Spatial and Temporal Control of Murine Melanoma Initiation from Mutant Melanocyte Stem Cells
Instructor Prep
concepts
Student Protocol
All animal procedures are performed in accordance with Cornell University Institutional Animal Care and Use Committee (IACUC).
1. Preparation
- Collect tail clips from 12 day postnatal mice and digest in 400 µL of 0.05 N NaOH at 95 °C for 1 h. Vortex and add 32 µL of 1 M Tris-HCl, pH 7. Genotype mice according to PCR protocols provided through the Jackson Laboratory and identify mice with genotype of interest6.
– Tyr-CreER – hemizygous (+/CreER)
– LSL-BrafV600E – heterozygous (+/LSL-V600E)
– Pten – homozygous flox (flox/flox)
– LSL-tdTomato – hemizygous (+/LSL-tdTomato)
NOTE: Primer sequences are provided in Table of Materials. - Use a hair clipper (electric trimmer) to shave the dorsal skin 2 to 3 days before all experiments described below, and when mice are approximately 7 weeks of age. Take care not to damage the thin mouse skin using the electric trimmer as any injury may elicit a wound healing response which will activate hair follicle stem cells, including MCSCs.
- Determine if the hair cycle is in telogen and whether the skin is wounded during the waiting period. If the skin shows any sign of injury, the mouse should not be used for these procedures.
NOTE: Although the hair cycle may slightly differ between genetic backgrounds, at this age the hair cycle in the dorsal skin is usually in the telogen state11. The Tyr-CreER transgene will target MCSCs in telogen skin, and the subsequent genetic alterations will be permanently expressed in all lineages of the MCSC, including differentiated melanocytes and melanocytic tumors.
2. Tamoxifen Treatment for Cre-Lox genetic Recombination
- Preparation of tamoxifen for intraperitoneal injection (IP) or topical administration for systemic or localized genetic recombination
NOTE: In order for tamoxifen to induce recombination, it must first be metabolized by the liver to 4-hydroxytamoxifen (4-OHT), which can bind to and activate the CreER. Topical application of tamoxifen will not be metabolized in this way and 4-OHT must directly be used. Only one method of tamoxifen delivery is required for sufficient recombination.- Tamoxifen: Prepare tamoxifen at a concentration of 10 mg mL-1 dissolved in corn oil. To aid in the dissolution of the drug into solution, add up to 10% total volume of 100% molecular grade ethanol to the drug in powder form, vortex for 3 min and then add remaining volume of corn oil. Continue to vortex until crystalline material is no longer apparent in solution. Tamoxifen solution can be sterilized by passing through a 0.2 µm filter. Proceed to step 2.2.1 for treatment.
- 4-OHT: Prepare a stock solution of hydroxytamoxifen up to 3 mg mL-1 in 100% ethanol. Then, a working solution can be applied as much as needed to cover the skin area of interest (generally a single application).
NOTE: A stock solution of hydroxytamoxifen can be dissolved in 100% ethanol for a final concentration of 5 mM. Then, to prepare a working solution, 5 µL of 4OH-tamoxifen stock solution can be diluted into 15 µL of 100% ethanol. For 4 mm2 area, 1 to 3 µL of working solution can be topically applied.
- Treating mice with tamoxifen either systemically or topically
- For systemic treatment, administer 2 mg of tamoxifen via IP injection once daily for 3 days using a 26 G needle.
NOTE: Using this model, approximately 93% ± 4% of quiescent MCSCs are labeled with tdTomato6. - For regional activation, perform one topical treatment with 4-OHT. Cover the skin area of interest with the working solution. The amount of working solution can be determined by the size of region of interest as described in step 2.1.2.
- For systemic treatment, administer 2 mg of tamoxifen via IP injection once daily for 3 days using a 26 G needle.
- At 48 h following the last dose of tamoxifen, proceed to step 3 for chemical depilation induced tumor initiation or to step 4 for UV-B induced tumor initiation
3. Chemical Depilation
- Equip a mouse treatment room containing an isoflurane system with the following required materials: hair removal cream, cottons swabs, paper cloth, and water.
- Place the mouse into the induction chamber and anesthetize with 3.5 to 4.5% isoflurane and 1 L/min oxygen.
- Once the respiratory rate has stabilized, confirm that the mouse is in the proper plane of anesthesia by performing a toe pinch and transfer the mouse to the main tube maintaining anesthesia with 1 to 1.5% isoflurane and 1 L/min oxygen. Apply eye ointment to keep the eyes from drying while under anesthesia.
- Use a cotton swab to apply a thin layer of hair removal cream to the shaved region of the mouse skin.
- Once the cream has been evenly applied, use clean cotton swabs to begin removal. The total time that the cream is in contact with the skin should not exceed 1 min.
- Gently wipe the skin with a damp paper cloth and ensure that any residual cream has been removed.
NOTE: Hair removal cream can cause irritation to the skin if it is not completely removed. Some mice, such as those on a C57BL/6 background, can be prone to developing dermatitis12. Although rare, some animals will develop dermatitis within 24 h after hair removal cream application or mechanical depilation. Make sure to check the mice the day after treatment to look for any signs of regional skin irritation. - Discard the removed hair shafts and any materials used into a biohazard bin.
- Place the mouse into an empty, clean mouse cage until the anesthesia has worn off.
- Return the mice to their cages.
4. UV-B Irradiation
- Equip a mouse treatment room containing an isoflurane system with the following required materials: UV-B light system, UV-B light meter, protective covering for the mouse, additional UV-B protective PPE for researcher, and a timer.
- Mice should be irradiated at the UV-B dose of interest (i.e., 0-180 mJ/cm2). Use the UV-B light meter to determine the appropriate length of time to irradiate the mice.
NOTE: mW/cm2 x time = dose - Anesthetize the mouse with isoflurane. While in the induction chamber, use 3.5 to 4.5% isoflurane and 1 L/min oxygen.
- Once the mouse is stabilized, confirm anesthesia effects with a toe pinch and transfer the mouse to the main anesthesia tube located in UV chamber. Apply eye ointment to prevent the eyes from drying while under anesthesia. Maintain anesthesia with 1 to 1.5% isoflurane and 1 L/min oxygen.
- Cover the dorsal skin with UV-B protective material so that only the desired region will be exposed.
NOTE: At this time, appropriate PPE should be utilized. The length of time needed to irradiate the mouse is dependent on the intensity of the light bulb used, but anesthesia may need to be monitored if the required time exceeds 30 s. - Cover the UV chamber using UV-resistant lid or any other suitable materials.
- Turn on the UV-B lamp and irradiate the mouse for the time determined by researchers for the specific aim.
- Turn off the UV system and isoflurane, then place the mouse to an empty mouse cage until the anesthesia has worn off.
- Return the mice to their cages.
5. Tissue Processing
- Skin isolation
- Obtain the following prior to starting: necropsy instruments, filter paper and paper towels.
- Euthanize the mice according to IACUC approved protocols, and confirm death prior to proceeding.
- Using a hair clipper, shave any hair from the dorsal skin area. Lightly brush the area with a lint free tissue to clear the area.
- Make a small incision with sharp scissors just above the base of the tail.
- Insert large blunt scissors into the incision and separate the subdermal connective tissues.
- Carefully isolate the skin region of interest by cutting with sharp scissors along the edge of the tissue.
- Place the skin tissue onto a clean paper towel and use dull forceps to stretch the skin so that it adheres to the towel and is taught. This step serves to provide additional support for the tissue so that it can be manipulated and processed while maintaining its shape.
NOTE: Be careful to only handle the outside regions of the skin tissue, as the forceps may cause damage to the skin which can be seen histologically.
- Tissue fixation
- Fold a piece of filter paper in half and label appropriately. Place the skin along with paper towel backing into the folded paper immediately adjacent to the crease, ensuring that the sample is smooth and not folded or crumpled. Seal the filter paper by stapling the open sides, and then trim so that the remaining paper can be used for future samples.
NOTE: This step is performed so that the skin retains its shape during fixation. - Fully submerge the tissue sample in 10% neutral buffered formalin for 3 to 5 h at room temperature or overnight at 4 °C.
- Following fixation, remove the samples from formalin, wash in deionized water (2x, 5 min each) and proceed to make tissue blocks. Carefully remove any residual material from the skin tissue (i.e., residual paper).
- Fold a piece of filter paper in half and label appropriately. Place the skin along with paper towel backing into the folded paper immediately adjacent to the crease, ensuring that the sample is smooth and not folded or crumpled. Seal the filter paper by stapling the open sides, and then trim so that the remaining paper can be used for future samples.
- Making frozen tissue blocks (Figure 1)
- Trim the edges of the skin sample so that they are not jagged and then make 3 sagittal cuts. The width of these strips should not be more than the height of the cryomold (5 mm). Next, make transverse cuts to have pieces of skin which are not longer than the width of the cryomold (20 mm) and can be embedded. Repeat with the remaining half of dorsal skin, or save the tissue for other uses (alternately, save half prior to fixation).
NOTE: It’s important to keep skin pieces in proper orientation. - Orient the cryomold (25 x 20 x 5 mm3) in a portrait orientation and place 4 to 5 pieces of skin on top of the OCT. Once all pieces are in place, use 2 pairs of fine forceps to bring the long edge of skin to the bottom of the cryomold. Skin should now be standing up in the OCT perpendicular to the base of the mold. Take care to minimize formation of air pockets within the OCT during this step (Figure 1).
- Fill the mold with OCT to the second lip, and place on the flat surface of dry ice. Use forceps to adjust any skin pieces that may have shifted during transfer as the block begins to freeze. Once the bottom layer is solidified, manipulation of tissues will be impossible.
- Cut 8-10 µm slides for analysis.
- Trim the edges of the skin sample so that they are not jagged and then make 3 sagittal cuts. The width of these strips should not be more than the height of the cryomold (5 mm). Next, make transverse cuts to have pieces of skin which are not longer than the width of the cryomold (20 mm) and can be embedded. Repeat with the remaining half of dorsal skin, or save the tissue for other uses (alternately, save half prior to fixation).
- Making Formalin Fixed Paraffin Embedded (FFPE) tissue blocks
- Place 2 pieces of fixed skin into a labeled cassette and dehydrate in increasing concentrations of ethanol (75% ethanol for 30 min, 85% for 30 min, 90% for 30 min, 95% for 30 min, 100% for 2x 30 min, Xylene or xylene substitute for 2x 30 min). Incubate with Paraffin at 58-60˚, first for 30 min, and then overnight.
- Embed the tissue in a 24 x 24 mm2 stainless steel tissue mold with liquid paraffin and solidify at -5 °C. The tissue orientation should be similar to that of frozen tissue blocks so that longitudinal sections across multiple hair follicles can be visualized (Figure 1).
- Once the paraffin has solidified, remove the mold and cut into 5-10 μm sections for analysis.
Spatial and Temporal Control of Murine Melanoma Initiation from Mutant Melanocyte Stem Cells
Learning Objectives
Cutaneous melanoma initiation induced by chemical depilation
The procedure of chemical depilation is depicted in Figure 2. When mice are 7-weeks postnatal, their dorsal skin is in telogen. During telogen, hair follicle stem cells and MCSCs are known to be in a quiescent, resting state. The skin should show no significant hair growth after shaving. On the other hand, chemical depilation can induce hair follicle stem cell activation, which in turn shows significant hair growth from the region of interest (Figure 2). Similarly, tumor-prone MCSCs expressing oncogenic mutations also need to be active for accelerated tumor formation. Chemical depilation can significantly induce the activation of both hair follicle stem cell and MCSCs. Melanoma-prone MCSCs in an active state can form tumors, and microscopic melanoma initiation can be observed within 2 weeks after chemical depilation using the lineage tracing allele LSL-tdTomato (Figure 3).
Cutaneous melanoma initiation induced by UV-B irradiation
Similar to chemical depilation, UV-B can induce tumor initiation from quiescent tumor-prone MCSCs (Figure 4). While the murine skin containing tumor-prone MCSCs in a quiescent state shows no significant tumor initiation and lack of significant pigmentation, the dorsal skin exposed to UV-B (twice, 180 mJ/cm2) shows black pigmented early melanocytic tumors which are macroscopically evident (Figure 4A, B). While UV-B can significantly induce macroscopically evident tumor initiation within 2 weeks, the application of sunscreen can protect UV-B-mediated melanoma initiation in the skin, similar to a UV-resistant cloth (Figure 4C, D).
Importantly, UV-B induces the direct translocation of MCSCs from their follicular stem cell niche to the interfollicular epidermis. Furthermore, UV-B can induce MCSC-originating cutaneous melanoma formation throughout the interfollicular epidermis, and the microscopic phenotype can be observed by the lineage tracing marker, tdTomato (Figure 5). As these tumor cells are malignant, they grow rapidly and invasively (Figure 5). Similar to the dorsal skin, UV-B-induced melanoma initiation can be notably observed in other skin areas, such as the ear skin (Figure 6). Compared to the control skin covered by UV-resistant cloth, the ear skin exposed to UV-B demonstrates higher pigmentation due to a higher burden of melanoma initiation (Figure 6).
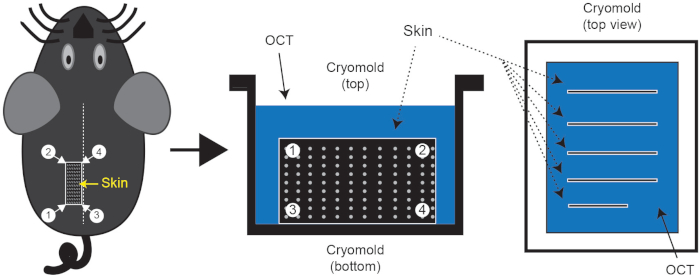
Figure 1: Skin tissue isolation and cryo-embedding. Following euthanasia, shave the mouse dorsal skin region of interest and embed skin in cryomold containing OCT. The dotted line on the mouse represents the midline. Typically, 3 or 4 pieces of skin can be isolated with line segment 3-4 along the midline; one such piece is indicated by rectangle 1/2/3/4. These skin pieces should be embedded in the OCT as shown in the figure. Please click here to view a larger version of this figure.

Figure 2: Macroscopic phenotypes after chemical depilation. (A) Diagram of chemical depilation. (B) After shaving, the hair cycle was confirmed to be in telogen. (C) Chemical depilation was performed to remove hair shafts from the hair follicles, which can activate both hair follicle and melanocyte stem cells. (D) Around a week later, early hair growth will be easily noticeable. (E) Then, significant hair growth can be observed over time. Please click here to view a larger version of this figure.
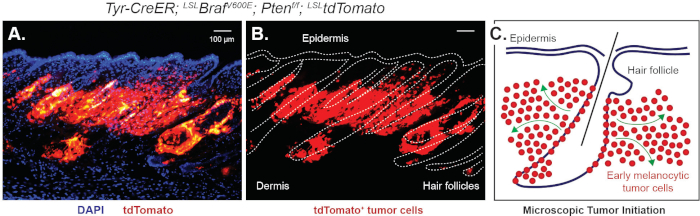
Figure 3: Microscopic observation of melanoma initiation controlled by chemical depilation. Chemical depilation can significantly induce the activation of quiescent tumor-prone MCSCs. Then, microscopic tumor initiation demonstrated by the lineage tracing marker tdTomato can be observed within 2 weeks. This figure demonstrates the microscopic phenotype 16 days post 3 days tamoxifen by IP injection followed by chemical depilation using different visualizations of the same field of view. (A) tdTomato+ tumor cells originating from the hair follicle merged with Dapi nuclear counter stain. (B) Hair follicles and interfollicular epidermis are outlined in white. (C) A schematic showing how tdTomato+ tumor cells invade into the dermis from the hair follicle, modified from Moon, H. et al.6. Please click here to view a larger version of this figure.
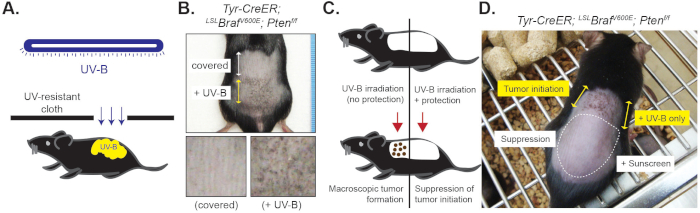
Figure 4: Macroscopic phenotype of cutaneous melanoma induced by UV-B irradiation. Mice were treated by tamoxifen for 3 days (day 1 to 3) followed by 2 UV-B exposures (day 4 and 6). (A) Diagram of UV-B irradiation on the dorsal skin containing mutant MCSCs. (B) Macroscopic phenotype between control and UV-B exposed dorsal skin. Significant black pigmentation related to melanocytic tumor formation can be observed within 2 weeks after the second UV-B irradiation. (C) Diagram of sunscreen application. (D) While UV-B can significantly induce tumor initiation from mutant MCSCs, application of sunscreen showed significantly suppressed UV-B-mediated tumor initiation (16 days post the second UV-B irradiation). Please click here to view a larger version of this figure.
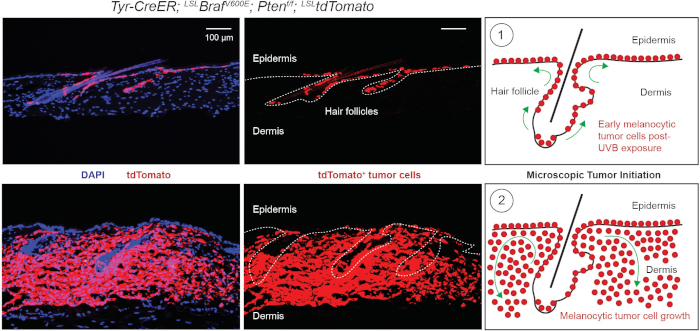
Figure 5: Microscopic observation of UV-B-induced cutaneous melanoma. Mice were treated with tamoxifen by IP injection for 3 days (day 1 to 3) followed by 2 UV-B exposures (day 4 and 5). (A-C) Early tumor initiation throughout the interfollicular epidermis demonstrated by lineage tracing tdTomato can be shown at day 17. (A) tdTomato+ tumor cells are shown migrating from the hair follicle into the interfollicular epidermis. Dapi is used as a nuclear counter stain. (B) Dapi staining is removed and dotted line indicates location of the hair follicles and interfollicular epidermis. (C) Schematic indicating tdTomato+ cell migration from the hair germ to the interfollicular epidermis following UV-B exposure. Modified from Moon et al.6. (D-F) Then, tumor cells grow rapidly and invade the dermal tissues below (day 25). (D) tdTomato+ tumor cells have continued to proliferate and invade into the dermal tissues. Dapi is used as a nuclear counter stain. (E) Dapi staining is removed and dotted line indicates the location of the hair follicles and interfollicular epidermis. (F) Schematic indicating tdTomato+ cell invasion into the dermis and melanocytic tumor growth. Modified from Moon et al.6. Please click here to view a larger version of this figure.
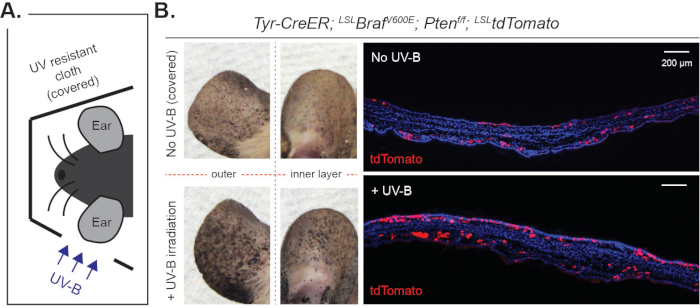
Figure 6: Macroscopic and microscopic observation of UV-B-induced melanoma in ear skin. (A) Experimental scheme. Mice were treated by tamoxifen for 3 days (day 1 to 3) followed by 3 exposures to UV-B irradiation (day 4, 6 and 8). (B) Significantly increased melanoma initiation by UV-B irradiation was observed macroscopically as well as microscopically at day 28. Please click here to view a larger version of this figure.
List of Materials
| Tamoxifen | Sigma | T5648-1G | For systemic injection |
| Tamoxifen | Cayman Chemical | 13258 | For systemic injection |
| Corn oil | Sigma | 45-C8267-2.5L-EA | |
| 4OH-tamoxifen | Sigma | H7904-25MG | For topical treatment |
| 26g 1/2" needles | various | Veterinary grade | |
| 1 mL syringe | various | Veterinary grade | |
| Pet hair trimmer | Wahl | 09990-502 | |
| Hair removal cream | Nair | n/a | Available at most drug stores |
| Cotton swabs | various | ||
| Ultraviolet light bulb | UVP | 95-0042-08 | model XX-15M midrange UV lamp |
| 200 proof ethanol | various | pure ethanol | |
| Histoplast PE | Fisher Scientific | 22900700 | paraffin pellets |
| Neutral Buffered Formalin, 10% | Sigma | HT501128-4L | |
| Clear-Rite 3 | Thermo Scientific | 6901 | xylene substitute |
| O.C.T. Compound | Thermo | 23730571 | |
| Tissue Cassette | Sakura | 89199-430 | for FFPE processing |
| Cryomolds | Sakura | 4557 | 25 x 20 mm |
| FFPE metal mold | Leica | 3803082 | 24 x 24 mm |
| Isoflurane | various | Veterinary grade | |
| Anesthesia inhalation system | various | Veterinary grade | |
| Fine scissor | FST | 14085-09 | Straight, sharp/sharp |
| Fine scissor | FST | 14558-09 | Straight, sharp/sharp |
| Metzenbaum | FST | 14018-13 | Straight, blunt/blunt |
| Forcep | FST | 11252-00 | Dumont #5 |
| Forcep | FST | 11018-12 | Micro-Adson |
| Tyr-CreER; LSL-BrafV600E; Pten-f/f | Jackson Labs | 13590 | |
| LSL-tdTomato | Jackson Labs | 007914 | ai14 |
| Cre-1 | n/a | GCATTACCGGTCGATGCAACGAGTGATGAG | |
| Cre-2 | n/a | GAGTGAACGAACCTGGTCGAAATCAGTGCG | |
| Braf-V600E-1 | n/a | TGAGTATTTTTGTGGCAACTGC | |
| Braf-V600E-2 | n/a | CTCTGCTGGGAAAGCGGC | |
| Kras-G12D-1 | n/a | AGCTAGCCACCATGGCTTGAGTAAGTCTGCA | |
| Kras-G12D-2 | n/a | CCTTTACAAGCGCACGCAGACTGTAGA | |
| Pten-1 | n/a | ACTCAAGGCAGGGATGAGC | |
| Pten-2 | n/a | AATCTAGGGCCTCTTGTGCC | |
| Pten-3 | n/a | GCTTGATATCGAATTCCTGCAGC | |
| tdTomato-1 | n/a | AAGGGAGCTGCAGTGGAGTA | |
| tdTomato-2 | n/a | CCGAAAATCTGTGGGAAGTC | |
| tdTomato-3 | n/a | GGCATTAAAGCAGCGTATCC | |
| tdTomato-4 | n/a | CTGTTCCTGTACGGCATGG |
Lab Prep
Cutaneous melanoma is well known as the most aggressive skin cancer. Although the risk factors and major genetic alterations continue to be documented with increasing depth, the incidence rate of cutaneous melanoma has shown a rapid and continuous increase during recent decades. In order to find effective preventative methods, it is important to understand the early steps of melanoma initiation in the skin. Previous data has demonstrated that follicular melanocyte stem cells (MCSCs) in the adult skin tissues can act as melanoma cells of origin when expressing oncogenic mutations and genetic alterations. Tumorigenesis arising from melanoma-prone MCSCs can be induced when MCSCs transition from a quiescent to active state. This transition in melanoma-prone MCSCs can be promoted by the modulation of either hair follicle stem cells' activity state or through extrinsic environmental factors such as ultraviolet-B (UV-B). These factors can be artificially manipulated in the laboratory by chemical depilation, which causes transition of hair follicle stem cells and MCSCs from a quiescent to active state, and by UV-B exposure using a benchtop light. These methods provide successful spatial and temporal control of cutaneous melanoma initiation in the murine dorsal skin. Therefore, these in vivo model systems will be valuable to define the early steps of cutaneous melanoma initiation and could be used to test potential methods for tumor prevention.
Cutaneous melanoma is well known as the most aggressive skin cancer. Although the risk factors and major genetic alterations continue to be documented with increasing depth, the incidence rate of cutaneous melanoma has shown a rapid and continuous increase during recent decades. In order to find effective preventative methods, it is important to understand the early steps of melanoma initiation in the skin. Previous data has demonstrated that follicular melanocyte stem cells (MCSCs) in the adult skin tissues can act as melanoma cells of origin when expressing oncogenic mutations and genetic alterations. Tumorigenesis arising from melanoma-prone MCSCs can be induced when MCSCs transition from a quiescent to active state. This transition in melanoma-prone MCSCs can be promoted by the modulation of either hair follicle stem cells' activity state or through extrinsic environmental factors such as ultraviolet-B (UV-B). These factors can be artificially manipulated in the laboratory by chemical depilation, which causes transition of hair follicle stem cells and MCSCs from a quiescent to active state, and by UV-B exposure using a benchtop light. These methods provide successful spatial and temporal control of cutaneous melanoma initiation in the murine dorsal skin. Therefore, these in vivo model systems will be valuable to define the early steps of cutaneous melanoma initiation and could be used to test potential methods for tumor prevention.
Procedure
Cutaneous melanoma is well known as the most aggressive skin cancer. Although the risk factors and major genetic alterations continue to be documented with increasing depth, the incidence rate of cutaneous melanoma has shown a rapid and continuous increase during recent decades. In order to find effective preventative methods, it is important to understand the early steps of melanoma initiation in the skin. Previous data has demonstrated that follicular melanocyte stem cells (MCSCs) in the adult skin tissues can act as melanoma cells of origin when expressing oncogenic mutations and genetic alterations. Tumorigenesis arising from melanoma-prone MCSCs can be induced when MCSCs transition from a quiescent to active state. This transition in melanoma-prone MCSCs can be promoted by the modulation of either hair follicle stem cells' activity state or through extrinsic environmental factors such as ultraviolet-B (UV-B). These factors can be artificially manipulated in the laboratory by chemical depilation, which causes transition of hair follicle stem cells and MCSCs from a quiescent to active state, and by UV-B exposure using a benchtop light. These methods provide successful spatial and temporal control of cutaneous melanoma initiation in the murine dorsal skin. Therefore, these in vivo model systems will be valuable to define the early steps of cutaneous melanoma initiation and could be used to test potential methods for tumor prevention.
