Adeno-Associated Virus-Mediated Delivery of CRISPR for Cardiac Gene Editing in Mice
Summary
Here we provide a detailed protocol to carry out in vivo cardiac gene editing in mice using recombinant Adeno-Associated Virus(rAAV)-mediated delivery of CRISPR. This protocol offers a promising therapeutic strategy to treat dystrophic cardiomyopathy in Duchenne muscular dystrophy and can be used to generate cardiac-specific knockout in postnatal mice.
Abstract
The clustered, regularly interspaced, short, palindromic repeat (CRISPR) system has greatly facilitated genome engineering in both cultured cells and living organisms from a wide variety of species. The CRISPR technology has also been explored as novel therapeutics for a number of human diseases. Proof-of-concept data are highly encouraging as exemplified by recent studies that demonstrate the feasibility and efficacy of gene editing-based therapeutic approach for Duchenne muscular dystrophy (DMD) using a murine model. In particular, intravenous and intraperitoneal injection of the recombinant adeno-associated virus (rAAV) serotype rh.74 (rAAVrh.74) has enabled efficient cardiac delivery of the Staphylococcus aureus CRISPR-associated protein 9 (SaCas9) and two guide RNAs (gRNA) to delete a genomic region with a mutant codon in exon 23 of mouse Dmd gene. This same approach can also be used to knock out the gene-of-interest and study their cardiac function in postnatal mice when the gRNA is designed to target the coding region of the gene. In this protocol, we show in detail how to engineer rAAVrh.74-CRISPR vector and how to achieve highly efficient cardiac delivery in neonatal mice.
Introduction
The clustered, regularly interspaced, short palindromic repeat (CRISPR) technology represents the most recent advancement in genome engineering and has revolutionized the current practice of genetics in cells and organisms. CRISPR-based genome editing utilizes a single guide RNA (gRNA) to direct a CRISPR-associated endonuclease such as CRISPR-associated protein 9 (Cas9) and Cpf1 to a genomic DNA target that is complementary in sequence to the protospacer encoded by the gRNA with a specific protospacer adjacent motif (PAM)1,2. The PAM varies depending on the bacterial species of the Cas9 or Cpf1 gene. The most commonly used Cas9 nuclease derived from Streptococcus pyogenes (SpCas9) recognizes a PAM sequence of NGG that is found directly downstream of the target sequence in the genomic DNA, on the non-target strand. At the target site, the Cas9 and Cpf1 proteins generate a double-strand break (DSB), which is then repaired via intrinsic cellular DNA repair mechanisms including non-homologous end joining (NHEJ) and homology-directed repair (HDR) pathways3,4. In the absence of a DNA template for homologous recombination, the DSB is primarily repaired by the error-prone NHEJ pathway, which can result in insertions or deletions (indels) of nucleotides causing frameshift mutations if the DSB were created in the coding region of a gene5,6. Thus, the CRISPR system has been widely used to engineer loss-of-function mutations in various cellular models and whole organisms, in order to investigate the function of a gene. Moreover, the catalytically inactive Cas9 and Cpf1 have been developed to transcriptionally and epigenetically modulate the expression of target genes7,8.
More recently, both SpCas9 and SaCas9 (derived from Staphylococcus aureus) have been packaged into recombinant adeno-associated virus (rAAV) vectors for postnatal gene correction in the animal model of human diseases, particularly, Duchenne muscular dystrophy (DMD)9,10,11,12,13,14,15. DMD is a fatal X-linked recessive disease caused by mutations in the DMD gene, which encodes dystrophin protein16,17, affecting approximately one in 3500 male births according to newborn screening18,19. It is characterized by progressive muscle weakness and wasting. The DMD patients usually lose the ability to walk between 10 and 12 years of age and die of respiratory and/or cardiac failure by the age of 20-30 years20. Notably, cardiomyopathy develops in >90% of DMD patients and represents the leading cause of death in DMD patients21,22. Although Deflazacort (an anti-inflammation drug) and Eteplirsen (an exon skipping medicine for skipping exon 51) have recently been approved for DMD by Food and Drug Administration (FDA)23,24, none of these treatments correct the genetic defect at the genomic DNA level. The mdx mouse, which carries a point mutation at exon 23 of the dystrophin gene, has been widely used as a DMD model25. Furthermore, we previously demonstrated that the functional dystrophin expression was restored by in-frame deletion of the genomic DNA covering exons 21, 22 and 23 in skeletal muscles of mdx mice using intramuscular Ad-SpCas9/gRNA delivery9, and in the heart muscles of mdx/utrophin+/- mice using intravenous and intraperitoneal rAAVrh.74-SaCas9/gRNA delivery26.
In this protocol, we describe in detail every step in postnatal cardiac gene editing to restore dystrophin in mdx/utrophin+/- mice using rAAVrh.74-SaCas9/gRNA vectors, from gRNA design to dystrophin analysis in the heart muscle sections.
Protocol
The animals used in this protocol were maintained at The Ohio State University Laboratory Animal Resources in accordance with animal use guidelines. All animal studies were authorized by the Institutional Animal Care, Use, and Review Committee of the Ohio State University.
1. Design and Cloning of gRNAs into the CRISPR Vector
- Select 2 gRNAs targeting dystrophin intron 20 and intron 23 (i20 and i23) for SaCas9: i20: 5'-GGGCGTTGAAATTCTCATTAC CAGAGT and i23: 5'-CACCGATGAGAGGGAAAGGTC CTGAAT (the PAM sequence is underlined and italicized).
NOTE: The target sites for i20 and i23 are about 23 kilo base pair (kbp) away. To disrupt a coding gene, two gRNAs targeting the common exons with the NNGRRT PAM sequence for SaCas9 (preferably NNGAAT, NNGGGT and NNGAGT) and distance within 20 kbp are recommended. - Use the following PCR parameters to anneal the gRNA oligos (100 µmol/L) in 1x annealing buffer (the 5x annealing buffer stock contains 0.5 mol/L K-acetate, 150 mmol/L HEPES-KOH, 10 mmol/L Mg-acetate, pH7.4): 95 °C 10 min, 90 cycles of 95 to 59 °C with a 0.4 °C decrease per cycle for 20 s, 90 cycles of 59 to 32 °C with a 0.3 °C decrease per cycle for 20 s, 20 cycles of 32 to 26 °C with a 0.3 °C decrease per cycle for 20 s.
- Ligate the annealed gRNA oligos into the pX601-CMV-SaCas9-mPA-U6-gRNA using BsaI and T4 DNA ligase in one reaction (1 µL 50 ng/µL pX601-CMV-SaCas9-mPA-U6-gRNA, 1 µL annealed oligos, 1.5 µL 10x T4 DNA ligase buffer, 0.5 µL T4 DNA ligase, 1 µL BsaI, 10 µL ddH2O), with the following reaction condition in a PCR cycler: 5 cycles of [37 °C 5 min and 16 °C 10 min]; 37 °C 20 min; 80 °C 5 min.
- Transform the 15 µL ligation product into 30 µL competent cells, plate the cells in agar plate with 100 µg/mL Ampicillin, and culture at 37 °C for 24 h.
- Pick up 5-10 colonies and culture in LB medium containing 100 µg/mL Ampicillin at 37 °C, 250 rpm in a shaker incubator for 3-4 h.
- Screen the colonies by PCR: 10 µL 2x PCR master mix, 8 µL ddH2O, 1 µL E. Coli culture, 1 µL 10 µmol/L U6-forward primer, 1 µL i20 or i23 gRNA reverse primer. The PCR cycling parameters: 95 °C 5 min, 35 cycles of 95 °C 30 s, 61 °C 30 s, 72 °C 30 s.
- Prepare 5 mL culture of the positive clones by adding 4 mL fresh LB medium containing100 µg/mL Ampicillin, and culture overnight at 37 °C, 250 rpm.
- Purify the plasmid DNA using the appropriate plasmid extraction kit, and verify the plasmid by Sanger sequencing with the U6-forward primer.
- Culture C2C12 cells in DMEM supplemented with 10% FBS and 1% Pen-Strep until reaching ~70% confluency for testing the gene editing efficacy of the plasmids.
- Electroporate a total of 5 µg SaCas9/gRNA plasmid mixture in 1:1 molar ratio into 1 x 106 C2C12 cells according to the manufacturer's protocol.
- After 48 h post-transfection, harvest the C2C12 cells and extract genomic DNA.
- Use 100 ng genomic DNA as template to perform the PCR reaction for mouse dystrophin locus: forward primer 5'-GGCCAAAGCAAACTCTGGTA (1 µL 10 µmol/L), reverse primer 5'-TTTAATCCCACGTCATGCAA (1 µL 10 µmol/L), 10 µL 2x the green PCR master mix, 7 µL ddH2O, 1 µL genomic DNA (100 ng). Use PCR cycling conditions as 95 °C 5 min, 35 cycles of [95 °C 30 s, 61 °C 30 s, 72 °C 30 s], 72 °C 2 min.
2. rAAV Production
- To culture the cells, seed AD293 cells onto 20 to 40 of 15-cm dishes and maintain in DMEM supplemented with 10% FBS and 1% Pen-Strep until reaching ~90% confluency for transfection.
- Dilute 3 plasmids with 200 µL reduced serum medium (per dish): 1)10 µg pHelper; 2) 5 µg pXR(rh.74 or other serotypes) and 3) A total of 5 µg of pX601-i20 and pX601-i23 mixture (1:1 ratio). Perform polyethylenimine (PEI) transfection by mixing with 80 µL PEI and incubate at room temperature for 10 min. Add the mixture on to the AD293 cell cultures in a dropwise manner.
- For AAV virus production, after 72 h post transfection, use a cell scraper to collect both the media and the AD293 cell pellets by centrifugation at 1100 x g for 10 mins.
- Resuspend the cell pellets in 15 mL rAAV lysis buffer (50 mmol/L Tris Base, 150 mmol/L NaCl, pH 8.5) followed by 3 freeze-thaw cycles with alternating incubation in liquid nitrogen and a 42 °C water bath. Store the cell lysates in -80 °C for future processing if they are not being processed immediately.
- Filter the culture media with a 0.45-µm filter and add a 40% PEG8000 in 2.5 mol/L NaCl to a final concentration of 8%. Incubate the mixture at 4 °C for 1 h and centrifuge at 1100 x g for 15 min.
- Resuspend the pellets in rAAV lysis buffer and combine with the cell lysates from step 2.3, followed by benzonase (50 U/mL) treatment at 37 °C for 1 h.
- Centrifuge the benzonase-treated rAAV containing mixture 1100 x g for 15 min at 4 °C, and save the supernatant (the crude rAAV preparation).
3. rAAV Purification and Concentration
- Load the crude rAAV preparation onto an iodixanol gradient in an ultracentrifuge tube in the following order from the top to the bottom: crude AAV lysate (~15 mL), 8 mL 15% Iodixanol, 6 mL 25% Iodixanol, 5 mL 40% Iodixanol, 5 mL 58% Iodixanol. Fill the remaining empty volume of the tube to the top using 1x DPBS.
- Centrifuge the samples at 230,000 x g in an ultracentrifuge rotor for 120 min at 10 °C and collect 3 – 4 mL of the 40% gradient fraction containing the rAAV particles with an 18 G needle.
- Dilute the rAAV particles with 1x DPBS and centrifuge using the centrifugal filter unit (MWCO 100 kDa). Repeat this step for 3-5 times and finally concentrate the rAAV preparation to 300-500 µL.
4. rAAV Titering
- Extract the genomic DNA from 2 µL concentrated rAAV, precipitate with 3 M NaOAc in ethanol, and resuspend in 20 µL ddH2O.
- Obtain the standard curve for rAAV quantitation by serial dilution of the pX601 plasmid: 1 x 109, 1 x 108, 1 x 107, 1 x 106, 1 x 105 copies in each 10 µL ddH2O. Dilute the DNAase-treated rAAV samples as 1:10, 1:50, 1:100, and 1:1000.
- Determine rAAV titer by quantitative real-time PCR (qPCR) using qPCR Kit according to manufacturer's instruction in a Real-Time PCR system: 1 µL 10 µmol/L of each primer (5'-GGATTTCCAAGTCTCCACCC and 5'-TCCCACCGT ACACGCCTAC), 5 µL 2x PCR master mix, 1 µL diluted rAAV samples and 2 µL ddH2O. Run the reaction with the following cycling parameters: 95 °C 3 min, 45 cycles of [95 °C 5 sec and 61 °C 20 sec].
5. Intravenous and Intraperitoneal Injection of rAAVrh.74-CRISPR into Neonatal mdx/utrophin+/- Mice
- Immobilize the mdx/utrophin+/- mouse pups (day 3) by placing on ice for 1 min, and then administer with 1 x 1012 viral particles in 50 µL per mouse systemically via a retro-orbital approach or an intraperitoneal injection.
- Allow the mice to recover and place back into their home cages. At ten weeks after rAAV administration, euthanize the mice by CO2 exposure for tissue collection.
- Snap-freeze tissues for genomic DNA and RNA extraction, and use cooling isopentane in liquid nitrogen to freeze tissues with optimal cutting temperature compound for cryosectioning.
6. Analysis of Target Gene Editing Outcomes
- Genomic DNA PCR analysis
- Isolate the total genomic DNA from the heart tissues of mdx/utrophin+/- mice treated with or without rAAV.
- Use the same protocol in Step 1.12 for PCR analysis.
- RT-PCR analysis of dystrophin expression
- Extract total RNA from the heart tissues with the RNA extraction kit following the manufacturer's manual.
- To perform reverse transcription, pre-treat the total RNA with an RNase-free DNase and use 5 µg of treated RNA as the template for first-strand cDNA synthesis with the reverse transcription kit.
- Use the reverse transcription product for RT-PCR analysis of dystrophin expression with the dystrophin cDNA primers: 5'-GGCTAGAGTATCAAACCAACATCAT and 5'- TGGAGGCTTACGGTTTTATCC.
- Estimate the gene editing efficacy by qPCR analysis
- Estimate the gene editing efficacy by quantitative RT-PCR to detect the reduction of exon 22-containing transcripts using the forward primer (exon 21): 5'-TTGTCAGCTCTTCAGCCTCAAA, and the reverse primer (exon 23): 5'-AAACTCTCCTTCACAGTGTCACT. Use Gapdh as a reference gene with the forward primer: 5'- AGGTTGTCTCCTGCGACTTC and the reverse primer: 5'- GGTGGTCCAGGGTTTCTTACT.
- Run the quantitative real-time PCR (qPCR) using the qPCR Kit according to manufacturer's instruction in a Real-Time PCR system and the 2-step reaction conditions: 95 °C 3 min, 45 cycles of 95 °C 5 sec and 61 °C 30 sec.
- Immunofluorescence staining to analyze dystrophin expression
- Cut the frozen tissues using a cryostat at a thickness of 10 µm. Fix the frozen sections with 4% paraformaldehyde at room temperature for 15 min.
- Wash 2x with PBS and incubate with blocking solution (10% horse serum) for 1 h.
- Incubate the anti-dystrophin primary antibody, at 4 °C overnight.
- Wash the slides with PBS 3 x 5 min and incubate with fluorescent secondary antibodies (1:500) at room temperature for 1 h.
- Mount the slides using the mounting medium with DAPI and imaging with an inverted confocal microscope.
7. Off-target analysis by T7E1 assay
- Predict the potential off-target sites using the online CRISPR design tools such as <http://crispr.mit.edu>.
- Amplify the genomic regions covering the top 5-10 potential off-target sites by PCR: genomic DNA 200 ng, 1 µL high-fidelity DNA polymerase, 5 µL 10x PCR Buffer mix, 1.5 µL 10 µmol/L of site-specific forward and reverse primers, adjusting the total volume to 50 µL with ddH2O.
- Run the PCR products by electrophoresis, extract and purify the DNA using a gel extraction kit. Measure the concentration using spectrophotometers.
- Promote heteroduplex formation by denaturing and re-annealing the purified amplicons slowly in the Buffer 2.1 using a PCR cycler. Use the same cycling conditions as detailed in step 1.2.
- Digest the re-annealed products for 30 min at 37 °C with 0.5 µL T7E1 enzyme, and analyze the reaction by electrophoresis using 1-2% agarose gel.
NOTE: The amplicons can also be analyzed by targeted deep sequencing.
Representative Results
The efficacy of the gRNAs to induce the deletion of the target genomic DNA region should be evaluated in cell cultures before packaging into rAAV for in vivo studies. To this end, the gRNAs and SaCas9-expressing constructs were electroporated into C2C12 cells and the genomic DNA was analyzed by PCR with the primers flanking the target sites (Figure 1). A PCR product of ~500 bp indicates successful deletion of the target genomic DNA resulting from gene editing while the control cells should not yield a band due to the large size of the genomic DNA.
Once the efficacy of the gRNAs has been confirmed in cell cultures, they can be packaged into rAAVrh.74 or other serotypes (such as AAV6, 8 or 9, all showing robust cardiac gene delivery) by using the three plasmids co-transfection into AAV293 cells. We found that it is not necessary to make two individual rAAV preparation for the two gRNAs, instead, we mixed the two gRNA constructs in an equal molar ratio and produced a single rAAV preparation. The rAAV viral particles were purified using density gradient ultracentrifugation and the purity was analyzed by SDS-PAGE. The highly purified rAAV preparation would yield only three bands corresponding to the capsid proteins VP1, VP2, and VP3, respectively, on SDS-PAGE as shown in Figure 2.
The purified rAAVrh.74 viral particles were injected into day 1-3 neonates of mdx/utrophin+/- mice via retro-orbital or intraperitoneal injection. We found both methods worked well for cardiac delivery of the rAAVrh.74-CRISPR. The injected mice can be analyzed for dystrophin expression by immunofluorescence staining (Figure 3) at 10 weeks of age.
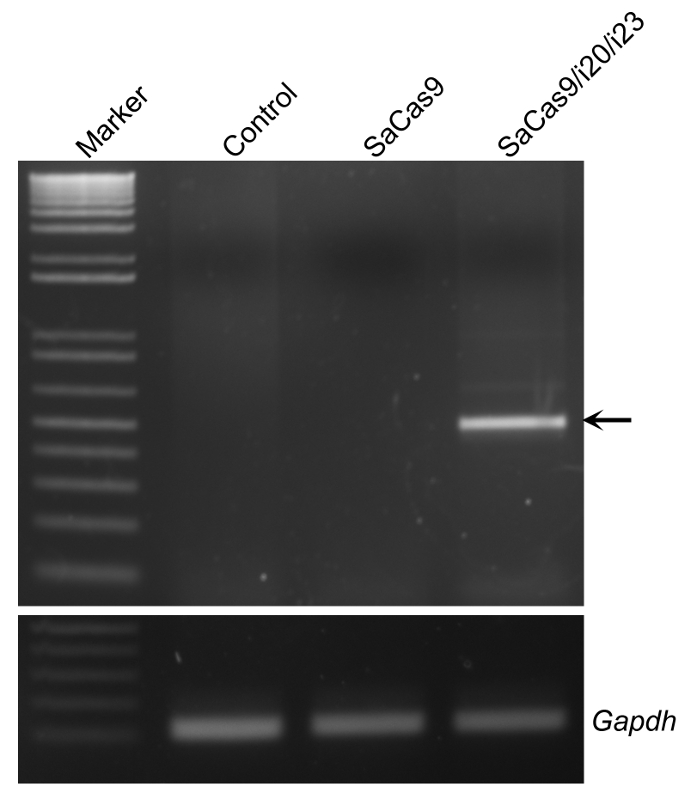
Figure 1: Validation of the gRNAs for SaCas9-mediated gene editing in C2C12 cells. The representative agarose gel electrophoresis showed the PCR products of genomic DNA extracted from electroporated C2C12 with or without SaCas9/gRNAs. The arrow indicates the PCR product resulted from gene editing. Gapdh served as a reference. Please click here to view a larger version of this figure.
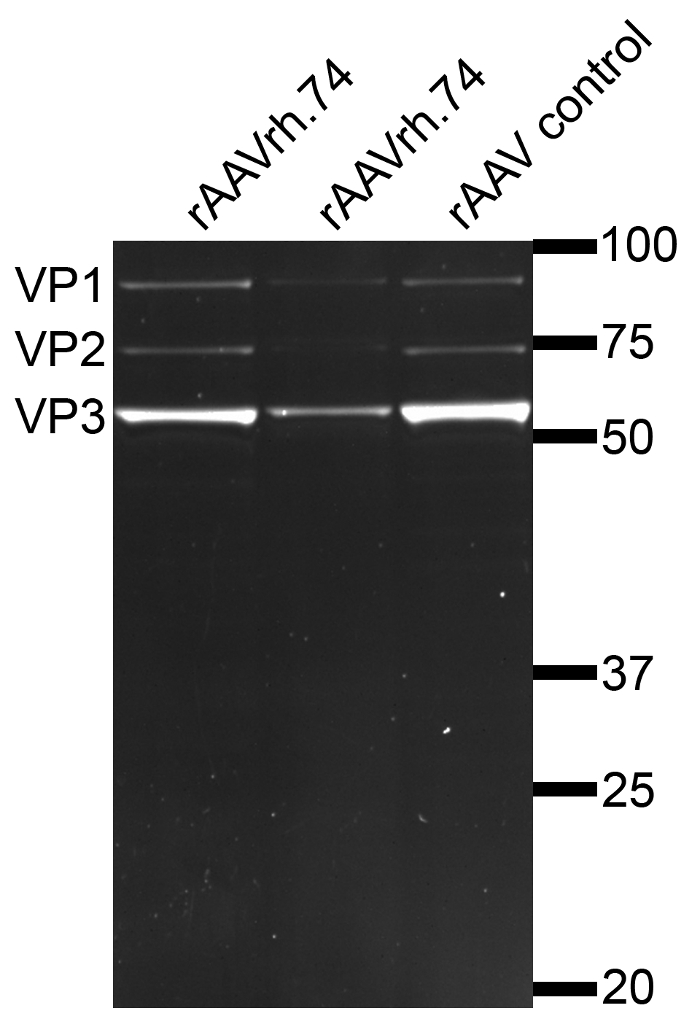
Figure 2: Analysis of rAAVrh.74 particles by SDS-PAGE. Purified rAAVrh.74 viral particles run on SDS-PAGE showed three bands, corresponding to the three capsid proteins of AAV, VP1 (87 kDa), VP2 (72 kDa) and VP3 (62 kDa), respectively. The presence of only these 3 bands indicates the high purity of the rAAV preparation without contaminating other cellular proteins. Please click here to view a larger version of this figure.
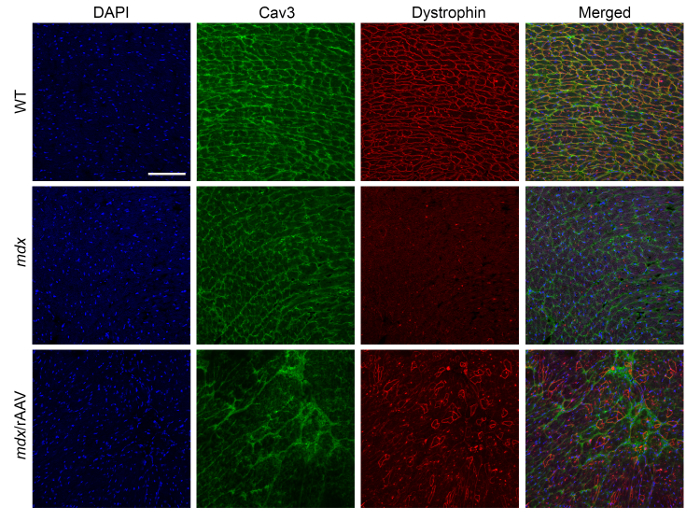
Figure 3: Immunofluorescence staining of the heart sections from mdx/utrophin+/- mice at 10 weeks following AAV-SaCas9/gRNAs treatment. The representative immunofluorescence images showed the expression of dystrophin (red) and caveolin-3 (green) in the heart sections of WT, mdx/utrophin+/- and mdx/utrophin+/- mice treated with 1 x 1012 vg AAV-SaCas9/gRNA systemically. DAPI was used to label the nuclei (blue). Scale bar: 100 µm. Please click here to view a larger version of this figure.
Discussion
In this protocol, we detail all the steps necessary to achieve in vivo cardiac gene editing in postnatal mice using rAAVrh.74-mediated delivery of SaCas9 and two gRNAs. As previously noted, this approach can be used to restore dystrophin expression in dystrophic mice as described in our work27, but it could also enable rapid reverse genetics studies of cardiac-related genes in mice without the lengthy process of the traditional knockout approach to generate and breed knockout mouse models.
In order to achieve high gene editing efficiency in the target tissue, there are several critical steps to keep in mind: 1) the efficacy of the SaCas9/gRNA to induce the deletion of targeted genomic DNA needs to be validated using appropriate cell lines; 2) it is very crucial to select a proper AAV serotype that can mediate a high level of gene transduction into the target tissue and 3) determine the optimal delivery route of AAV vectors for gene transfer to the desired tissue.
In the current protocol, the designed SaCas9/gRNAs were packaged into AAVrh.74 rather than previously reported AAV8 and AAV9 to edit Dmd gene12,28. AAVrh.74 exhibits about 93% homology to AAV829 and has been shown to be non-pathogenic and effective in transducing skeletal muscle following isolated limb delivery29,30,31. In this protocol, we use retro-orbital and intraperitoneal injection of rh.74 into neonatal mice for cardiac gene transfer. We found that the neonate administration of rAAVrh.74 is highly efficient for the restoration of dystrophin expression in about 30-40% of cardiomyocytes in the mice receiving a high dosage of rAAVrh.74 (1 x 1012 vg). Interestingly, we found that the skeletal muscle dystrophin expression was not effectively rescued. This is likely due to the low skeletal muscle tropism of rh.74 serotype when administered via intravenous or intraperitoneal injection, although previous studies showed that isolated limb delivery of rAAVrh.74 can effectively transduce mouse and monkey skeletal muscle29,30,31. Also, when the rAAVrh.74 was administered into adult mice, we observed very low efficiency in restoring dystrophin expression. It would be interesting to determine the minimum dosage which is required to achieve the highest dystrophin rescue. Furthermore, other serotypes and delivery methods can be tested and compared for achieving optimal cardiac gene editing.
Successful deletion of a large genomic DNA piece requires simultaneous delivery of two gRNAs. We found it is not necessary to make two separate rAAV preparations for delivering two gRNAs. In this protocol, we simply mixed the two gRNA plasmids in an equal amount during transfection for rAAV production, and this combined production of rAAV was found to be effective in delivering both gRNAs. This combined approach is, therefore, cost- and labor-saving and recommended when simultaneous delivery of two gRNAs are required.
In many cases, it may be necessary to analyze the gene editing efficiency by genetic approaches when a good antibody is not available for the target gene product. However, it is technically challenging to accurately quantify the gene editing efficacy with two gRNAs targeting the same locus since the gene editing products would include target sequence deletion, target sequence inversion, and small indels at only one target site. Instead, it is more meaningful and feasible to quantify the efficiency of the desired targeted sequence deletion as in our case where the reduction of the transcripts containing exons 21-23 can be quantified by real-time RT-PCR.
In this protocol, we also describe the use of T7E1 assay and computational prediction to analyze the potential off-target activity. For the assay to work well, it is necessary to use a high-fidelity DNA polymerase. Also, it is always a good idea to prepare for a negative control experiment (e.g. control DNA products amplified from the cells/tissues without gene editing, and treated the same way as the test samples) and a positive control experiment (e.g. a known sample with well-characterized gene editing). However, it is of note that the detection limit of the T7E1 assay is about 5%32, and thus it may not be detectable by the T7E1 assay if the off-target activity is below 5%. GUIDE-seq33 can be used to unbiasedly analyze the off-target activity of the SaCas9/gRNA-mediated gene editing. The identified off-target sites from T7E1 assay, computational prediction and/or GUIDE-seq approaches can further be quantified for indel frequencies following in vivo gene editing by high-throughput sequencing.
Taken together, we have provided an optimized cardiac gene editing approach using rAAVrh.74. This established protocol can be further tested for therapeutic applications and reverse genetic studies.
Divulgaciones
The authors have nothing to disclose.
Acknowledgements
R.H. is supported by US National Institutes of Health grants (R01HL116546 and R01 AR064241).
Materials
| Alexa Fluor 555 goat anti-rabbit IgG | Thermo Fisher Scientific | A-21428 | |
| Benzonase Nuclease | Sigma-Aldrich | E1014 | |
| Bio Safety Cabinets | Thermo Fisher Scientific | 1347 | 1300 Series A2 Class II, Type A2 |
| BsaI | New England BioLabs Inc | RO535S | |
| Competent cells | Agilent Technologies | 200314 | |
| Cell culture dishes, 150mm | Nest Scientific USA | 715001 | |
| Cryostat | Leica Biosystems | Leica CM3050S | |
| DMEM | Thermo Fisher Scientific | MT10017CV | Corning cellgro |
| Dystrophin antibody | Spring Bioscience | E2660 | |
| Fast-Ion Midi Plasmdi Kits | IBI Scientific | IB47111 | |
| FBS | Thermo Fisher Scientific | 26-140-079 | |
| Filter Unit, 0.45μm | Thermo Fisher Scientific | 166-0045 | |
| GoTaq Master Mixes | Promega | M7122 | |
| High-speed plasmid mini kit | IBI Scientific | IB47102 | |
| Horse serum | Abcam | ab139501 | |
| Isotemp 2239 Water Bath | Thermo Fisher Scientific | 15-460-11Q | |
| Isotemp Heat Block | Thermo Fisher Scientific | 11-718 | |
| Inverted confocal microscope | Carl Zeiss Microscopy, LLC | LSM 780 | |
| LightCycler 480 instrument | Roche | 5015243001 | LightCycler 480 Instrument II |
| Large Capacity Centrifuge | Thermo Fisher Scientific | 46-910 | Thermo Scientific Sorvall RC 6 Plus |
| Microcentrifuge | Thermo Fisher Scientific | 75002445 | Sorvall Legend Micro 21R |
| Nanodrop spectrophotometers | Thermo Scientific | ND2000CLAPTOP | NanoDrop™ 2000/2000c |
| Oligos for i20-F | Integrated DNA Technologies | CACCgGGCGTTGAAATTCTCATTAC | |
| Oligos for i20-R | Integrated DNA Technologies | AAAC GTAATGAGAATTTCAACGCCc; | |
| Oligos for i23-F | Integrated DNA Technologies | CACCgCACCGATGAGAGGGAAAGGTC | |
| Oligos for i23-R | Integrated DNA Technologies | GACCTTTCCCTCTCATCGGTGc | |
| Oligos | Integrated DNA Technologies | ||
| OptiPrep Density Gradient Medium | Sigma-Aldrich | D1556-250ML | |
| OptiMEM | Thermo Fisher Scientific | 31985070 | |
| PEG8000 | Sigma-Aldrich | 89510-1kg-F | |
| Polyethylenimine | Polysciences | 23966 | |
| Proteinase K | Sigma-Aldrich | P2308 | |
| Quick-Seal centrifuge tube | Beckman Coulter Inc | 342414 | |
| QIAquick Gel Extraction kit | Qiagen | 28704 | |
| Radiant SYBR Green Hi-ROX qPCR Kits | Alkali Scientific | QS2005 | |
| RevertAid RT Reverse Transcription Kit | Thermo Fisher Scientific | K1691 | |
| Rotor | Beckman Coulter Inc | 337922 | Type 70Ti |
| T4 DNA ligase | New England BioLabs Inc | M0202S | |
| Thermal Cycler | Bio-rad | 1861096 | |
| TRIzol Reagent | Thermo Fisher Scientific | 15596026 | |
| Ultracentrifuge | Beckman Coulter Inc | 8043-30-1192 | Optima le-80k |
| Ultra centrifugal filter unit | MilliporeSigma | UFC510096 | |
| VECTASHIELD Mounting Medium with DAPI | Vector Laboratories, Inc | H-1200 |
Referencias
- Makarova, K. S., Koonin, E. V. Annotation and Classification of CRISPR-Cas Systems. Methods Mol Biol. 1311, 47-75 (2015).
- Zetsche, B., et al. Cpf1 is a single RNA-guided endonuclease of a class 2 CRISPR-Cas system. Cell. 163, 759-771 (2015).
- Cong, L., et al. Multiplex genome engineering using CRISPR/Cas systems. Science. 339, 819-823 (2013).
- Mali, P., et al. RNA-guided human genome engineering via Cas9. Science. 339, 823-826 (2013).
- Lieber, M. R. The mechanism of double-strand DNA break repair by the nonhomologous DNA end-joining pathway. Annual review of biochemistry. 79, 181-211 (2010).
- Ran, F. A., et al. Genome engineering using the CRISPR-Cas9 system. Nature protocols. 8, 2281-2308 (2013).
- Zhang, X., Wang, J., Cheng, Q., Zheng, X., Zhao, G. Multiplex gene regulation by CRISPR-ddCpf1. Cell discovery. 3, 17018 (2017).
- Zhang, X. H., Tee, L. Y., Wang, X. G., Huang, Q. S., Yang, S. H. Off-target Effects in CRISPR/Cas9-mediated Genome Engineering. Molecular therapy. Nucleic acids. 4, e264 (2015).
- Xu, L., et al. CRISPR-mediated genome editing restores dystrophin expression and function in mdx mice. Mol Ther. 24, 564-569 (2015).
- Long, C., et al. Postnatal genome editing partially restores dystrophin expression in a mouse model of muscular dystrophy. Science. 351, 400-403 (2016).
- Long, C. Z., et al. Prevention of muscular dystrophy in mice by CRISPR/Cas9-mediated editing of germline DNA. Science. 345, 1184-1188 (2014).
- Nelson, C. E., et al. In vivo genome editing improves muscle function in a mouse model of Duchenne muscular dystrophy. Science. 351, 403-407 (2016).
- Ousterout, D. G. K., Thakore, A. M., Majoros, P. I., Reddy, T. E. W. H., Gersbach, C. A. Multiplex CRISPR/Cas9-based genome editing for correction of dystrophin mutations that cause Duchenne muscular dystrophy. Nat Commun. 6, 6244 (2015).
- Tabebordbar, M., et al. In vivo gene editing in dystrophic mouse muscle and muscle stem cells. Science. 351, 407-411 (2016).
- Bengtsson, N. E., et al. Muscle-specific CRISPR/Cas9 dystrophin gene editing ameliorates pathophysiology in a mouse model for Duchenne muscular dystrophy. Nat Commun. 8, 14454 (2017).
- Hoffman, E. P., Brown, R. H., Kunkel, L. M. Dystrophin: the protein product of the Duchenne muscular dystrophy locus. Cell. 51, 919-928 (1987).
- Nowak, K. J., Davies, K. E. Duchenne muscular dystrophy and dystrophin: pathogenesis and opportunities for treatment. EMBO reports. 5, 872-876 (2004).
- Mendell, J. R., et al. Evidence-based path to newborn screening for Duchenne muscular dystrophy. Annals of neurology. 71, 304-313 (2012).
- Mendell, J. R., Lloyd-Puryear, M. Report of MDA muscle disease symposium on newborn screening for Duchenne muscular dystrophy. Muscle Nerve. 48, 21-26 (2013).
- Spurney, C. F. Cardiomyopathy of Duchenne muscular dystrophy: current understanding and future directions. Muscle Nerve. 44, 8-19 (2011).
- Moriuchi, T., Kagawa, N., Mukoyama, M., Hizawa, K. Autopsy analyses of the muscular dystrophies. Tokushima J Exp Med. 40, 83-93 (1993).
- McNally, E. M. New approaches in the therapy of cardiomyopathy in muscular dystrophy. Annual review of medicine. 58, 75-88 (2007).
- Baker, D. E. Approvals, Submission, and Important Labeling Changes for US Marketed Pharmaceuticals. Hosp Pharm. 52, 306-307 (2013).
- Baker, D. E. Eteplirsen. Hosp Pharm. 52, 302-305 (2017).
- Sicinski, P., et al. The molecular basis of muscular dystrophy in the mdx mouse: a point mutation. Science. 244, 1578-1580 (1989).
- Xu, L., et al. CRISPR-mediated Genome Editing Restores Dystrophin Expression and Function in mdx Mice. Molecular therapy : the journal of the American Society of Gene Therapy. 24, 564-569 (2016).
- El Refaey, M., et al. In Vivo Genome Editing Restores Dystrophin Expression and Cardiac Function in Dystrophic Mice. Circ Res. 121, 923-929 (2017).
- Tabebordbar, M., et al. In vivo gene editing in dystrophic mouse muscle and muscle stem cells. Science. 351, 407-411 (2016).
- Pozsgai, E. R., Griffin, D. A., Heller, K. N., Mendell, J. R., Rodino-Klapac, L. R. beta-Sarcoglycan gene transfer decreases fibrosis and restores force in LGMD2E mice. Gene therapy. 23, 57-66 (2016).
- Chicoine, L. G., et al. Plasmapheresis eliminates the negative impact of AAV antibodies on microdystrophin gene expression following vascular delivery. Mol Ther. 22, 338-347 (2014).
- Chicoine, L. G., et al. Vascular delivery of rAAVrh74.MCK.GALGT2 to the gastrocnemius muscle of the rhesus macaque stimulates the expression of dystrophin and laminin alpha2 surrogates. Mol Ther. 22, 713-724 (2014).
- Vouillot, L., Thelie, A., Pollet, N. Comparison of T7E1 and surveyor mismatch cleavage assays to detect mutations triggered by engineered nucleases. G3. 5, 407-415 (2015).
- Tsai, S. Q., et al. GUIDE-seq enables genome-wide profiling of off-target cleavage by CRISPR-Cas nucleases. Nat Biotechnol. 33, 187-197 (2015).

