Orthotopic Aortic Transplantation in Mice for the Study of Vascular Disease
Summary
We describe a technique in which a section of the abdominal aorta from a mouse is transplanted orthotopically to just below the renal arteries in an allogeneic or syngeneic recipient. This technique can be useful in studies in which transplantation of large arteries of uniform size is deemed advantageous.
Abstract
Vascular procedures involving anastomoses in the mouse are generally thought to be difficult and highly dependent on the skill of the individual surgeon. This is largely true, but there are a number of important principles that can reduce the difficulty of these procedures and enhance reproducibility. Orthotopic aortic transplantation is an excellent procedure in which to learn these principles because it involves only two end-to-end anastomoses, but requires good suturing technique and handling of the vessels for consistent success. This procedure begins with the procurement of a length of abdominal aorta from a donor animal, followed by division of the native aorta in the recipient. The procured aorta is then placed between the divided ends of the recipient aorta and sutured into place using end-to-end anastomoses. To accomplish this objective successfully requires a high degree of concentration, good tools, a steady hand, and an appreciation of how easily the vasculature of a mouse can be damaged, resulting in thrombosis. Learning these important principles is what occupies most of the beginner’s time when learning microsurgery in small rodents. Throughout this protocol, we refer to these important points. This model can be used to study vascular disease in a variety of different experimental systems1-8. In the context shown here, it is most often used for the study of post-transplant vascular disease, a common long-term complication of solid organ transplantation in which intimal hyperplasia occurs within the allograft. The primary advantage of the model is that it facilitates quantitative morphometric analyses and the transplanted vessel lies contiguous to the endogenous vessel, which can serve as an additional control9. The technique shown here is most often used for mice weighing 18-25 grams. We have accumulated most of our experience using the C57BL/6J, BALB/cJ, and C3H/HeJ strains.
Protocol
1. Presurgical Preparation
- Surgical procedures, no matter how carefully done, result in significant stress. To minimize the effects of such stress and to maximize reserves, animals should be maintained in the vivarium for at least 72 hr before use10,11.
- Surgical tools, gauze, and swabs must be sterile. It is not necessary to use sterile gloves providing only the tips of the instruments touch suture or the operative field.
- For hemostasis, it is imperative that clamping pressures are the minimum necessary. Damage to the vessel due to excessive pressure or rough handling will cause thrombosis, resulting in hind-leg paralysis, bowel ischemia, and death within 24 hr. We have found that using clamps with a pressure of no more than 2 gm/cm2 eliminates the problem. These clamps are noted in Table 1. Clamp pressures are indicated in the specifications for clamps made by high quality surgical instrument manufacturers. We have found that inexpensive clamps or disposable clamps universally exert too much pressure.
- The location of the collateral vessels as well as the relative locations of the abdominal aorta and the inferior vena cava (IVC) are strain dependent. You will also find small variations between individual mice, especially in the location of collateral vessels.
2. Donor Operation
- Mice can be anesthetized with isoflurane (1.5-2.5 % induction, 1.0 % maintenance in air) or with pentobarbital (55-65 mg/kg). Pentobarbital imposes a time limit (up to 1.5 hr), but provides the surgeon with the option of rotating the animal for better access, if desired. Anesthesia time can be extended to up to 2 hr by placing 2-3 drops of the anesthetic when needed onto the intestine or liver during the surgery. Ketamine/xylazine (100 mg/kg body weight for Ketamine and 7 mg/kg for Xylazine, ip) can also be used by well-trained surgeons who can finish the procedure within 1 to 1.5 hr, but it has a narrower safe dose range. In the US, the use of pentobarbital has become more problematic in recent years because of short supplies. It is imperative that the depth of anesthesia be monitored to be certain that it is sufficient. The toe-pinch reflex is the best indicator of adequate depth but one should also note respiration rate and other movements as well.
- After anesthesia induction, use a depilatory gel or a shaver to remove hair on the ventral abdomen.
- Mount the animal on the operating board using laboratory tape to hold it in a spread-eagled position. A metallic operating board permits the use of a heating pad underneath it to provide temperature support during the surgery. We manually control the temperature of the heating pad, which is a consumer-grade unit for home-use. The temperature is maintained between 35-38°. The metallic operating board is also easy to sterilize between procedures.
- Disinfect the area using a disinfectant like chlorhexidine, followed by an ethanol wash. Repeat this procedure three times, to insure adequate disinfection. This will also remove any residual cut pieces of hair, if present.
- Using scissors make an incision through the body wall to expose the abdominal viscera.
- Use a cotton swab, push the viscera off to the side to expose the great vessels of the abdomen. The viscera are placed on a piece of gauze that is wetted with warm saline to keep it moist.
- Using blunt dissection, very carefully dissect the abdominal aorta away from the surrounding tissue. Be very careful in dissecting the vessel away from the inferior vena cava. There will be one or more branches, such as the lumbar artery, that must be carefully lifted and cauterized. The section of dissected aorta should be below the renal vessels and above the bifurcation of the aorta into the femoral arteries. This section can serve as donor material for as many as two recipients, depending on how many branching vessels exist and the size of the donor animal.
- Tie a suture just below the renal arteries and another just above the bifurcation. Using scissors divide the aorta, fully rinse the graft with saline containing 200 U/ml heparin. Very carefully hold one end of the vessel and drip the saline solution so that it runs through the vessel by gravity. Do not let air bubbles run through the vessel. Handle the ends of the vessel as little as possible otherwise the intima can be damaged resulting in thrombosis later. Remove it from the field and immediately place it in a container of ice-cold saline. Allow the donor animal to exsanguinate.
3. Recipient Operation
- Anesthetize the animal as above and apply a depilatory gel or use a shaver to remove hair on the abdomen. Just prior to the incision we administer a dose of buprenorphine subcutaneously at 0.1 mg/kg to establish analgesia.
- Mount the animal on the operating field using laboratory tape. The animal must be mounted on a warm operating stage that will help maintain body temperature during the operation.
- Apply gentamycin ophthalmic ointment to the eyes to prevent them from drying out.
- Disinfect the abdomen, followed by an ethanol wash. Repeat this procedure three times, to insure adequate disinfection.
- Check the adequacy of anesthesia once again before proceeding to the next step.
- Make a midline incision through the skin and the body wall in two stages, being careful not to impinge on the tissues underneath.
- Insert a spreader to hold the abdominal incision open. Point the screw assembly towards the hind end to keep the bulk of the clamp out of the way.
- Wet a square of sterile gauze with saline and place it over the intestines. With your finger gently on the gauze, insert a cotton swab under the intestines and gently reflect the intestines over to the side so they are sitting on top of the gauze. Take another piece of gauze, place it on the intestines and wet it with saline.
- Remove any fatty tissue covering the aorta and the inferior vena cava (IVC). Be gentle. The IVC is very fragile.
- Gently use forceps to dissect the infrarenal aorta from the IVC. Clear an area large enough to provide room for two clamps with enough vessel between them such that, when divided, there will be enough vessel to which the graft can be sutured. When dissecting, look for vessel branches. If necessary cauterize them with a low temperature cauterizer.
- Insert a vascular clamp just below the renal artery and another one just above the bifurcation.
- Cut the aorta. The ends of the aorta will usually retract, leaving a space of about 5 mm. Check for hemostasis. If the clamps are working correctly, only a small amount of blood should escape after the cut. If bleeding continues, check the clamps to be sure nothing is interfering with them. Note that none of the aorta is removed. It is merely divided.
- Rinse the cut ends with heparinized saline (200 U/ml) and then remove the extra solution with a swab.
- Tack the graft into place using three discontinuous sutures at each end. Just suture one side of the anastomosis at each end, and then fill in the rest with a continuous running suture or more discontinuous sutures. Seven to eight discontinuous sutures is sufficient. If you use a running suture, be sure that the vessel walls remain relaxed and that you do not cause stenosis at the anastomosis.
- Carefully remove the clamp at the cranial end and look for leaks at the suture lines. A small amount of seepage is OK providing it stops within about a minute. If there is more than that, it is possible to use a single stitch to stop the bleeding.
- Gently press on the aorta above the anastomosis site with a wetted cotton applicator, and then remove the second clamp. Gently press and release a couple of times and check that the aorta appears patent. The graft will be perfused immediately and a pulse should be visible.
- Remove the gauze covering the intestines and move them back into place. Avoid twisting of the intestine and maintain normal anatomical orientation.
- Close the muscle layer using 5-0 Vicryl.
- Close the skin using 5-0 or 6-0 prolene.
- Treat the mouse subcutaneously with carprophen at 5 mg/kg to augment analgesia before terminating anesthesia.
- Give the mouse 0.5-0.8 ml saline subcutaneously.
- Place the mouse in a heated cage to recover. Monitor it carefully during recovery to be sure that it is recovering normally. 2-3 hr after surgery, the mouse should be behaving relatively normally. At 12 hr post-op, treat the animal with buprenorphine subcutaneously at 2 mg/kg for analgesia. If, at any time, the animal is hunched, making noise or exhibiting a limited range of motion, investigate the problem. If a definitive cause cannot be established and fixed, the animal should be euthanized per institutional protocols. At 24 hr the animal should receive another dose of carprophen subcutaneously at 5 mg/kg. Hind leg paralysis indicates a failed anastomosis or a thrombus. In this situation, the animal should be euthanized.
Representative Results
Figure 1 shows an aortic graft. The white arrows denote the suture lines. A patent graft will show a visible pulse. Figure 2 indicates a typical experiment in which recipient survival was followed for a period of 56 days. One group consisted of wild-type (C57BL/6 x FVB) recipient mice transplanted with BALB/c aorta. The other group, designated “KO” consists of recipients (C57BL/6 x FVB) deficient in expression of heme oxygenase-1, which results in thrombosis of the grafts within 24 hr. Notably, this results in the death of all recipients as shown in the figure. Figure 3 shows echo measurements of the IVC and abdominal aorta in a normal animal and in a transplant recipient. Note that the graft is patent and similar in appearance to the non-transplanted aorta.
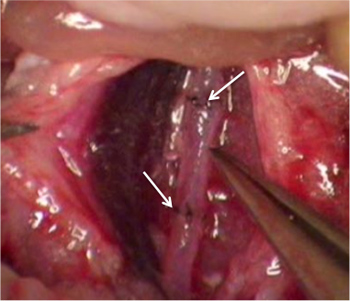
Figure 1. A view of a transplanted aorta. The white arrows denote the suture lines.
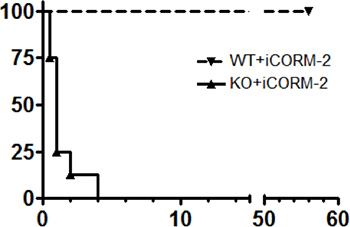
Figure 2. Kaplan-Meier depictions of survival after aortic transplantation in two groups of mice transplanted with aorta from a BALB/c mouse. “KO” designates recipients deficient in the expression of heme oxygenase-1, which results in thrombosis of the aortic grafts within 24-48 hr. “WT” designates the wild-type littermates. Reprinted from: Carbon monoxide rescues heme oxygenase-1-deficient mice from arterial thrombosis in allogeneic aortic transplantation, Chen B, Guo L, Fan C, Bolisetty S, Joseph R, Wright MM, Agarwal A, George JF. Am J Pathol. 2009 Jul;175(1):422-9 with permission from Elsevier.
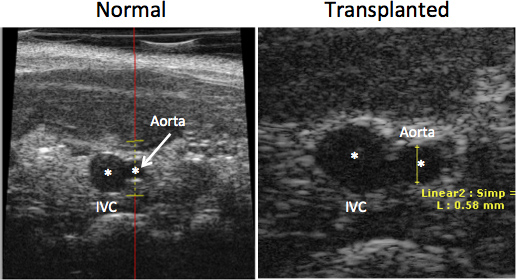
Figure 3. Echo imaging of the inferior vena cava (IVC) and abdominal aorta in vivo in a normal mouse (left panel) and in an aortic transplant recipient (right panel). The images were produced with a Visualsonics Vevo 660 instrument. Asterisks denote the lumen of the vessels.
Discussion
Mouse models of aortic transplantation provide a number of advantages because mice are very well defined immunogenetically9,12,13, and they can be easily manipulated to alter their expression of specific genes, if desired. As noted in the introduction, vascular surgery in the mouse is more difficult than most models because of the size of the vessels. Even the great arteries, such as the aorta are usually no more than 100-200 μm in inner diameter; so manipulating these vessels requires a significant amount of skill and dexterity8,14. The most common complication observed in this surgery by those just beginning to implement the system is hind-leg paralysis, usually caused by thrombosis resulting from injury of intima by rough handling or clamping. Most of the clamps marketed by companies that sell tools suitable for microsurgery create too much pressure to be used for mice, with pressures usually greater than 25 gm/cm2. The clamps we use are smooth, with a pressure of 2 gm/cm2, a pressure that is just enough to achieve hemostasis and not damage the vessels. Some surgeons prefer to use suture rather than clamps. We do not recommend this practice because it is a method that is highly dependent on the ability of the surgeon to gauge the correct pressure and, in our view, can contribute to a lower success rate. With practice, survival rates of 90% or better should be expected.
Suturing technique for the end-to-end anastomoses is a critical skill that is acquired with experience. Depending on prior experience with surgical techniques and manual dexterity, full proficiency may be achieved after 50-100 procedures. More recent studies suggest alternative technologies for anastomoses may become available in the future15.
The primary limitation of this procedure is, like most microsurgical procedures in mice, that a high level of skill is required for successful execution and individuals without excellent fine motor skills may never achieve a high level of proficiency. However, with practice, most individuals can achieve an acceptable survival rate. The small size of the tissues also results in additional constraints because the size of the graft is very small, so the amount of material for subsequent analysis is limiting. The largest advantage of the model is the well characterized immunogenetics of mice, the wide availability of numerous inbred strains, transgenic mice, and knockout mice, allowing for very useful experiments to address molecular mechanisms in vivo.
Disclosures
The authors have nothing to disclose.
Acknowledgements
This work was funded by the core resource of the NIH P30 O’Brien center (DK 079337).
Materials
| Item | Company | Catalog No. |
| Vascular Clamps | Fine Science Tools | 00396-01 (Size B-1) |
| Dumont Forceps | Fine Science Tools | 11293-00 |
| 10-0 Needled microsuture | AROSurgical | TK-107038 |
| Straight scissors | Roboz Surgical Instrument Co. | RS-5620 |
| Low temperature cauterizer | Beaver-Visitec International | 8441000 |
| Self retaining retractor | World Precision Instruments | 14240 |
References
- Sun, H., et al. Improved surgical technique for the establishment of a murine model of aortic transplantation. Microsurgery. 18, 368-371 (1998).
- Dambrin, C., Calise, D., Pieraggi, M. T., Thiers, J. C., Thomsen, M. Orthotopic aortic transplantation in mice: a new model of allograft arteriosclerosis. J. Heart Lung Transpl. 18, 946-951 (1999).
- Calise, D., et al. Orthotopic aortic transplantation in rodents by the sleeve technique: a model system for the study of graft vascular disease. Transpl. Proc. 33, 2369-2370 (2001).
- Thomsen, M., et al. An orthotopic aortic graft mouse model to study the immunopathology of chronic vascular rejection. Transpl. Proc. 34, 2833-2835 (2002).
- Benza, R. L., George, J. F. Aortic graft transplantation in mice. J. Heart Lung Transpl. 21, 1319-1321 (2002).
- Chereshnev, I., et al. Mouse model of heterotopic aortic arch transplantation. The Journal of surgical research. 111, 171-176 (2003).
- Cho, H. R., et al. Improved surgical technique for heterotopic aortic transplantation in mice. J. Korean Med. Sci. 22, 12-15 (2007).
- Zhong, R. Organ transplantation in mice: current status and future prospects. Microsurgery. 19, 52-55 (1999).
- George, J. F., Pinderski, L. J., Litovsky, S., Kirklin, J. K. Of mice and men: mouse models and the molecular mechanisms of post-transplant coronary artery disease. J. Heart Lung Transpl. 24, 2003-2014 (2005).
- Gorska, P. Principles in laboratory animal research for experimental purposes. Med Sci Monit. 6, 171-180 (2000).
- Landi, M. S., Kreider, J. W., Lang, C. M., Bullock, L. P. Effects of shipping on the immune function in mice. Am. J. Vet. Res. 43, 1654-1657 (1982).
- Sun, H., et al. Improved surgical technique for the establishment of a murine model of aortic transplantation. Microsurgery. 18, 368-371 (1998).
- Chen, B., et al. Carbon monoxide rescues heme oxygenase-1-deficient mice from arterial thrombosis in allogeneic aortic transplantation. Am. J. Pathol. 175, 422-429 (2009).
- Corry, R. J., Winn, H. J., Russell, P. S. Heart transplantation in congenic strains of mice. Transpl. Proc. 5, 733-735 (1973).
- Chang, E. I., et al. Vascular anastomosis using controlled phase transitions in poloxamer gels. Nature medicine. 17, 1147-1152 (2011).

