Real-time Visualization and Analysis of Chondrocyte Injury Due to Mechanical Loading in Fully Intact Murine Cartilage Explants
Summary
We present a method to assess the spatial extent of cell injury/death on the articular surface of intact murine joints after application of controlled mechanical loads or impacts. This method can be used to investigate how osteoarthritis, genetic factors and/or different loading regimens affect the vulnerability of in situ chondrocytes.
Abstract
Homeostasis of articular cartilage depends on the viability of resident cells (chondrocytes). Unfortunately, mechanical trauma can induce widespread chondrocyte death, potentially leading to irreversible breakdown of the joint and the onset of osteoarthritis. Additionally, maintenance of chondrocyte viability is important in osteochondral graft procedures for optimal surgical outcomes. We present a method to assess the spatial extent of cell injury/death on the articular surface of intact murine synovial joints after application of controlled mechanical loads or impacts. This method can be used in comparative studies to investigate the effects of different mechanical loading regimens, different environmental conditions or genetic manipulations, as well as different stages of cartilage degeneration on short- and/or long-term vulnerability of in situ articular chondrocytes. The goal of the protocol introduced in the manuscript is to assess the spatial extent of cell injury/death on the articular surface of murine synovial joints. Importantly, this method enables testing on fully intact cartilage without compromising native boundary conditions. Moreover, it allows for real-time visualization of vitally stained articular chondrocytes and single image-based analysis of cell injury induced by application of controlled static and impact loading regimens. Our representative results demonstrate that in healthy cartilage explants, the spatial extent of cell injury depends sensitively on load magnitude and impact intensity. Our method can be easily adapted to investigate the effects of different mechanical loading regimens, different environmental conditions or different genetic manipulations on the mechanical vulnerability of in situ articular chondrocytes.
Introduction
Articular cartilage (AC) is a load bearing tissue that covers and protects bones in synovial joints, providing smooth joint articulation. Tissue homeostasis is dependent on the viability of chondrocytes, the sole cell type residing in AC. However, exposure of cartilage to extreme forces due to trauma (e.g., falls, vehicle accident or sports injuries) or due to post-traumatic joint instability can induce chondrocyte death, leading to irreversible breakdown of the joint (osteoarthritis)1. Furthermore, in osteochondral grafting procedures that aim to repair local defects in damaged cartilage, graft insertion-associated mechanical trauma reduces chondrocyte viability and has detrimental effects on surgical outcomes2.
Cartilage explant models are commonly used to study the susceptibility of articular chondrocytes to mechanically-induced cell death. These models typically use explants from large animals to study the effects of loading conditions, environmental conditions and other factors on cell vulnerability3,4,5,6,7,8,9,10,11,12,13,14,15. However, due to the large size of the native joints, these models generally require removal of a plug from the articular surface of an intact joint, thereby compromising native boundary conditions. Moreover, they generally require application of large mechanical loads to induce cell injury. Alternatively, murine cartilage explant models provide several advantages over larger animal models in studying the mechanical vulnerability of in situ chondrocytes. In particular, due to their smaller dimensions, these models facilitate testing of fully intact articular cartilage without altering native tissue integrity. In addition, loading of murine cartilage occurs over small contact areas such that chondrocyte death/injury can be induced with small loads (<1 N). Finally, the mouse genome is easily manipulated, enabling testing of how specific genes impact the susceptibility of in situ chondrocytes to mechanical injury.
The overall goal of the method introduced in this manuscript is to quantify and visualize-in real-time-the spatial extent of in situ cell death/injury due to applied mechanical loads on fully intact mouse cartilage-on-bone explants in vitro. This method requires careful dissection of mouse synovial joints without compromising chondrocyte viability, followed by mechanical testing of vitally stained explants using a microscope-mounted device similar to a testing platform that we recently developed to quantify murine cartilage mechanical properties16. During mechanical testing, a large portion of the (intact) articular surface of the dissected bone is visible on a single fluorescence micrograph, enabling rapid analysis of cell viability after a load is applied. A similar analysis of surface cell viability in murine cartilage explants has been performed previously, but without simultaneous application of load17. Potential applications of our method include comparative studies to investigate the vulnerability of articular chondrocytes to different controlled environmental and mechanical conditions, as well as screening of treatments aimed at reducing the sensitivity of chondrocytes to mechanical loading.
Protocol
All animal work was approved by the University of Rochester Committee on Animal Resources.
1. Solutions
- Prepare Hank's Balanced Salt Solution (1X HBSS) containing calcium, magnesium and no phenol red. Adjust the pH to 7.4 by adding small amounts of HCl or NaOH.
- Adjust the osmolarity to 303 mOsm by adding NaCl or deionized water. Use the buffer during the dissections, specimen preparation and mechanical testing.
2. Dissections of the Distal Femur and Proximal Humerus with Fully Intact Articular Cartilage
- Euthanize the mouse according to institutional guidelines using a CO2 inhalation chamber followed by cervical dislocation. Follow aseptic technique throughout.
NOTE: Mice between 8 and 81 weeks old of any strain and sex may be used. - Use the following surgical tools for dissections: micro-scissors (used for making incisions), standard dissecting scissors (used for cutting the bone), scalpel with #11 scalpel blade (used for cutting soft tissue), jeweler's forceps (used for removal of soft tissue) and standard forceps (used for peeling the soft tissue and the skin).
- For dissections of the distal femur follow the instructions below.
- Position the mouse in a supine position.
- Make a small (~5 mm) incision in the skin on the anterior portion of the knee.
- Extend the incision all the way around the knee and pull back the skin to expose the knee joint and leg muscles.
- Starting from the proximal end of the femur, use a scalpel blade (#11) to cut along the distal direction. Position the blade between the quadriceps muscle-tendon unit and the anterior side of the femoral shaft. Extend the cut towards and past the patella and finish by cutting through the middle of the patellar tendon, thereby removing the quadriceps muscle-tendon unit.
- Starting from the proximal end of the femur, use a scalpel blade (#11) to cut along the distal direction. Position the blade between the hamstring muscle-tendon unit and the posterior side of the femoral shaft. Once the cut approaches the knee joint, begin cutting through the soft tissue, avoiding contact between the scalpel and the articular surface on the distal condyles of the femur. Finish the cut past the knee joint.
- Pull back the quadriceps and hamstring muscles to expose the femur. Cut away any excess muscle on the lateral and medial sides of the femur.
- Cut the calf muscle on the posterior side of the proximal tibia and flip the leg to visualize the posterior side of the femur.
- Expose the distal condyles of the femur and posterior surface of the proximal tibia by removing excess tissue around the knee joint.
- Cut the anterior and posterior cruciate ligaments using a scalpel, cutting away from the femoral condyles. Pull the tibia away from the femur and cut all the ligaments to separate the lower leg from the femur.
- Using standard dissecting scissors, cut through the femur at the proximal end of the bone (8 mm above the tibio-femoral joint) from the lateral side. After cutting the bone, wipe away any visible marrow on the outside of the bone to avoid possible contamination from bone marrow cells.
NOTE: Cutting from lateral side reduces the risk of crack propagation along the bone. - Remove surrounding soft tissues (i.e., ligaments and excess muscle) from the femur using jeweler's forceps and expose the cartilage on both condyles at the distal end of the femur. Avoid contact between the cartilage and forceps.
- For dissections of the humerus follow the instructions below.
- Position the mouse in a supine position.
- Make an incision (~5 mm) in the skin on the posterior side of the elbow using micro-scissors, extend the incision around the elbow and pull back the skin to expose the muscles of the arm and shoulder.
- Starting from the proximal end of the humerus, use a scalpel blade (#11) to cut along the distal direction. Position the blade between the triceps muscle-tendon unit and the posterior side of the humerus. Extend the cut towards the distal end of the humerus and finish by cutting through the triceps tendon.
- Then pull back the triceps towards the proximal end of the humerus until the humeral head is exposed.
- Cut the connective tissue around the humeral head using a scalpel blade (#11) without touching the articular surface and remove the limb (arm and shoulder) from the body. Periodically hydrate the articular surface of the humeral head with HBSS.
- Disconnect the humerus from the arm by first breaking off the proximal end of the ulna on the posterior side of the arm using forceps and then cutting the connective tissue around the distal end of the humerus. Remove any excess tissue on the humerus.
- Cut off the deltoid tuberosity on the posterior side of the humerus using standard dissecting scissors.
- Place the dissected specimen(s) into a 1.5 mL microcentrifuge tube containing HBSS buffer.
3. Live (Calcein AM) / Dead (Propidium Iodide) Staining Protocol
- Stain using calcein AM as follows.
- Make a stock solution of calcein AM by adding 12.5 µL of dimethyl sulfoxide (DMSO) into a vial of 50 µg of calcein AM, resulting in a stock concentration of 4 mg/mL (4.02 mM).
- Dilute the stock calcein solution 1:400 in HBSS to reach a concentration of 10 µg/mL (10.05 µM) of calcium AM (for example, add 1.25 µL of stock solution to 500 µL of HBSS).
- Centrifuge the diluted staining solution for 5 s at 2,000 x g to ensure that all of the dye, which may occasionally stick to the walls of the tube, mixes with the buffer. Do not remove the supernatant. Vortex the solution to ensure proper mixing.
- Put the dissected specimen into a 1.5 mL microcentrifuge tube containing 500 µL of the diluted staining solution. Incubate the specimen at 37 °C for 30 min in a thermomixer while agitating at 800 rpm. Make sure the tubes are covered in aluminum foil to protect from exposure to light.
- Transfer the specimen into calcein AM-free HBSS buffer. At this point, the specimen is ready for mechanical testing.
- Prepare propidium iodide (PI) staining solution as follows.
- Prepare the PI staining solution by diluting the purchased stock solution (1 mg/mL, 1.5 mM) 1:25 in HBSS to reach 40 µg/mL (60 µM) of PI. For example, add 40 µL of stock solution to 1000 µL of HBSS.
- Keep the PI staining solution covered in aluminum foil to protect it from exposure to light. Use the staining solution immediate after the mechanical test to detect injured cells (see section 4).
NOTE: One can perform the PI staining prior to loading as well to more rigorously assess the quality of the dissections.
4. Mechanical Testing Protocol
- Place the specimen (femur or humerus) onto the glass slide of a custom microscope-mounted mechanical testing device such that the articular surface on the posterior femoral condyles or humeral head is sitting on the glass (Figure 1).
NOTE: Fasten a 10 mm-long wooden applicator (diameter = 2 mm) to the distal side of the humerus using cyanoacrylate glue before placing it onto the device in order to stabilize the specimen on the flat surface. For a detailed description of the mechanical testing platform see Supplementary File 1: section 1. - Hydrate the specimen with HBSS and place the device with the specimen onto a fluorescence microscope.
- Image the articular chondrocytes stained with calcein AM (excitation/emission wavelengths = 495/515 nm) before (baseline) application of the load under a fluorescence microscope with a 4X dry lens (NA = 0.13). Adjust the acquisition settings to optimize the image quality.
- Apply mechanical loading on top of the specimen such that articular cartilage is compressed against the cover glass.
- For static loading, apply a prescribed static load (e.g., 0.5 N) on top of the specimen (femur or humerus). Hold the load for 5 min and then remove it.
- For impact, apply a prescribed impact energy (e.g., 1 mJ) onto the specimen by dropping a cylindrical impactor of known weight (e.g., 0.1 N) from a prescribed height (e.g., 1 cm). Release the load 5 s after the impact.
NOTE: The weight of the impactor (mg) and the prescribed height (h) can be converted into impact energy (E) using the following equation: E = mgh.
- Incubate the specimen in PI staining solution for 5 min at room temperature.
- Image the articular chondrocytes stained with calcein AM and PI (excitation/emission wavelengths = 535/617 nm) under a fluorescence microscope (Figure 2).
5. Data Analysis
- Quantify the area of injured/dead cells due to the applied mechanical loading regimen.
- Open the micrographs of the articular surface acquired before and after application of mechanical load in ImageJ18.
- Combine the images into a stack.
- Set the scale of the images based on the image resolution.
- Use the Polygon tool to define the area where the cells became calcein-negative (loss of green fluorescence) and PI-positive (gain of red fluorescence). These cells are considered to be injured or dead.
- Determine the area of injured/dead cells using the Measurement tool.
Representative Results
Six different applied loading protocols (static loading: 0.1 N, 0.5 N and 1 N for 5 min; and impact loading: 1 mJ, 2 mJ and 4 mJ) reproducibly induced quantifiable localized areas of cell injury in femoral and humeral cartilage obtained from 8-10-week-old BALB/c mice (Figure 2). Importantly, the spatial extent of chondrocyte injury on the articular surface was measured quickly and easily in ImageJ. Representative results demonstrate that the mechanical vulnerability of articular chondrocytes was affected by load magnitude and impact intensity. In particular, higher load magnitudes and higher impact intensities significantly exacerbated the spatial extent of cell injury in both femurs and humeri (Figure 3).
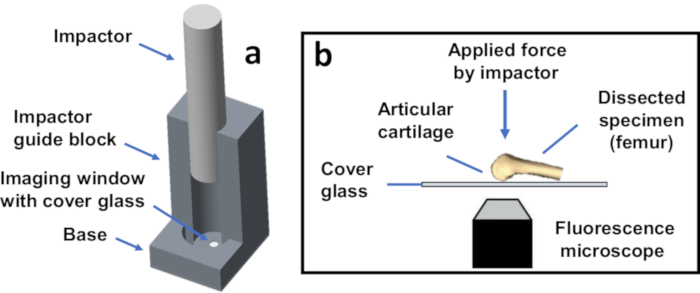
Figure 1: Schematic representation of the custom mechanical testing device. (a) Assembled device with the cylindrical impactor used to apply prescribed mechanical loads and/or impact energies to a specimen. The device is shown without a specimen. (b) Schematic representation of the experiment. Controlled static (e.g., 0.1 N) and/or impact (e.g., 1 mJ) loading can be applied on top of the dissected specimen such that articular cartilage is compressed against the cover glass. Please click here to view a larger version of this figure.
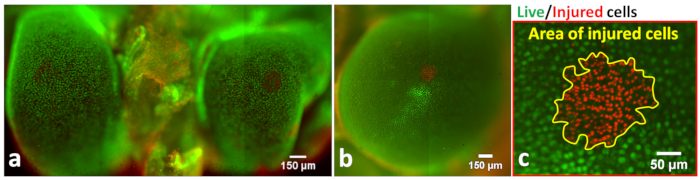
Figure 2: Representative micrograph of the articular surface after injurious mechanical loading. Representative micrograph of the articular surface on (a) the distal femoral condyles and (b) the humeral head after injurious mechanical loading (impact [1 mJ] and static loading [1 N], respectively). Green cells are vitally stained chondrocytes with intact cell membranes while red nuclei indicate injured cells with permeabilized cell membranes. (c) Zoomed-in view of the area of injured/dead cells (yellow contours) on the articular surface of the humeral head. Please click here to view a larger version of this figure.
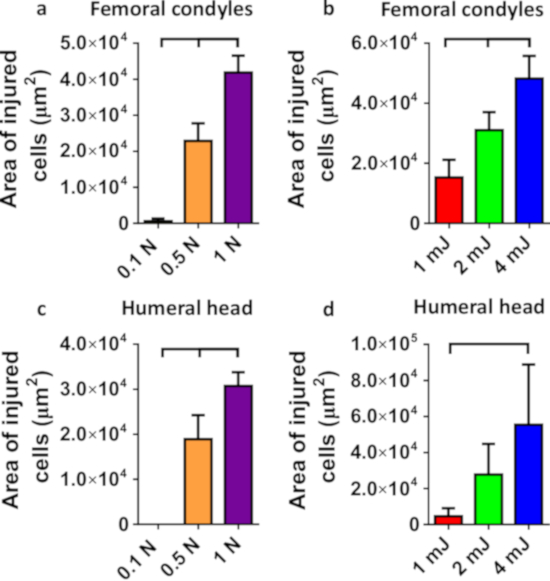
Figure 3: Area of injured/dead cells. Area of injured/dead cells on the articular surface of the distal femoral condyles (a, b) and the humeral head (c, d) after (a, c) static (0.1 N, 0.5 N and 1 N; n = 6 per group) and (b, d) impact (1 mJ, 2 mJ and 4 mJ; n = 6 per group) loading. All cartilage-on-bone specimens were obtained from female BALB/c 8-10 weeks old mice. Data are mean + standard deviation; brackets denote statistical significance at α=0.05 determined by analysis of variance (ANOVA) test with Tukey post hoc comparisons. Please click here to view a larger version of this figure.
Supplementary File 1. Please click here to download this file.
Discussion
The methods described above were successfully employed to visualize viable and injured/dead in situ articular chondrocytes from mouse joints after prescribed mechanical loads or impacts. In particular, we were able to analyze the mechanical vulnerability of chondrocytes within fully intact articular cartilage from two different synovial joints: the knee joint (distal femurs) and shoulder (humeri). Our representative results show that the spatial extent of cell injury on the articular surface depends sensitively on load magnitude and impact intensity (Figure 3). Importantly, the use of this method facilitates investigations of the cellular response to mechanical loading under physiologically relevant conditions. That is, it enables testing of articular cartilage on an intact joint under physiological and supraphysiological loads (see Supplementary File 1: section 2).
Given the steep learning curve in performing dissections of murine synovial joints and challenges in preserving viable in situ chondrocytes at baseline, some protocol modification and troubleshooting may be required. The greatest risk of damaging articular chondrocytes during dissection occurs during steps 2.3.5 through 2.3.10 and 2.4.5 through 2.4.7. To minimize cell injury/death during dissections, the researcher should avoid any contact between surgical tools (e.g., the scalpel when cutting the tissue or the jeweler's forceps when removing the soft tissue) and the articular surface of the specimen. However, touching the articular surface with a glove induces little cell injury/death. In order to improve baseline cell viability, it may also be necessary to reduce the amount of soft tissue removed from the joint. Additionally, using finer tools will generally reduce dissection-induced cell death. Ultimately, to rigorously confirm the absence of dissection-associated damage of articular chondrocytes at baseline, it is advisable to stain the specimens with both permeability dyes (calcein AM and PI, prior to loading) especially while the researcher is becoming familiar with the dissection procedure (see Supplemental File 1: section 3).
Given the small size of the mouse joint and the genetic manipulability of the mouse genome, murine models provide multiple advantages over large animal models to study vulnerability of articular chondrocytes to mechanical loading. However, to the authors' knowledge, no studies have previously been conducted to quantify injury/death of in situ articular chondrocytes due to mechanical loading of intact murine cartilage. Investigators typically use explants removed from joints of large animal models to investigate the extent of cell death due to mechanical injury3,4,5,6,7,8,9,10,11,12. In contrast, mouse models facilitate 1) visualization of nearly the entire articular surface on a given bone; and 2) analyses of post-loading or post-impact chondrocyte viability in intact joints without compromising native boundary conditions. Furthermore, while compressed, mouse specimens generate substantially smaller contact areas compared to large animal models; therefore, stresses are dramatically higher than in large animal models for a given load magnitude. Hence, cell injury can be induced by smaller loads. Additionally, murine models facilitate research on cartilage degeneration and, in particular, osteoarthritis, as this disease can be easily induced in mice through genetic19,20,21,22, dietary23,24 or surgical manipulations25,26,27,28. Moreover, spontaneous osteoarthritis occurs in several mouse strains including BALB/c and C57BL/629,30.
In our representative data, we used viability (live/dead) stains to quantify cell injury/death 5 min after the removal of mechanical loading. We acknowledge that these experiments may not discriminate injured cells (cells with temporarily ruptured membranes) from dead cells (cells with permanently ruptures membranes). That is, cells that are calcein negative and PI positive- indicating that the membrane was previously permeabilized-may repair their membranes over time scales ranging from seconds to several minutes31. In fact, in a separate set of experiments, we have determined that the fraction of "injured" cells that survive mechanical trauma is small (~5%) but significant (see Supplementary File 1: section 4). Therefore, live/dead staining is a direct measure of membrane integrity that is not always indicative of cell viability. In particular, PI-positive and calcein-negative cells are most appropriately defined as "injured", where injury is defined as a (potentially temporary) loss of plasma membrane integrity due to mechanical trauma. We also acknowledge that our representative data likely reflects only immediate (necrotic) cell death. Imaging specimens at later time points (e.g., 48 h after removal of load) should enable quantification of both necrotic and apoptotic cell death.
Several limitations must be considered when using these methods. In our trial experiments for this manuscript, we used 8-10-weeks-old female BALB/c mice to demonstrate the capabilities of the testing platform (Figure 3). However, due to noticeable alterations in cell density in femurs of older mice, the assessment of load-induced cell injury becomes more challenging (though feasible) in older femurs (success rate = 40%). In contrast, no noticeable alterations in cell density on humeri were observed in 61-81-week-old C57BL/6 mice (see Supplementary File 1: section 5), thereby making the testing platform useful for ages 8-81 weeks. Another limitation is that the method was used to analyze the spatial extent of cell injury only on the articular surface of femoral condyles and humeral head. However, the method could further be extended to analyze depth-dependent spatial extent of cell injury through use of laser scanning confocal microscopy. Note that the latter method would require usage of more expensive equipment, longer image acquisition time, and more involved and time-consuming analysis. Finally, although the contact location between the articular cartilage and the cover glass was physiologically relevant for mice32,33, this location was not varied. However, our testing platform allows for variation of the contact location if the femur or humerus is gripped with a rotating armature.
In conclusion, we have developed an in vitro murine injury model that enables application of controlled mechanical loads and/or impacts onto the articular surface of intact articular cartilage. This model enables visualization of fluorescently labeled articular chondrocytes in real-time and rapid single image-based analysis of cell injury/death. Importantly, the effects of different mechanical loading regimens, different environmental conditions or different genetic manipulations on short- and/or long-term chondrocyte viability can be tested using this methodology. Thus, our platform provides a tool to interrogate basic science questions and screen therapeutic targets related to the mechanical vulnerability of chondrocytes.
Divulgaciones
The authors have nothing to disclose.
Acknowledgements
The authors would like to thank Dr. Richard Waugh and Luis Delgadillo for the generous use of their pH meter and osmometer. Additionally, the authors would like to thank Andrea Lee for contributing to the initial development of the mechanical testing system. This study was funded by NIH P30 AR069655.
Materials
| Calcein, AM | Invitrogen by Thermo Fisher Scientific | C3100MP | 20x50mg , Eugene, OR, USA |
| Propidium Iodide | Invitrogen by Thermo Fisher Scientific | P3566 | 1 mg/mL solution in water, 10mL, Eugene, OR, USA |
| Dimethyl sulfoxide (DMSO) | Sigma-Aldrich | 276855 | 1L DMSO, anhydrous, ≥99.9%, St. Louis, MO, USA |
| HBSS (calcium, magnesium, no phenol red) | Gibco by Thermo Fisher Scientific | 14025-092 | 1X, 500mL, Grand Island, NY, USA |
| Feather surgical blade (#11) | VWR | 102097-822 | Hatfield, PA, USA |
| Vapor pressure osmometer, VAPRO | ELITechGroup | Model 5520 | Puteaux, France |
| pH meter | Beckman | Model Phi 32 | Brea, CA, USA |
| Eppendorf thermomixer | Eppendorf AG | Model 5350 | Hamburg, Germany |
| Motorized inverted research microscope | Olypmus | Model IX-81 | Center Valley, PA, USA |
| Wooden applicator | Puritan Medical Products Company, LLC | 807 | 6"x100, Guilford, ME, USA |
| 1.5 Glass coverslips | Warner Instruments, LLC | 64-1696 | #1.5, 0.17mm thick, 40mm diameter, Hamden, CT, USA |
Referencias
- Lotz, M. K., Kraus, V. B. New developments in osteoarthritis. Posttraumatic osteoarthritis: pathogenesis and pharmacological treatment options. Arthritis Research & Therapy. 12 (3), 211 (2010).
- Pallante, A. L., et al. The in vivo performance of osteochondral allografts in the goat is diminished with extended storage and decreased cartilage cellularity. American Journal of Sports Medicine. 40 (8), 1814-1823 (2012).
- Delco, M. L., Bonnevie, E. D., Bonassar, L. J., Fortier, L. A. Mitochondrial dysfunction is an acute response of articular chondrocytes to mechanical injury. Journal of Orthopaedic Research. 36 (2), 739-750 (2018).
- Ewers, B. J., Dvoracek-Driksna, D., Orth, M. W., Haut, R. C. The extent of matrix damage and chondrocyte death in mechanically traumatized articular cartilage explants depends on rate of loading. Journal of Orthopaedic Research. 19 (5), 779-784 (2001).
- Goodwin, W., et al. Rotenone prevents impact-induced chondrocyte death. Journal of Orthopaedic Research. 28 (8), 1057-1063 (2010).
- Issa, R., Boeving, M., Kinter, M., Griffin, T. M. Effect of biomechanical stress on endogenous antioxidant networks in bovine articular cartilage. Journal of Orthopaedic Research. 36 (2), 760-769 (2018).
- Bartell, L. R., Fortier, L. A., Bonassar, L. J., Cohen, I. Measuring microscale strain fields in articular cartilage during rapid impact reveals thresholds for chondrocyte death and a protective role for the superficial layer. Journal of Biomechanics. 48 (12), 3440-3446 (2015).
- Levin, A. S., Chen, C. T., Torzilli, P. A. Effect of tissue maturity on cell viability in load-injured articular cartilage explants. Osteoarthritis and Cartilage. 13 (6), 488-496 (2005).
- Lee, W., et al. Synergy between Piezo1 and Piezo2 channels confers high-strain mechanosensitivity to articular cartilage. Proceedings of the National Academy of Sciences of the United States of America. 111 (47), E5114-E5122 (2014).
- Jeffrey, J. E., Gregory, D. W., Aspden, R. M. Matrix damage and chondrocyte viability following a single impact load on articular cartilage. Archives of Biochemistry and Biophysics. 322 (1), 87-96 (1995).
- Chen, C. T., Bhargava, M., Lin, P. M., Torzilli, P. A. Time, stress, and location dependent chondrocyte death and collagen damage in cyclically loaded articular cartilage. Journal of Orthopaedic Research. 21 (5), 888-898 (2003).
- Morel, V., Mercay, A., Quinn, T. M. Prestrain decreases cartilage susceptibility to injury by ramp compression in vitro. Osteoarthritis and Cartilage. 13 (11), 964-970 (2005).
- Sauter, E., Buckwalter, J. A., McKinley, T. O., Martin, J. A. Cytoskeletal dissolution blocks oxidant release and cell death in injured cartilage. Journal of Orthopaedic Research. 30 (4), 593-598 (2012).
- Martin, J. A., Buckwalter, J. A. Post-traumatic osteoarthritis: the role of stress induced chondrocyte damage. Biorheology. 43 (3,4), 517-521 (2006).
- Martin, J. A., Brown, T., Heiner, A., Buckwalter, J. A. Post-traumatic osteoarthritis: the role of accelerated chondrocyte senescence. Biorheology. 41 (3-4), 479-491 (2004).
- Kotelsky, A., Woo, C. W., Delgadillo, L. F., Richards, M. S., Buckley, M. R. An Alternative Method to Characterize the Quasi-Static, Nonlinear Material Properties of Murine Articular Cartilage. Journal of Biomechanical Engineering. 140 (1), (2018).
- Zhang, M., et al. Induced superficial chondrocyte death reduces catabolic cartilage damage in murine posttraumatic osteoarthritis. Journal of Clinical Investigation. 126 (8), 2893-2902 (2016).
- Schneider, C. A., Rasband, W. S., Eliceiri, K. W. NIH Image to ImageJ: 25 years of image analysis. Nature Methods. 9 (7), 671-675 (2012).
- Habouri, L., et al. Deletion of 12/15-lipoxygenase accelerates the development of aging-associated and instability-induced osteoarthritis. Osteoarthritis and Cartilage. 25 (10), 1719-1728 (2017).
- Higuchi, Y., et al. Conditional knockdown of hyaluronidase 2 in articular cartilage stimulates osteoarthritic progression in a mice model. Scientific Reports. 7 (1), 7028 (2017).
- Zhu, M., et al. Activation of beta-catenin signaling in articular chondrocytes leads to osteoarthritis-like phenotype in adult beta-catenin conditional activation mice. Journal of Bone and Mineral Research. 24 (1), 12-21 (2009).
- Hu, K., et al. Pathogenesis of osteoarthritis-like changes in the joints of mice deficient in type IX collagen. Arthritis & Rheumatology. 54 (9), 2891-2900 (2006).
- Mooney, R. A., Sampson, E. R., Lerea, J., Rosier, R. N., Zuscik, M. J. High-fat diet accelerates progression of osteoarthritis after meniscal/Ligamentous injury. Arthritis Research & Therapy. 13 (6), R198 (2011).
- Griffin, T. M., Huebner, J. L., Kraus, V. B., Yan, Z., Guilak, F. Induction of osteoarthritis and metabolic inflammation by a very high-fat diet in mice: effects of short-term exercise. Arthritis & Rheumatology. 64 (2), 443-453 (2012).
- Kamekura, S., et al. Osteoarthritis development in novel experimental mouse models induced by knee joint instability. Osteoarthritis and Cartilage. 13 (7), 632-641 (2005).
- Glasson, S. S., Blanchet, T. J., Morris, E. A. The surgical destabilization of the medial meniscus (DMM) model of osteoarthritis in the 129/SvEv mouse. Osteoarthritis and Cartilage. 15 (9), 1061-1069 (2007).
- Huang, H., Skelly, J. D., Ayers, D. C., Song, J. Age-dependent Changes in the Articular Cartilage and Subchondral Bone of C57BL/6 Mice after Surgical Destabilization of Medial Meniscus. Scientific Reports. 7, 42294 (2017).
- Hamada, D., Sampson, E. R., Maynard, R. D., Zuscik, M. J. Surgical induction of posttraumatic osteoarthritis in the mouse. Methods in Molecular Biology. 1130, 61-72 (2014).
- Wilhelmi, G., Faust, R. Suitability of the C57 black mouse as an experimental animal for the study of skeletal changes due to ageing, with special reference to osteo-arthrosis and its response to tribenoside. Pharmacology. 14 (4), 289-296 (1976).
- Stoop, R., et al. Type II collagen degradation in spontaneous osteoarthritis in C57Bl/6 and BALB/c mice. Arthritis & Rheumatism. 42 (11), 2381-2389 (1999).
- McNeil, P. L., Kirchhausen, T. An emergency response team for membrane repair. Nature Reviews Molecular Cell Biology. 6 (6), 499-505 (2005).
- Adebayo, O. O., et al. Kinematics of meniscal- and ACL-transected mouse knees during controlled tibial compressive loading captured using roentgen stereophotogrammetry. Journal of Orthopaedic Research. 35 (2), 353-360 (2017).
- Vahedipour, A., et al. Uncovering the structure of the mouse gait controller: Mice respond to substrate perturbations with adaptations in gait on a continuum between trot and bound. Journal of Biomechanics. , (2018).

