Encapsulation Thermogenic Preadipocytes for Transplantation into Adipose Tissue Depots
Summary
Here, we present a protocol for encapsulation of catabolic cells, which consume lipids for heat production in intra-abdominal adipose tissue and increase energy dissipation in obese mice.
Abstract
Cell encapsulation was developed to entrap viable cells within semi-permeable membranes. The engrafted encapsulated cells can exchange low molecular weight metabolites in tissues of the treated host to achieve long-term survival. The semipermeable membrane allows engrafted encapsulated cells to avoid rejection by the immune system. The encapsulation procedure was designed to enable a controlled release of bioactive compounds, such as insulin, other hormones, and cytokines. Here we describe a method for encapsulation of catabolic cells, which consume lipids for heat production and energy dissipation (thermogenesis) in the intra-abdominal adipose tissue of obese mice. Encapsulation of thermogenic catabolic cells may be potentially applicable to the prevention and treatment of obesity and type 2 diabetes. Another potential application of catabolic cells may include detoxification from alcohols or other toxic metabolites and environmental pollutants.
Introduction
Increasing incidence of chronic diseases1 has stimulated studies on transplantation of therapeutic cell populations2. Syngenic or allogenic stem cells are the most commonly used cell types for these applications2. However, these treatments do not allow control of differentiation and migration of stem cells after implantation and are not cost efficient. Transplantation of genetically modified cells with beneficial functions anticipates improving the treatment of many diseases. However, genetic cell modifications are recognized by the host’s immune system, therefore, these treatments require immunosuppression3. Encapsulation of cells producing insulin has been developed by Dr. Chang4. The technique is based on encapsulation of cells in alginate droplets that are immersed into a calcium chloride solution. Alginate molecules consist of mannuronic (M) and guluronic acid (G) and can be connected by Ca2+. After gelation, the beads are suspended a poly-L-lysine (PLL) solution. During this step, PLL binds to G and M in the alginate molecules which establishes the capsule’s membrane. The porosity of the capsule’s membrane can be modulated by varying the M and PLL concentrations, the incubation time, and temperature. The binding of PLL also depends on the type and concentration of alginate. Alginate matrices crosslinked with Ca2+ ions, are unstable in the physiological environment or in common buffer solutions with high concentration of phosphate and citrate ions. These buffers can extract Ca2+ from the alginate and liquefy the core. Liquefaction of the alginate core provides space inside the capsules for cellular movement and growth. Cells encapsulated in polyanionic alginate with polycationic poly-L-lysine (APL) are impermeable for immunoglobulins but have influx of nutrients and efflux of toxins. These APL's properties enable the long term survival of encapsulated cells after transplantation into genetically different hosts. Elliott et al. reported the survival of functioning encapsulated porcine pancreatic cells in a human patient nine years after implantation5.
Encapsulation techniques can be classified into microencapsulation (3-800 µm) and macroencapsulation (larger than 1,000 µm). Microcapsules are more durable than macrocapsules6. Since its discovery by Dr. Chang and colleagues in 1964, microencapsulation has been widely used for the encapsulation of anabolic cells producing insulin, other hormones, and bioactive molecules7. These treatments faced several challenges in the host tissue including fibrosis and immune response8. Initially, the side effects related to the quality of biopolymers have been resolved. However, transplantation of anabolic cells still initiates side effects, such as fibrosis, as a result of hormone overproduction outside of a specialized gland.
In recent decades, obesity and type 2 diabetes has reached epidemic proportions9. More than 30% of adult people worldwide are overweight and obese10. Increased intra-abdominal (iAb) fat formation increases incidence of chronic inflammation and promotes type 2 diabetes, cardiovascular disease, certain cancers, and other morbidities11-13. Several lines of evidence suggested that pathogenesis associated with iAb fat can be averted by specific adipocytes. Recent studies have shown that transplantation of subcutaneous adipocytes into iAb region can improve metabolism and decrease obesity and insulin resistance in rodents in vivo14. Effective reduction of obesity and insulin resistance has been associated with thermogenic adipocytes capable of dissipating energy in form of heat15,16. Thermogenic modification of adipocytes can be achieved by stable transfection of genes participating in the mitochondrial proton uncoupling, such as uncoupling protein 1 (Ucp1) or of genes regulating expression of Ucp1 and other thermogenic genes15,16. Our recent studies showed that deficiency in aldehyde dehydrogenase 1 a1 (Aldh1a1) leads to the thermogenic remodeling of iAb fat that reduces obesity and insulin resistance in these mice17,18. Notably, encapsulation of thermogenic Aldh1a1 deficient (Aldh1a1-/-) preadipocytes mediates same therapeutic effect in iAb fat in obese wild type mice, suggesting new therapeutic opportunities for treatment of iAb fat18. In experimental settings, encapsulated cells enable researchers to study effects of specific cell populations in a cost effective manner19. Here we discuss the method of encapsulation of a thermogenic catabolic cell line and its laboratory and therapeutic application in a mouse model of obesity. The protocol describes three phases for microcapsule production (Figure 1): the formation of the alginate microbeads (Figure 1A), the formation of the polycationic poly-L-lysine (PLL) membranes on the surface of microbeads (Figure 1B), and the removal of the alginate cores (Figure 1C).
Protocol
The study protocol was approved by The Ohio State University Ethics Committees. Animal experiments were approved by IACUC protocol. All procedures were performed under the level 2 biosafety cabinet with laminar flow. We followed all standard safety requirements and procedures. The microencapsulation technique for preparation of microcapsules has been performed as described17,18.
1. Preparations of Materials
- Prepare 10 ml of 2% sodium alginate solution in autoclaved physiological saline (0.9% NaCl in water). Prepare 0.05% PLL solution in physiological saline. Prepare these solutions the day before and stir overnight. Filter the solutions with 0.22 μm filter before use.
- Prepare 50 mM sodium citrate and 100 mM CaCl2 in 0.9% NaCl solutions and autoclave them.
- Autoclave all solutions, needles, electrodes, and beakers. Thoroughly clean needles with wires to avoid clogging.
- Determine the appropriate amount of cells to use for the capsules (102 microcapsules are needed for every cm2 of well and there are approximately 500 cells per capsule18). Use 1 ml of sodium alginate per two million cells.
- Prepare a ‘Fibroblast Growth Medium’ containing 10% calf serum, and 100 U/ml penicillin/streptomycin in a high glucose (4,500 mg/l glucose) Dulbecco's Modified Eagle's medium (DMEM).
- Prepare a ‘Differentiation Medium I’ containing 10% fetal bovine serum, 10 µg/ml insulin, 1 µM dexamethasone, 0.5 mM 3-isobutyl-1-methyl xanthine, and 100 U/ml penicillin/streptomycin in DMEM.
- Prepare a ‘Differentiation Medium II’ containing 10% fetal bovine serum, 10 µg/ml insulin, and 100 U/ml penicillin/streptomycin in DMEM.
- Prepare lysis buffer containing one protease inhibitor tablet per 10 ml Radio-Immunoprecipitation Assay buffer (RIPA buffer).
2. Alginate Microbeads Preparation (Figure 1A)
- Remove old medium from cell culture flask. Rinse cells with 10 ml of PBS. Bring cells in suspension with 0.25% trypsin-EDTA (2 ml per one confluent T175 flask).
- Count cells in cell suspension using a hemocytometer. Use 10 ml aliquot from the cell suspension and count cells according to manufacturer’s instructions. Centrifuge the remaining cells in centrifugation medium at 480 x g at room temperature for 5 min.
- Suspend the cell pellet in sodium alginate as described in step 1.4. Transfer the sodium alginate-cell solution to a 5 ml syringe.
- Remove air bubbles in the solution, add a 23-gauge needle and invert the syringe to create a 1 ml pocket of air.
- Place a small beaker (180 ml) containing 144 ml of 100 mM CaCl2 solution under the needle spout of the encapsulator. Attach the electrode to the encapsulator with the tip approximately 2.5 cm above the surface of the 100 mM CaCl2 solution.
- Place tightly the syringe containing the sodium alginate-cell solution in the syringe pump. Attach the rubber tube to the opening of the syringe. Push the plunger until the sodium alginate-cell solution enters halfway through the tube. Adjust the voltage to 5.4 kV. Set the 12.06 mm diameter on the syringe pump. Adjust the speed to 3 ml/hr. Start the pump. Turn on the encapsulator and maintain the voltage at 5.4 kV.
- Close the window of the hood and avoid any unnecessary vibration till the end of formation of the alginate microbeads. After all solution passes through the needle, solidify the alginate ball-shaped microbeads in the 100 mM CaCl2 solution for additional 20 min before coating with PLL.
3. Coating Microbeads with PLL (Figure 1B)
- Remove the beaker containing the sodium alginate-cell ball-shaped microbeads and transfer these microbeads into a 50 ml centrifugation tube. Remove CaCl2 solution from the microbead pellet.
- Wash the alginate-cell ball-shaped microbeads by adding 30 ml of 0.9% NaCl. Shake the tube by hand gently. Remove 0.9% NaCl with 25 ml pipette after the microbeads have precipitated by gravitation. Repeat two more times for a total of 3 washes.
- Use 10 ml of 0.05% PLL solution for every 1 ml of sodium alginate solution. Add the 0.05% PLL and vortex at 1,000 rotations per min for 10 min.
Note: Usually, 10 min is sufficient for PLL coating. - After PLL coat is formed, remove the PLL solution and wash the capsules 3 times as described in 3.2.
4. Removal of Alginate Core (Figure 1C)
- Add 30 ml of 50 mM sodium citrate solution. Wait 5 min or until all the sodium alginate is dissolved. Wash capsules three times as described in step 3.1.1.
- Remove the 0.9% NaCl, add 20 ml of culture medium to the 50 ml tube and transfer all capsules containing the cells to a cell culture flask. Handle encapsulated cells under standard cell culture conditions18.
5. In Vitro Applications to Study Xenograft and Host Cell Interactions or Kinetics of Metabolite Influx/Efflux between Cells (Figure 2)
- Culture host cells on 24 well plate until confluent for co-cultures. Use the ‘Fibroblast Growth Medium’ for culturing preadipocytes.
- Transfer microcapsules into 24 well plate containing confluent host cells. Add microcapsules to achieve a monolayer (102 microcapsules/cm2 of well).
- Induce pre-adipocyte differentiation with Differentiation Medium I. Every 48 hr, change media to Differentiation Medium II for six days. Lyse cells in RIPA buffer. Use 50 µg protein per condition to analyze protein expression using Western blot.
6. In Vivo Application for Treatment of Obesity (Figure 3)
- Mix 4% isoflurane with oxygen for induction of anesthesia and 2% isoflurane with oxygen for maintenance of anesthesia. Confirm adequate anesthetic depth by toe pinch.
- Apply anti-itch ointment on eyes to protect the corneas from drying out.
- Use encapsulated cells that are permanently labeled with artificial fluorescence protein, such as green fluorescence protein (GFP).
- Use a 3 ml syringe and a 20 gauge needle to inject encapsulated cells.
- Inject 0.5 x 106 cells suspended in 0.2 ml of PBS into each iAb fat depot that is located in the intraperitoneal area between a gonad and kidney as shown in Figure 3. Use this volume of PBS and cell number for mice with an average weight of 40 g. Adjust cell number and volume in mice that have different weight or for determination of dose-dependent effects.
Representative Results
Figure 1 shows that every step of microbeads production could be controlled under the microscope. Figure 2A shows how to co-culture adipocytes with a monolayer of encapsulated cells. Figure 2B is a representative example of a quantitative study using adipocyte/microcapsules co-cultures that were described in section 5. Lysates of adipocytes were analyzed using Western blot. Encapsulated cells were not analyzed in this experiment. Primary ATGL and β-actin antibodies were used at a 1:1,000 dilution. The ratio of ATGL to β-actin are shown as mean S.D. of three independent experiments. Similar co-culture approaches could be used to analyze mRNA and study effects of encapsulated cell and adipocyte interactions in co-cultures. Data shows that encapsulated thermogenic Aldh1a1-/- adipocytes induce significantly higher levels of ATGL lipase and lipolysis18 in adipocytes compared to encapsulated WT adipocytes.
Figure 3 shows that GFP indicates location and the integrity of capsules in the host adipose tissue. The expression of GFP is also an indicator of cell viability.
Qualitative evaluation
The quality of encapsulation or implantation of encapsulated cells could be evaluated by microscopy, MRI, and using immunohistochemical analysis of treated adipose tissues18.
Quantitative approach
Given the unique expression of GFP in surviving transplanted cells, the measurement of GFP expression levels allow encapsulated cells to be quantified in tissue as described18.GFP and other proteins can be detected using specific anti-GFP antibodies in a homogenate of a whole adipose tissue depot. The GFP levels of protein in this homogenate provide information about the number of viable implanted cells.
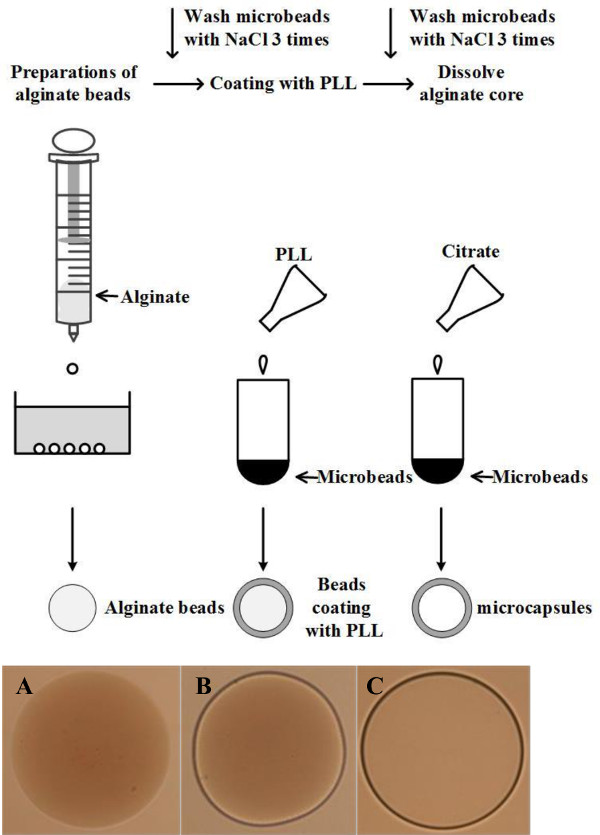
Figure 1: A schematic of the procedure for microcapsule production. (A) alginate microbeads under a microscope (20X). (B) the outer layer after coating with PLL (20X). (C) the final microcapsule after dissolving the alginate core (20X). Please click here to view a larger version of this figure.
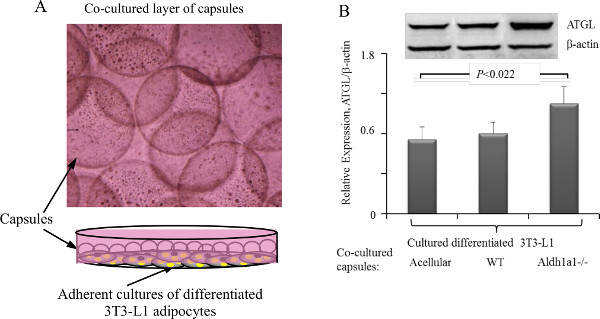
Figure 2: Microcapsule co-cultures with adherent cell line cultures. (A) Schematic (lower panel) of a co-culture of microcapsules (circles floating in media) with adherent adipocytes. (B) Comparison of expression of ATGL from 3T3-L1 adipocytes co-cultured with acellular, WT, or Aldh1a1-/- adipocyte containing microcapsules. Significance P value was determined using Mann-Whitney U test. Upper inserts shows one representative Western blot. Please click here to view a larger version of this figure.
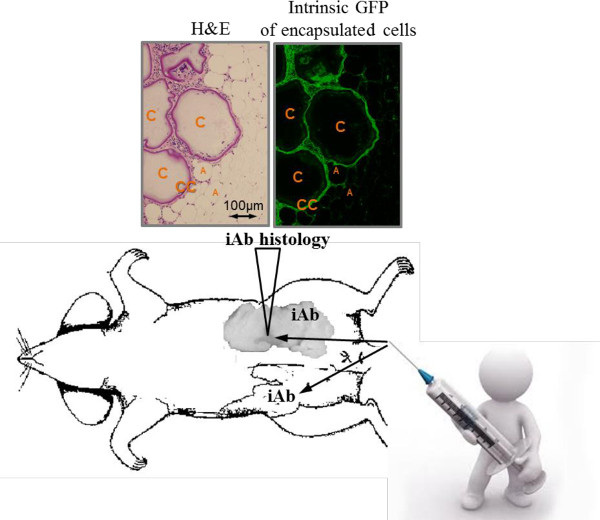
Figure 3: Schematic of an intra-abdominal (iAb, visceral) fat injection with encapsulated cells. The photographic image of the injected iAb fat pad shows discolored clusters of encapsulated cells 80 days after the transplantation (triangle). These iAb fad pad were embedded into paraffin and analyzed. Hematoxylin and eosin (H&E) staining shows clusters of encapsulated cells (arrows), implanted microcapsules (C), encapsulated cells (CC), host adipocytes (A). Same image analyzed under fluorescent light shows GFP-labeled transplanted cells on the inside of round intact capsules. Please click here to view a larger version of this figure.
Discussion
Various methods have been used to encapsulate cells, including drying, extrusion, and emulsion19. In this method, the alginate beads are extruded through a needle, then coated with PLL and the alginate core will be dissolved to complete the encapsulation. Although this method has been used for years, formation of the beads with the desired size and spherical shape is still challenging. The size of the capsules is highly dependent on the viscosity of sodium alginate solution, the extruder diameter and the distance between the needle tip and the CaCl2 solution20. The shorter the distance between the needle tip and the surface of CaCl2 solution is, the smaller beads are produced. The described protocol leads to the production of APL with pores (<32 kD). The pore size could be experimentally tested using fluorescent immunoglobulins (>32 kD) and fluorescent small peptides as previously described18. This size of the pore is sufficient to support survival of cells in in vitro cultures for 2 weeks and after in vivo implantation into adipose tissue for at least 80 days18. The microbead shape is an important factor influencing the survival of encapsulated cells. Cracked beads with many satellite microbeads increase protrusion of cells21. Satellite formation occurred when lower concentration and low viscosity intermediate-G alginate solutions are applied22, while formation of the tail is due to high-G alginate23. As the shearing force may also contribute to cracked beads, we recommend a gentle washing procedure for microbeads and propose optimized conditions for production of robust capsules suitable for in vitro co-cultures and engrafting into tissues in vivo.
Microcapsulation has been proven to be an effective method for implantation for protecting biologicals such as cells, cytokines, enzymes, hormones, and bioabsorbents from the environment and immune response24. The controlled release of hormones or cytokines from encapsulated cells was tested for its therapeutic effects in a wide variety of diseases, including obesity18, diabetes mellitus5, liver failure25 and anemia26. However, the efficacy of engrafted encapsulated anabolic cells is often diminished due to fibrosis. Here we describe the new application of thermogenic catabolic cells for treatment of obesity and insulin resistance18. Encapsulation of Aldh1a1-/- adipocytes, and possibly other catabolic cells, have several advantages compared to encapsulation of anabolic cells. Aldh1a1-/- preadipocytes spontaneously adhere to the inner surface of the APL membrane18 and differentiate in the adipogenic environment of iAb fat that prevents their excessive proliferation in capsules and fast rupture of capsules. In addition, these cells have diminished inflammatory responses and catabolic properties27. Immunohistochemical data shows that the implantation of APL encapsulated Aldh1a1-/- adipocytes into iAb fat for 80 days was not accompanied by pronounced immune response and fibrosis in mice18; however, more studies need to be performed in the future to assess potential inflammatory effects. Primarily, Aldh1a1 deficiency increases expression of thermogenic Ucp1 in adipocytes. Aldh1a1 deficiency reduces autocrine production of retinoic acid increasing levels of retinaldehyde28. This pathway influences numerous transcriptional pathways28,29, which could be involved in the production of intrinsic and paracrine thermogenic factors. Notably, encapsulated Aldh1a1-/- adipocytes cells produce similar thermogenic response in host obese WT mice. Encapsulated Aldh1a1-/- adipocytes appear to produce paracrine factor(s) increasing number of Ucp1-positive cells in the host WT adipose tissue18. Together, these thermogenic responses resulted in a preferential reduction of injected iAb fat. A phenotype of immortalized Aldh1a1-/- preadipocytes exerts many properties that make this gene modification a promising candidate for encapsulation technology reducing diet-induced obesity.
Recently many other biologicals including cytokine irisin, miR-133a30, meteorin like hormone31, parathyroid-hormone-related protein32 were reported to exert thermogenic remodeling of subcutaneous white adipose tissue. More studies are needed to determine if these thermogenic factors are suitable for encapsulation technologies and therapies against obesity in subcutaneous and/or iAb fat. Microcapsules containing catabolic cells can be potentially suitable to remove an excess of deleterious metabolites in a toxic environment. For example, patients may benefit from facilitated catabolism of alcohol or deoxyglucosone, a first reactive metabolite in patients with diabetes. However, future studies are needed to determine if these applications could have therapeutic benefits.
In summary, these studies provide a proof-of-concept that amplification of the thermogenic response in iAb fat could be initiated by a small subset of encapsulated Aldh1a1-/- preadipocytes. Moreover, these studies have demonstrated feasibility of tissue-specific treatment with injected implants of encapsulated catabolic thermogenic cells.
Disclosures
The authors have nothing to disclose.
Acknowledgements
We would like to thank Jennifer Petrosino and David DiSilvestro for editorial help. This research was supported by Award Number 20020728 from the American Egg Board and Award Number 10040042 from Novo Nordisk Pharmaceuticals as well as by the Food Innovation Center, Office for International Affairs, Center for Advanced Functional Foods Research, and Entrepreneurship at OSU as well as the National Science Foundation grant EEC-0914790 (L.J.L). The project described was supported by Award Number R21OD017244 (O.Z.) and UL1RR025755 (OSUCCC) from the National Center for Research Resources, funded by the Office of the Director, National Institutes of Health (OD) and supported by the NIH Roadmap for Medical Research and NCI P30CA16058. The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Center for Research Resources or the National Institutes of Health.
Materials
| Encapsulation device (VAR V1) | Nisco | LIN-0042 | None |
| KD scientific syringe pump | KD scientific | 780100Y | None |
| Olympus microscope | Olympus Optical | IX70-S8F2 | None |
| Sodium alginate | Sigma | MKBP8122V | None |
| Poly-l-lysine hydrobromide (PLL) | Sigma | 020M5006V | None |
| Calcium chloride | Sigma | SLBJ2662V | None |
| Sodium citrate tribasic dihydrate | Sigma | 030M0200 | None |
| Sodium chloride | Sigma | SLBD2595V | None |
| Mini-PROTEAN TGX Gels | Bio-Rad | 456-1093 | None |
| ATGL primary antibody (from rabbit) | Cell Signaling | 2138S | None |
| Secondary anti body (anti rabbit) | LI-COR | 926-68071 | None |
| Radio-Immunoprecipitation Assay (RIPA) buffer | Boston BioProducts | D25Y6Z | None |
| Phosphate buffered saline (PBS) | Sigma | RNBD2893 | None |
| Trypsin | Gibco | 25200-056 | None |
| Cortizone 10 anti-itch ointment | Cortizone 10 | C4029138 | None |
| Dulbecco's Modified Eagle Medium (DMEM) | Gibco | 11965-092 | None |
| Newborn calf serum (CS) | Sigma | N4762 | None |
| Fetal bovine serum (FBS) | Sigma | F4135 | None |
| 3-Isobutyl-1-methylxanthine (IBMX) | Sigma | I0516 | None |
| Dexamethasone | Sigma | D4902 | None |
| Insulin (bovine) | Sigma | I5879 | None |
| Protease inhibitor cocktail tablets | Roche | 4693159001 | None |
References
- Vogeli, C., et al. Multiple chronic conditions: prevalence, health consequences, and implications for quality, care management, and costs. Journal of general internal medicine. 22, 391-395 (2007).
- Vija, L., et al. Mesenchymal stem cells: Stem cell therapy perspectives for type 1 diabetes. Diabetes & metabolism. 35, 85-93 (2009).
- Acarregui, A., Orive, G., Pedraz, J. L., Hernandez, R. M. Therapeutic applications of encapsulated cells. Methods in molecular biology. 1051, 349-364 (2013).
- Chang, T. M. Semipermeable Microcapsules. Science. 146, 524-525 (1964).
- Elliott, R. B., et al. Live encapsulated porcine islets from a type 1 diabetic patient 9.5 yr after xenotransplantation. Xenotransplantation. 14, 157-161 (2007).
- Lim, F., Sun, A. M. Microencapsulated islets as bioartificial endocrine pancreas. Science. 210, 908-910 (1980).
- Vos, P., Spasojevic, M., Faas, M. M. Treatment of diabetes with encapsulated islets. Advances in experimental medicine and biology. 670, 38-53 (2010).
- Cotton, C. K. Engineering challenges in cell-encapsulation technology. Trends in biotechnology. 14, 158-162 (1996).
- Yach, D., Stuckler, D., Brownell, K. D. Epidemiologic and economic consequences of the global epidemics of obesity and diabetes. Nature medicine. 12, 62-66 (2006).
- Ng, M., et al. Global, regional, and national prevalence of overweight and obesity in children and adults during 1980-2013: a systematic analysis for the Global Burden of Disease Study 2013. Lancet. 384, 766-781 (2014).
- Kissebah, A. H., et al. Relation of body fat distribution to metabolic complications of obesity. The Journal of clinical endocrinology and metabolism. 54, 254-260 (1982).
- Bray, G. A., et al. Relation of central adiposity and body mass index to the development of diabetes in the Diabetes Prevention Program. The American journal of clinical nutrition. 87, 1212-1218 (2008).
- Klein, J., et al. What are subcutaneous adipocytes really good for. Experimental dermatology. 16, 45-70 (2007).
- Tran, T. T., Yamamoto, Y., Gesta, S., Kahn, C. R. Beneficial effects of subcutaneous fat transplantation on metabolism. Cell metabolism. 7, 410-420 (2008).
- Seale, P., Kajimura, S., Spiegelman, B. M. Transcriptional control of brown adipocyte development and physiological function–of mice and men. Genes & development. 23, 788-797 (2009).
- Kozak, L. P. Genetic variation in brown fat activity and body weight regulation in mice: lessons for human studies. Biochimica et biophysica acta. 1842, 370-376 (2014).
- Zhang, X., He, H., Yen, C., Ho, W., Lee, L. J. A biodegradable, immunoprotective, dual nanoporous capsule for cell-based therapies. Biomaterials. 29, 4253-4259 (2008).
- Yang, F., et al. The prolonged survival of fibroblasts with forced lipid catabolism in visceral fat following encapsulation in alginate-poly-L-lysine. Biomaterials. 33, 5638-5649 (2012).
- Chang, T. M. Artificial cells with emphasis on bioencapsulation in biotechnology. Biotechnology annual review. 1, 267-295 (1995).
- Chang, T. M. Hybrid artificial cells: microencapsulation of living cells. ASAIO journal. 38, 128-130 (1992).
- Koo, J., Chang, T. M. Secretion of erythropoietin from microencapsulated rat kidney cells: preliminary results. The International journal of artificial organs. 16, 557-560 (1993).
- Weidenauer, U., Bodmer, D., Kissel, T. Microencapsulation of hydrophilic drug substances using biodegradable polyesters. Part I: evaluation of different techniques for the encapsulation of pamidronate di-sodium salt. Journal of microencapsulation. 20, 509-524 (2003).
- Smidsrod, O., Skjak-Braek, G. Alginate as immobilization matrix for cells. Trends in biotechnology. 8, 71-78 (1990).
- Lewinska, D., Rosinski, S., Werynski, A. Influence of process conditions during impulsed electrostatic droplet formation on size distribution of hydrogel beads. Artificial cells, blood substitutes, and immobilization biotechnology. 32, 41-53 (2004).
- Chan, E. S., Lee, B. B., Ravindra, P., Poncelet, D. Prediction models for shape and size of ca-alginate macrobeads produced through extrusion-dripping method. Journal of colloid and interface science. 338, 62-72 (2009).
- Bhujbal, S. V., Paredes-Juarez, G. A., Niclou, S. P., de Vos, P. Factors influencing the mechanical stability of alginate beads applicable for immunoisolation of mammalian cells. Journal of the behavior of biomedical materials. 37, 196-208 (2014).
- Gushchina, L. V., Yasmeen, R., Ziouzenkova, O. Moderate vitamin A supplementation in obese mice regulates tissue factor and cytokine production in a sex-specific manner. Archives of biochemistry and biophysics. 539, 239-247 (2013).
- Ziouzenkova, O., et al. Retinaldehyde represses adipogenesis and diet-induced obesity. Nature. 13, 695-702 (2007).
- Yasmeen, R., Jeyakumar, S. M., Reichert, B., Yang, F., Ziouzenkova, O. The contribution of vitamin A to autocrine regulation of fat depots. Biochimica et biophysica acta. 1821, 190-197 (2012).
- Liu, W., et al. miR-133a regulates adipocyte browning in vivo. PLoS genetics. 9, e1003626 (2013).
- Rao, R. R., et al. Meteorin-like is a hormone that regulates immune-adipose interactions to increase beige fat thermogenesis. Cell. 157, 1279-1291 (2014).
- Kir, S., et al. Tumour-derived PTH-related protein triggers adipose tissue browning and cancer cachexia. Nature. 513, 100-104 (2014).

