An Approach to Constructing Multispecies Biofilm Communities from Rhizosphere Soil
Summary
A rapid and standardized procedure for establishing synergistic multispecies biofilm communities from various rhizosphere soils is presented here. It is a unique protocol designed to probe and simulate the complex rhizosphere soil microbiota.
Abstract
The multispecies biofilm is a naturally occurring and dominant lifestyle of bacteria in nature, including in rhizosphere soil, although the current understanding of it is limited. Here, we provide an approach to rapidly establish synergistic multispecies biofilm communities. The first step is to extract cells from rhizosphere soil using the differential centrifugation method. Afterward, these soil cells are inoculated into the culture medium to form pellicle biofilm. After 36 h of incubation, the bacterial composition of the biofilm and the solution underneath are determined using the 16S rRNA gene amplicon sequencing method. Meanwhile, high-throughput bacterial isolation from pellicle biofilm is conducted using the limiting dilution method. Then, the top 5 bacterial taxa are selected with the highest abundance in the 16S rRNA gene amplicon sequencing data (pellicle biofilm samples) for further use in constructing multispecies biofilm communities. All combinations of the 5 bacterial taxa were quickly established using a 24-well plate, selected for the strongest biofilm formation ability by the crystal violet staining assay, and quantified by qPCR. Finally, the most robust synthetic bacterial multispecies biofilm communities were obtained through the methods above. This methodology provides informative guidance for conducting research on rhizosphere multispecies biofilm and identifying representative communities for studying the principles governing interactions among these species.
Introduction
Biofilms represent intricate microbial communities either affixed to surfaces or linked with interfaces. There is broad acknowledgment that the majority of bacteria encountered in various environments, such as natural habitats, clinical settings, and industrial contexts, endure within biofilm communities1. In soil, the rhizosphere—encompassing the root surface and its adjacent 2 mm thick region—forms an ecological niche with heightened nutrient availability. Specific bacteria have evolved strategies to exploit this niche, fostering microbial proliferation and fostering the formation of biofilm communities in the rhizosphere2.
Rhizosphere microbes are considered the 'second genome' of plants3. Plant growth-promoting microorganisms (PGPM) engage in mutualistic symbiosis with plants, providing significant growth promotion and biocontrol functions4. On the one hand, PGPM can supply nutrients to plant roots, enhance nutrient uptake, defend against pathogens, and degrade pollutants in the rhizosphere soil5. On the other hand, PGPM utilizes plant root exudates as a nutrient source for growth and evolution, thereby influencing the rhizosphere soil and root system through their own metabolism. It is worth noting that the prerequisite for all these functions is the effective colonization of PGPM in the plant rhizosphere, forming stable biofilms6.
Rhizosphere biofilm affects the assembly process of rhizosphere microbes, and the temporal and spatial characteristics of rhizosphere microbe assembly are closely related to the interaction of multispecies biofilm7. It serves as the hub of plant-microbiome interactions, providing a stable and controllable niche for them to interact effectively. Therefore, studying rhizosphere biofilm is also an important direction to gain insight into the interaction between plants and microbes.
However, there is currently little research on rhizosphere multispecies biofilms. The high complexity of microbiome composition makes it challenging to answer fundamental ecological questions surrounding natural microbial communities8. In the process of experiments, many conditions, such as soil type, plant type, and the large number of microbial communities, need to be considered. Various factors limit the research of rhizosphere multispecies biofilms.
To further investigate multispecies biofilm communities from rhizosphere soil, such as interspecies synergistic interactions or metabolite cross-feeding8, it is desirable to artificially construct a simplified, representative, stable, and tractable synthetic microbial community under laboratory conditions9,10. Here, a standardized protocol combines several of the latest microbiological and bioinformatic techniques.
Protocol
This protocol is generally applicable to rhizosphere soil microbiota from various plants. Here, cucumber is used as an example. Details of the reagents and equipment used for the study are listed in the Table of Materials.
1. Bacterial isolation
- Rhizosphere soil isolation
- Cucumber culture
- Disinfect cucumber seeds with 75% ethanol and 2% sodium hypochlorite (NaClO) solution, then rinse them in sterile water11.
- Germinate the seeds under sterile conditions and select those that reach the two-leaf stage12. Subsequently, transplant the consistently sprouted cucumber seedlings into pots containing 2 kg of black soil.
NOTE: Two types of black soil from different locations in Northeast China are used to reduce systematic errors.
- Soil suspension preparation
- Prepare PBS-S buffer using the formulation: 6.33 g of NaH2PO4·H2O, 16.5 g of Na2HPO4·7H2O, 200 µL of Silwet L-77, and 1 L distilled water.
- When the seedlings reach the four-leaf stage12, invert the pot and isolate the roots along with 2 mm-thick rhizosphere soil using sterilized gloves.
- Place the roots in 30 mL of PBS-S buffer in a 50 mL centrifuge tube. After shaking for 20 min, filter the suspension through a 100 µm nylon cell filter to remove roots and large sediments.
- Transfer the filtrate to another 50 mL centrifuge tube, centrifuge it at 3200 x g for 15 min at room temperature, and temporarily store it at 4 °C.
- Cucumber culture
- Extraction of rhizosphere soil bacterial cells.
NOTE: This methodology adopts the differential centrifugation method13, although the Nycodenz density gradient centrifugation method14 also applies for this step.- Dilute the soil suspension with a 0.2% Na4P2O7 solution to 500 mL and centrifuge at 1,000 x g for 10 min to initially separate organisms from the soil.
- Re-homogenize the precipitate, centrifuge it at the same low speed for 60 s, and repeat the process three times. Combine the supernatants, centrifuge them at 12,000 x g for 20 min, and then discard the supernatant.
- Resuspend the precipitate in 50 mL of PBS-S buffer, constituting the rhizosphere soil bacterial cell suspension.
2. Sequencing and identification of rhizosphere biofilm microbiota
- Media preparation
NOTE: Tryptic soy broth (TSB) and Minimal salts glycerol glutamate (MSgg) media are commercially available. The formulations below are for reference only.- Prepare TSB medium using the following formulation: 15 µg/mL tryptone, 5 µg/mL soy peptone, 5 µg/mL NaCl, pH = 7.
- Prepare MSgg medium using the following formulation: 5 mM KH2PO4, 100 mM 3-(N-morpholino) propane sulfonic acid (MOPS), 2 mM MgCl2, 700 µM CaCl2, 50 µM MnCl2, 50 µM FeCl3, 1 µM ZnCl2, 2 µM thiamine, 0.5% glycerol, 0.5% glutamate, 50 µg/mL tryptophan, 50 µg/mL phenylalanine, pH = 7.
- To prepare TSB-MSgg media, mix TSB media with MSgg media in a 1:1 ratio (v/v)15.
- Pellicle biofilm coculture
- Incubate the cell suspension in TSB medium at 30 °C overnight (usually 12 h) to activate the bacteria.
- Place 100 µm nylon cell filters on 24-well plates. Incubate the bacteria in TSB media and set up three repetitions. Cultivate the pellicle biofilm for 36 h at 30 °C.
NOTE: If the pellicle biofilm from the last step is not well-cultivated, the TSB-MSgg medium is an effective substitute for the TSB medium. - Remove the filter containing the pellicle biofilm. Add 2 mL of PBS-S buffer to a new 24-well cell plate, place the 24-well filter into it to wash the biofilm, and remove the free cells. Repeat the washing process three times.
- Extraction of pellicle biofilm DNA
- Extract the biofilm genome using a DNA kit following the manufacturer's instructions.
- High-throughput sequencing
NOTE: Biotech companies can help complete sequencing experiments for most labs. If microbial composition in the remaining solution is needed, sequence it at this step. The sequencing step varies depending on the instruments. Please follow the manufacturer's instructions. Step 2.4.1 is for reference only.- Amplify the V3-V4 regions of the 16S rRNA gene based on the database with full-length 16S rRNA gene sequences. Create species OTUs according to the minimum number of sequences per sample16. Cluster the species classification annotations and acquire the compositions of communities from each sample.
- Based on the sequencing data, select the top five strains in terms of their relative abundance.
- High-throughput bacterial isolation and culture
NOTE: This step is designed according to the latest microbiological and bioinformatic methodology17. The dilution-coated plate method is available for bacterial isolation here, although it is more time-consuming and less targeted compared to high-throughput techniques.- Mix the cultivated biofilm. Use limiting dilution to ensure that no more than 30% of the wells in the 24-well plate show bacterial growth.
NOTE: To determine the most suitable dilution, perform preliminary experiments using a wide range of dilution rates. The optimal dilution should contain no more than two culturable bacteria per milliliter, representing bacterial incubation in less than 30% of the wells. - Amplify the 16S rRNA gene of the bacteria while adding labels for the wells and plates and sequence them using the high-throughput Illumina platform17.
- Conduct bioinformatic analyses using commercially available software to obtain purity and species taxonomic information for each culture.
- Cultivate the top five abundant pure bacteria by continuous line culture and use glycerol to preserve the bacteria at -80 °C.
- Mix the cultivated biofilm. Use limiting dilution to ensure that no more than 30% of the wells in the 24-well plate show bacterial growth.
3. Construction of synthetic microbial communities
- Re-constructed biofilm culture
NOTE: For details, please refer to Ren et al.18. The 5 strains correspond to 31 synthetic combinations, including 5 single-species, 10 two-species, 10 three-species, 5 four-species, and 1 five-species consortia.- Incubate the 5 strains in the TSB medium until their OD600 reaches 1 to activate them.
- Prepare several 24-well plates with TSB medium and place 100 µm nylon cell filters on them.
NOTE: If the biofilm from the last step is not well-cultivated, the TSB-MSgg medium is an effective substitute for the TSB medium. - Incubate the 31 combinations of synthetic communities in the medium. Ensure the inoculation number of bacteria is consistent in each well. Set up three repetitions for each combination.
- Cultivate the biofilm at 30 °C for 36 h.
- Quantification of pellicle biofilm
- Follow the same procedure as in step 2.2.3.
- Transfer the biofilm to a new 24-well plate containing 180 µL of crystal violet solution and stain it for 20 min.
- Transfer the stained sample to another 24-well plate containing 200 µL of 96% ethanol, elute it for 30 min, and measure OD590.
4. Pellicle cell number quantification
NOTE: To determine the proportion of each species and the cell counts in the pellicle and remaining solution, quantitative PCR is necessary. For details, refer to Sun et al.16.
- Design strain-specific primers for the selected synthetic microbial communities.
- Use Roary to perform genome comparisons and find out the strain-specific single-copy genes of each isolate. Ensure that primers are targeted to these genes.
- Ligase the amplified fragments to PMD19T plasmids. Generate standard curves using the plasmids containing corresponding fragments as templates.
- Follow the same steps as mentioned in step 2.2.
- Prepare reaction components as follows: 7.2 µL of H2O, 10 µL of 2x qPCR Master mix, 0.4 µL of 10 µM of each primer, and 2 µL template DNA.
- Perform the PCR with a Real-time PCR Instrument under the following conditions: 95 °C for 10 min, 40 cycles of 95 °C for 30 s, and 60 °C for 45 s, followed by a standard melting curve segment.
- Perform six biological replicates for each treatment.
Representative Results
Following the mentioned procedure, significant gradients in biofilm-forming capacity from cucumber rhizosphere soil microbiota are observed (Figure 1). Biofilm amplicon sequencing confirmed the species in the pellicle (Figure 2). Based on the heat mapping of sequencing data, the top five bacterial species whose abundance in the pellicle is larger than that in the remaining solution were selected (Figure 3). Here, a typical group of synthetic communities was taken as an example. Crystal violet staining and quantitative PCR directly visualize the differences in pellicle biomass and the relative abundance of each species in the communities. Increased coculture biomass indicates the success of constructing synergistic multispecies biofilm communities from rhizosphere soil (Figure 4 and Figure 5).
The results show that Bacillus velezensis SQR9, Acinetobacter baumannii XL-1, and Burkholderia cenocepacia XL-2 can form prominent synergistic consortia, with the most robust community consisting of all three species. They are predicted to exhibit strong synergism in biofilm formation. It is also noteworthy that XL-1 serves as a 'dedicator.' It promotes the biofilm formation of XL-2 and SQR9, despite its poor growth and minimal abundance in the consortia (Figure 5). While a number of multispecies coculture biofilms represent a synergistic phenotype, some species, such as Chryseobacterium sp., are always negatively related to other strains by reducing the biomass of synthetic biofilm (Figure 2). Although this phenomenon is not related to the target of this protocol, it might be worth further investigation in the future.
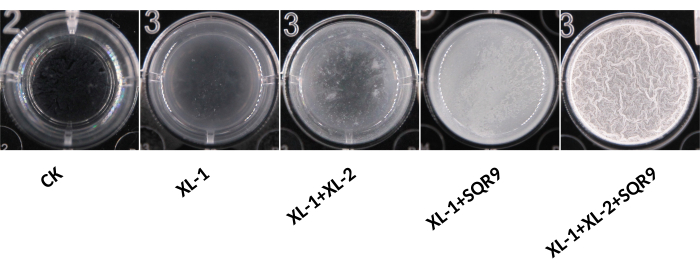
Figure 1: Biofilm formation of different synthetic communities (SynComs). Here, the samples are in standard 24-well plates. The photographs are captured without any magnification. Please click here to view a larger version of this figure.
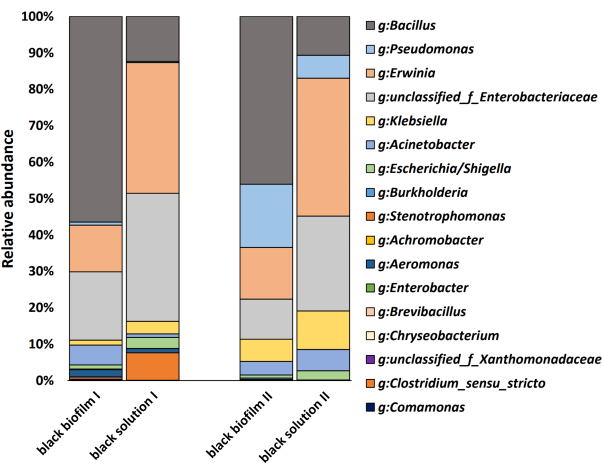
Figure 2: Bacterial composition in pellicle and solution. Graphs are based on 16S rRNA gene amplicon sequencing data, mentioned in protocol step 2.3. Please click here to view a larger version of this figure.
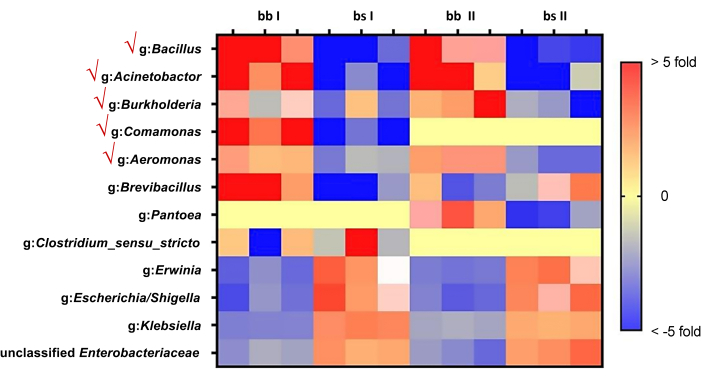
Figure 3: Heatmap of biofilm-forming capacity in pellicle and solution. Red represents the abundance in biofilm, which is higher than the solution, and blue represents the opposite. Genus showed no difference are not shown. Species of the top 5 abundance are ticked on the left. bb: black biofilm; bs: black solution. Please click here to view a larger version of this figure.
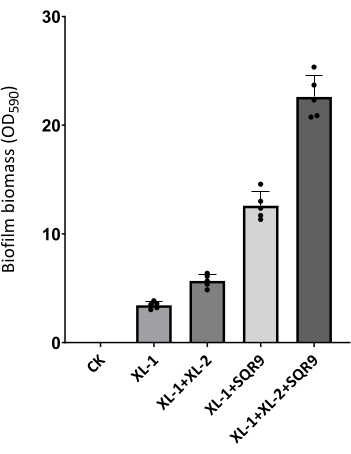
Figure 4: Biofilm biomass quantification of synthetic communities in Figure 1. Data is from crystal violet quantification with OD = 590 nm. The data presented are the mean ± SD, n = 5. Please click here to view a larger version of this figure.
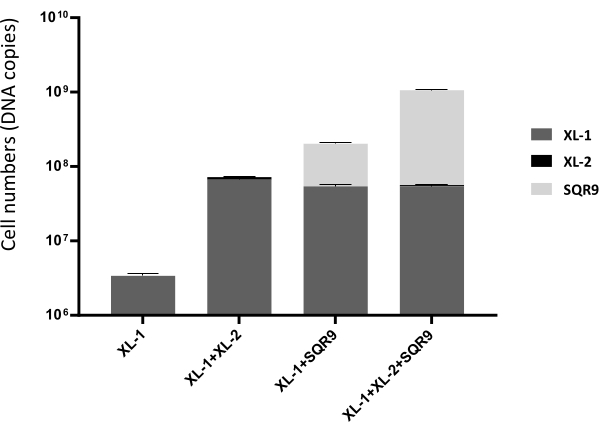
Figure 5: Cell numbers and species relative abundance of synthetic communities in Figure 1. Data is from quantitative PCR. The data presented are the mean ± SD, n = 6. Please click here to view a larger version of this figure.
Discussion
Following the protocol, a series of robust synthetic multispecies biofilm communities are constructed based on the microbiota in rhizosphere soil from different plants. Using quantitative PCR techniques, the composition of each community is deciphered clearly. The biomass of the biofilm indicates the strength of their potential metabolic interactions, although various properties and mechanisms behind the cooperation could not be revealed here19. Given the size and complexity of natural microbial communities, synthetic communities (SynComs) represent a model system for overcoming existing obstacles14. This protocol highlights the systematic and fundamental methodology of constructing synthetic biofilm communities from complex rhizosphere soil microbiota.
It is evident from the last part that SQR9, XL-1, and XL-2 show strong potential for stable colonization in the actual rhizosphere. Researchers can start investigating the metabolic relationships and co-colonization mechanisms based on these strains. Subsequent research on microbial inoculants for cucumber is also accessible thereafter.
Despite the advantages mentioned, it is important not to overlook the drawbacks of the protocol. Due to differences between laboratory culture and in situ conditions, the results of each synthetic community might not accurately represent the microbiota in real rhizosphere soil20. Confirmation through further rhizosphere inoculation is necessary. Additionally, the reproducibility of these results is relatively poor. Pellicle biofilm species in the laboratory may have undergone mutations over many generations compared to wild types in the soil, leading to some uncontrollable inaccuracies.
There are several alternatives to certain steps in the protocol. For instance, in step 1.2, the Nycodenz density gradient centrifugation method is relatively more efficient for collecting soil cell samples13. Additionally, in step 2.4, the latest high-throughput cultivation and identification of bacteria significantly increase experimental efficiency, albeit at a higher cost. Therefore, the traditional dilution-coated plate method remains suitable for selecting a few species. Finally, in step 3.2, the biomass of cultivated biofilm can be assessed both by crystal violet staining and by directly weighing it on an analytical balance.
This protocol extends beyond merely creating basic experimental materials for microbial and environmental research. Identifying the most robust synthetic bacterial multispecies biofilm communities is just the first step in unraveling the open and complex rhizosphere ecosystem. By focusing on the core species of SynComs, researchers can gain insight into real in situ situations in a more targeted and rapid way. This approach may also offer a novel perspective for exploring biofilms on a larger scale, such as rhizosphere soil aggregates, and for precise management of rhizosphere microbial communities for plant growth and health21.
Most significantly, this methodology will shed light on further research into interspecies interactions that govern the assemblage and dynamics of rhizosphere soil microbiota. For instance, when dealing with a complex rhizosphere community, other analytical methods such as co-occurrence network analysis or genome-scale metabolic modeling15 are often combined with these fundamental steps to artificially construct a representative biofilm community. This includes considerations of interspecies relationships.
Disclosures
The authors have nothing to disclose.
Acknowledgements
This work was financially supported by the National Natural Science Foundation of China (42307173 and 42107328) and the National Key Research and Development Program (2022YFD1500202 and 2022YFF1001800). P.W, Z.X designed the study. P.W, B.X, Z.C, J.X, and N.Z analyzed the data and created the figures. P.W wrote the first draft of the manuscript. Z.X., R.Z., and Q.S. revised the manuscript.
Materials
| 0.2 % Na4P2O7 solution | Sinopharm Chemical Reagent Co.,Ltd. | 20041418 | Used to dilute the soil suspension. |
| 100 μm Nylon Cell Filter | Sangon Biotech (Shanghai) Co.,Ltd. | F613463-0001 | Used to filter culture solution and separate pellicle biofilm. |
| 2% NaClO solution | Sinopharm Chemical Reagent Co.,Ltd. | L03336903 | Used to sterilize the seeds. |
| 24-well Plate | Merck & Co., Inc. | PSRP010 | Used for micobial cultivation and pellicle biofilm separation. |
| 3-(N-morpholino) propane sulfonic acid (MOPS) | Bioolook Scientific Research Special | 2360010 | Used to prepare MSgg media. |
| 50 mL Centrifuge Tube | Beijing Su-bio Biotech Co., Ltd. | 62.547.254 | Properly-sized centrifuge tubes for our protocol. |
| 75% C2H5OH solution | Sinopharm Chemical Reagent Co.,Ltd. | 801769680 | Used to sterilize the seeds. |
| Acinetobacter baumannii XL-1 | Nanjing Agricultural University | N/A | Strains isolated from rhizosphere soil. |
| Bacillus velezensis SQR9 | Nanjing Agricultural University | N/A | Strains isolated from rhizosphere soil. |
| Bacteria DNA Kit | Nanjing Dinsi Biotechnology Co.,Ltd. | D3350020000K04U021 | Used to extract bacterial DNA. |
| Black Soil I | Nanjing Agricultural University | N/A | Black soil from Jilin Province. |
| Black Soil II | Nanjing Agricultural University | N/A | Black soil from Heilongjiang Province. |
| Burkholderia cenocepacia XL-2 | Nanjing Agricultural University | N/A | Strains isolated from rhizosphere soil. |
| CaCl2 | Xilong Scientific Co.,Ltd. | 10035048 | Used to prepare MSgg media. |
| Centrifuge | Sangon Biotech (Shanghai) Co.,Ltd. | G508009-0001 | High-speed centrifuge. |
| ChamQ SYBR qPCR Master Mix | Vazyme Biotech Co.,Ltd. | Q311-02 | Used for DNA amplification in qPCR. |
| Clean Bench | Suzhou Antai Airtech Co., Ltd. | NB026143 | Used for aseptic operation. |
| Constant Temperature Incubator | ShangHai CIMO Medical Instrument Co.,Ltd | GNP-9080BS- |
Used for microbial cultivation. |
| Cristal Violet Dye | Sangon Biotech (Shanghai) Co.,Ltd. | A600331 | Used for pellicle biofilm staining and quantifivation. |
| Cucumber Seed | Nanjing Agricultural University | N/A | "Jinchun I" cucumber seed. |
| Culturome v 1.0 | The Institute of Genetics and Developmental Biology of the Chinese Academy of Sciences | N/A | Used for high-throughput rhizosphere soil bacterial cultivation and identification |
| FeCl3 | Sinopharm Chemical Reagent Co.,Ltd | 10011918 | Used to prepare MSgg media. |
| Fridge | Hefei Midea Refrigerator Co.,Ltd. | BCD-556WKPM(Q) | Used for strain preservation. |
| Glutamate | Bioolook Scientific Research Special | 2180020 | Used to prepare MSgg media. |
| Glycerol | Sinopharm Chemical Reagent Co.,Ltd | 10010618 | Used to prepare MSgg media. |
| Hydroponic Planting Basket | Yulv Furniture Store | 6526262626 | Used for cucumber hydroponics. |
| KH2PO4 | Xilong Scientific Co.,Ltd. | 10017618 | Used to prepare MSgg media. |
| MgCl2 | Xilong Scientific Co.,Ltd. | 7791186 | Used to prepare MSgg media. |
| MnCl2 | Xilong Scientific Co.,Ltd. | 10400101 | Used to prepare MSgg media. |
| Msgg Medium | Shanghai Bioesn Biotechnology Co.,Ltd. | BES20791KB | Pellicle biofilm culture medium. |
| Na2HPO4·7H2O | Merck & Co., Inc. | S9390-100G | Used to prepare TSB media. |
| NaCI | Chinasun Specialty Products co., Ltd. | HS0416 | Used to prepare TSB media. |
| NaH2PO4·H2O | Merck & Co., Inc. | S9638-25G | Used to prepare TSB media. |
| PBS-S Buffer | Sangon Biotech (Shanghai) Co.,Ltd. | E607008-0001 | Basic buffer for living tissues. |
| Phenylalanine | RYON | RT3486L005 | Used to prepare MSgg media. |
| Pipette | Beijing Labgic Technology Co.,Ltd. | BS-1000-T | Used for strain inoculation. |
| PMD19T Plasmid | Takara Biomedical Technology (Beijing) Co.,Ltd. | D102A | Used to generate qPCR standard curves. |
| Pot | Sinopharm Chemical Reagent Co.,Ltd. | YHHWS2401 | Used for cucumber soil culture. |
| Primer for qPCR | Sangon Biotech (Shanghai) Co.,Ltd. | N/A | Used for DNA amplification in qPCR. |
| Real-Time PCR Instrument | Thermo Fisher Scientific (China) Co.,Ltd. | 4484073 | Used to perform qPCR on selected pellicle biofilm. |
| Roary | The Wellcome Trust SangerInstitute | N/A | Used to design primers of qPCR |
| Shaker | Shanghai Zhichu Instrument Co.,Ltd | ZQTY-50S | Used for solution mixing and microbial cultivation. |
| Silwet L-77 | Cytiva Bio-technology(Hangzhou) Co., Ltd. | SL77080596 | Used to prepare TSB media. |
| Soy peptone | Qingdao Hi-tech Industrial Park Hope Bio-technology Co., Ltd | HB8275 | Used to prepare TSB media. |
| Spectrophotometer | Shanghai Yidian Analysis Instrument Co.,Ltd. | 76713100010 | Used for pellicle biofilm cristal violet quantifivation. |
| Sterile Water | Sinopharm Chemical Reagent Co.,Ltd. | SW150302 | Used to wash the pellicle biofilm. |
| Sterilized Nitrile Gloves | Beijing Labgic Technology Co.,Ltd. | 223016852LLZA | Used for basic experimental operations. |
| Template DNA | Sangon Biotech (Shanghai) Co.,Ltd. | N/A | Used for DNA amplification in qPCR. |
| Thiamine | Bioolook Scientific Research Special | 2180020 | Used to prepare MSgg media. |
| Tryptone | Thermo Fisher Scientific | LP0042B | Used to prepare TSB media. |
| Tryptophan | Bioolook Scientific Research Special | 2190020 | Used to prepare MSgg media. |
| TSB Medium | Guangdong Huankai Microbial Sci. & Tech. Co.,Ltd. | 024048 | Broad-spectrum bacterial medium. |
| TSB-Msgg Meidium | Nanjing Agricultural University | N/A | Mixed medium for pellicle biofilm culture with wider applicability. |
| ZnCl2 | Nanjing Chemical Reagent Co.,Ltd | C0310520123 | Used to prepare MSgg media. |
References
- Davey, M. E., O’Toole, G. A. Microbial biofilms: from ecology to molecular genetics. Microbiol Mol Biol Rev. 64 (4), 847-867 (2000).
- Campbell, R., Greaves, M. P., Lynch, J. Anatomy and community structure of the rhizosphere. Rhizosphere. , 11-34 (1990).
- Berendsen, R. L., Pieterse, C. M., Bakker, P. A. The rhizosphere microbiome and plant health. Trends Plant Sci. 17 (8), 478-486 (2012).
- Lugtenberg, B., Kamilova, F. D. Plant-growth-promoting rhizobacteria. Annu Rev Microbiol. 63, 541-556 (2009).
- Compant, S., Clément, C., Sessitsch, A. Plant growth-promoting bacteria in the rhizo- and endosphere of plants: Their role, colonization, mechanisms involved and prospects for utilization. Soil Biol Biochem. 42, 669-678 (2010).
- Bais, H. P., Fall, R. R., Vivanco, J. M. Biocontrol of Bacillus subtilis against infection of Arabidopsis roots by Pseudomonas syringae is facilitated by biofilm formation and surfactin production. Plant Physiol. 134 (1), 307-319 (2004).
- Trivedi, P., Leach, J. E., Tringe, S. G., Sa, T., Singh, B. K. Plant-microbiome interactions: From community assembly to plant health. Nat Rev Microbiol. 18, 607-621 (2020).
- Sun, X., et al. Bacillus velezensis stimulates resident rhizosphere Pseudomonas stutzeri for plant health through metabolic interactions. ISME J. 16, 774-787 (2021).
- Pandhal, J., Noirel, J. Synthetic microbial ecosystems for biotechnology. Biotechnol Lett. 36, 1141-1151 (2014).
- Grosskopf, T., Soyer, O. S. Synthetic microbial communities. Curr Opin Microbiol. 18, 72-77 (2014).
- Lundberg, D. S., et al. Defining the core Arabidopsis thaliana. root microbiome. Nature. 488, 86-90 (2012).
- Bai, Y., et al. Functional overlap of the Arabidopsis leaf and root microbiota. Nature. 528, 364-369 (2015).
- Bakken, L. R. Separation and purification of bacteria from soil. Appl Environ Microbiol. 49 (6), 1482-1487 (1985).
- Yang, O., et al. Direct cell extraction from fresh and stored soil samples: Impact on microbial viability and community compositions. Soil Biol Biochem. 155, 108178 (2021).
- Ren, D., et al. High-throughput screening of multispecies biofilm formation and quantitative PCR-based assessment of individual species proportions, useful for exploring interspecific bacterial interactions. Microb Ecol. 68, 146-154 (2013).
- Sun, X., et al. Metabolic interactions affect the biomass of synthetic bacterial biofilm communities. mSystems. 8, e01045-e01023 (2023).
- Zhang, J., et al. High-throughput cultivation and identification of bacteria from the plant root microbiota. Nat Protoc. 16, 988-1012 (2021).
- Ren, D., Madsen, J. S., Sørensen, S. J., Burmølle, M. High prevalence of biofilm synergy among bacterial soil isolates in cocultures indicates bacterial interspecific cooperation. ISME J. 9, 81-89 (2015).
- Liu, Y., et al. Root colonization by beneficial rhizobacteria. FEMS Microbiol Rev. 48 (1), (2024).
- Palková, Z. Multicellular microorganisms: Laboratory versus nature. EMBO Rep. 5, 470-476 (2004).
- Xu, Z., et al. Chemical communication in plant-microbe beneficial interactions: a toolbox for precise management of beneficial microbes. Curr Opin Microbiol. 72, 102269 (2023).

.
