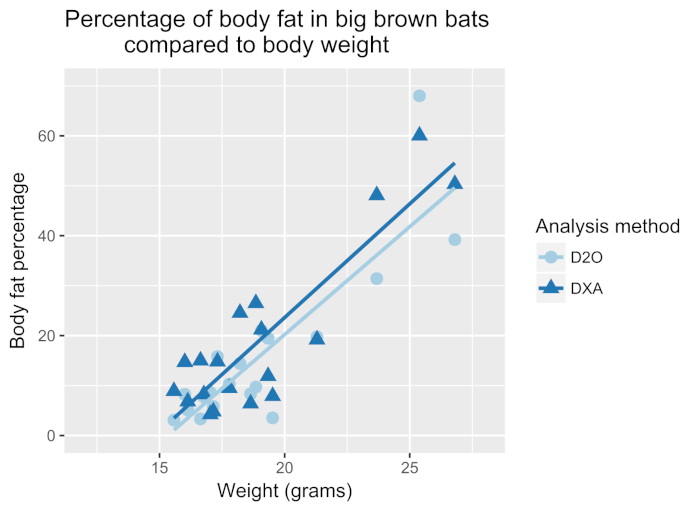
Figure 3: Percentage of body fat in big brown bats compared to body weight. Body weight of each bat plotted against the body fat percentage determined by D2O or DXA. A strong correlation exists between the body weight and body fat as determined by DXA (dark blue line, Pearson's r = 0.88) and D2O (blue line, Pearson's r = 0.86). Please click here to view a larger version of this figure.
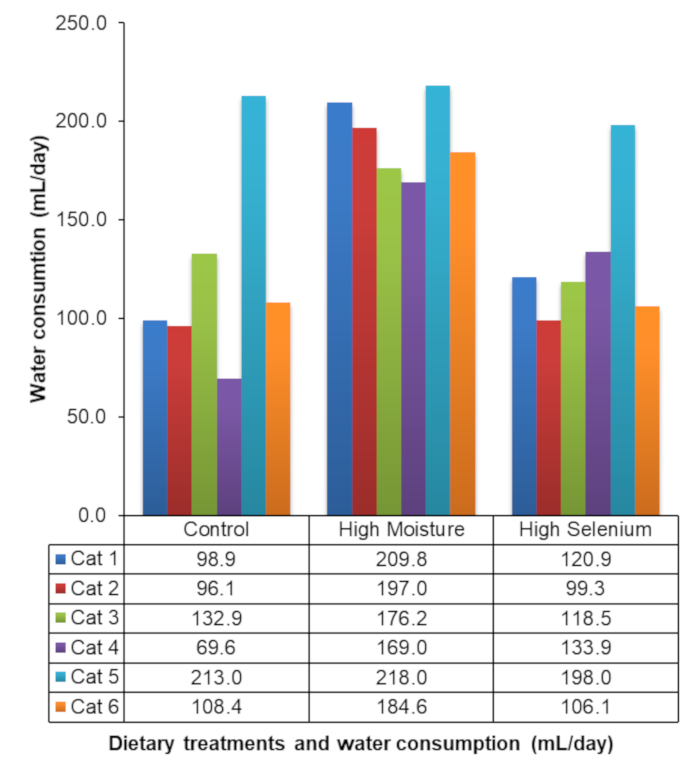
Figure 4: Water consumption of socially housed cats. Representative results of the daily water consumption of socially housed cats during an experiment evaluating the effects of dietary constituents on water consumption. This figure has been modified from Hooper et al.9. Please click here to view a larger version of this figure.
| Parameter | Setting |
| Number of scans | 64 |
| Resolution | 2 |
| Data spacing | 0.946 cm-1 |
| Final format | Absorbance |
| Correction | None |
| Use fixed Y-axis limits in collection window | Min -0.01, Max 0.03 |
| Bench range | Max 6.38, Min -5.02, Loc 1024 |
| Total absorbing peak sensitivity | 50 |
| fringes or channeling sensitivity | 80 |
| Derivative peaks sensativity | 51 |
| Baseline error sensitivity | 50 |
| CO2 levels sensitivity | 19 |
| H2O levels sensitivity | 19 |
| Apodization mode | Happ-Genzel |
| Phase correction | Mertz |
| Filters set based upon | velocity |
| low pass filter | 11,000 |
| high pass filter | 20 |
Table 1: Spectral software settings. Parameter settings used for spectral recording software.
| Animal | Species | Body weight (kg) |
D2O injected (g) |
Total body water (g) |
Lean body mass (g) |
Body fat mass (g) |
Body fat mass (%) |
DXA lean + bmc (g) |
DXA fat (g) |
DXA fat (%) |
| 1 | Eptesicus fuscus | 0.01715 | 0.0740 | 11.80 | 16.15 | 1.00 | 5.80 | 14.65 | 0.75 | 4.80 |
| 2 | Eptesicus fuscus | 0.01950 | 0.0920 | 13.80 | 18.83 | 0.69 | 3.50 | 16.20 | 1.40 | 7.90 |
| 3 | Eptesicus fuscus | 0.01677 | 0.08 | 11.33 | 15.47 | 1.30 | 7.74 | 11.33 | 1.30 | 7.74 |
| 4 | Eptesicus fuscus | 0.02129 | 0.097 | 12.51 | 17.09 | 4.20 | 19.7 | 15.9 | 19.65 | 19.2 |
Table 2: Body composition of big brown bats. The representative results of total body water, lean body mass, and body fat as determined by deuterium oxide dilution in big brown bats are shown in columns 5−8. Representative results of the lean body mass plus bone mineral content and body fat as determined by DXA in the same big brown bats are shown in columns 9−11.
| Animal | Species | Body weight (kg) |
D2O injected (g) |
Total body water (g) |
Lean body mass (g) |
Body fat mass (g) |
Body fat mass (%) |
DXA lean + bmc (g) |
DXA fat (g) |
DXA fat (%) |
Comment |
| 1 | Eptesicus fuscus | 0.0277 | 0.1299 | 34.18 | 46.69 | -19.02 | -68.74 | 9.90 | 26.55 | 62.80 | Equili-bration time insufficient |
| 2 | Eptesicus fuscus | 0.0185 | 0.0810388 | 64.23 | 87.75 | -69.25 | -374.33 | 14.20 | 17.30 | 17.95 | Full dose not injected |
| 3 | Eptesicus fuscus | 0.0164 | 0.0719 | 17.38 | 23.74 | -7.33 | -44.68 | 14.15 | 14.40 | 1.70 | Less than 3% fat |
| 4 | Eptesicus fuscus | 0.0212 | 0.0994 | 54.57 | 74.54 | -53.37 | -252.0 | 16.41 | 19.01 | 13.65 | Bat became torpid (cool to touch) |
Table 3: Body composition of big brown bats. Representative results from bats that did not receive the entire dose of deuterium oxide, became torpid during the equilibration phase, bats with abnormally large fat mass and minimal lean mass, or bats under 3%−5% body fat as determine by DXA. The representative results of total body water, lean body mass, and body fat as determined by deuterium oxide dilution are shown in columns 5−8. Representative results of the lean body mass plus bone mineral content and body fat as determined by DXA are shown in columns 9−11.
| Block | Species | Body weight (kg) |
D2O injected (g) |
Total body water (kg) |
Lean body mass (kg) |
Body fat mass (kg) |
Body fat mass (%) |
Daily water consumption (mL/day) |
Dietary Treatment |
| 1 | Felis Catus | 4.830 | 3.36 | 2.69 | 3.68 | 1.149 | 23.8 | 96.8 | Control |
| 2 | Felis Catus | 4.764 | 3.45 | 2.66 | 3.63 | 1.136 | 23.8 | 217.5 | High Moisture |
| 3 | Felis Catus | 4.727 | 3.25 | 2.50 | 3.41 | 1.314 | 27.8 | 125.1 | High Selenium |
Table 4: Body composition and water consumption in a single feline. Representative results of deuterium oxide dilutional technique for assessing the lean body mass, fat mass, and water consumption of one cat at three different time points during the study conducted by Hooper et al.9.
List of Materials
| 0.2 micron non-pyrogenic disk filter | Argos Technologies | FN32S | nylon, 30mm diameter, 0.22um, sterile |
| 1.5 mL conical microcentrifuge tubes | USA Scientific | 1415-9701 | 1.5 ml self-standing microcentrifuge tube, natural with blue cap |
| 10 mL sterile glass vial for injection | Mountainside Medical Equipment | MS-SEV10 | clear, sterile glass injection unit |
| 10 mL syringe | Becton Dickinson | 305219 | sterile 10 mL syringe individually wrapped |
| 100 mL sterile glass vial for injection | Mountainside Medical Equipment | AL-SV10020 | clear, sterile glass injection unit |
| 20 gauge needle | Exel | 26417 | needles hypodermic 20g x 1" plastic hub (yellow) / regular bevel |
| 22 gauge needle | Exel | 26411 | needles hypodermic 22g x 1" plastic hub (black) / regular bevel |
| deuterium oxide | Sigma-Aldrich | 151882-25G | 99.9 atom % D |
| isofluorane | Vetone | 3060 | fluriso isoflurane, USP |
| OMNIC Spectra Software | ThermoFisher Scientific | 833-036200 | FT-IR standard software |
| petroleum jelly | Vaseline | 305212311006 | Vaseline, 100% pure petroleum jelly, original, skin protectant |
| plastic capillary tubes | Innovative Med Tech | 100050 | sodium heparin anticoagulant, 50 μL capacity, 30 mm length |
| Sealed liquid spectrophotometer SL-3 FTIR CAF2 Cell | International Crystal Laboratory | 0005D-875 | 0.05 mm Pathlength |
| sodium chloride | EMD Millipore | 1.37017 | suitable for biopharmaceutical production |
| Thermo Electron Nicolet 380 FT-IR Spectrometer | ThermoFisher Scientific | 269-169400 | discontinued model, newer models available |
Lab Prep
Body condition scoring systems and body condition indices are common techniques used for assessing the health status or fitness of a species. Body condition scoring systems are evaluator dependent and have the potential to be highly subjective. Body condition indices can be confounded by foraging, the effects of body weight, as well as statistical and inferential problems. An alternative to body condition scoring systems and body condition indices is using a stable isotope such as deuterium oxide to determine body composition. The deuterium oxide dilution method is a repeatable, quantitative technique used to estimate body composition in humans, wildlife, and domestic species. Additionally, the deuterium oxide dilution technique can be used to determine the water consumption of an individual animal. Here, we describe the adaption of the deuterium oxide dilution technique for assessing body composition in big brown bats (Eptesicus fuscus) and for assessing water consumption in cats (Felis catis).
Body condition scoring systems and body condition indices are common techniques used for assessing the health status or fitness of a species. Body condition scoring systems are evaluator dependent and have the potential to be highly subjective. Body condition indices can be confounded by foraging, the effects of body weight, as well as statistical and inferential problems. An alternative to body condition scoring systems and body condition indices is using a stable isotope such as deuterium oxide to determine body composition. The deuterium oxide dilution method is a repeatable, quantitative technique used to estimate body composition in humans, wildlife, and domestic species. Additionally, the deuterium oxide dilution technique can be used to determine the water consumption of an individual animal. Here, we describe the adaption of the deuterium oxide dilution technique for assessing body composition in big brown bats (Eptesicus fuscus) and for assessing water consumption in cats (Felis catis).
Procedure
Body condition scoring systems and body condition indices are common techniques used for assessing the health status or fitness of a species. Body condition scoring systems are evaluator dependent and have the potential to be highly subjective. Body condition indices can be confounded by foraging, the effects of body weight, as well as statistical and inferential problems. An alternative to body condition scoring systems and body condition indices is using a stable isotope such as deuterium oxide to determine body composition. The deuterium oxide dilution method is a repeatable, quantitative technique used to estimate body composition in humans, wildlife, and domestic species. Additionally, the deuterium oxide dilution technique can be used to determine the water consumption of an individual animal. Here, we describe the adaption of the deuterium oxide dilution technique for assessing body composition in big brown bats (Eptesicus fuscus) and for assessing water consumption in cats (Felis catis).
