Cell Dissociation from the Tongue Epithelium and Mesenchyme/Connective Tissue of Embryonic-Day 12.5 and 8-Week-Old Mice
Instructor Prep
concepts
Student Protocol
Animal use (C57BL/6 mice throughout the study) was approved by the University of Georgia Institutional Animal Care and Use Committee and was in accordance with the National Institutes of Health Guidelines for care and use of animals for research.
1. Animal usage
NOTE: Mice were bred and maintained in the animal facility of the Animal and Dairy Science department at the University of Georgia at 22 °C under 12-h day/night cycles.
- Designate noon of the day of vaginal plug detection in mice as embryonic (E) day 0.5. Use embryos at E12.5 and postnatal mice at 8 weeks of age for the following experiments.
2. Preparation before experiment
NOTE: The instruments required for this protocol are listed in the Table of Materials.
- Autoclave instruments before the experiment. Sterilize instruments using a bead sterilizer during the experiment.
- Clean the surgical area, the dissecting microscope, and the biosafety cabinet using 70% ethanol wipes. Turn on the UV light of biosafety cabinet and keep it on for 20 min prior to the experimental procedure.
- Prepare an enzyme mixture of 1:1 dispase (5.0 mg/mL) and collagenase (2.0 mg/mL) to a final concentration of 2.5 and 1.0 mg/mL respectively, and filter the solution using 0.22 µm syringe filter7.
- Prepare 1 mL of enzyme mixture for an adult tongue or 0.5 mL for a specific region of tongue (e.g., posterior, or anterior tongue).
- Prepare 2 mL of enzyme mixture for embryonic tongues.
- Make 10 mL of 2.5% BSA in 0.1 M PBS and filter the solution using 1 mL syringe and 0.22 µm syringe filter.
- Make 500 µL of 5% FBS in DMEM/F12.
- Make 3 mL of DMEM/F12 containing 10% FBS and 1% BSA and filter the solution using 1 mL syringe and 0.22 µm syringe filter.
3. Separation of the tongue epithelium from the mesenchyme/underlying connective tissue
- Separation of the epithelium from the mesenchyme of an E12.5 mouse tongue
- Euthanize timed pregnant female mice carrying E12.5 embryos by placing it in a CO2 chamber followed by cervical dislocation.
NOTE: Mouse embryos at E12.5 are collected after 12 pm (afternoon) on the 12th day after detection of vaginal plug in the pregnant female mice. - Transfer mice to the surgical area. Wet the mouse abdomen using 70% ethanol to prevent fur from getting into the operating site.
- Open the abdomen using dissecting scissors to expose the uterine horns carrying embryos. Dissect the uterine horns using dissecting scissors and transfer it to 15 mL of fresh Tyrode's solution in a 100 mm culture dish.
- Dissect the embryos (Figure 1A1) out from uterine horns under a dissecting microscope using mini-scissors and fine forceps.
- Carefully open the mouth cavity wide by using fine forceps and dissect the tongue off from the mandible using mini-scissors (Figure 1A2).
- Wash the tongues using 15 mL of fresh sterile Tyrode's solution in a 100 mm culture dish.
- Transfer the tissues to 2 mL of enzyme mixture of dispase (2.5 mg/mL) and collagenase (1.0 mg/mL) in a 35 mm culture dish with a spatula and fine forceps in a biosafety cabinet. Incubate for 20 min at 37 °C.
- Transfer tongues to 15 mL of fresh sterile Tyrode's solution in a 100 mm culture dish and gently remove the mesenchyme from the epithelium from the ventral side using fine forceps.
NOTE: Epithelial sheets may be separated without mechanical force during the incubation. - Wash the separated epithelia and mesenchyme twice in 15 mL of fresh sterile Tyrode's solution in a 100 mm culture dish.
NOTE: The activities of dispase and collagenase will be inhibited by EDTA in the cell dissociation procedure (step 4.1).
- Euthanize timed pregnant female mice carrying E12.5 embryos by placing it in a CO2 chamber followed by cervical dislocation.
- Separation of the tongue epithelium from the underlying connective tissue of adult mice
- Euthanize the mouse at 8 weeks of age by placing it in a CO2 chamber. Confirm that the mouse is euthanized with no breaths and forepaw-pinch response.
- Transfer the mice to the surgical area. Wet the mouse head using 70% ethanol to prevent fur from getting into the oral cavity.
- Cut the corners of the mouth along the cheek using dissecting scissors to open the oral cavity. Dissect the tongue with mandible (Figure 1B1) and place it in a plastic dish with a layer of plastic wrap.
- Using surgical forceps to hold the tongue under a dissecting microscope, inject the enzyme mixture of dispase (2.5 mg/mL) and collagenase (1.0 mg/mL) in the sub-epithelial space of tongue through the cutting edge (Figure 1B1, arrows) of the posterior tongue.
- Inject 1 mL of enzyme mixture evenly to the whole tongue for tissue collection from both anterior and posterior tongue.
- Inject 0.5 mL of enzyme mixture locally to the anterior tongue for tissue collection from tongue tip or to the posterior tongue for circumvallate papilla tissue collection.
NOTE: The tongue will swell as the enzyme accumulates (Figure 1B2). Gentle injection of the enzyme can prevent pressure from damaging the epithelium and keep as much enzyme as possible in the tongue.
- Wrap the tongue with plastic wrap and incubate the tongue for 30 min at 37 °C.
- Use mini-scissors to dissect the tongue tip and/or circumvallate papilla, and use spatula and fine forceps to transfer tissue to 15 mL of fresh sterile Tyrode's solution in a 100 mm culture dish.
- Separate the epithelium from the underlying connective tissue in the enzyme-digested sub-epithelial space using mini-scissors. Trim the tissues to a proper size according to the requirement of downstream experiments.
- Wash the separated epithelium and underlying connective tissue twice in 15 mL of fresh sterile Tyrode's solution in a 100 mm culture dish.
NOTE: The activities of dispase and collagenase will be inhibited by EDTA in the cell dissociation procedure (step 4.1).
4. Cell dissociation
NOTE: The cell dissociation protocol described here can be applied to the tongue epithelium and mesenchyme/connective tissue in both E12.5 embryonic and 8-week-old mice. To reduce the cell loss during agitation and transfer of cell suspension, use commercial low retention pipette tips or pre-coated pipette tips with 2.5% BSA in 0.1 M PBS at pH 7.48.
- Transfer the tissues using spatula and fine forceps to 3 mL of 0.25% trypsin-EDTA in a new 35 mm culture dish for 30 min at 37 °C. Gently agitate tissues every 5 min with 1 mL pipette tips.
NOTE: Do not cut the pipette tip, as the cutting edge can physically damage the dissociated cells. - Add 500 µL of 5% FBS in DMEM/F12 to stop the reaction and transfer the medium to a 5 mL low binding centrifuge tube.
- Centrifuge cell suspension at 200 x g for 8 min at room temperature and remove the supernatant.
- Gently re-suspend cells in 3 mL of DMEM/F12 containing 10% FBS and 1% BSA using 1 mL pipette tips and filter the cells using a 70 µm cell strainer, followed by a 35 µm cell strainer.
- Centrifuge cell suspension at 200 x g for 8 min at room temperature. Remove most of the medium and leave 50-300 µL as the final volume to re-suspend cells.
NOTE: Adjust the concentration of cells according to requirements of downstream experiments by changing the final volume of single cell suspension.
5. Cell counting and viability test using hemocytometer
NOTE: To improve measurement accuracy, 3 technical replicates are recommended for each sample.
- Gently mix 5-10 µL of the single cell suspension with an equal volume of Trypan blue and add onto hemocytometer.
NOTE: Clean the hemocytometer thoroughly using 70% ethanol before use. Dusts particles on the hemocytometer will be stained in dark blue and affect the accuracy of viability test. Check the hemocytometer under a microscope. - Count the total cell number, the number of live cells (white), and the number of dead cells (dark blue) stained by Trypan blue (Figure 2, arrows) respectively in 4 squares with 16 grids (Figure 2) using inverted microscope with imaging system.
- Calculate the cell concentration:
Cell concentration =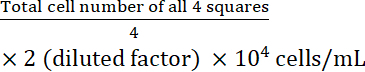
- Calculate the viability:
Viability =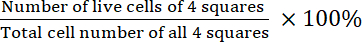
- Calculate the viability:
Cell Dissociation from the Tongue Epithelium and Mesenchyme/Connective Tissue of Embryonic-Day 12.5 and 8-Week-Old Mice
Learning Objectives
Separation of the tongue epithelium from the underlying mesenchyme/connective tissue
In the embryonic mouse tongue, a gap in the sub-epithelial space is visible after proper enzyme digestion. Epithelial sheets of some tongues are separated without mechanical force during the incubation.
In the adult mouse tongue, a successful enzyme injection is indicated by the swelling in the injected areas (Figure 1B2), which suggests that the enzyme can be held by the tongue. Insufficient enzyme and/or deep needle insertion to the mesenchyme and/or tongue epithelial penetration by needle will induce a partial swelling of the injection area or no swelling at all. After enzyme digestion, the underlying connective tissues with proper enzyme digestion become loose and sticky. A gap in the sub-epithelial space is visible when gently lifting the edge of the epithelial sheet.
Effect of cell dissociation on total cell number and viability
With step 4, E12.5 tongues, the epithelial sheets, and thin layers of mesenchyme immediately under the epithelium of tongues were pooled, respectively. Manual cell counting using a hemocytometer (Figure 2) demonstrated that the protocol yielded 63,917 cells in total with a viability of 95.2% from the epithelial sheets (around 0.3 mm2 in size per tongue) (Figure 1A3), and 294,333 cells in total with a viability of 96.3% from the mesenchyme (around 0.3 mm2 in size per tongue) (Figure 1A4).
Using 10 adult tongues at 8 weeks of age, the pieces of the epithelial sheets of the tongue tip (where taste buds are densely distributed), epithelial sheets of circumvallate papillae, and thin layers of connective tissue immediately under the epithelium of circumvallate papillae were pooled, respectively. A manual cell count using the hemocytometer (Figure 2) demonstrated that the protocol yielded 187,333 cells in total with a viability of 95.4% from epithelial sheets of tongue tip (around 0.075 mm2 in size per tongue) (Figure 1B3), 544,000 cells in total with a viability of 96.3% from epithelial sheets of circumvallate papillae (around 0.1 mm2 in size per tongue) (Figure 1B5), and 150,500 cells in total with a viability of 93% from connective tissues (around 0.1 mm2 in size per tongue) (Figure 1B6).
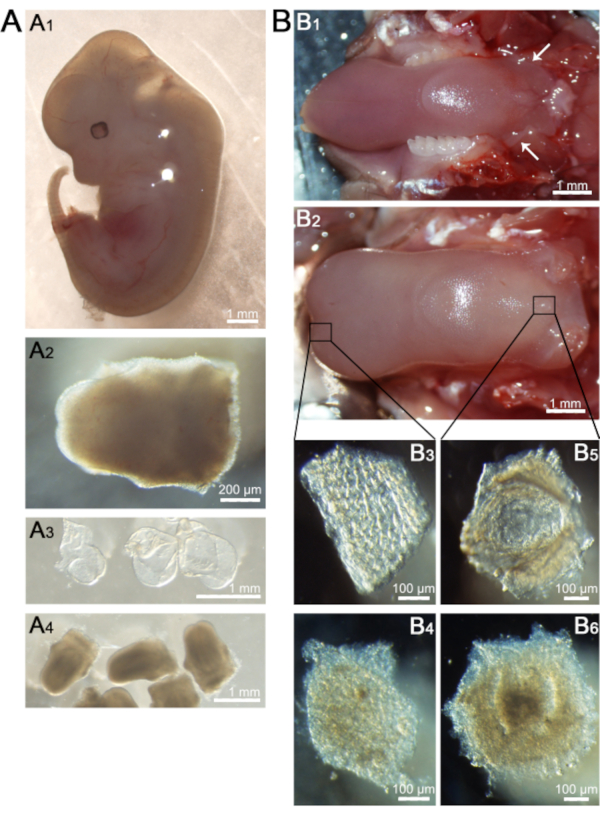
Figure 1. Tissue preparation for cell dissociation. A) Representative images of an E12.5 whole embryo (A1), dorsal view of the dissected tongue (A2), and epithelial sheets (A3) and mesenchyme (A4) separated from tongues. B) Representative images of an adult tongue before (B1) and after enzyme injection (B2). Dorsal view of an epithelial sheet (B3) and mesenchyme (B4) of tongue tip, and an epithelial sheet (B5) and underlying connective tissue (B6) of circumvallate papilla. Scale bars: 1 mm in A1, A3, A4, B1, B2; 200 µm in A2; 100 µm in B3, B4, B5, and B6. Please click here to view a larger version of this figure.
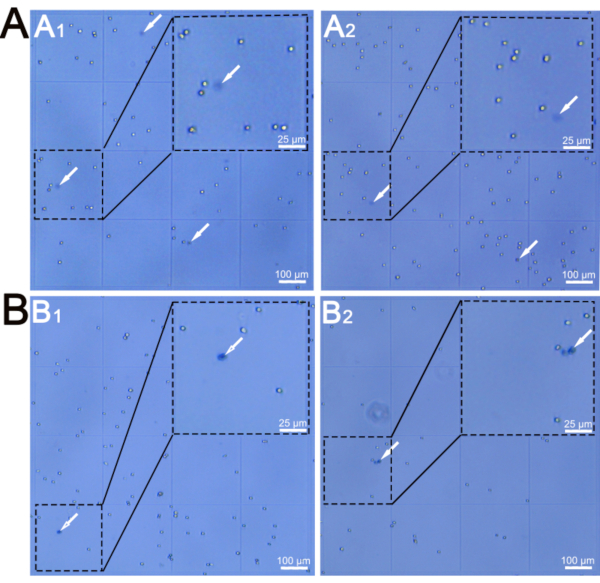
Figure 2. Representative images of isolated cells visualized in hemocytometer. A) Isolated cells from epithelial sheets (A1) and mesenchyme (A2) of embryos at E12.5. B) Isolated cells from epithelial sheets (B1) and underlying connective tissue cores (B2) of circumvallate papillae at 8 weeks of age. Dashd lines encircle the grids amplified in top right corner. Arrows point to dead cells stained by Trypan blue. Scale bars: 100 µm for B1,B2, B3, and B4; 25 µm for high power images in top right corner. Please click here to view a larger version of this figure.
List of Materials
| bovine serum albumin (BSA) | Gold Biotechnology | A-420-100 | |
| C57BL/6 mouse (C57BL/6J) | The Jackson Laboratory | 000664 | |
| collagenase (Collagenase A) | Sigma-Aldrich | 10103586001 | |
| culture dish (35 mm in diameter) | Genesee Scientific | 32-103G | |
| culture dish (100 mm in diameter) | Genesee Scientific | 32-107G | |
| dispase (Dispase II) | Sigma-Aldrich | 04942078001 | |
| dissecting scissors (Student Fine Scissors) | Find Science Tool | 91460-11 | |
| DMEM/F12 | Gibco | 11320033 | |
| fetal bovine serum (FBS) | Hyclone | C838U82 | |
| fine forceps (Dumount #3 Forceps) | Find Science Tool | 11293-00 | |
| hemocytometer | Hausser Scientific | 3520 | |
| inverted microscope with imaging system (EVOS XL Core Cell Imaging System) | Life Technologies | AMEX1000 | |
| low retention pipette tips | METTLER TOLEDO | 17014342 | |
| mini-scissors (Evo Spring Scissors) | Fine Science Tool | 15800-01 | |
| plastic warp | VWR | 46610-056 | |
| spatula (Moria Spoon) | Fine Science Tool | 10321-08 | |
| surgical forceps (Dumount #2 Laminectomy Forceps) | Fine Science Tool | 11223-20 | |
| Trypan blue | Gibco | 15250061 | |
| Tyrode’s solution | Sigma-Aldrich | T2145-10L | made from Tyrode's salts |
| 0.25% typsin-EDTA | Gibco | 25200056 | |
| 0.1 M Phosphate-Buffered Saline (PBS) | Hoefer | 33946 | made from 1 M PBS |
| 0.22-μm syringe filter | Genesee Scientific | 25-243 | |
| 70% ethanol | Koptec | 233919 | made from 100% ethanol |
| 1-mL syringe | BD | 8194938 | |
| 5-mL low binding microcentrifuge tube | Eppendorf | 30122348 | |
| 30-G needle | BD | 9193532 | |
| 35-μm cell strainer | Falcon | 64750 | |
| 70-μm cell strainer | Falcon | 64752 |
Lab Prep
Cell dissociation has been an essential procedure for studies at the individual-cell level and/or at a cell-population level (e.g., single cell RNA sequencing and primary cell culture). Yielding viable, healthy cells in large quantities is critical, and the optimal conditions to do so are tissue dependent. Cell populations in the tongue epithelium and underlying mesenchyme/connective tissue are heterogeneous and tissue structures vary in different regions and at different developmental stages. We have tested protocols for isolating cells from the mouse tongue epithelium and mesenchyme/connective tissue in the early developmental [embryonic day 12.5 (E12.5)] and young adult (8-week) stages. A clean separation between the epithelium and underlying mesenchyme/connective tissue was easy to accomplish. However, to further process and isolate cells, yielding viable healthy cells in large quantities, and careful selection of enzymatic digestion buffer, incubation time, and centrifugation speed and time are critical. Incubation of separated epithelium or underlying mesenchyme/connective tissue in 0.25% Trypsin-EDTA for 30 min at 37 °C, followed by centrifugation at 200 x g for 8 min resulted in a high yield of cells at a high viability rate (>90%) regardless of the mouse stages and tongue regions. Moreover, we found that both dissociated epithelial and mesenchymal/connective tissue cells from embryonic and adult tongues could survive in the cell culture-based medium for at least 3 h without a significant decrease of cell viability. The protocols will be useful for studies that require the preparation of isolated cells from mouse tongues at early developmental (E12.5) and young adult (8-week) stages requiring cell dissociation from different tissue compartments.
Cell dissociation has been an essential procedure for studies at the individual-cell level and/or at a cell-population level (e.g., single cell RNA sequencing and primary cell culture). Yielding viable, healthy cells in large quantities is critical, and the optimal conditions to do so are tissue dependent. Cell populations in the tongue epithelium and underlying mesenchyme/connective tissue are heterogeneous and tissue structures vary in different regions and at different developmental stages. We have tested protocols for isolating cells from the mouse tongue epithelium and mesenchyme/connective tissue in the early developmental [embryonic day 12.5 (E12.5)] and young adult (8-week) stages. A clean separation between the epithelium and underlying mesenchyme/connective tissue was easy to accomplish. However, to further process and isolate cells, yielding viable healthy cells in large quantities, and careful selection of enzymatic digestion buffer, incubation time, and centrifugation speed and time are critical. Incubation of separated epithelium or underlying mesenchyme/connective tissue in 0.25% Trypsin-EDTA for 30 min at 37 °C, followed by centrifugation at 200 x g for 8 min resulted in a high yield of cells at a high viability rate (>90%) regardless of the mouse stages and tongue regions. Moreover, we found that both dissociated epithelial and mesenchymal/connective tissue cells from embryonic and adult tongues could survive in the cell culture-based medium for at least 3 h without a significant decrease of cell viability. The protocols will be useful for studies that require the preparation of isolated cells from mouse tongues at early developmental (E12.5) and young adult (8-week) stages requiring cell dissociation from different tissue compartments.
Procedure
Cell dissociation has been an essential procedure for studies at the individual-cell level and/or at a cell-population level (e.g., single cell RNA sequencing and primary cell culture). Yielding viable, healthy cells in large quantities is critical, and the optimal conditions to do so are tissue dependent. Cell populations in the tongue epithelium and underlying mesenchyme/connective tissue are heterogeneous and tissue structures vary in different regions and at different developmental stages. We have tested protocols for isolating cells from the mouse tongue epithelium and mesenchyme/connective tissue in the early developmental [embryonic day 12.5 (E12.5)] and young adult (8-week) stages. A clean separation between the epithelium and underlying mesenchyme/connective tissue was easy to accomplish. However, to further process and isolate cells, yielding viable healthy cells in large quantities, and careful selection of enzymatic digestion buffer, incubation time, and centrifugation speed and time are critical. Incubation of separated epithelium or underlying mesenchyme/connective tissue in 0.25% Trypsin-EDTA for 30 min at 37 °C, followed by centrifugation at 200 x g for 8 min resulted in a high yield of cells at a high viability rate (>90%) regardless of the mouse stages and tongue regions. Moreover, we found that both dissociated epithelial and mesenchymal/connective tissue cells from embryonic and adult tongues could survive in the cell culture-based medium for at least 3 h without a significant decrease of cell viability. The protocols will be useful for studies that require the preparation of isolated cells from mouse tongues at early developmental (E12.5) and young adult (8-week) stages requiring cell dissociation from different tissue compartments.
